Production of Nurr-1 Specific Polyclonal Antibodies Free of Cross-reactivity Against Its Close Homologs, Nor1 and Nur77
Summary
Here, we selectively target antibodies against a specific member of a highly conserved family of proteins by immunizing animals with their most divergent regions followed by removing cross reactive antibodies by pre-adsorption.
Abstract
The nuclear receptor subfamily 4 (NR4A) is composed of 3 related proteins sharing a DNA binding domain (DBD) and a ligand-binding domain (LBD). The nuclear receptor related 1 protein (Nurr1 or NR4A2) plays a key role in the maintenance of the dopaminergic system. Dopamine dysfunctions associated with the Nurr1 gene include Parkinson’s disease, schizophrenia and manic depression among others. Furthermore, recent evidence indicates that Nurr1 is also expressed in other brain areas such as the hippocampus and plays critical roles for learning and memory. The other members of the family are nerve growth factor IB (Nur77 or NR4A1) and neuron-derived orphan receptor 1 (NOR1 or NR4A3). To help investigate the precise functional roles of Nurr1 in dopaminergic and other brain region-related neuronal dysfunctions antibodies devoid of cross-reactivities against Nur77 and NOR1 were needed. Since the proteins are more divergent in their LBDs than in their DNA binding domains immunization with purified LBDs should yield antibodies specific for Nurr1 with minimal reactivities against Nur77 and/or NOR1. Although anti-Nurr1 antibodies were successfully generated these showed significant immunoreactivity against the other members of the family. Affinity chromatography over immobilized Protein A followed by pre-adsorption against immobilized Nur77 and NOR1 LBDs yielded Nurr1 specific antibodies free of cross-reactivity. Here, we selectively target antibodies against a specific member of a highly conserved family of proteins by immunizing animals with their most divergent regions followed by removing cross reactive antibodies by pre-adsorption. The goal of the protocol is to increase polyclonal antibodies specificity through pre-adsorption against cross-reactive antigens.
Introduction
The transcription factor Nurr1 and its homologs (Nur77 and Nor1) belong to the nuclear receptor subfamily 4A (NR4A)1. They are also orphan receptors because their endogenous ligands are not identified yet. Nurr1 was first cloned in 1992 and although known to be expressed in the brain2, its essential role for development and maintenance of midbrain dopamine neurons was revealed by knockout mouse studies3. In addition, its important role for maintenance of midbrain dopamine neurons was recently demonstrated by a conditional knockout study4. Due to these elegant studies showing Nurr1’s critical roles for midbrain dopamine neurons, many subsequent studies have largely focused on the midbrain dopamine neurons and dopamine-related neurodegenerative disorder, Parkinson’s disease (PD)5.
Notably, Nurr1 is expressed not only in the midbrain dopamine (mDA) neurons, but also in diverse brain areas2, suggesting that it may have functional roles in many non-DA areas, which is strongly supported by more recent studies showing that Nurr1 plays important roles in various brain functions. For examples, it was shown that memory-inducing activities such as learning, or other hippocampus-dependent tasks result in up-regulation of Nurr1 expression in the hippocampus6,7. In addition, knock down of Nurr1 expression in the hippocampus was sufficient to impair long-term memory and/or synaptic plasticity8-11, strongly suggesting that Nurr1 plays diverse roles in many brain areas. Thus, to further understand the cell-type-specific and subcellular expression of Nurr1, it is desirable to use Nurr1-specific antibodies, which do not exhibit any cross-reactivity to its homologs Nur77 or Nor1. This paper describes a pre-adsorption protocol to generate Nurr1-specific antibodies and present additional data showing its specificity.
Protocol
Note: The composition of all solutions cited below can be found in the Materials/Equipment Table.
1. Protein A Column Antibody Purification
- Equilibrate a protein A spin column and all required buffers to room temperature for 15 min prior to starting the procedure.
- Dilute the immune serum 1:1 with Protein A IgG Binding Buffer. (5 ml of serum is recommended).
- Gently tap a protein A spin column on the bench top to dislodge any resin that may be lodged in the cap. Remove the top cap and gently snap off the bottom closure. Place the column in a 15 ml collection tube and allow storage solution to drain.
- Add 5 ml of Protein A IgG Binding Buffer and allow the solution to drain.
- Apply the diluted immune serum to the column and collect the flow-through. For best results, add a sample volume that contains a concentration of antibodies less than 80% of the column’s antibody-binding capacity.
- Wash the column with 15 ml of Protein A IgG Binding Buffer or until the absorbance at 280 nm is below 0.1.
- Add 100 µl of Neutralization Buffer to five labeled collection tubes.
- Add 5 ml of IgG Elution Buffer to the Protein A column and collect 1 ml fractions in each of the tubes containing the 100 µl of Neutralization Buffer.
- Measure the absorbance of each fraction at 280 nm and pool fractions with an absorbance greater than 0.5. Keep the pooled fractions on ice until ready for dialysis.
- Regenerate the column by adding 8 ml of IgG Elution Buffer and allow the solution to flow through the column.
- Rinse the column with Protein A IgG Binding solution until the eluent pH returns to 7.4.
- Store the column by adding 5 ml of storage solution (0.02% sodium azide in PBS). When approximately 3 ml remain in the column, cap bottom and secure the top cap. Store the column at 4 °C.
- Determine the length of dialysis tubing needed according to the manufacturer’s instructions. Using 16 mm flat diameter tubing use 5.47 cm of tube length per ml of solution to be dialyzed. Cut the tubing and rinse with PBS pH 7.4 for 15 to 30 min.
- Fold and clip one end of the dialysis tubing, transfer the pooled IgG containing fractions to dialysis tubing equilibrated in PBS pH 7.4 and close the other end.
- Dialyze at room temperature against 200 volumes of PBS pH 7.4 for 2 hr.
- Change the dialysis buffer (fresh PBS pH 7.4) and dialyze for another 2 hr at room temperature.
- Change the dialysis buffer (fresh PBS pH 7.4) and dialyze overnight at 4 °C.
- Keep the dialyzed antibody solution at 4 °C until ready to proceed with further purification steps.
- Assess the antibody concentration using its absorbance at 280 nm and the molar absorption coefficient of antibodies at 280 nm of 210,000 M-1 cm-112.
2. Coupling of LBD Proteins to AminoLink Coupling Resin
- Prepare two columns, one for Nor1 and the other for Nur77. Follow the same protocol for both proteins.
- Thaw the protein to be coupled to the resin on ice. Determine the protein concentration using its molar extinction coefficient and its absorbance at 280 nm12.
- Alternatively, use a protein determination assay such as the bicinchoninic acid assay13. If the protein is dissolved in an amine-containing buffer dialyze or buffer exchange against the Coupling Buffer. If the protein is in a suitable buffer (no free amines) dilute it 4-fold in Coupling Buffer.
- Use 2 ml of resin for 2 mg of protein. All steps that follow are for 2 ml resin in a 10 ml column. Up to 10 mg of protein can be linked per 1 ml of resin (2 ml of 1:1 slurry).
- Prepare a 10 ml column by pushing a frit to the bottom and running PBS pH 7.4 through it. Cap the bottom of the column and add 2 ml of Coupling Buffer.
- Add the desired volume of slurry (4 ml to obtain 2 ml of resin) to the column, remove the bottom cap and let the column drain. Rinse the column with a total of 6 ml (3 resin-bed volume) of Coupling Buffer and allow the column to drain. After the Coupling buffer has drained, replace the bottom cap.
- Add 2-4 ml of protein suspended in Coupling Buffer to the column (keep an aliquot of the protein solution to assess the coupling efficiency by comparing the protein concentration of the eluate in step 2.11 using either the absorbance at 280 nm or the bicinchoninic assay). Cap and mix end over end to have a homogeneous solution.
- Weigh an empty 1.8 ml microcentrifuge tube and move to the fume hood. Carefully transfer NaCNBH3 (about 0.05 g) in the pre-weighed tube. Close the tube and weigh the tube containing NaCNBH3. Do not open the tube outside the hood.
- Based on the weight of NaCNBH3 transferred into the tube, make a 5 M solution by adding 1 M NaOH. The molecular weight of NaCNBH3 is 62.84 g therefore 0.05 g is equal to (0.05/62.84 = 7.96 x 10-4 moles) 7.96 x 10-4 moles. To make a 5 M solution add (7.96 x 10-4 /5) 159 µl of 1 M NaOH.
- Measure the reaction’s total volume taking the resin volume into consideration. Add 10 µl of 5 M NaCNBH3 per ml of reaction.
- For example: for a 4 ml reaction volume add 40 µl of 5 M NaCNBH3. For 1 ml of resin and 3 ml of added protein, the total volume is 4 ml.
- Cap the column and mix end over end. Secure the column on a tube rotator and let the reaction continue overnight at 4 °C.
- In the morning, bring the column to the chemical hood and carefully remove the cap as some gas may have formed. Drain the column into a clean tube and save the eluate to measure the protein concentration using the absorbance at 280 nm method12 or the bicinchoninic acid assay and compare it with the starting protein concentration in step 2.6.
- Wash the resin with 4 ml of Coupling Buffer and let the column drain.
3. Blocking Remaining Sites
- Wash the resin with 4 ml of quenching buffer. Drain and replace the bottom cap. Add 2 ml of quenching buffer and suspend the resin.
- Assess the total reaction volume by measuring the resin volume and the quenching buffer volume then add 10 µl of 5 M NaCNBH3 per ml of reaction volume.
- Mix gently at room temperature for 30 min end-over-end. After 30 min, bring the column in the fume hood. Carefully remove the top and then remove the bottom cap and let the column drain into a waste tube.
- Wash the column with 5 resin volumes of Wash solution. Monitor the washes for the presence of residual proteins by measuring the eluate’s absorbance at 280 nm. Uncoupled proteins are washed out by the high salt concentration.
- Wash the resin with 6 ml of degassed PBS pH 7.4 containing 0.02% sodium azide. Keep the column at 4 °C until needed.
4. Purification of Nurr1 Specific Antibodies
- Rinse the Nur77 LBD coupled column with 10 ml of PBS pH 7.4 and let the column drain. Cap the bottom of the column and place the drained Nur77 LBD coupled column in a new 15 ml collection tube.
- Add 5 ml of protein A purified anti-Nurr1 antibodies, cap the column and place on a rotator at 4 °C for 1 hr. Remove the top and bottom caps and collect the flow through. Set aside on ice until ready to add to the Nor1 LBD coupled column.
- Rinse the Nor1 LBD coupled column with 10 ml of PBS pH 7.4 and let the column drain. Cap the bottom of the column and place the Nor1 LBD coupled column in a 15 ml collection tube.
- Add the flow through from the Nur77 LBD coupled column, cap the column and place on a rotator at 4 °C for 1 hour. Remove the top and bottom caps and collect the flow through.
- Measure the antibody concentration by measuring the absorbance at 280 nm.
5. ELISA-based Analysis of Purified Anti-Nurr1 Antibody
- Add 100 µl of protein (2.5 µg/ml) to the wells of a 96 well plate. If testing 3 different proteins, add 100 µl of protein 1 to wells A1 to H4, 100 µl of protein 2, to wells A5 to H8 and 100 µl of protein 3 to wells A9 to H12. Cover the plate and incubate overnight at 4 °C.
- Wash the coated wells twice with 300 µl per well of PBS pH 7.4 and add 300 µl of ELISA blocking buffer to the entire antigen coated wells and incubate 2 hours at room temperature.
- While the plate is blocking prepare a desired starting dilution of purified anti-Nurr1 antibody (ranging from 10 to 100 µg/ml) in ELISA blocking buffer.
- After 2 hr of blocking, replace the ELISA block buffer with 100 µl of fresh ELISA block buffer to all wells.
- Add 100 µl of diluted purified anti-Nurr1 antibody to all the wells in row A. Serially dilute the antibodies by transferring 100 µl of solution from row A to the wells in row B followed by transferring 100 µl of solution from row B to row C continuing down to row G. After mixing the solutions in wells in row G discard the extra 100 μl. Wells in row H only receive the initial 100 µl of blocking buffer. Incubate at room temperature for 1 hr.
- Wash the plate three (3) times with 300 µl per well of 0.05% Tween 20 in PBS pH 7.4 and blot the plate to remove excess wash buffer.
- Add 100 µl of Horse Radish Peroxidase (HRP) conjugated Goat anti-rabbit IgG diluted in ELISA block buffer (1:8,000 dilution; the typical range is from 1:5,000 to 1:25,000). Incubate at room temperature for 1 hr.
- Wash the plate three (3) times as described in 5.6. Rinse all wells by filling them with 300 μl of deionized water. Blot excess water by inverting the plate onto absorbing paper.
- Add 100 µl of 3, 3’, 5, 5’ tetramethylbenzidine (warmed to room temperature) to all wells and incubate 15 to 30 min in the dark at room temperature.
- Stop the reaction by adding 50 µl of 2 N H2SO4.
- Read absorbance at 450 nm subtracting background at 650 nm.
Representative Results
A comparison of Protein A column purified Nurr1-specific antibodies with Protein A purified anti-Nurr1 antibodies followed by passage through Nur77 LBD and Nor1 LBD columns is shown in Figure 1. As can be seen, Protein A-purified Nurr1 antibody exhibited a strong binding to Nurr1 LBD. However, it also showed significant binding to Nur77 LBD and to Nor1 LBD. When Protein A-purified Nurr1 antibodies were further purified against Nur77 LBD and Nor1 LBD, the final affinity purified Nurr1 antibodies exhibited specific binding to Nurr1 LBD with undetectable binding to Nur77 LBD or Nor1 LBD, demonstrating that its cross-reactivity to Nur77 and Nor1 was completely removed.
This specificity was further demonstrated by Western blot analysis of extracts from CHO cell transfected with expression vectors carrying full length Nurr1, Nor1, or Nur77 fused to Myc tagging protein (Figure 2). As expected, anti-myc antibodies revealed that each full-length protein was expressed at its expected molecular weight. In agreement with the ELISA results, Protein A-purified Nurr1 specific antibody robustly detected full-length Nurr1 but also detected Nor1 and Nur77 although less efficiently. In contrast, the fully purified Nurr1 antibody did not exhibit any detectable cross-reactivity to Nor1 and Nur77 by ELISA or Western blot analyses.
Finally, the fully purified Nurr1 antibody was used for immunohistochemical analysis of mDA neurons. It is well documented that Nurr1, but not its close homologs Nor1 and Nur77, is prominently expressed in rodent and human mDA neurons at both mRNA and protein levels14,15. The use of the fully purified Nurr1 antibody confirmed that Nurr1 is almost exclusively expressed in the nucleus of mDA neurons in the substantia nigra area (Figure 3).
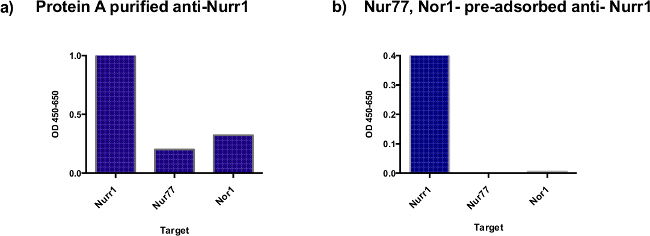
Figure 1: ELISA comparison of cross-reactivities of anti-Nurr1 antibodies before and after purification by passage through Nur77 and Nor1 LBDs coupled columns. Protein A purified anti-Nurr1 antibodies (0.156 μg/ml) (a) or protein A purified followed by chromatography onto Nor1 and Nur77 LBD-coupled columns anti-Nurr1 antibodies (0.156 μg/ml) (b) were added to wells of 96 well plate coated with 100 μl of 2.5 μg/ml of either Nurr1 LBD, Nor1 LBD or Nur77 LBD. The ELISA was carried out as described in the method section.
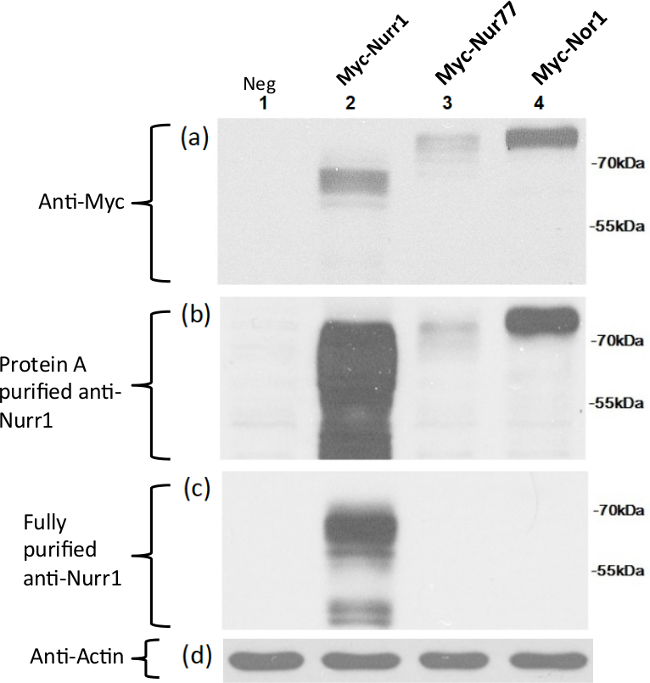
Figure 2: Specific detection of full-length Nurr1 expressed in CHO cells using the fully purified Nurr1-specific antibody. Full-length Nurr1, Nor1, and Nur77 proteins were expressed as myc tagged fusion proteins in CHO cells and detected by Myc specific antibodies (a), Protein A-purified Nurr1 antibody (b), and fully purified Nurr1 antibody (c). Anti-actin antibodies used for protein loading control (d). Each Myc-tagged full length DNA (molecular weight of 65, 66, and 69 kDa for Nurr1, Nur77, Nor1, respectively) was transiently transfected into CHO cells and the same amount of cell extracts were loaded in each lane. Lane 1: negative control consisting of CHO cells extracts transfected with an empty vector; lane 2: Myc-Nurr1; lane 3: Myc-Nur77; lane 4: Myc-Nor1.
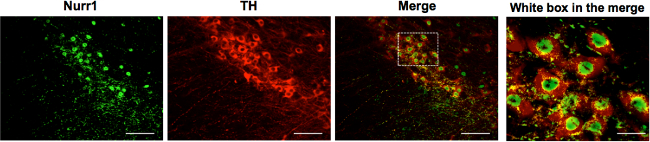
Figure 3: The fully purified Nurr1-specific antibody specifically detects tyrosine-hydroxylase-positive dopamine neurons. Midbrain dopamine neurons in the substantia nigra are positive for Nurr1, as examined by immunohistochemistry using the fully purified Nurr1-specific antibodies. Notably, Nurr1 was localized in the nucleus of mDA neurons. TH (tyrosine hydroxylase). Scale bar = 100 μm. Scale bar in white box merge is 10 μm. Please click here to view a larger version of this figure.
Discussion
The success of this protocol relies on the availability of pure proteins for characterization of the antibodies raised against a specific member of the protein family of interest. There is no need to purify the antibodies using protein A/G until it is clearly established that there are significant cross-reactivities by ELISA and Western blotting.
As stated in the text, it is important to avoid suspending the protein(s) to be cross-linked to the column in an amine-containing buffer such as Tris or glycine. Furthermore the optimal protein concentration used for cross-linking must be determined empirically. Too much protein on the column could result in steric hindrance while too little would reduce the efficiency of adsorption. Therefore, it is a good idea to test protein concentrations ranging from 2 to 10 mg/ml and to assess the quality of the resulting antibodies.
Several factors must be considered when this technique fails to yield highly specific antibodies. These include: cross-linking efficiency, loss of immunoreactive domain, and cross-linked protein denaturation. As stated in the protocol, monitoring cross-linking efficiency allows the determination that too much or too little protein is being immobilized on the column. If cross-linking using reactive amines is suspected of destroying immunoreactive domain(s), immobilization through the carboxylic group or through carbohydrates should be considered.
It is important to monitor the efficiency of the procedure to consistently yield highly specific antibodies. For regeneration purposes, the columns must be stripped of the cross-reactive antibodies bound to them using harsh conditions (high or low pH). This causes an increasingly high percentage of cross-linked proteins to be effectively denatured, reducing the column performance.
The addition of blocking agents in ELISA and Western blotting is commonly used with a fair level of success. However, the approach described in this video increases the probability of success and reduces the need for maintaining large amounts of the cross-reactive proteins on hand. As the reagents and methods to immobilize proteins to chromatography media keep improving, this approach will contribute to the identification of circulating transformed cells specific antibodies.
This approach is limited by the availability of pure proteins. Another limitation of this protocol is the dependence on proper folding when using protein domains. If the protein domains do not fold as in the whole protein, the pre-adsorption strategy may not yield antibodies that can readily identify the protein member of interest when analyzing cell or tissue extracts.
Many proteins of interests belong to (sub)families of proteins with high amino acid sequence homologies. Functional characterization and identification of the distribution of proteins often rely on the availability of specific antibodies against each family member. The high amino acid homologies between protein members in the same family contribute to the difficulties in generating highly specific antibodies. Using these cross-reactive antibodies often lead to conflicting results.
One such example is the orphan nuclear receptor Nurr1 that belongs to the NR4A subfamily of nuclear receptors, together with Nur77 and Nor1. Since these three factors share a high degree of amino acid homology, antibodies against each factor show significant cross reactivities, which hamper precise analysis of each factor.
Using these NR4A members as an example, it was possible to generate highly specific Nurr1 antibodies free of reactivities to Nor1 or Nur77. Since the LBDs share less homology than the DNA binding domains, the LBD of each protein was expressed and purified and then used to improve the specificity of the anti-Nurr1 antibodies. Initially, antibodies against Nurr1’s LBD exhibited modest but significant levels of cross reactivity to the other proteins, as examined by ELISA and Western blot analyses. However, when Protein A-purified Nurr1 antibodies were further purified through pre-adsorption against cross-reactive LBD domains of Nur77 and Nor1, the resulting Nurr1 antibody did not show any cross reactivity, as examined by Western blot and ELISA analyses of prominently detected Nurr1-expressing DA neurons in the midbrain areas. The Western blot data and ELISA analyses closely agree with each other, indicating that these analyses can be complementary and support each other.
Taken together, this antibody purification strategy will be extremely useful in generating a specific antibody by eliminating the cross reactivity to homologous protein members.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This work was supported by NIH grants (NS070577 and NS084869).
Materials
| Purified Ligand Binding Domain from Nurr1, Nor 1 and Nur77 | Column Storage solution | ||
| AminoLink Coupling Resin and Kit | Thermo Scientific | 44890 | |
| Protein A spin column | Thermo Scientific | 89978 | |
| Dulbecco's Phosphate-Buffered Saline | Corning | 21-031-CV | |
| 96 well Flate-bottom plates, High Flange, 330μL | Thermo Scientific | 3455 | |
| 10% Normal Goat Serum | KPL | 50-675-69 | |
| Milk Diluent/ Blocking Concentrate | KPL | 50-82-01 | |
| IgG Elution Buffer | Thermo Scientific | 21004 | |
| UltraPure 1 M Tris-HCI Buffer, pH 7.5 | Life Technologies | 15567-‐027 | |
| Micro BCA Protein Assay Kit | Thermo Scientific | 23235 | |
| Horse Radish Peroxidase conjugated Goat anti-Rabbit IgG (H+L) | Thermo Scientific | 31460 | |
| Ni-NTA Resin | Thermo Scientific | 88221 | |
| 3, 3', 5, 5' Tetramethylbenzydine | Thermo Scientific | 34028 | |
| 2N H2SO4 | Macron Fine Chemicals | H381 05 | |
| Protein A IgG Binding Buffer | Thermo Scientific | 21001 | |
| IgG Elution Buffer | Thermo Scientific | 21004 | |
| Neutralization Buffer | 1M Tris-‐HCl pH 8.5 | ||
| Column Storage solution | Phosphate Buffered Saline containing 0.02% sodium azide | ||
| Spectra/Por 7 pre-treated dialysis tubing | Spectrum Labs | 132128 | |
| 10 ml Disposable columns | Thermo Scientific | 29924 | |
| AminoLink Coupling Buffer | 0.1M sodium phosphate, 0.05% NaN3, pH 7.0 | ||
| Quenching buffer | 1M Tris. HCI, 0.05% NaN3, pH 7.4 |
||
| Wash Solution | 1M NaCl, 0.05% NaN3 | ||
| ELISA Blocking buffer | 1% Normal Horse serum (NHS), 1% Normal Goat Serum (NGS) in 1X KPL milk diluent | ||
| Storage Solution | PBS pH 7.4 containing 0.02% sodium azide | ||
| Micropipettes: 10 μl; 20 μl; 200 μl; 1000 μl |
References
- Hawk, J. D., Abel, T. The role of NR4A transcription factors in memory formation. Brain Res. Bull. 85 (1-2), 21-29 (2011).
- Law, S. W., Conneely, O. M., DeMayo, F. J., O’Malley, B. W. Identification of a new brain-specific transcription factor, NURR1. Mol. Endocrinol. 6 (12), 2129-2135 (1992).
- Zetterström, R. H., et al. Dopamine neuron agenesis in Nurr1-deficient mice. Science. 276 (5310), 248-250 (1997).
- Kadkhodaei, B., et al. Nurr1 is required for maintenance of maturing and adult midbrain dopamine neurons. J. Neurosci. 29 (50), 15923-15932 (2009).
- Decressac, M., Volakakis, N., Björklund, A., Perlmann, T. NURR1 in Parkinson disease–from pathogenesis to therapeutic potential. Nat Rev Neurol. 9 (11), 629-636 (2013).
- Peña de Ortiz, S. Hippocampal Expression of the Orphan Nuclear Receptor Gene hzf-3/nurr1 during Spatial Discrimination Learning. Neurobiol Learn Mem. 74 (2), 161-175 (2000).
- Vecsey, C. G., et al. Histone deacetylase inhibitors enhance memory and synaptic plasticity via CREB:CBP-dependent transcriptional activation. J. Neurosci. 27 (23), 6128-6140 (2007).
- Colón-Cesario, W. I., et al. Knockdown of Nurr1 in the rat hippocampus: implications to spatial discrimination learning and memory. Learn Mem. 13 (6), 734-744 (2006).
- McQuown, S. C., et al. HDAC3 is a critical negative regulator of long-term memory formation. J. Neurosci. 31 (2), 764-774 (2011).
- Hawk, J. D., et al. NR4A nuclear receptors support memory enhancement by histone deacetylase inhibitors. J. Clin. Invest. 122 (10), 3593-3602 (2012).
- Bridi, M. S., Abel, T. The NR4A orphan nuclear receptors mediate transcription-dependent hippocampal synaptic plasticity. Neurobiol Learn Mem. 105, 151-158 (2013).
- Gill, S. C., von Hippel, P. H. Calculation of protein extinction coefficients from amino acid sequence data. Anal. Biochem. 182 (2), 319-326 (1989).
- Smith, P. K., et al. Measurement of protein using bicinchoninic acid. Anal. Biochem. 150 (1), 76-85 (1985).
- Zetterström, R. H., Williams, R., Perlmann, T., Olson, L. Cellular expression of the immediate early transcription factors Nurr1 and NGFI-B suggests a gene regulatory role in several brain regions including the nigrostriatal dopamine system. Brain Res. Mol. Brain Res. 41 (1-2), 111-120 (1996).
- Xiao, Q., Castillo, S. O., Nikodem, V. M. Distribution of messenger RNAs for the orphan nuclear receptors Nurr1 and Nur77 (NGFI-B) in adult rat brain using in situ hybridization. Neurosciences. 75 (1), 221-230 (1996).

