Time-Lapse Video Microscopy for Assessment of EYFP-Parkin Aggregation as a Marker for Cellular Mitophagy
Summary
Herein, we describe in detail a time-lapse video microscopy approach to measuring the temporal recruitment of EYFP-Parkin during the selective removal of damaged mitochondria. This dynamic process of EYFP-Parkin-dependent removal of damaged mitochondria can be used as an indicator of cellular health under different experimental conditions.
Abstract
Time-lapse video microscopy can be defined as the real time imaging of living cells. This technique relies on the collection of images at different time points. Time intervals can be set through a computer interface that controls the microscope-integrated camera. This kind of microscopy requires both the ability to acquire very rapid events and the signal generated by the observed cellular structure during these events. After the images have been collected, a movie of the entire experiment is assembled to show the dynamic of the molecular events of interest. Time-lapse video microscopy has a broad range of applications in the biomedical research field and is a powerful and unique tool for following the dynamics of the cellular events in real time. Through this technique, we can assess cellular events such as migration, division, signal transduction, growth, and death. Moreover, using fluorescent molecular probes we are able to mark specific molecules, such as DNA, RNA or proteins and follow them through their molecular pathways and functions. Time-lapse video microscopy has multiple advantages, the major one being the ability to collect data at the single-cell level, that make it a unique technology for investigation in the field of cell biology. However, time-lapse video microscopy has limitations that can interfere with the acquisition of high quality images. Images can be compromised by both external factors; temperature fluctuations, vibrations, humidity and internal factors; pH, cell motility. Herein, we describe a protocol for the dynamic acquisition of a specific protein, Parkin, fused with the enhanced yellow fluorescent protein (EYFP) in order to track the selective removal of damaged mitochondria, using a time-lapse video microscopy approach.
Introduction
Macro autophagy is an intracellular process that involves the catabolic degradation of both damaged and dysfunctional cellular components, such as organelles and proteins for the purpose of either recycling or energy production. To initiate this metabolic process, the cell engulfs the damaged cellular components into a double-membrane structure, known as an autophagosome, which fuses with a lysosome and its content is degraded and recycled 1,2. There are two major types of autophagy, the non-selective and selective. The non-selective autophagy process occurs when the cell is under nutrient deprivation conditions and needs to scavenge for both essential nutrients and energy. However, selective autophagy occurs to mediate the removal of both dysfunctional/damaged organelles and proteins that otherwise could be toxic. One of the most studied selective autophagy process is the removal of mitochondria, termed mitophagy 1,3-5.
Mitochondria are the central organelles for cell metabolism and the primary source of adenosine triphosphate (ATP) via oxidative phosphorylation through the electron transport chain, fatty acid oxidation, and tricarboxylic acid (TCA) cycle. Moreover, mitochondria regulate reactive oxygen species (ROS) production and release proteins that participate in cell death pathways 6-8.
PTEN-induced putative kinase 1 (PINK1) and Parkin RBR E3 ubiquitin ligase (Parkin) are the key proteins implicated in the mitophagy process. Parkin can protect against cell death by keeping the cell healthy through mitochondrial quality control9. Upon the loss of mitochondrial membrane potential, cytosolic Parkin is recruited to the mitochondria by PINK1. This recruitment triggers the sequential events of mitophagy 10. There is a broad range of evidence that mitophagy is a fundamental mitochondria quality control process and abnormalities in this process drive disease 7. For instance, autosomal recessive Parkinson’s disease has been associated with mutations in the genes that encode for Parkin and PINK1 (PARK2 and PINK1, respectively) 11. The quality control of mitochondrial health is essential for the removal of mitochondria that contribute to the accumulation of ROS12. Excessive presence of intracellular ROS can lead to damage of both nuclear and mitochondrial DNA (DNA and mt DNA, respectively).
Herein, we show a time-lapse video microscopy approach to follow the aggregation of Parkin after the induction of Parkin-mediated mitophagy in immortalized mouse embryonic fibroblasts via in vitro administration of carbonyl cyanide 4-(trifluoromethoxy)-phenylhydrazone (FCCP), an uncoupling agent. FCCP disrupts ATP synthesis by short circuiting protons across the outer mitochondria membrane and hence uncoupling oxidative phosphorylation from the electron transport chain 13. Triggering the depolarization of the mitochondrial membrane leads to the disruption of mitochondria and selective Parkin-dependent removal. Therefore, transfecting the cells of interest with an expression vector encoding Parkin fused with a fluorescent marker (enhanced yellow fluorescent protein, EYFP) can be used as a fluorescent tag to follow the recruitment of Parkin during the mitophagic process. In order to visualize the mitochondria, we co-transfected pDsRed2-Mito, which encodes red fluorescent protein (DsRed2) that contains a mitochondrial targeting sequence of cytochrome c oxidase subunit VIII (Mito). pDsRed2-Mito is designed for fluorescent labeling of mitochondria14. The time required for Parkin translocation into the mitochondrial membrane can be measured and gives an indirect measure of cellular health. For example, we can say that if a cell line knocked-out for a particular gene of interest shows either a faster or slower recruitment of Parkin after the induction of mitophagy by FCCP, that gene product would be a key player in order to keep the metabolic rates of the cell at the physiological status and prevent the development of diseases. Therefore, the time-lapse video microscopy provides a very powerful tool for both basic and clinical research applications in following the dynamic of labeled proteins during their molecular processes and understanding how these processes are affected during a pathological condition.
Protocol
1. Electroporation of Fibroblast with Both Expression Vectors EYFP-Parkin and pDsRed2-Mito
- Grow the immortalized mouse embryonic fibroblast cells on 10 centimeter tissue-culture plate using DMEM (Dulbecco's modified Eagle's medium) medium supplemented with 10% fetal bovine serum, 2 mmol/l L-glutamine, 100 U/ml penicillin, and 100 mg/ml streptomycin in humidified atmosphere containing 5% CO2 at 37 °C.
- At 80% cell confluency, discard the complete DMEM medium by sterile suction and add 10 ml of sterile 1x phosphate buffer solution (PBS) (80 g of NaCl, 2.0 g of KCl, 14.4 g of Na2HPO4, 2.4 g of KH2PO4 and 1 L distilled H2O, PH: 7.4).
- Discard the PBS by sterile suction and add 1 ml of Trypsin-EDTA 0.25%. Incubate the plate at 37 °C until the cells are detached (2 – 3 min). Add 4 ml of complete DMEM medium, resuspend the cells and take out 10 μl of the cell suspension for the hemocytometer counting.
NOTE: The hemocytometer is designed so that the number of cells in one set of 16 corner squares is equivalent to the number of cells x 104/ml.
- Seed 1 x 106 cells onto 10 cm tissue culture dishes 24 hours prior to the electroporation process.
- Discard the complete DMEM medium by sterile suction and add 10 ml of sterile PBS. Discard the PBS by sterile suction and add 1 ml of Trypsin-EDTA 0.25%. Incubate the plate at 37 °C until the cells are detached (2 – 3 min). Add 4 ml of complete DMEM medium and resuspend the cells in a 15 ml tube.
- Spin the cells down at 250 x g for 5 min using a refrigerated-centrifuge (4 °C). Discard the supernatant by sterile suction and resuspend the pellet in 1 ml of sterile PBS. Spin the cells down at 250 x g for 5 min at 4 °C.
- Discard the supernatant by sterile suction, and add 100 µl of solution mix for electroporation (82 µl of electroporation Solution V + 18 µl of Supplemental solution 1) to the cell pellet and gently resuspend the pellet by pipetting (For more details refer to user's guide). Add 2 µg of EYFP-Parkin (excitation/emission 514/527) and 1 µg pDsRed2-Mito (excitation/emission 565/620) at the cell suspension.
- Transfer the solution to the sterile cuvette using a disposable pasteur (both tools are provided with the kit) and electroporate using the pre-set program NIH/3T3 U-030 (Single pulse, voltage 200 V, capacitance 960 µF, pulse time 20 milliseconds, pulse number: 1).
- Immediately after the electroporation, add 500 µl of fresh pre-warmed complete DMEM medium and seed the cell on a 6 cm tissue-culture plate live-imaging-grade and incubate them in a humidified atmosphere containing 5% CO2 at 37 °C for 24 hours.
2. Time-Lapse Video Microscopy
- Set the temperature of the microscope's chamber at 37 °C prior to use.
- At this point, prepare a specific live imaging medium to proceed with the experimental protocol. Prepare the live imaging medium as follows; mix DMEM phenol-free supplemented with 10% fetal bovine serum, 2 mmol/l L-glutamine, 100 U/ml penicillin, and 100 mg/ml streptomycin. Pre-warm the live imaging medium at 37 °C in a water bath.
- Discard the medium of the electroporated cells by sterile suction and add 1 ml of pre-warmed live imaging medium, using a P1000 pipet and incubate the plate for 30 min at 37 °C.
- During the plate incubation period, influx 5% CO2 in the microscope's chamber already at a stable temperature of 37 °C.
- Place the plate with the electroporated cells into the microscope's chamber avoiding major movements or oscillations.
- Using the software interface, set the microscope in order to detect both fluorescence signals from the fusion proteins encoded by the co-transfected vectors, EYFP-Parkin (Excitation range 495 to 510 nm and emission range 520 to 550 nm, green) and the pDsRed2-Mito (Excitation maximum 558 nm and emission maximum 583 nm, red).
- Open the time-lapse video microscopy software. In the upper menu select the FITC for EYFP-Parkin and Rhodamine for pDsRed2-Mito. In the upper menu select the magnification (20X).
- Using the software interface, look for the single cell expressing both co-transfected vectors and register the position. Repeat this step until a minimum of 10 cells is collected for every experimental condition (Experimental group).
- Select the menu "Apps" and click on Multi Dimensional Acquisition. In the Multi Dimensional Acquisition windows select the parameters needed, such as the number of acquisitions, the interval of time between each acquisition and the position of the recorded cell. Click on "Acquire" to start the acquisition process.
- Start the basal acquisition of both EYFP-Parkin and DsRed2-Mito fluorescent signal collecting images every 5 min for a total interval of 15 min.
- Prepare pre-warmed live imaging medium with a concentration of FCCP twice higher than the final working concentration (0.1 – 10 µM, cell type-dependent).
- Interrupt the acquisition process and add gently 1 ml of pre-warmed live imaging medium with FCCP into the plate inside the microscope's chamber using a P1000 pipet.
- Restart the acquisition process as previously described, for a total period of 3 hours, using the recorded position. Save all the acquired images in order to analyze them and create a video of the mitophagic process when the acquisition is over.
3. Analysis
- Collect all the acquired images in ".tiff " format on a personal computer
- Open the images with a graphic software and define the temporal interval occurred from the mitochondrial induced-depolarization with FCCP to the first image showing the recruitment of Parkin into the mitochondrial membrane (Images are acquired every 5 min). Repeat this analysis for each cell in the experimental group.
- Label the first cell of the column on a spreadsheet with the experimental group name. For each experimental group, measure the temporal intervals for Parkin recruitment of a minimum of 10 cells. Make an average of the calculated temporal intervals for Parkin recruitment and define the standard deviation for each experimental group (Mean ± S.D., N = 10).
- Organize into columns the single calculated temporal intervals. Every column contains the n = 10 measurements of the experimental group.
- Select the function "average" from the Formula Builder menu. Select the data in the column. Select the function "Standard Deviation" from the Formula Builder menu. Select the data in the column.
- Using a statistical approach, compare the experimental groups applying the appropriate statistic in order to define a significant difference in the dynamic of mitophagy between the experimental groups. (For instance, two groups = Student's t-Test, three or more groups = ANOVA plus a post-hoc test)
Representative Results
Herein, we show how time-lapse video microscopy is a powerful technique that can be used to follow molecular events of fluorescently-tagged proteins in a single cell. The representative results also show how this technique allows for the acquisition of high quality images. When images of the molecular process, are obtained, we have the opportunity to analyze them in different ways. Here, we analyze the interval of time between initiation of the induced mitophagy process, which is the crucial molecular process that maintain the cellular homeostasis selectively removing the damaged mitochondria.
This technique founds a broad range of applications. It can be used either for monitoring macro cellular process such as cell migration or micro molecular events through the labeling of the key molecules involved in the process that has to be studied.
Parkin-mediated Mitophagy
The selective Parkin-mediated removal of damaged and dysfunctional mitochondria process is summarized in Figure 1, where a membrane depolarization induced by the uncoupling agent FCCP triggers selective Parkin-mediated mitophagy, recruiting PINK1 to the outer mitochondrial membrane. Following, Parkin is recruited to the damaged mitochondria and interacts with PINK1. This complex Parkin-PINK1 leads to the ubiquitination of the damaged organelle and its incorporation into an autophagosome, which fuses with a lysosome. This process known as mitophagy is considered to be a crucial metabolic process in order to keep the cellular environment healthy and allow the regeneration of functional mitochondria 8,15.
Experimental Protocol
Figure 2 shows a schematic representation of the experimental protocol and the microscope set-up used for following the recruitment of EYFP-Parkin before and after FCCP-induced mitophagy process.
Parkin Recruitment to Depolarized Mitochondrial Membrane
In Figure 3A, we show Parkin distribution prior and post FCCP administration. During the basal time points (BT5, BT10 and BT15 min) EYFP-Parkin (Green) is homogeneously diffused throughout the cell and the mitochondria network (Red) appears to be well interconnected. Following FCCP administration (FT = 0 min) (Figure 3B), we observe mitochondria fractionation (FT = 5 min). However, EYFP-Parkin is still homogeneously diffused though out the cytoplasm. Mitophagy is stimulated at 55 min post FCCP administration, EYFP-Parkin is recruited at the damaged mitochondrial membranes to trigger the mitophagy process (FT = 55).
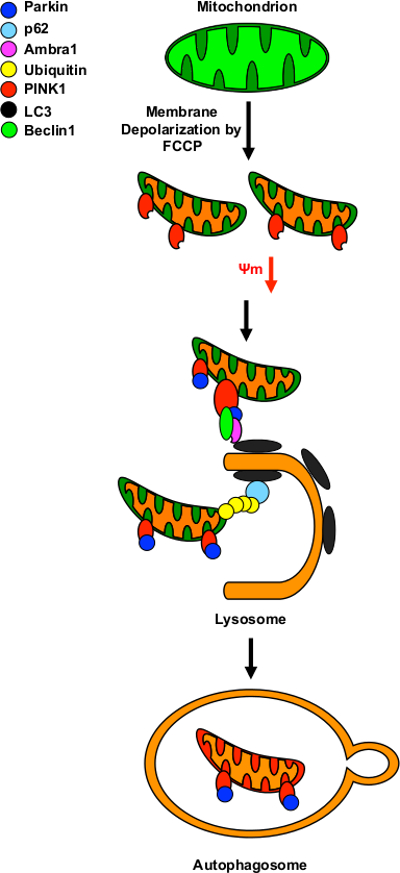
Figure 1. Schematic Representation of Parkin-mediated Mitophagy. A decreasing of membrane potential (Ψm) occurs in damaged or dysfunctional mitochondria and can be induced by the uncoupling agent FCCP. Loss of membrane potential triggers selective Parkin-mediated mitophagy, recruiting PINK1 (Red) to the outer mitochondrial membrane. Parkin (Blue) is recruited to the damaged mitochondria by directly interacting with PINK1. The presence of Parkin leads to the ubiquitination (Yellow) of the damaged organelle and its incorporation into an autophagosome, which fuses with a lysosome. Please click here to view a larger version of this figure.

Figure 2. Microscope Set-up and Representation of the Experimental Protocol. (A) Overview of the experimental events described in the protocol section. The reported experimental events allow the operator to transiently co-transfect the cells with EYFP-Parkin and pDsRed2-Mito by electroporation in order to follow the FCCP-induced mitophagy process using a (B) set-up for time-lapse video microscopy approach. BT = basal time point; FT = FCCP time point. Please click here to view a larger version of this figure.
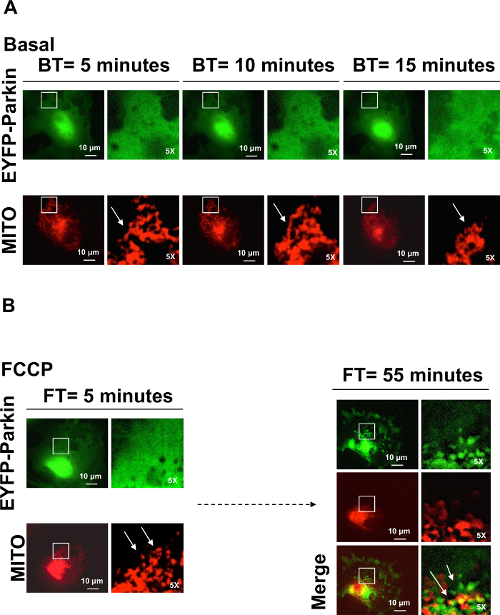
Figure 3. Fibroblast Cell Showing Parkin Recruitment after Mitochondrial Membrane Depolarization Induced by FCCP. EYFP-Parkin (green) translocation into the mitochondria (red) was monitored by time-lapse video microscopy during the basal time point (BT) and post FCCP administration (FT). From BT = 0, EYFP-Parkin and mitochondrial fluorescence were detected every 5 min. (A) EYFP-Parkin is homogenously diffused across the whole basal period and the mitochondrial network is intact (white arrows). (B) Mitochondrial fractionation due to membrane depolarization by FCCP is recorded at FT = 5 min (white arrows) and EYFP-Parkin recruitment to damaged mitochondrial membranes started at FT = 55 min (white arrows). The white boxes represent the magnified area of the panels (5X). For more details concerning the application of this experimental protocol refer to reference8. (Scale bar: 10 µm). Please click here to view a larger version of this figure.
Discussion
Time-lapse microscopy can be defined as the technique that extends live cell imaging from a single observation in time to the observation of cellular dynamics over long periods of time. This methodology is distinguished from a simple confocal or live cell microscopy because it allows the observer to identify in real time a single fluorescent-tagged protein and follow its dynamic inside a single live cell. In fact, the confocal microscopy can easily identifies immuno-labeled proteins using fluorescent antibody, but it does not allow observing the cell and the molecular event in its live environment.
The time-lapse video microscopy is primarily used in research monitoring molecular processes in different cell types such as HEK293 16, primary neurons 17 and Hela cells 18 but has also clinical application such as pregnancy and predicting aneuploidy 19,20. Time-lapse microscopy provides embryologists with additional information about embryo development for a clinical pregnancy, and it is possible that this information can be used to improve our ability to select viable embryos for transfer. Additional advantages of the use of Time-lapse microscopy include less handling of embryos and their dishes for intermediate observations, which would reduce the risk of loss or contamination within the laboratory 19.
Cells are able to maintain a healthy mitochondrial homeostasis by degrading damaged and dysfunctional mitochondria through mitophagy. Defective mitophagy is linked to Parkinson's disease 21, apoptosis 22, aging 23 and cancer 24. Mitophagy is driven by the ubiquitin ligase Parkin and PINK121. Due to the importance of this process to maintain a healthy mitochondrial status, we found it important to study the dynamic of this event using a time-lapse video microscopy. The proposed protocol will provide evidence on how mitochondria homeostasis is affected under different cellular conditions, including changes in gene expression and protein functions.
In this study, we transiently transfected fibroblasts by electroporation. The electroporation method applies an electrical pulse at an optimized voltage lasting a few microseconds to the cell suspension in order to increase the permeability of the cell membrane by disturbing the phospholipid bilayer of the membrane. This action results in the formation of temporary pores. The membrane permeabilization allows the DNA to be introduced into the cell 25. We chose this process to transiently transfect fibroblasts in order to have higher transfection efficiency of both plasmids EYFP-Parkin and pDsRed2-Mito. In fact, the electroporation process is approximately ten times more effective than chemical transfection 26. The Images acquisition process can be compromised by both external factors; temperature fluctuations, vibrations, humidity and internal factors; pH, cell motility. The electroporation and fluorescent tracking are two steps considered to be the most critical for the success of this experimental protocol. Even if the transfection efficiency is higher than other methods, the electroporation kills ~ 20% of the electroporated cells. Another major issue is the acquisition quality of the images during the monitoring step. Since the cellular environment is dynamic, the focus plane needs continuous adjustments in order to maintain the focus throughout the acquisition process.
Mitochondrial damage can be caused by membrane mitochondrial depolarization and oxidative stress. These factors induce a mitophagic response. Evidence that mitochondria depolarization can induce selective mitophagy comes from photodamage experiments which lead to ROS production and mitochondrial injury 27-30. In order to induce mitochondrial membrane depolarization and mitophagic response, FCCP has been used as protonophore 10. FCCP is an ionophore that allows protons to cross lipid bilayers inducing membrane depolarization. Using this approach we induced mitophagy and followed the dynamic of the molecular marker.
The concentration of FCCP is crucial, as it can be cytotoxic and promote cellular death. Since this approach relies on the observation of living cells, the final concentration of FCCP has to be adjusted in a cell type dependent manner in order to induce mitophagy without trigger the mechanisms that lead to cellular death.
Overall, we think that following the dynamic of mitophagy, using EYFP-Parkin as a fluorescent marker, will provide new insights into mitochondrial function. The selective removal of mitochondria via mitophagy is a crucial process in maintaining mitochondrial quality, cell viability and homeostasis. This live imaging approach will help to understand the mitophagic process under healthy and stressed conditions.
Modern approaches are further extending time-lapse microscopy observations beyond making movies of cellular dynamics. Increasingly this boundary is blurred as cytometric techniques are being integrated with imaging techniques for monitoring and measuring dynamic activities of cells and subcellular structures 31.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This work was supported in part by NIH grants (1R01CA137494, R01CA132115, R01CA086072 to R.G.P.), the Kimmel Cancer Center NIH Cancer Center Core grant P30CA056036 (R.G.P.), a grant from the Breast Cancer Research Foundation, generous grants from the Dr. Ralph and Marian C. Falk Medical Research Trust (R.G.P.) and a grant from the Pennsylvania Department of Health (R.G.P.). In part this work was supported by an American Italian Cancer Foundation postdoctoral fellowship (G.D.) and Bioimaging Shared Resource of the Sidney Kimmel Cancer Center (NCI 5 P30 CA-56036).The Department specifically disclaims responsibility for an analysis, interpretations or conclusions. There are no conflicts of interest associated with this manuscript.
Materials
| DMEM | Corning Life Science | 10-013-CV | Pre-warm at +37 C before use |
| PHENOL-FREE DMEM | Corning Life Science | 17-205-CV | Pre-warm at +37 C before use |
| FETAL BOVINE SERUM | Sigma-Aldrich | F2442 | Pre-warm at +37 C before use |
| L-GLUTAMINE | Gibco | 25030 | Pre-warm at +37 C before use |
| PENICILLIN/ STREPTOMYCIN | Corning Life Science | 30-002-CI | Pre-warm at +37 C before use |
| EYFP-PARKIN EXPRESSION VECTOR | Addgene | 23955 | |
| pDsRed2-Mito EXPRESSION VECTOR | Clontech | 632421 | |
| NUCLEOFECTOR 2B DEVICE | LONZA | AAD-1001S | |
| NUCLEOFECTOR FOR KIT R NIH/3T3 | LONZA | VCA-1001 | |
| ZEISS AXIOVERT 200M INVERTED MICROSCOPE | CARL ZEISS | ||
| Carbonyl Cyanide 4-(trifluoromethoxy)-Phenylhydrazone (FCCP) | Sigma-Aldrich | C2920 | |
| MetaMorph | Molecular Devices | Experimental Builder | |
| ImageJ | National Institute of Health | Experimental Builder |
References
- Youle, R. J., Narendra, D. P. Mechanisms of mitophagy. Nat Rev Mol Cell Biol. 12, 9-14 (2011).
- Zhi, X., Zhong, Q. Autophagy in cancer. F1000Prime Rep. 7, 18 (2015).
- Ding, W. X., Yin, X. M. Mitophagy: mechanisms, pathophysiological roles, and analysis. Biol Chem. 393, 547-564 (2012).
- Lemasters, J. J. Selective mitochondrial autophagy, or mitophagy, as a targeted defense against oxidative stress, mitochondrial dysfunction, and aging. Rejuvenation Res. 8, 3-5 (2005).
- Kissova, I., Deffieu, M., Manon, S., Camougrand, N. Uth1p is involved in the autophagic degradation of mitochondria. J Biol Chem. 279, 39068-39074 (2004).
- Kubli, D. A., Gustafsson, A. B. Mitochondria and mitophagy: the yin and yang of cell death control. Circ Res. 111, 1208-1221 (2012).
- Redmann, M., Dodson, M., Boyer-Guittaut, M., Darley-Usmar, V., Zhang, J. Mitophagy mechanisms and role in human diseases. Int J Biochem Cell Biol. 53, 127-133 (2014).
- Di Sante, G., et al. Loss of sirt1 promotes prostatic intraepithelial neoplasia, reduces mitophagy, and delays park2 translocation to mitochondria. Am J Pathol. 185, 266-279 (2015).
- Tanaka, A. Parkin-mediated selective mitochondrial autophagy, mitophagy: Parkin purges damaged organelles from the vital mitochondrial network. FEBS Lett. 584, 1386-1392 (2010).
- Shiba-Fukushima, K., et al. PINK1-mediated phosphorylation of the Parkin ubiquitin-like domain primes mitochondrial translocation of Parkin and regulates mitophagy. Sci Rep. 2, 1002 (2012).
- Trinh, J., Farrer, M. Advances in the genetics of Parkinson disease. Nat Rev Neurol. 9, 445-454 (2013).
- Murphy, M. P. How mitochondria produce reactive oxygen species. Biochem J. 417, 1-13 (2009).
- Heytler, P. G., Prichard, W. W. A new class of uncoupling agents–carbonyl cyanide phenylhydrazones. Biochem Biophys Res Commun. 7, 272-275 (1962).
- Karan, G., Yang, Z., Zhang, K. Expression of wild type and mutant ELOVL4 in cell culture: subcellular localization and cell viability. Mol Vis. 10, 248-253 (2004).
- Jin, S. M., Youle, R. J. PINK1- and Parkin-mediated mitophagy at a glance. J Cell Sci. 125, 795-799 (2012).
- Kerr, M. C., et al. Visualisation of macropinosome maturation by the recruitment of sorting nexins. J Cell Sci. 119, 3967-3980 (2006).
- Edin, F., et al. 3-D gel culture and time-lapse video microscopy of the human vestibular nerve. Acta Otolaryngol. 134, 1211-1218 (2014).
- Ramsden, A. E., Mota, L. J., Munter, S., Shorte, S. L., Holden, D. W. The SPI-2 type III secretion system restricts motility of Salmonella-containing vacuoles. Cell Microbiol. 9, 2517-2529 (2007).
- Meseguer, M., et al. Embryo incubation and selection in a time-lapse monitoring system improves pregnancy outcome compared with a standard incubator: a retrospective cohort study. Fertil Steril. 98, 1481-1489 (2012).
- Campbell, A., et al. Modelling a risk classification of aneuploidy in human embryos using non-invasive morphokinetics. Reprod Biomed Online. 26, 477-485 (2013).
- Bingol, B., et al. The mitochondrial deubiquitinase USP30 opposes parkin-mediated mitophagy. Nature. 510, 370-375 (2014).
- Carroll, R. G., Hollville, E., Martin, S. J. Parkin sensitizes toward apoptosis induced by mitochondrial depolarization through promoting degradation of Mcl-1. Cell Rep. 9, 1538-1553 (2014).
- Batlevi, Y., La Spada, A. R. Mitochondrial autophagy in neural function, neurodegenerative disease, neuron cell death, and aging. Neurobiol Dis. 43, 46-51 (2011).
- Frank, M., et al. Mitophagy is triggered by mild oxidative stress in a mitochondrial fission dependent manner. Biochim Biophys Acta. 1823, 2297-2310 (2012).
- Neumann, E., Schaefer-Ridder, M., Wang, Y., Hofschneider, P. H. Gene transfer into mouse lyoma cells by electroporation in high electric fields. EMBO J. 1, 841-845 (1982).
- Sugar, I. P., Neumann, E. Stochastic model for electric field-induced membrane pores. Electroporation. Biophys Chem. 19, 211-225 (1984).
- Lemasters, J. J. Variants of mitochondrial autophagy: Types 1 and 2 mitophagy and micromitophagy (Type 3). Redox Biol. 2, 749-754 (2014).
- Aggarwal, B. B., Quintanilha, A. T., Cammack, R., Packer, L. Damage to mitochondrial electron transport and energy coupling by visible light. Biochim Biophys Acta. 502, 367-382 (1978).
- Alexandratou, E., Yova, D., Handris, P., Kletsas, D., Loukas, S. Human fibroblast alterations induced by low power laser irradiation at the single cell level using confocal microscopy. Photochem Photobiol Sci. 1, 547-552 (2002).
- Kim, I., Lemasters, J. J. Mitophagy selectively degrades individual damaged mitochondria after photoirradiation. Antioxid Redox Signal. 14, 1919-1928 (2011).
- Coutu, D. L., Schroeder, T. Probing cellular processes by long-term live imaging–historic problems and current solutions. J Cell Sci. 126, 3805-3815 (2013).

