Isolation of CD146+ Resident Lung Mesenchymal Stromal Cells from Rat Lungs
Summary
This protocol describes an isolation technique for obtaining primary lung resident mesenchymal stromal cells from rats, through the use of enzymatic digestion, density gradient separation, plastic adherence and CD146+ magnetic bead selection.
Abstract
Mesenchymal stromal cells (MSCs) are increasingly recognized for their therapeutic potential in a wide range of diseases, including lung diseases. Besides the use of bone marrow and umbilical cord MSCs for exogenous cell therapy, there is also increasing interest in the repair and regenerative potential of resident tissue MSCs. Moreover, they likely have a role in normal organ development, and have been attributed roles in disease, particularly those with a fibrotic nature. The main hurdle for the study of these resident tissue MSCs is the lack of a clear marker for the isolation and identification of these cells. The isolation technique described here applies multiple characteristics of lung resident MSCs (L-MSCs). Upon sacrifice of the rats, lungs are removed and rinsed multiple times to remove blood. Following mechanical dissociation by scalpel, the lungs are digested for 2-3 hr using a mix of collagenase type I, neutral protease and DNase type I. The obtained single cell suspension is subsequently washed and layered over density gradient medium (density 1.073 g/ml). After centrifugation, cells from the interphase are washed and plated in culture-treated flasks. Cells are cultured for 4-7 days in physiological 5% O2, 5% CO2 conditions. To deplete fibroblasts (CD146–) and to ensure a population of only L-MSCs (CD146+), positive selection for CD146+ cells is performed through magnetic bead selection. In summary, this procedure reliably produces a population of primary L-MSCs for further in vitro study and manipulation. Because of the nature of the protocol, it can easily be translated to other experimental animal models.
Introduction
Mesenchymal stromal cells (MSCs) are increasingly recognized for their therapeutic potential in a wide range of diseases, including lung diseases. Besides the use of bone marrow and umbilical cord MSCs for exogenous cell therapy, there is also increasing interest in the repair and regenerative potential of resident tissue MSCs. During development, including in the lung, the mesenchyme is an important source of developmental cues, and resident MSCs are a likely candidate to be at the center of this. Moreover, evidence is emerging that resident MSCs are perturbed in adult diseases, including cancer1,2 and fibrosis3. The main hurdle for the study of these resident tissue MSCs is the lack of a clear marker for the isolation and identification of these cells4. Stem cell antigen-1 (Sca-1) was identified in mice as a marker for a variety of tissue stem cells, and can be used for the isolation of L-MSCs5, but has unfortunately no known orthologs in other species6. Researchers have reported a variety of different isolation methods for the isolation of L-MSCs from either lung tissue or fluid. These vary from fluorescence activated cell sorting (FACS) based methods selecting for CD31–/CD45–/CD90+ cells7, CD31–/CD45–/epithelial cell adhesion molecule (EpCAM)-/Sca-1+ cells8, multidrug resistance transporter ATP binding cassette G (ABCG2) positive cells9 or Hoechst 33342 dye efflux10, to plastic adherence11,12 and migration out of minced tissue13.
The advantages of the herein presented method are several fold. By using a gentle enzymatic digestion and density gradient14, one obtains all the cells of the density range that include MSCs but exclude epithelial or endothelial cells. The subsequent plastic adherence step ensures that only the mesenchymal cells adhere and stay in culture, eliminating leukocytes. Most importantly however, the CD146+ selection step allows for the elimination of fibroblasts, as these cells do not express CD146. Expression of the cell adhesion molecule CD146 is positively correlated with multipotency, and is therefore a good marker to weed out fibroblasts from a mesenchymal cell population15-19. This is an advantage over using CD90 as a selection marker, as it is not only expressed in MSCs but also in lipofibroblasts5,20. In this protocol we have explicitly chosen a magnetic bead selection, as it is gentler on the cells, and the entire procedure can be done in sterile conditions. Another important advantage of this isolation method as opposed to the outgrowth method, is that it is relatively fast, 6-10 days as opposed to a month or more for the outgrowth method. Three to five days after the initial isolation the mesenchymal population is ready for CD146+ selection; after another three to five days the CD146+ cells are ready to be used for experiments or can be frozen for later use. The decreased time of culture improves the quality of the cells as MSCs transdifferentiate towards fibroblasts in prolonged ex vivo culture19. Lastly, because of the nature of the protocol, it is possible to apply this method to other species by simply choosing appropriate antibodies, or even to other organ systems by adjusting the choice of digestion enzymes and incubation time.
A detailed protocol of this isolation method is given below, and a schematic overview of the isolation and subsequent selection of the CD146+ subpopulation is provided in Figure 1A and 1B respectively. Additionally, details are included for passaging, freezing and thawing these cells.
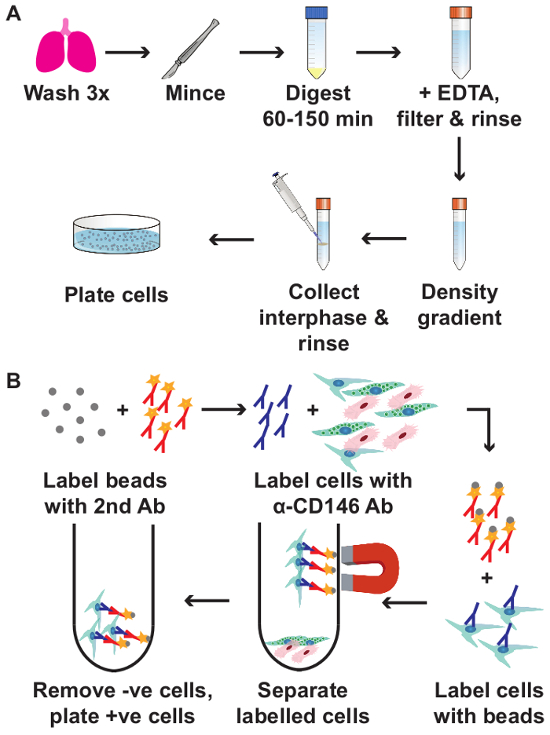
Figure 1. Schematic overview of the isolation of pulmonary mesenchymal cells (A) and subsequent CD146+ cell selection (B). min = minutes; EDTA = Ethylenediaminetetraacetic acid; 2nd Ab = secondary antibody; α-CD146 Ab = primary anti-CD146 antibody; -ve cells = CD146 negative cells; +ve cells = CD146 positive cells. Please click here to view a larger version of this figure.
Protocol
All procedures were approved by the Animal Care Committee of the University of Ottawa (animal ethics protocol OHRI-1696). Animal care was performed in accordance with institutional guidelines.
1. Isolation of Lung Mesenchymal Stromal Cells
- Prepare the enzyme mix in a 50 ml tube: weigh in 30 U Neutral Protease, 2,500 U Collagenase I and 500 U DNAse I. These amounts suffice for the lungs of an adult mouse or rat pup. Prepare on day of isolation and store at 4 °C until use.
- Sacrifice rat pups at day 13 by an intra-peritoneal injection of pentobarbital sodium (0.2 ml, 65 mg/ml). Use the toe pinch reflex to establish unconsciousness. Death of the animal is ensured by opening the chest, as outlined below.
- Sanitize the skin by spraying the animal with 70% ethanol and carefully open the rib cage using surgical scissors, starting at the diaphragm and cutting towards the rostral side, being very careful not to damage the lungs. Spread the ribcage open using hemostatic clamps, or alternatively cut away the ribcage to provide access to the thorax.
- Exsanguinate the animal by removing the heart. To remove the heart, grasp the thymus and heart with small forceps and cut these away with surgical scissors. Immediately afterwards, absorb the blood with a gauze until no more blood comes out of the severed aorta and pulmonary artery.
- Remove the lungs from the thorax as follows: hold the trachea with small forceps, then sever the trachea with surgical scissors on the rostral side. While gently pulling the lung package out of the thorax, cut away any connective tissue on the dorsal side along the ribcage to free the lungs.
- Sever the lungs from the aorta and esophagus by cutting along the diaphragm with surgical scissors. Now that the lungs are completely free from the thorax remove any remaining blood gently with a gauze.
- Remove the trachea and bronchi with surgical scissors and carefully transfer the lung lobes to a 50 ml tube containing cold 35 ml 30% Citrate-Phosphate-Dextrose Adenine (CPDA-1) anticoagulant (26.30 g trisodium citrate dihydrate, 3.27 g ascorbic acid monohydrate, 2.22 g monosodium dihydrogen phosphate, 31.80 g D-glucose, 0.275 g adenine in 1 L purified H2O; sterile filter the solution using a 0.22 µm membrane filter before use) in phosphate buffered saline (PBS) to remove blood and debris.
- After a 5 min rinse in 30% CPDA-1/PBS, transfer the lung to a new 50 ml tube containing 35 ml sterile phosphate buffered saline (PBS) (RT), invert gently to remove citrate. Transfer to a tube containing 35 ml Dulbecco's PBS + Sodium-pyruvate + Glucose (DPBS++) (RT). Each rinsing step should take approximately 5 min.
Note: DPBS++ can be ordered commercially or made up as follows21: calcium chloride dihydrate 132.4 mg, magnesium chloride hexahydrate 100 mg, potassium chloride (anhydrous) 200 mg, potassium phosphate monobasic (anhydrous) 200 mg, sodium chloride (anhydrous) 8,000 mg, sodium phosphate dibasic (anhydrous) 1,144.5 mg, D-glucose 1000 mg, sodium pyruvate 36 mg in 1 L purified H2O, pH 7.3. Sterile filter the solution using a 0.22 µm membrane filter before use. - Dissolve the enzyme mix by adding 10 ml DPBS++ (pre-warmed to 37 °C) and inverting gently until dissolved.
- Transfer the lungs to a new 50 ml tube and chop the lung on the wall of the tube using a scalpel until finely minced. Add the enzyme mix to the tissue (10 ml enzyme mix/ per adult mouse/rat pup lung), close the tube tightly and incubate at 38 °C with gentle agitation (either in a thermal mixer, a shaking water bath, or a water bath with regular manual agitation). The duration depends on the type of tissue: fetal tissue 60 min, juvenile/ adult tissue 90-120 min, fibrotic adult tissue 120-150 min.
- Stop the digestion by chelating the bivalent cations with 200 µl ethylenediaminetetraacetic acid (EDTA, 0.15 M, pH 7.4. Sterile filter the solution using a 0.22 µm membrane filter before use).
- Add 20 ml 5% fetal bovine serum (FBS)/PBS and pass the entire suspension through 100 µm cell strainer into a new 50 ml tube. Rinse the tube and cell strainer with 10 ml 5% FBS/PBS, bringing the total volume to 40 ml.
- Centrifuge for 5 min at 500 x g, RT.
- Remove the supernatant (pellet should be clearly visible), resuspend in 40 ml 5% FBS/PBS and repeat centrifugation step 1.13.
- Resuspend in 4 ml 5% FBS/PBS (RT) and layer carefully over 3 ml density gradient media (1.073 g/cm3) in a 15 ml tube14. To obtain a good interphase, it is crucial that the layering of the single cell suspension occurs at a >45° angle at very low speed.
- Centrifuge 20 min at 900 x g, medium (5/10) acceleration, no deceleration, 19 °C.
- Collect the cells present in the interphase and resuspend them in 10 ml sterile PBS.
- Centrifuge for 5 min at 500 x g, 21 °C.
- Remove the supernatant and resuspend the cell pellet in 10 ml pre-warmed completed L-MSC culture medium (minimum essential media Eagle, alpha modification (αMEM), supplemented with 2 mmol L-Glutamine per 500 ml αMEM, 20% vol/vol FBS and 1% vol/vol Penicillin/ Streptomycin/Amphotericin B.
- Repeat centrifugation step 1.18. Subsequently, remove supernatant and resuspend in 10 ml pre-warmed fresh culture medium.
- Count cells using a hemocytometer or automated cell counter and plate at a high density, since the majority of cells at this stage are not plastic adherent. The exact plating density depends on the animal species and age, e.g., the cells of an adult mouse lung are seeded at 105 cells/ cm2, whereas cells from a rat pup lung are sufficiently dense at a seeding density of 1-2 x 104 cells/ cm2. Use 0.2 ml L-MSC culture medium/cm2.
- Culture L-MSCs in a humidified 5% O2, 5% CO2 atmosphere at 37 °C.
- Change medium 24 hr after the initial seeding, and subsequently every 3 days after rinsing once with PBS. Cells should be cultured until 80-90% confluence, which takes 3-5 days on average depending on the animal of origin. Higher confluence will promote differentiation into fibroblasts.
2. Selection of CD146+ Lung Mesenchymal Stromal Cells
- Coating of Magnetic Beads with Secondary Antibody
- Coat the magnetic beads with biotinylated secondary antibody, at a ratio of 10 µg secondary antibody/ 100 µl magnetic beads in a round bottom sterile 2 ml cryovial in sterile conditions. Use 10 µl secondary antibody per expected 0.5 x 106 cells, and use a minimum of 100 µl magnetic beads and 10 µg antibody.
Note: The ratio of antibodies used per volume of beads may differ between manufacturers, please consult the manufacturer's instructions for the beads of choice. For this protocol, please refer to table 1 for the beads and antibodies that were used in the optimization of this protocol. - Incubate in a rotating sample mixer for 30 min at 30 rotations/min (rpm), at RT.
- Resuspend the beads in 1.5 ml sterile 0.1 % bovine serum albumin (BSA)/PBS and transfer to a 3 ml round bottom tube (flow cytometry tube). Rinse the cryovial with an additional 1.5 ml 0.1 % BSA/PBS and transfer to the 3 ml tube.
- Place the tube on an appropriate magnet and wait 2 min until all beads are attached to the rear wall of the tube and the solution remains clear. Remove runoff and repeat wash with 3 ml 0.1 % BSA/PBS two times.
- Resuspend the beads in 3 ml 0.1 % BSA/PBS and store at 4 °C, protected from light. Use within 5 days.
- Coat the magnetic beads with biotinylated secondary antibody, at a ratio of 10 µg secondary antibody/ 100 µl magnetic beads in a round bottom sterile 2 ml cryovial in sterile conditions. Use 10 µl secondary antibody per expected 0.5 x 106 cells, and use a minimum of 100 µl magnetic beads and 10 µg antibody.
- Labelling CD146+ L-MSCs
- Harvest the mesenchymal cells after rinsing with PBS using a gentle non-trypsin solution.
Note: Volumes depend on the size of culture flask (0.2 ml/cm2 medium or PBS).- Remove culture medium and rinse cells three times with sterile PBS.
- Add the appropriate volume of gentle non-trypsin solution (see Table 1). Incubate at 37 °C in the incubator until the cells have detached, approximately 10 min when using the solution in Table 1.
- Inactivate the enzyme by adding L-MSC culture medium, and centrifuge for 5 min at 500 x g.
- Remove supernatant, resuspend cells in 0.1 % BSA/PBS and count the cells with a hemocytometer or automated cell counter.
Note: The volume of 0.1% BSA/PBS depends on the size of culture flask: aim for 5 or 10 ml 0.1% BSA/PBS. - Repeat step 2.2.2 and resuspend the dedicated amount of cells in 3 ml 0.1 % BSA/PBS (blocks unspecific binding sites and keeps the cells alive). Keep cells on ice.
- Prepare a 3 ml round bottom tube with the primary anti-CD146 antibody, and keep on ice.
Note: The concentration of primary antibody that is added to the dedicated amount of cells depends on the antibody that is used. For a suggested anti-rat CD146 antibody see Table 1. - Add the total volume of cells from step 2.2.3 to the tube with the primary antibody. Close tightly and incubate 30 min on a rotating sample mixer at 15 rpm, 4 °C.
- Centrifuge cells for 5 min at 500 x g, 4 °C, remove supernatant and resuspend cell pellet in 3 ml 0.1 % BSA/PBS. Repeat centrifuge step.
- Resuspend the cells in the 3 ml solution containing secondary antibody coated beads that was prepared according to the instructions in section 2.1 and incubate 30 min on a rotating sample mixer at 15 rpm, 4 °C.
- Harvest the mesenchymal cells after rinsing with PBS using a gentle non-trypsin solution.
- Selecting CD146+ L-MSCs
- Place the tube containing the labelled CD146+ L-MSCs on the magnet. The positive cells will be drawn to the back of the tube. Without moving the tube or touching the beads, harvest the supernatant with the negative cells slowly with a sterile Pasteur pipette (these can be either collected or discarded based on the experimental design).
- Remove the tube from the magnet and resuspend the cells in 3 ml 10 % BSA/PBS. Repeat the selection procedure described in step 2.3.1. four more times (Σ 5 selection steps).
- Resuspend the CD146+ L-MSCs in pre-warmed L-MSC culture medium and return to the magnet.
- Remove supernatant, resuspend in L-MSC culture medium and plate in an appropriately sized culture flask (0.2 ml/cm2). As a rule of thumb, plate cells in the same size culture flask from which the cells were lifted prior to bead-selection. The number of CD146+ L-MSCs, and therefore the plating density, will depend on the age, species and strain of the source animal. Always aim for a plating density of ~5,000 cells/cm2.
Note: The beads will not disturb the cell growth and will gradually disappear as the cells proliferate.
3. Freezing Cells
- Use the following freezing media: 60% pentastarch solution (10% pentastarch in 0.9% NaCl), 20% FBS, 12.5% CPDA-1, 2.5% NaCl (0.9%), 5% dimethyl sulfoxide (DMSO)22.
Note: If not all ingredients are available, a solution containing 5% DMSO, 30% FBS and 65% αMEM is a good alternative. - Resuspend cells at 0.5 x 106 cells/ml, aliquot in cryovials of 1 ml each and freeze O/N at -80 °C using a freezing container.
- Transfer to a liquid nitrogen storage tank the next day.
4. Thawing Cells
- Thaw a vial of cells in a 37 °C water bath for 5 min.
- Resuspend cells in 10 ml pre-warmed culture medium and centrifuge for 5 min at 500 x g, 21 °C.
- Remove supernatant and resuspend the cell pellet in 10 ml culture medium. Seed cells in a vented cap cell culture treated flasks at 5,000 cells/cm2 in 0.2 ml/cm2 media (e.g., 15 ml for a 75 cm2 flask).
- Culture cells in 5% O2, 5% CO2 until 80-90% confluent for harvesting and/or experimental use. Over-confluency will induce differentiation. When seeding at 5,000 cells/cm2, L-MSCs will reach 80-90% confluency within 3-4 days of culture.
Representative Results
Two of the most reliable physical characteristics of MSCs, density and plastic adherence, are used in the first part of this protocol to obtain the mesenchymal cell fraction of the lung that contains L-MSCs. Although the density gradient interphase will include monocytes and macrophages in addition to lung mesenchymal cells, the plastic adherence followed by 3-5 days of culture ensures that only the lung mesenchymal cells remain. Indeed this cell population expresses the classic MSC surface markers CD73, CD90 and CD146 and is negative for the markers CD34, CD45, major histocompatibility complex type II (MHCII)-RT1B, CD11b and CD79a, indicating that there are no longer any leukocytes present in the cell population (Figure 2A). Of interest is that out of the CD146+ subset, a range of 44.4-65.7% is also CD73+ and CD90+ (data not shown). Moreover, this cell population is capable of a colony forming unit (CFU) efficiency that at ~30% is much higher than generally reported for bone marrow MSCs or even other MSC types (Figure 2B), and differentiates along the three classic MSC lineages (Figure 2C).
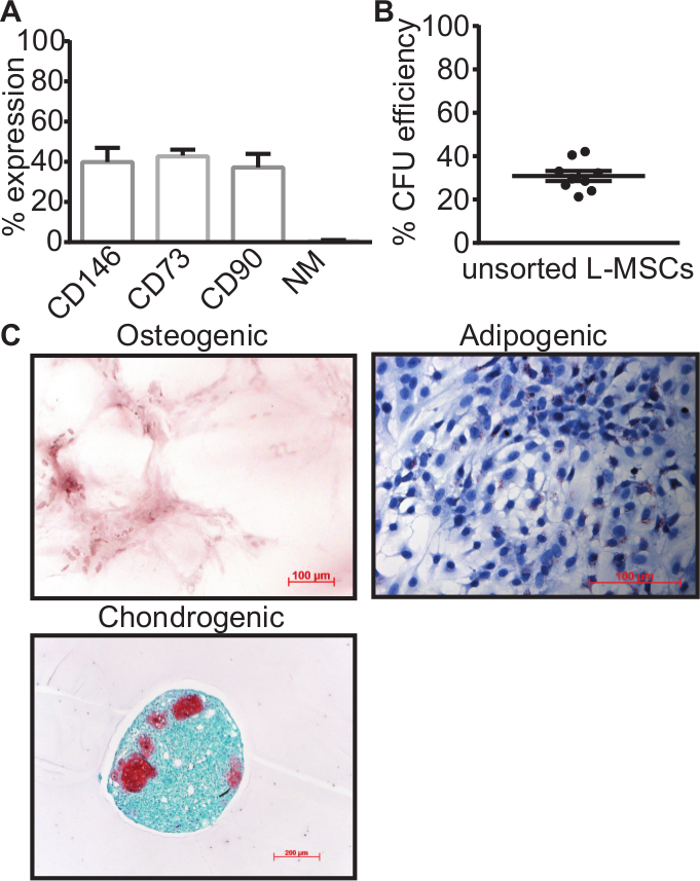
Figure 2. MSC characteristics after enzymatic digestion, density gradient separation and plastic adherence. (A) Expression of MSC-related surface markers CD146, CD73 and CD90 is present in part of the plastic adherent cell population, whereas the negative leukocyte markers CD11b, CD79a, CD34, CD45 and MHCII-RT1B were virtually not expressed. (B) Colony forming unit assay through limiting dilution assay (5 cells/cm2) indicated that ~30% of plastic adherent cells had a clonal capacity, indicative of MSCs. (C) Plastic adherent cells were capable of osteogenic matrix production (red), contained a higher number of small lipid vesicles (red) when induced with adipogenic differentiation medium compared to non-differentiated controls and formed chondrogenic like spheres containing chondrocytes (red). Data is presented as mean ± standard error of mean (SEM). All data were generated with passage 1-3 cells. NM = negative markers; CFU = colony forming units. Please click here to view a larger version of this figure.
However, the main caveat is that this population likely contains both L-MSCs and different subtypes of lung fibroblasts, the L-MSCs differentiated progeny23. Since multipotency is positively correlated with CD146 expression, and fibroblasts should have no expression of this marker, positive selection of the CD146+ subpopulation ostensibly resulted in a cell population that is highly enriched in L-MSCs. This is clearly demonstrated by a higher percentage of the cell population expressing MSC associated surface markers (Figure 3A), a considerably higher colony forming potential of ~80% (Figure 3B) and a stronger differentiation response in particularly the chondrogenic lineage (Figure 3C) compared to the total mesenchymal population that is obtained before CD146 selection. Of note is that these L-MSCs form only very few true adipocytes, but show many more spindle shaped cells that are filled with small lipid vesicles. These could reflect the lipofibroblast lineage that is crucial for both lung development and alveolar type II cell support. After CD146 selection, more adipocyte like cells filled with large lipid vesicles can be observed (Figure 3C) compared to before CD146 selection, yet the majority of cells are still the lipofibroblasts-like cells containing small lipid vesicles.
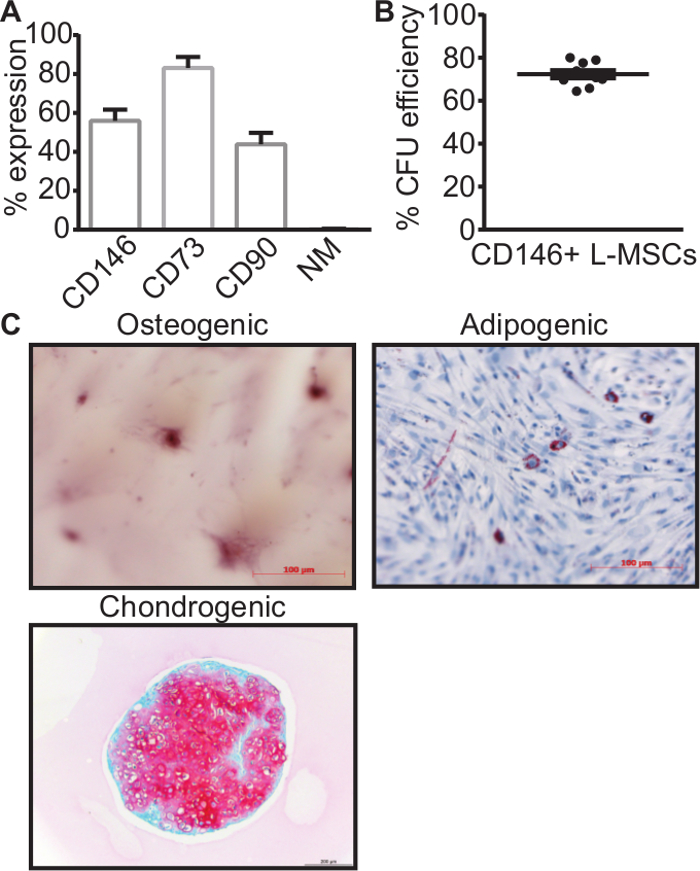
Figure 3. MSC characteristics after additional CD146+ magnetic bead selection. After positive selection of the CD146+ subpopulation, these cells showed a higher expression of MSC-related surface markers, CD73 in particular (A), a considerably higher CFU efficiency after single cell plating (B), and a stronger differentiation response for particularly the chondrogenic lineage and to a lesser extent the adipogenic lineage (C). Data is presented as mean ± SEM. All data were generated with passage 3 cells. NM = negative markers; CFU = colony forming units. Please click here to view a larger version of this figure.
Discussion
The isolation and culture of primary L-MSCs presents an opportunity to better understand their function and their interaction with other cell populations at a cellular level, and their role in lung development, health and disease. This is especially important as the lack of a specific single marker of these cells makes it nearly impossible to study these cells in situ. As with all primary cell populations, one should keep in mind that these cells are more likely to change their character the longer they are kept in culture19 (reviewed in24). To keep L-MSCs in a state that is as close as possible to their natural state, it is very important that they are cultured in 5% O2, 5% CO2, in a 37 °C humidified incubator. In respiratory physiology it is generally accepted that based on Dalton's law of partial pressures, the partial oxygen pressure of ambient air (21% O2) is 160 mmHg, and drops to ~101 mmHg in the alveolar airspace25-27. In a mature lung the alveolar-arterial PaO2 gradient is ~3-4 mmHg, but this may be increased significantly in a diseased or immature lung where the alveolar-arterial distance is increased25,28. Inside the body, the mesenchymal niche is normally exposed to an oxygen concentration of 2-8%29,30. Only few papers have actually measured the PO2 in the lungs, but one study measured this in the lungs of 20 patients, finding a range in normal lung PO2 from 23 to 656 mmHg, with a median of 42.8 mmHg27,31. According to Dalton's law, a humidified incubator with a temperature of 37 °C and 5% O2 has a pO2 of ~36 mmHg, which falls perfectly within the range measured in normal lung tissue and is very close to the median. Additionally, low oxygen culture conditions promote the maintenance of progenitor populations in culture29. Taken together, it is hard to tell exactly what oxygen concentration L-MSCs are exposed to in situ at different stages during normal postnatal development or disease, but it appears that 5-10% O2 is likely the most accurate from a physiological perspective27,31. Based on the information cited above, we chose to maintain 5% O2 in our culture conditions, and we found that when these conditions are maintained in vitro, the L-MSCs will grow faster, display fewer stress fibers, and have a smaller likelihood of spontaneous differentiation or senescence. Additional factors that will prevent spontaneous differentiation or other abnormalities is by using a gentle non-trypsin dissociation agent for passaging, passaging L-MSCs when they reach 80-90% confluency (dense conditions will promote fibroblast differentiation), and keeping the passage number low (<P4). In a study by Hoffmann and colleagues, trypsin was shown to have adverse effects on L-MSCs function and phenotype, whereas trypsin-free dissociation agents could preserve this13.
To optimize the yield during the isolation process, it is important to mince the lungs as finely as possible, while keeping in mind that the tissue should not be allowed to dry. During the enzymatic digestion, it is important that the tubes are agitated frequently if not using a heated shaking device, as the tissue pieces will sediment and will not get optimum exposure to the enzymes. Another crucial step is the density gradient separation: if the layering is not done very slowly or at an angle <45°, it will lead to the mixing of the two liquids instead of layering, causing the loss of the entire cell population. Moreover, the density of the density gradient medium is very dependent on the surrounding temperature. Any other temperature than 19 °C (or RT), will result in suboptimal or no density gradient separation.
Of interest to future users of this protocol is that we have successfully isolated viable L-MSCs from lungs that were harvested up to 24 hr before isolation, provided that the lungs are directly stored in cold L-MSC media upon harvest, and kept at 4 °C. This allows for more flexible experimental planning, or even shipment of lungs between different labs. The yield is generally similar to that of freshly harvested lungs, and cells grow equally well.
Another modification that would be possible, is to use FACS instead of magnetic bead selection, even though this method subjects the cells to more stress as the procedure takes more time and as cells are subjected to high pressures and speeds, and harbors the risk of contamination if not done under sterile conditions. The magnetic beads used here do not interfere with cell growth, and disappear within a few days of culture. A preference for FACS or magnetic bead sorting may also depend on the facilities available to the end user. Additionally, it would be possible to modify this protocol to isolate resident MSCs from other tissues. In this case it would be important to tailor the enzymes to the organ of interest, as the matrix composition varies between organs.
An important limitation of only using the density gradient and subsequent plastic adherence for obtaining mesenchymal cells, is that the resulting population contains both L-MSCs and fibroblasts. Selecting for CD146+ cells strongly remedies this caveat, but despite the CD146+ selection procedure and the careful culturing conditions, the nature of L-MSCs and the effects of in vitro cell culture make it impossible to avoid some degree of differentiation into fibroblasts. MSCs in general have been reported to contain a hierarchy of multipotency, in which cells further down the hierarchy slowly lose their multilineage potential until they become terminally differentiated fibroblasts23. In all likelihood this also occurs in the L-MSC niche4, and seems to be reflected in the cultured CD146+ population as already one passage after CD146+ selection roughly 40% of cells have lost CD146 expression. Additionally, in this population there is only 80% CD73 and 40% CD90 expression. In part, this could be explained by the MSC hierarchy of multipotency. It would be possible that CD146+ undergo asymmetric proliferation, giving rise to both CD146 positive MSCs and negative, more differentiated, daughter cells. Resident MSCs of the lung could also reasonably be expected to have a somewhat different phenotype and function from bone marrow MSCs, on which the definition of CD73 and CD90 expression was based. The International Society for Cell Therapy has recognized that not all MSCs express the classic MSC markers, including CD73 and CD90, and that surface marker expression is not the best way to define MSCs32. The exceptionally high CFU efficiency of ~70% of the CD146+ L-MSC population supports this. Because the CD146+ population performs better with respect to MSC characteristics than the unsorted mesenchymal population, the authors have not further characterized the CD146– population. We strived to enrich our mesenchymal population with MSCs and deplete them of fibroblasts, but we do not claim that this is the only true L-MSC population. L-MSCs are likely very heterogeneous, both in origin, phenotype and function.
Although CD146 is widely acknowledged to be a marker of multipotency in MSCs and pericytes16,17,33,34, very little is known about the CD146+ MSC population in vivo. CD146 is a cell adhesion molecule that is important for angiogenesis and endothelial cell function, and is expressed in endothelial cells, smooth muscle cells, pericytes, MSCs and T-lymphocytes15,16,35,36. In recent years, evidence has emerged that MSCs inhabit the perivascular niche in multiple organs, as CD146+ cells co-stain for MSC-markers CD73 and CD10537. Indeed, cultured pericytes and MSCs have a very similar gene expression profile38, strengthening the view that MSCs are derived from pericytes4,39. Although we have not performed immunohistochemical analyses to determine the location of our CD146+ L-MSC population, it is very likely that they would also be located in a perivascular location. Future studies will be needed to verify if this is indeed the case. It is possible that a subset of our cell population are pericytes, although it should be pointed out that all cells look phenotypically very similar after CD146+ selection.
Mesenchymal stromal cells are notoriously hard to identify due to the lack of a single all-encompassing marker. Various studies have reported different methods for the isolation of L-MSCs, and have convincingly shown that these cell populations are similar to each other with respect to MSC marker expression. Based on the characterization of the CD146+ L-MSC as presented here, the CD146+ L-MSC population is very similar to previously reported L-MSC populations as achieved by outgrowth or various forms of FACS using stem cell associated surface markers such as Sca-1, ABCG2 and CD90. When compared to outgrowth L-MSCs13, CD146+ L-MSCs appear to have a greater ability to give rise to colonies from single cells, indicating that our method yields a more enriched multipotent L-MSC population. This advantage is also true when compared to methods that isolate L-MSCs based on CD907 or platelet-derived growth factor receptor α (PDGFRα)40,41, as both surface markers also select for more terminally differentiated lipofibroblasts and alveolar/structural fibroblasts and can therefore only reasonably be identified as stromal cells20,42. In contrast, both Sca-1 and ABCG2 are well documented markers that correlate with stemness and MSC expression9,43-46. ABCG2+ L-MSCs however have a more endothelial nature as they are 99% positive for CD3146, whereas the method reported here selects against endothelial cells through the Ficoll gradient and plastic adherence. The main advantage of our method compared to the Sca-1 based isolation protocol of McQualter and colleagues5,8 is that it is widely applicable to species other than mice, in which Sca-1 cannot be used.
Although it is very challenging, probably nigh on impossible, to obtain a "pure" population of L-MSCs from a late developing or adult lung and maintain it in an undifferentiated state, the protocol presented here provides a fast, consistent and reliable method of obtaining a cell population highly enriched in multipotent self-renewing lung resident MSCs. Using this method will enable a more defined study of the role of L-MSCs in a wide variety of disease models and their role in the developing lung.
Divulgations
The authors have nothing to disclose.
Acknowledgements
JJPC is supported by a Canadian Institutes of Health Research (CIHR) postdoctoral fellowship. MAM receives a merit scholarship from the German National Academic Foundation – Studienstiftung des Deutschen Volkes and is supported by a grant from the EFCNI (European Foundation for the Care of the Newborn Infant). BT is supported by CIHR, the Canadian Lung Association, the Stem Cell Network and the Children’s Hospital of Eastern Ontario Research Institute.
Materials
| Neutral Protease | Worthington Biochemical Corporation | LS02104 | Prepare on day of isolation and store at 4°C until use |
| Collagenase I | Worthington Biochemical Corporation | LS004196 | Prepare on day of isolation and store at 4°C until use |
| DNAse I | Sigma-Aldrich | D5025 | Prepare on day of isolation and store at 4°C until use |
| Pentobarbital sodium (Euthanyl) | Bimeda-MTC Animal Health Inc, Dublin, Ireland | – | Use 0.2 mL for rat pups between 20-30g; larger animals may need more. |
| Dulbecco’s PBS + Sodium-pyruvate + Glucose | Life Technologies | 14287-072 | Very crucial to use this type of D-PBS, as the calcium and magnesium that are present in this D-PBS facilitate facilitate the enzymatic digestion |
| Ficoll-Paque PREMIUM (1.073 g/cm3) | GE Healthcare | 17-5446-52 | Density gradient media. To obtain a good interphase, it is crucial that the layering of the single cell suspension occurs at a >45o angle at very low speed, and that the subsequent centrifugation is done at 19oC. |
| αMEM | Sigma-Aldrich | M8042 | Warm in 37oC water bath before use |
| 200 mM L-Glutamine | Life Technologies | 25030-164 | |
| 100x Antibiotic-Antimycotic | Life Technologies | 15240-062 | Penicillin/Streptomycin/Fungizone |
| M-280 Dynabeads | Life Technologies | 11205D | Magnetic beads |
| biotinylated polyclonal rabbit-anti-mouse IgG | Dako, Agilent Technologies | E0464 | Biotinylated secondary antibody that matches with the anti-rat CD146 antibody described below |
| DynaMag5-magnet | Life Technologies | 12303D | Magnet that is recommended for use with Dynabeads. NOTE: magnet should be chosen based on the manufacturer’s instructions of the magnet beads of choice. |
| TrypLE express | Life Technologies | 12605-028 | Gentle non-trypsin alternative, use 1 mL of TrypLE express/ 25cm2 surface area. After detachment, 10 mL L-MSC culture medium is sufficient to inactivate 3 mL TrypLE |
| anti-rat CD146 antibody | Lifespan Biosciences Inc. | C35841 | 12.5 μl per 0.5 x 106 cells |
| Pentaspan (pentastarch solution) | Bristol-Myers Squibb Canada | – | Can be obtained through local blood donation services or hematology departments. Alternatively, one could use the PSI 20% Pentastarch solution (Preservation Solutions, PST001) diluted 1:1 with 0.9% NaCl. |
| Mr.Frosty freezing container | ThermoFisher Scientific | 5100-0001 | Improves viability when freezing L-MSCs overnight at -80oC |
References
- Bhowmick, N. A., Neilson, E. G., Moses, H. L. Stromal fibroblasts in cancer initiation and progression. Nature. 432, 332-337 (2004).
- Wei, H. J., et al. FOXF1 mediates mesenchymal stem cell fusion-induced reprogramming of lung cancer cells. Oncotarget. 5, 9514-9529 (2014).
- Marriott, S., et al. ABCG2pos lung mesenchymal stem cells are a novel pericyte subpopulation that contributes to fibrotic remodeling. Am J Physiol Cell Physiol. 307, 684-698 (2014).
- Collins, J. J., Thebaud, B. Lung mesenchymal stromal cells in development and disease: to serve and protect. Antioxid Redox Signal. 21, 1849-1862 (2014).
- McQualter, J. L., et al. Endogenous fibroblastic progenitor cells in the adult mouse lung are highly enriched in the sca-1 positive cell fraction. Stem Cells. 27, 623-633 (2009).
- Holmes, C., Stanford, W. L. Concise review: stem cell antigen-1: expression, function, and enigma. Stem Cells. 25, 1339-1347 (2007).
- Gottschling, S., et al. Mesenchymal stem cells in non-small cell lung cancer–different from others? Insights from comparative molecular and functional analyses. Lung Cancer. 80, 19-29 (2013).
- Bertoncello, I., McQualter, J. Isolation and clonal assay of adult lung epithelial stem/progenitor cells. Current protocols in stem cell biology. , (2011).
- Jun, D., et al. The pathology of bleomycin-induced fibrosis is associated with loss of resident lung mesenchymal stem cells that regulate effector T-cell proliferation. Stem Cells. 29, 725-735 (2011).
- Majka, S. M., et al. Identification of novel resident pulmonary stem cells: form and function of the lung side population. Stem Cells. 23, 1073-1081 (2005).
- Hennrick, K. T., et al. Lung cells from neonates show a mesenchymal stem cell phenotype. Am J Respir Crit Care Med. 175, 1158-1164 (2007).
- Salama, M., et al. Endothelin-1 governs proliferation and migration of bronchoalveolar lavage-derived lung mesenchymal stem cells in bronchiolitis obliterans syndrome. Transplantation. 92, 155-162 (2011).
- Hoffman, A. M., et al. Lung-derived mesenchymal stromal cell post-transplantation survival, persistence, paracrine expression, and repair of elastase-injured lung. Stem Cells Dev. 20, 1779-1792 (2011).
- Grisendi, G., et al. GMP-manufactured density gradient media for optimized mesenchymal stromal/stem cell isolation and expansion. Cytotherapy. 12, 466-477 (2010).
- Bardin, N., et al. S-Endo 1, a pan-endothelial monoclonal antibody recognizing a novel human endothelial antigen. Tissue antigens. 48, 531-539 (1996).
- Covas, D. T., et al. Multipotent mesenchymal stromal cells obtained from diverse human tissues share functional properties and gene-expression profile with CD146+ perivascular cells and fibroblasts. Exp Hematol. 36, 642-654 (2008).
- Russell, K. C., et al. In vitro high-capacity assay to quantify the clonal heterogeneity in trilineage potential of mesenchymal stem cells reveals a complex hierarchy of lineage commitment. Stem Cells. 28, 788-798 (2010).
- Sorrentino, A., et al. Isolation and characterization of CD146+ multipotent mesenchymal stromal cells. Exp Hematol. 36, 1035-1046 (2008).
- Halfon, S., Abramov, N., Grinblat, B., Ginis, I. Markers distinguishing mesenchymal stem cells from fibroblasts are downregulated with passaging. Stem Cells Dev. 20, 53-66 (2011).
- McGowan, S. E., Torday, J. S. The pulmonary lipofibroblast (lipid interstitial cell) and its contributions to alveolar development. Annu Rev Physiol. 59, 43-62 (1997).
- Dulbecco, R., Vogt, M. Plaque formation and isolation of pure lines with poliomyelitis viruses. The Journal of experimental medicine. 99, 167-182 (1954).
- Hayakawa, J., et al. 5% dimethyl sulfoxide (DMSO) and pentastarch improves cryopreservation of cord blood cells over 10% DMSO. Transfusion. 50, 2158-2166 (2010).
- Sarugaser, R., Hanoun, L., Keating, A., Stanford, W. L., Davies, J. E. Human mesenchymal stem cells self-renew and differentiate according to a deterministic hierarchy. PLoS One. 4, 6498 (2009).
- Prockop, D. J. Repair of tissues by adult stem/progenitor cells (MSCs): controversies, myths, and changing paradigms. Molecular therapy : the journal of the American Society of Gene Therapy. 17, 939-946 (2009).
- Respiratory Physiology. UCL Available from: https://www.ucl.ac.uk/anesthesia/people/RespPhysiolLong.pdf (2015)
- Boron, W. F., Boulpaep, E. L. . Medical Physiology. 2nd edn. , (2009).
- Carreau, A., El Hafny-Rahbi, B., Matejuk, A., Grillon, C., Kieda, C. Why is the partial oxygen pressure of human tissues a crucial parameter? Small molecules and hypoxia. J Cell Mol Med. 15, 1239-1253 (2011).
- Heller, H., Brandt, S., Schuster, K. D. Determination of alveolar-capillary O2 partial pressure gradient by using 15NO. Nitric oxide : biology and chemistry / official journal of the Nitric Oxide Society. 12, 127-128 (2005).
- Mohyeldin, A., Garzon-Muvdi, T., Quinones-Hinojosa, A. Oxygen in stem cell biology: a critical component of the stem cell niche. Cell stem cell. 7, 150-161 (2010).
- Simon, M. C., Keith, B. The role of oxygen availability in embryonic development and stem cell function. Nature reviews. Molecular cell biology. 9, 285-296 (2008).
- Le, Q. T., et al. An evaluation of tumor oxygenation and gene expression in patients with early stage non-small cell lung cancers. Clin Cancer Res. 12, 1507-1514 (2006).
- Krampera, M., et al. Immunological characterization of multipotent mesenchymal stromal cells-The International Society for Cellular Therapy (ISCT) working proposal. Cytotherapy. , (2013).
- Corselli, M., et al. Identification of perivascular mesenchymal stromal/stem cells by flow cytometry. Cytometry. Part A : the journal of the International Society for Analytical Cytology. 83, 714-720 (2013).
- Russell, K. C., et al. Cell-Surface Expression of Neuron-Glial Antigen 2 (NG2)and Melanoma Cell Adhesion Molecule (CD146) in Heterogeneous Cultures of Marrow-Derived Mesenchymal Stem Cells. Tissue engineering. Part A. , (2013).
- Lv, F. J., Tuan, R. S., Cheung, K. M., Leung, V. Y. Concise review: the surface markers and identity of human mesenchymal stem cells. Stem Cells. 32, 1408-1419 (2014).
- Dagur, P. K., et al. Secretion of interleukin-17 by CD8+ T cells expressing CD146 (MCAM). Clinical immunology. 152, 36-47 (2014).
- Crisan, M., et al. A perivascular origin for mesenchymal stem cells in multiple human organs. Cell stem cell. 3, 301-313 (2008).
- da Silva Meirelles, L., et al. Cultured human adipose tissue pericytes and mesenchymal stromal cells display a very similar gene expression profile. Stem Cells Dev. , (2015).
- Caplan, A. I. All MSCs are pericytes. Cell stem cell. 3, 229-230 (2008).
- Barkauskas, C. E., et al. Type 2 alveolar cells are stem cells in adult lung. J Clin Invest. 123, 3025-3036 (2013).
- McGowan, S. E., McCoy, D. M. Regulation of fibroblast lipid storage and myofibroblast phenotypes during alveolar septation in mice. Am J Physiol Lung Cell Mol Physiol. 307, 618-631 (2014).
- Chen, L., Acciani, T., Le Cras, T., Lutzko, C., Perl, A. K. Dynamic regulation of platelet-derived growth factor receptor alpha expression in alveolar fibroblasts during realveolarization. Am J Respir Cell Mol Biol. 47, 517-527 (2012).
- Zhou, S., et al. The ABC transporter Bcrp1/ABCG2 is expressed in a wide variety of stem cells and is a molecular determinant of the side-population phenotype. Nat Med. 7, 1028-1034 (2001).
- Bonyadi, M., et al. Mesenchymal progenitor self-renewal deficiency leads to age-dependent osteoporosis in Sca-1/Ly-6A null mice. Proc Natl Acad Sci U S A. 100, 5840-5845 (2003).
- Ito, C. Y., Li, C. Y., Bernstein, A., Dick, J. E., Stanford, W. L. Hematopoietic stem cell and progenitor defects in Sca-1/Ly-6A-null mice. Blood. 101, 517-523 (2003).
- Chow, K., et al. Dysfunctional resident lung mesenchymal stem cells contribute to pulmonary microvascular remodeling. Pulmonary circulation. 3, 31-49 (2013).

