Development of an In Vitro Ocular Platform to Test Contact Lenses
Summary
Current in vitro models for evaluating contact lenses (CLs) and other eye-related applications are severely limited. The presented ocular platform simulates physiological tear flow, tear volume, air exposure and mechanical wear. This system is highly versatile and can be applied to various in vitro analyses with CLs.
Abstract
Currently, in vitro evaluations of contact lenses (CLs) for drug delivery are typically performed in large volume vials,1-6 which fail to mimic physiological tear volumes.7 The traditional model also lacks the natural tear flow component and the blinking reflex, both of which are defining factors of the ocular environment. The development of a novel model is described in this study, which consists of a unique 2-piece design, eyeball and eyelid piece, capable of mimicking physiological tear volume. The models are created from 3-D printed molds (Polytetrafluoroethylene or Teflon molds), which can be used to generate eye models from various polymers, such as polydimethylsiloxane (PDMS) and agar. Further modifications to the eye pieces, such as the integration of an explanted human or animal cornea or human corneal construct, will permit for more complex in vitro ocular studies. A commercial microfluidic syringe pump is integrated with the platform to emulate physiological tear secretion. Air exposure and mechanical wear are achieved using two mechanical actuators, of which one moves the eyelid piece laterally, and the other moves the eyeballeyepiece circularly. The model has been used to evaluate CLs for drug delivery and deposition of tear components on CLs.
Introduction
Two significant areas of interest within the contact lens (CL) arena include discomfort and the development of novel CL applications. Elucidating the mechanisms underlying CL discomfort is an issue that has eluded the field for decades.8 The development of novel, functional CLs, such as drug-delivery devices1,3,9 and biosensors,10-12 is an area of growing interest, with substantial potential markets. In both circumstances, a sophisticated in vitro model would provide relevant information to assist with selecting appropriate lens materials or design characteristics during the development phase. Unfortunately, current in vitro models for evaluating CLs and other eye related applications are relatively crude and unsophisticated. Traditionally, in vitro CL studies evaluating tear film deposition or drug delivery are performed in static, large volume vials containing a fixed fluid volume, which greatly exceeds physiological amounts. Furthermore, this simple model lacks the natural tear flow component and the blinking reflex, both of which are defining factors of the ocular environment.
The development of a sophisticated, physiologically relevant eye "model" will necessitate a multi-disciplinary approach and require substantial in vivo validation. For these reasons, the fundamental framework for our in vitro eye model is highly versatile, such that the model can be continually improved through future upgrades and modulations. To date, the model is capable of simulating tear volume, tear flow, mechanical wear and air exposure. The aim is to create an in vitro model that will provide meaningful results, which is predictive and complimentary to in vivo and ex vivo observations.
Protocol
All experiments were completed in accordance and compliance with all relevant guidelines outlined by the University of Waterloo's animal research ethics committee. The bovine eyes are generously donated from a local abattoir.
1. Eye Model
- Design and Production of Molds13
- Design the eye models according to the average physiological dimensions of human adult eyes.13
- Leave a gap of 250 µm between the eyeball and the eyelid pieces of the eye model. Design the respective molds using computer-aided design (CAD) software.
- Create new .cad file or .sldprt file with AutoCAD or Solidworks. Create 3D models of the human eyeball/eyelid. Create molds of the models and save the molds as .stl files.
- Import .stl files into 3D printer software (e.g., makeware for replicator2). Specify parameters of the print (location, sparseness, scale, orientation, smoothness, etc.)13.
- Save the file as G-code file for 3D printers to read. Select materials such as PLA (polylactic acid), ABS (acrylonitrile butadiene styrene), PC (polycarbonate), or a combination thereof, to print the molds13.
- Install desired filament of the material of choice. Import the G-code file into the 3D printer to read. Print the mold.
NOTE: Alternatively, produce the eye molds using a computer numerical controlled (CNC) machine, if a smoother surface on the eye model is desired. For CNC mold production, materials for molds are no longer limited to thermal plastics, but extend to metal, ceramics, and chemically resistive polymers such as Polytetrafluoroethylene. - Open the CNC software interface that is connected to a cutting drill. Construct 3D molds according to front, top, side, and perspective views of the previously-constructed eyeball/eyelid model molds in control software interface. Select appropriate parameters for the machining (bit size, substrate material, material thickness) and proceed to cut the mold.
- Synthesis of Eyepieces Using PDMS
- Using a syringe, measure 10 ml volume of PDMS (polydimethylsiloxane) base and fill it into a 15-50 ml centrifuge tube. Add 10% w/v of the elastomer solution by total weight of PDMS. Using a stirring rod, mix the solutions well.
- Pour the PDMS solution into the eyeball and eyelid molds. Allow the PDMS to settle at RT O/N (or for at least 12 hr) to start the polymerization and to allow bubbles to dissolve out of the polymer.
NOTE: Ensure that there are no bubbles left in the PDMS that might rise or expand. - Subsequently, put the molds into a 75 °C (167 °F) oven for 1 hr, or 150 °C (302 °F) for 5 min. For a softer gel, let the PDMS sit at RT for at least 48 hr to completely polymerize.
- Put the samples in a freezer for a few min; this will shrink the PDMS and simplify the removal of the samples from the molds. Extract the eyepieces from the molds using a thin spatula.
- For the delivery of solution into the space between the eyeball and eyelid pieces, connect a 1/16" x 1/8" polytetrafluoroethylene tube with a 1/16" equal leg coupler tube connector and attach it to the eyelid piece at the tubing hole.
- Synthesis of Eyeball Piece Using Agarose
NOTE: The eyeball piece can be synthesized using other polymers such as agarose. The following procedure can also be modified to produce eye pieces from a variety of agar types, such as PDA (potato dextrose agar) or SDA (sabouraud dextrose agar).- To produce a 2% (2 g/100 ml) gel, measure 2 g of agarose and mix with 100 ml of ultrapure water. Bring the solution to a boil (100 ºC) such that the agarose dissolves completely. Let the solution cool down for 5 min.
- Pour the solution into the eyeball mold and allow the solution to cool for 30 min at RT. Remove the eyeball pieces with a spatula. Store the eyeball agar in a -20 °C freezer for later use. For microbiology studies, sterilize the eyeball molds by autoclaving and/or UV-irradiation.
- Incorporation of Bovine Cornea on PDMS Eyeball
NOTE: This protocol has been adapted from Parekh et al.14- Perform the dissection and incorporation of the bovine corneas in sterile conditions under a laminar flow hood. Acquire the eyes and dissect them on the same day.
- Turn the flow hood on for 10 min prior to use and sanitize with 70% ethanol alcohol. Ensure that all materials and instruments are sterile by autoclaving at 273 °F/133 °C for 45 min, and positioned no less than 4 inches from the flow hood entrance.
- Immerse the bovine eye in a beaker containing a diluted povidone-iodine solution for 2 min. Rinse the eye in a beaker containing phosphate buffered saline (PBS) pH 7.4. Using forceps gently place the eye on a glass petri dish, corneal face up.
- Remove the excess muscle and fatty tissue by cutting at the scleral attachment points with blunt end dissection scissors. Dispose of the excess tissue into a sterile beaker designated for animal waste.
- Using micro-scissors, remove the conjunctiva from the eye. Wrap the eye with sterile gauze, maintaining a distance of at least 1 cm from the limbus.
- Using a scalpel, incise the sclera approximately 2 mm from the limbus region and superficially so as to avoid penetration of the underlying choroid and vitreous body. Carefully extend the incision by 360° using a scalpel or dissection scissors without deforming the cornea from its natural curvature.
- With fine forceps, remove the cornea from the eye. Using forceps, carefully remove any adhering uveal tissue and rinse cornea with PBS.
- Store the cornea at 31ºC in a sterile container with culture medium (such as Medium 199) containing 3% Fetal Bovine Serum to maintain tissue moisture and cell nourishment.
- Prior to experimentation, rest the excised cornea on the PDMS eyeball, and clamp the two pieces together with a specialized clip-on.
2. Blink-platform
- Design and Production of the Blink-platform
NOTE: The blink-platform is composed of three functional parts: eye model (described in section 1), gear system, and electronic system.- Design and manufacture the blink platform using CAD and 3D printing, similar to that described for the eye model (section 1.1). Design the gear system such that it translates simple rotation of motors into the lateral and rotational motions of the eyepieces.15
- Using the pinion and gear mechanism, translate rotational motion of a stepper motor into the lateral motion of a pinion, which is connected to the eyelid pieces.
- Using the conjugate gear system, amplify one rotational motion from a stepper motor into three (or more) rotational motions for three different eyeball pieces.
- Align the two gear systems, one for the eyelid and one for eyeball, so that the distance between the two are constant. Assemble the electronic system with a microcontroller, motor shield, and two motors.
NOTE: Use two stepper motors to provide rotational motors, which is translated by the gear system into a blinking motion. - Connect the two stepper motors with a system consisting of a motor shield stacked on the microcontroller. Connect and configure the electronic components to work with open source software products.
- Program the system to control motor parameters such as rounds per minute (RPM), number of rounds forward, number of rounds backward, and turning style.
NOTE: Refer to the supplementary "Arduino code file" for details. - Download the system software from the manufacturers' website.
- Install the software and open it. Write the code to control stepper motors in the desired configuration. Connect the system with a source to power the electronic system so that the motors move in the desired manner as defined by the researcher.
NOTE: Refer to the supplementary "Arduino code file".
- Assembly with Microfluidics (Artificial Tear Solution)
- Take the synthesized eyeball and eyelid pieces and slip them onto their corresponding clip-ons for the eye-model. Connect the tubing that is joined with a syringe and positioned on the microfluidic pump with the eyelid piece (section 1.2.5). Test run the platform and check for consistent movement.
- Prime the tubing and check for a steady flow of artificial tear solution (ATS). The recipe for ATS has been previously reported.16
- Manually move the eye-model parts together on a level plane, such that the eyeball and the eyelid are in contact. Set the flow rate of the microfluidic pump to desired values. Set physiological flow rates to 1-1.5 µl/min.17
- Start the pump and the actuators to begin experiment. For drug delivery experiments, place the drug-containing contact lens on the eyeball piece.
- Allow the flow-through fluid to drip into a 12-well plate. At the desired set time intervals, quantify the analyte or drug concentration using common detection methods such as UV-Vis spectroscopy or fluorescence.1,4,18
- For studies evaluating deposition of tear components on contact lenses, place the contact lens on the "eyeball" piece. Collect the flow-through fluid, which can be discarded.
- After the desired time intervals, remove the contact lens from the eyeball piece and prepare the lens for further analysis such as confocal microscopy.
Representative Results
The synthesized eye molds obtained from the machine shop and from 3-D printing are shown in Figure 1. These molds can be used with a variety of polymers, such as PDMS and agarose, to produce eyepieces with the desired properties. The motioned assembly of the eye model platform with a microfluidic syringe pump is shown in Figure 2. The platform simulates mechanical wear via the rotation of the eyeball piece, and air exposure through the lateral in and out motion of the eyelid piece. Tear fluid is infused into the eyelid from a microfluidic pump at the desired flow rate, and the flow-through fluid can be collected in a 12-well plate.
The procedure for dissection of a bovine lens, and mounting onto a PDMS eyepiece is depicted in Figure 3. The excess tissues are separated from the eye and discarded, followed by the removal of the conjunctiva. The removal of the cornea begins with an incision into the sclera near the limbus. Figure 4 shows the variety of eyepieces that could be used for various in vitro analyses. The mounted eyeball pieces shown are synthesized from PDMS, agar, and an ex-vivo bovine cornea mounted on a PDMS eyeball piece.
Figure 5 depicts a study evaluating the release of an antibiotic, moxifloxacin, from CLs.18 When measured in the traditional vial model, drug release occurs within the first 2 hr followed by a plateau phase. In contrast, the novel eye model shows drug release to be slow and sustainable for up to 24 hr.18 A study evaluating the deposition of cholesterol on CLs is shown in Figure 6. The cholesterol in the study was fluorescently tagged in the form of NBD-cholesterol (7-nitrobenz-2-oxa-1,3-diazol-4-yl-cholesterol), and deposition was imaged using laser scanning confocal microscopy. The results indicate that there are substantial differences when the deposition studies are performed in a vial as compared to the eye model.

Figure 1. Eyepiece molds. (A) Eyeball piece mold from machine shop. (B) Eye lid mold from 3-D printing. Please click here to view a larger version of this figure.

Figure 2. An in vitro ocular platform. (A) Circular motion simulates mechanical wear. (B) Lateral motion produces intermittent air exposure. (C) Tear fluid infusion into eyelid. (D) Collecting well plate. Please click here to view a larger version of this figure.
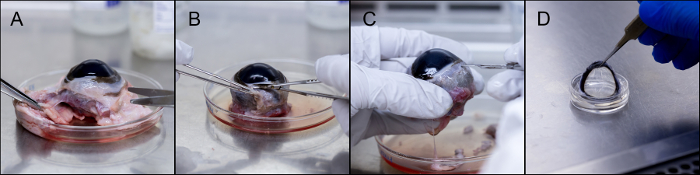
Figure 3. Dissection and incorporation of bovine cornea. (A) Removal of excess tissue. (B) Removal of conjunctiva. (C) Incision into the limbus region. (D) The excised cornea can be stored or mounted on a PDMS eye ball piece. Please click here to view a larger version of this figure.

Figure 4. Sample eyepieces. Sample of PDMS eye piece with a contact lens, an agar eye piece, and ex vivo bovine cornea mounted eye piece. Please click here to view a larger version of this figure.
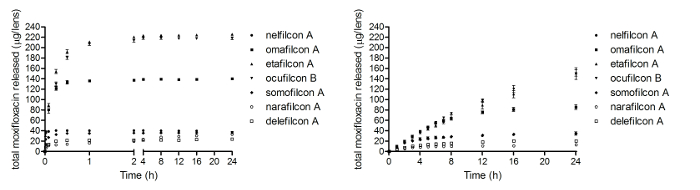
Figure 5. Drug delivery using the in vitro ocular platform. Release of moxifloxacin from daily disposable contact lenses from (A) a large volume static vial and (B) the eye model (Re-print with permission from the Association for Research in Vision and Ophthalmology).18 All data are reported as mean ± standard deviation. Please click here to view a larger version of this figure.

Figure 6. Cholesterol deposition using the in vitro ocular platform. Confocal images showing a cross-section of etafilcon A, nelfilcon A, nesofilcon A, ocufilcon B, delefilcon A, somofilcon A, narafilcon A after 4 hr incubation with NBD-cholesterol in the vial and eye model. Please click here to view a larger version of this figure.
Discussion
There are three critical steps within the protocol that require special attention: design and production of molds (section 1.1), platform assembly (section 2.2.1-2.2.3), and monitoring the experimental run (section 2.2.4-2.2.7). In terms of the design and production of molds (section 1.1), the eyeball piece should be designed according to the dimensions of a human cornea. However, it may require multiple prototypes of the mold before an eyeball piece can be created that perfectly fits a commercial contact lens (CL). In addition, the 250 µm needs to be maintained when the eyeball and eyelid piece are in contact to ensure the tear fluid flows smoothly throughout the entire eye model when a CL is present. This distance could be changed in future iterations, but should not be less than 150 µm to allow for enough spacing to fit a CL. The platform assembly (section 2.2.1-2.2.3) requires careful attention such that the eyeball and eyelid piece come into contact during the blink motion. If the eyepieces are not in perfect contact, then simulation of a closed eyelid and mechanical rubbing fails. The operator should observe the platform in motion for a few cycles to ensure that both the eyeball and eyelid are in contact, and that rubbing occurs as programmed. The current platform is designed to run continuously over one month, but an operator should always check on the stability of the system every 24 hr when running an experiment (section 2.2.4-2.2.7). This is important as the current platform does not possess a temperature or humidity control, and fluctuations in these parameters could dry up the CLs. If this occurs, place the eye model within a controlled humidity and temperature chamber. In addition, for drug delivery experiments, the collected flow-through fluid should be analyzed or stored at least every 2 hr to avoid significant evaporation of the sample.
There are currently two limitations of the presented eye model. The first limitation is in regards to exposure to the surrounding environment. Currently, because the eye pieces are not enclosed in a controlled chamber, changes such as temperature and humidity in the work area will influence various aspects of the experiments. For instance, if the environment is too dry, then the CLs dry up quicker and could separate from the eyeball piece, or the flow-through fluid could evaporate. To address this problem, future iterations will house the eye model in a controlled temperature and humidity chamber. The second limitation pertains to the complexity eyeball piece. Currently, the eyepieces are simple, consisting of either PDMS or agarose, neither of which truly represents corneal surface properties. Future work will aim to produce eye models which closer mimics the corneal surface structures.
In vitro ocular research is generally viewed as the preceding testing phase to in vivo research. However, it is important to keep in mind that in vitro research can also be complementary to in vivo data, providing critical insights that otherwise cannot be achieved from in vivo studies alone. Regrettably, the current in vitro models for testing CLs are rudimentary and lack several key components to adequately mimic the in vivo environment. For instance, in vitro CL studies are performed in vials containing 2-5 ml of phosphate buffered saline,1-6 which greatly exceeds physiological tear volumes at 7.0 ± 2 µl.7 Moreover, two important factors of the ocular environment, natural tear flow and the blinking reflex, are absent from the simple static vial model. The limitations of the conventional vial model have been recognized by researchers, and attempts have been made to create unique in vitro eye models simulating the ocular environment, by including a microfluidic tear replenishment component20-24 and/or intermittent air exposure.25,26 Not surprisingly, the results generated from these experiments are very different than those obtained with the conventional vial model, and may more closely resemble in vivo data.20-25 Thus, developing an intricate in vitro eye model to examine CLs will provide new insights on the interaction of lens materials with the ocular surface, and help facilitate the development of new materials and new applications for CLs in the coming decades.
Arguably, one of the most debated aspects of the in vitro eye model is whether the eye resembles an infinite sink, which is particularly important when it comes to drug delivery from CLs. Under infinite sink conditions, the volume of the surrounding solution is significantly higher than the drug saturation volume, such that drug release is not affected by the drug's solubility.27 Advocates for the vial as an acceptable eye model argue that the cornea, conjunctiva, and surrounding ocular tissues together function as an infinite sink. While in theory this may be true, the drug must first dissolve into the tear fluid. This rate limiting step is likely not a sink condition, and will be dependent on both tear volume and flow as simulated by our model.
The unique identity of the presented model lies in its ability to emulate the tear film. By adopting a two-piece design, a "corneal/scleral" eyeball section and an "eyelid", it is possible to create an evenly spread thin layer of tear film across the eyeball piece when both pieces come into contact. To further simulate the ocular surface, mechanical wear and air exposure is incorporated into the model through two mechanical actuators. As the eyelid piece moves laterally, it simulates the closing of the eye and intermittent air exposure. The rotation of the eyeball simulates the mechanical wear produced during blinking. The system is coupled with a microfluidic pump, which infuses the eye model with tear fluid at a physiological flow rate or any other desired flow rate. The tear film is formed each time the two pieces come into contact, and tear break-up occurs when the two pieces separate.
The aim is to create a universal testing platform to evaluate CLs for various in vitro analyses. In order to be versatile, the eyeball pieces can be synthesized from various polymers, such as polydimethylsiloxane (PDMS) or agar. For simple ocular studies, these polymers, which represent hydrophobic and hydrophilic surfaces respectively, will suffice. However, as more complex analyses are required, for example ocular drug penetration or toxicity studies, the eye pieces will need to be further modified. These additional modifications to the model, such as the inclusion of an ex vivo cornea as shown, are relatively feasible. However, further validation studies are required, and future work will aim to improve the validity of this model by comparing it with in vivo models.
Divulgations
The authors have nothing to disclose.
Acknowledgements
The authors would like to acknowledge our funding source NSERC 20/20 Network for the Development of Advanced Ophthalmic Materials.
Materials
| Arduino Uno R3 (Atmega328 – assembled) | Adafruit | 50 | Board |
| Stepper motor | Adafruit | 324 | Motor and Motor shield |
| Equal Leg Coupler 1.6mm 1/16" | VWR | CA11009-280 | 50 pcs of tube connector |
| Tubing PT/SIL 1/16"x1/8" | VWR | 16211-316 | Case of 50feet |
| PDMS | Dow Corning | Sylgard 184 Solar Cell Encapsulation | |
| Agarose, Type 1-A, low EEO | Sigma-Aldrich | A0169-25G | |
| PHD UltraTM | Harvard Apparatus | 703006 | MicroFluidic Pump |
| Bovine cornea | Cargill, Guelph/ON | ||
| Soldidworks | Dassault Systemes | Software | |
| 3-D printing | University of Waterloo – 3D Print Centre | ||
| Dissection tools | Fine Science Tools | General dissection tools | |
| Medium 199 | Sigma-Aldrich | Culture medium storage for cornea | |
| Fetal bovine serum | Thermo Fisher | Add to culture medium, 3% total volume |
References
- Phan, C. M., Subbaraman, L. N., Jones, L. In vitro drug release of natamycin from beta-cyclodextrin and 2-hydroxypropyl-beta-cyclodextrin-functionalized contact lens materials. J Biomater Sci Polym Ed. 25, 1907-1919 (2014).
- Peng, C. C., Kim, J., Chauhan, A. Extended delivery of hydrophilic drugs from silicone-hydrogel contact lenses containing vitamin E diffusion barriers. Biomaterials. 31, 4032-4047 (2010).
- Hui, A., Willcox, M., Jones, L. In vitro and in vivo evaluation of novel ciprofloxacin-releasing silicone hydrogel contact lenses. Invest Ophthalmol Vis Sci. 55, 4896-4904 (2014).
- Boone, A., Hui, A., Jones, L. Uptake and release of dexamethasone phosphate from silicone hydrogel and group I, II, and IV hydrogel contact lenses. Eye Contact Lens. 35, 260-267 (2009).
- Lorentz, H., Heynen, M., Trieu, D., Hagedorn, S. J., Jones, L. The impact of tear film components on in vitro lipid uptake. Optom Vis Sci. 89, 856-867 (2012).
- Hall, B., Phan, C. M., Subbaraman, L., Jones, L. W., Forrest, J. Extraction versus in situ techniques for measuring surface-adsorbed lysozyme. Optom Vis Sci. 91, 1062-1070 (2014).
- Mishima, S., Gasset, A., Klyce, S. D., Baum, J. L. Determination of tear volume and tear flow. Invest Ophthalmol Vis Sci. 5, 264-276 (1966).
- Nichols, J. J., et al. The TFOS international workshop on contact lens discomfort: executive summary. Invest Ophthalmol Vis Sci. 54, 7-13 (2013).
- Peng, C. C., Burke, M. T., Carbia, B. E., Plummer, C., Chauhan, A. Extended drug delivery by contact lenses for glaucoma therapy. J Control Release. 162, 152-158 (2012).
- Faschinger, C., Mossbock, G. Continuous 24 h monitoring of changes in intraocular pressure with the wireless contact lens sensor Triggerfish. First results in patients. Der Ophthalmologe : Zeitschrift der Deutschen Ophthalmologischen Gesellschaft. 107, 918-922 (2010).
- Shaw, A. J., Davis, B. A., Collins, M. J., Carney, L. G. A technique to measure eyelid pressure using piezoresistive sensors. IEEE transactions on bio-medical engineering. 56, 2512-2517 (2009).
- Liao, Y. T., Yao, H. F., Lingley, A., Parviz, B., Otis, B. P. A 3-mu W CMOS glucose sensor for wireless contact-lens tear glucose monitoring. Ieee J Solid-St Circ. 47, 335-344 (2012).
- Coster, D. J. . Cornea. , (2002).
- Parekh, M., et al. A simplified technique for in situ excision of cornea and evisceration of retinal tissue from human ocular globe. Journal of visualized experiments : JoVE. , e3765 (2012).
- Way, S. Gear and pinion. US patent. , (1942).
- Lorentz, H., et al. Contact lens physical properties and lipid deposition in a novel characterized artificial tear solution. Molecular vision. 17, 3392-3405 (2011).
- Furukawa, R. E., Polse, K. A. Changes in tear flow accompanying aging. American journal of optometry and physiological optics. 55, 69-74 (1978).
- Bajgrowicz, M., Phan, C. M., Subbaraman, L., Jones, L. Release of ciprofloxacin and moxifloxacin from daily disposable contact lenses from an in vitro eye model. Invest Ophthalmol Vis Sci. , (2015).
- Luensmann, D., Zhang, F., Subbaraman, L., Sheardown, H., Jones, L. Localization of lysozyme sorption to conventional and silicone hydrogel contact lenses using confocal microscopy. Current eye research. 34, 683-697 (2009).
- Tieppo, A., Pate, K. M., Byrne, M. E. In vitro controlled release of an anti-inflammatory from daily disposable therapeutic contact lenses under physiological ocular tear flow. Eur J Pharm Biopharm. 81, 170-177 (2012).
- Ali, M., et al. Zero-order therapeutic release from imprinted hydrogel contact lenses within in vitro physiological ocular tear flow. J Control Release. 124, 154-162 (2007).
- White, C. J., McBride, M. K., Pate, K. M., Tieppo, A., Byrne, M. E. Extended release of high molecular weight hydroxypropyl methylcellulose from molecularly imprinted, extended wear silicone hydrogel contact lenses. Biomaterials. 32, 5698-5705 (2011).
- Kaczmarek, J. C., Tieppo, A., White, C. J., Byrne, M. E. Adjusting biomaterial composition to achieve controlled multiple-day release of dexamethasone from an extended-wear silicone hydrogel contact lens. J Biomater Sci Polym Ed. 25, 88-100 (2014).
- Mohammadi, S., Postnikoff, C., Wright, A. M., Gorbet, M. Design and development of an in vitro tear replenishment system. Ann Biomed Eng. 42, 1923-1931 (2014).
- Lorentz, H., Heynen, M., Khan, W., Trieu, D., Jones, L. The impact of intermittent air exposure on lipid deposition. Optom Vis Sci. 89, 1574-1581 (2012).
- Peng, C. C., Fajardo, N. P., Razunguzwa, T., Radke, C. J. In vitro spoilation of silicone-hydrogel soft contact lenses in a model-blink cell. Optom Vis Sci. 92, 768-780 (2015).
- Liu, P., et al. Dissolution studies of poorly soluble drug nanosuspensions in non-sink conditions. AAPS PharmSciTech. 14, 748-756 (2013).

