In Vitro Permeation of FITC-loaded Ferritins Across a Rat Blood-brain Barrier: a Model to Study the Delivery of Nanoformulated Molecules
Summary
A method to establish an in vitro model of blood-brain barrier based on a co-culture of rat brain microvascular endothelial cells and astrocytes is described and validated. This system proved to be a valid tool to study the effect of nanoformulation on the trans-barrier permeation of fluorescent molecules.
Abstract
Brain microvascular endothelial cells, supported by pericytes and astrocytes endfeet, are responsible for the low permeation of large hydrosoluble drugs through the blood-brain barrier (BBB), causing difficulties for effective pharmacological therapies. In recent years, different strategies for promoting brain targeting have aimed to improve drug delivery and activity at this site, including innovative nanosystems for drug delivery across the BBB. In this context, an in vitro approach based on a simplified cellular model of the BBB provides a useful tool to investigate the effect of nanoformulations on the trans-BBB permeation of molecules. This study describes the development of a double-layer BBB, consisting of co-cultured commercially available primary rat brain microvascular endothelial cells and astrocytes. A multiparametric approach for the validation of the model, based on the measurement of the transendothelial electrical resistance and the apparent permeability of a high molecular weight dextran, is also described. As proof of concept for the employment of this BBB model to study the effect of different nanoformulations on the translocation of fluorescent molecules across the barrier, we describe the use of fluorescein isothiocyanate (FITC), loaded into ferritin nanoparticles. The ability of ferritins to improve the trans-BBB permeation of FITC was demonstrated by flux measurements and confocal microscopy analyses. The results suggest this is a useful system for validating nanosystems for delivery of drugs across the BBB.
Introduction
The resistance of central nervous system (CNS) diseases (i.e. cancer, epilepsy, depression, schizophrenia and HIV-associated neurological disorder) to pharmacological therapies is due to various different mechanisms, including arduous drug permeation across the blood-brain barrier (BBB). The BBB is the boundary that isolates brain tissues from the substances circulating in the blood. Within this barrier, a layer of brain microvascular endothelial cells (BMECs), supported by pericytes and astrocytes endfeet, is responsible for the high selectivity of the BBB to those hydrosoluble drugs with a molecular weight higher than 400 Da1. Another drug-related resistance mechanism is linked to the presence on BMECs of drug efflux transporters (P-glycoprotein and multidrug resistance proteins), which co-operate to reduce drug penetration into the CNS and facilitate their extrusion from the brain2.
In the last decade, a large number of nanotechnological approaches have been developed to meet the clinical and biological challenge of delivering drugs across the BBB3-6. In this context, ferritin nanospheres (FnN) represent a completely innovative and promising solution. FnN are 12 nm spheres of 24 self-assembling ferritin (Fn) monomers, which are arranged in a hollow spherical structure of 8 nm inner diameter. Ferritin subunits can be disassembled at acidic pH and reassembled in a shape-memory fashion by bringing the pH to neutrality, allowing various organic molecules to be encapsulated. Therefore, FnN represent an interesting model for the development of multifunctional drug delivery systems7,8. Moreover, FnN may interact with BMECs thanks to the specific recognition of Transferrin Receptor (TfR) 1, which is expressed on the luminal membrane of these cells9.
So far, different in vitro models of the BBB have been developed in order to elucidate trans-BBB permeability to various drugs, toxicity toward the BBB, or the interaction of molecules with efflux transporters. Indeed, these models are considered valid in vitro approaches for a rapid screening of active molecules before proceeding with in vivo studies. These models consist of a single endothelial layer of BMECs or co-cultured BMECs and astrocytes (more rarely pericytes), obtained from animal (rat, mouse, pig and bovine) or human cell lines10,11,12. The TransEndothelial Electrical Resistance (TEER) and the apparent permeability (Papp) of tracers with a defined molecular weight represent two critical parameters that are used to determine the quality of the in vitro model. Here we describe the employment of a BBB in vitro model, based on a co-culture of rat BMECs (RBMECs) and rat cortical astrocytes (RCAs) to study the trans-BBB permeation of ferritin nanocages encapsulating fluorescein isothiocyanate (FITC).
Protocol
1. Establishing the BBB Model
Note: For establishing the BBB model we suggest using commercially available primary RBMECs and RCAs. All steps must be performed with sterile reagents and disposables, handled in a laminar flow hood.
- Cell Culture
- Coat cell culture flasks with poly-L-lysine 100 µg/ml (1 hr at RT) or fibronectin 50 µg/ml (1 hr at 37 °C) to promote the attachment of RCAs or RBMECs, respectively. Then, thaw 1 x 106 RCAs and 5 x 105 RBMECs in Endothelial Cell Medium, supplemented with 5% fetal bovine serum, 1% endothelial cell growth supplement and 1% penicillin/streptomycin (sECM). Seed RCAs in a T175 flask in 20 ml sECM and RBMECs in a T75 flask in 10 ml sECM.
Note: The thawing and seeding passages of RCAs and RBMECs must be adjusted, in terms of cell density and time in culture, according to the number of experimental conditions to be tested with the BBB model. By starting with 1 vial of 1 x 106 RCAs and 1 vial of 5 x 105 RBMECs, it is possible to obtain up to 20 BBB-bearing inserts. - Maintain the cells at 37 °C and 5% CO2 in a humidified atmosphere for approximately 6 days, until about 80% confluence for RCAs and over 90% confluence for RBMECs. Detach cells using trypsin-EDTA solution (trypsin 1:250) for 5 min (1 ml trypsin for T75 flasks and 2 ml for T175 flasks). Stop trypsin activity with sECM (2:1), centrifuge cell suspensions at 750 × g for 5 min and resuspend the pellets in 60 ml sECM.
- Split the RBMECs into 3 T175 flasks and culture in sECM for another 3 days before seeding onto inserts. Count the total number of living RCAs, by observing a cell suspension diluted 1:1 with trypan blue under an optical microscope in a Burker chamber.
- Coat cell culture flasks with poly-L-lysine 100 µg/ml (1 hr at RT) or fibronectin 50 µg/ml (1 hr at 37 °C) to promote the attachment of RCAs or RBMECs, respectively. Then, thaw 1 x 106 RCAs and 5 x 105 RBMECs in Endothelial Cell Medium, supplemented with 5% fetal bovine serum, 1% endothelial cell growth supplement and 1% penicillin/streptomycin (sECM). Seed RCAs in a T175 flask in 20 ml sECM and RBMECs in a T75 flask in 10 ml sECM.
- Cell Seeding onto Inserts
- Treat one side of the Polyethylene terephthalate (PET) membrane of 6 multi-well transparent inserts with poly-L-lysine 100 µg/ml and the other side with fibronectin 50 µg/ml, to allow attachment of RCAs and BMECs. Handle the inserts with tweezers in order to avoid contact with the PET membrane.
- Place the inserts into a 6 well plate and add fibronectin solution (minimum 500 µl) into the upper chamber. After 1 hr of incubation at 37 °C, remove the fibronectin solution, take the inserts off the multi-well plate and put them upside down on the bottom of a 150 cm2 Petri dish, which is used here as a sterile support for the already coated inserts.
- Gently add 800 µl of poly-L-lysine onto the bottom side of the insert (as shown in Figure 1A; referred to as RCAs seeding), and keep the solution on the inserts for 1 hr at RT.
- Aspirate the solution and let the inserts dry at RT for 15-30 min. The inserts are now ready for cell seeding, but can also be stored in the multi-well plate for several days at 4 °C, before proceeding with the BBB construction.
Note: Remember to keep at least 3 coated inserts free from cells, to be used as controls to validate BBB establishment by FD40 flux measurements.
- Seed RCAs (35,000/cm2) onto the bottom side of the poly-L-lysine-coated inserts, by dropping 800 µl of cell suspension onto the upside down insert (Figure 1A). Leave the RCA suspension on the inserts for 4 hr at RT, in order to allow efficient attachment of the cells to the membrane.
- Aspirate the residual solution, place the inserts into wells containing 2 ml of sECM and maintain the multi-well plate at 37 °C and 5% CO2 in a humidified atmosphere, changing the sECM every 2 days.
- After 3 days, when the RCAs have coated the lower face of the insert, seed RBMECs onto the upper side of the insert, following these steps:
- Detach RBMECs from T175 flasks and count the total living cells, as reported in steps 1.1.2 and 1.1.3.
- Seed RBMECs (60,000/cm2) onto the upper surface of the insert in sECM (1,000 µl) (Figure 1B), leaving 3 inserts with the astrocytes only, to be used as TEER background. Place the multi-well plate in standard culture conditions. Maintain the system to culture for at least 3 days, changing the sECM in the inner and lower chamber every 2 days.
- Treat one side of the Polyethylene terephthalate (PET) membrane of 6 multi-well transparent inserts with poly-L-lysine 100 µg/ml and the other side with fibronectin 50 µg/ml, to allow attachment of RCAs and BMECs. Handle the inserts with tweezers in order to avoid contact with the PET membrane.
2. BBB Validation
- TEER Measurement
- On the 3rd day of co-culture, check the TEER by inserting the BBB-bearing inserts into an appropriate chamber(that has a cap and where both chamber and cap contain a pair of voltage-sensing and current electrodes), filled with 4 ml of sECM. Connect to an epithelial tissue volt/ohmmeter.
- Together with the TEER measurements of the cell BBB-systems, record the TEER values of the 3 inserts bearing the RCAs that will be subtracted from the values obtained with the BBBs. Multiply the resulting TEER values by the surface of the insert (4.2 cm2) in order to express the results as Ω x cm2.
- From the 3rd day of co-culture, measure the TEER every day. Note: After an initial period where the TEER increases, the recorded values should remain stable for at least two consecutive days (usually between the 5th and the 7th day of co-culture). At that point the BBB is ready for the second step of validation (section 2.2) and/or for the trans-BBB experiments.
Note: The procedure described in section 2.1 can be performed on the same BBB-systems devoted to the following permeability experiments (section 3). Operate under sterile conditions.
- Trans-BBB Flux of FITC-dextran 40 (FD40)
- Measure the FD40 flux from the upper to the lower chamber of the BBB models compared to that across 3 empty inserts (see the note of section 1.2.1) according to the following steps:
- Add 1 mg/ml FD40 (diluted in sECM) into the upper compartment of the BBBs, and after 1, 2 and 3 hr withdraw 200 µl sECM from the lower chamber and measure the fluorescence intensity by spectrofluorimeter (λex 488 nm, λem 515 nm, slit 5).
- Obtain the values for sECM background fluorescenceby measuring 500 µl of virgin medium using the same analytical parameters. Subtract the sECM background fluorescence from all FD40 fluorescence values.
- Determine the amount of permeated FD40 by comparison of the observed fluorescence values with a calibration curve produced with known concentrations (i.e. 0.312, 0.625, 1.25, 2.5 µg/ml) of the fluorescent tracer dissolved in 500 µl of sECM.
- Calculate the apparent permeability coefficient (Papp) from the mean flux values according to:
Papp = J/AC
where J is the flux of the molecule (moles/sec), A is the permeation area (cm2) and C is the concentration of the molecule in the upper compartment (moles/cm3).
Note: Three or more BBB-systems that have reached suitable TEER values, must be exclusively devoted to this validation procedure, and should not be employed for the following permeability experiments (section 3). Sterility is not mandatory.
- Measure the FD40 flux from the upper to the lower chamber of the BBB models compared to that across 3 empty inserts (see the note of section 1.2.1) according to the following steps:
3. Trans-BBB Permeation of FITC-loaded Ferritins (FnN)
Note: A recombinant variant of human ferritin (Fn), produced in Escherichia coli and assembled in nanocages (FnN) for the encapsulation of different fluorescent molecules, is available from the NanoBioLab of Prof. Prosperi (University of Milan-Bicocca, Italy). FnN are loaded with FITC, according to a previously described protocol13 and the concentrations of both Fn and the loaded molecules are accurately determined.
- Trans-BBB Flux of FITC and FITC-FnN
- Measure the FITC-FnN flux from the upper to the lower chamber of validated BBB models (usually at the 5th – 7th day of co-culture), compared to that of the free dye after 7 and 24 hr of incubation, according to the following steps:
- Add FITC-FnN (50 µg/ml FnN, 1.1 µM FITC) or an equal amount of free FITC into the upper chamber of at least 6 BBB systems for each formulation, in order to withdraw 2 ml of sECM solution from the lower chamber of at least 3 inserts after 7 hr, and from the lower chamber of other 3 inserts after 24 hr.
- Measure the fluorescence intensity of 500 µl of the collected samples by spectrofluorimeter (λex 488 nm, λem 515 nm, slit 5).
- Obtain the sECM background fluorescence by measuring 500 µl of medium using the same analytical parameters. Subtract the sECM background fluorescence from all FITC or FITC-FnN fluorescence values.
- Determine the concentration of permeated FITC or FITC-FnN by comparing the obtained fluorescence values with two different calibration curves produced with known concentrations (i.e. 6.87, 13.75, 27.5, 55 nM) of the free or nanoformulated dye dissolved in 500 µl sECM.
- Measure the FITC-FnN flux from the upper to the lower chamber of validated BBB models (usually at the 5th – 7th day of co-culture), compared to that of the free dye after 7 and 24 hr of incubation, according to the following steps:
- FITC-FnN Localization in RBMECs
- Remove sECM from the upper chamber of the BBB systems, wash the inserts with PBS and fix the RBMECs on at least two inserts for each experimental group by adding 500 µl paraformaldehyde (4% in phosphate buffer saline-PBS) in the upper compartment for 10 min at RT.
- Wash the inserts three times with PBS to remove residual paraformaldehyde.
Note: From this point on, sterility is not necessary. - Cut suitable pieces of the PET membrane, and proceed with the immunodecoration of the cells according the following steps:
- Permeabilize RBMECs with 0.1% Triton X-100 in PBS for 10 minutes. Perform a blocking step for 1 hr at RT with a solution containing 2% bovine serum albumin (BSA), 2% goat serum in PBS. Incubate the samples with an anti-Von Willebrand Factor (VWF) at 1:20 dilution, for 2 hr at RT.
- After washing three times with PBS, expose the cells for 2 hr at RT to a 2% BSA, 2% goat serum solution containing a suitable secondary antibody at a 1:300 dilution in order to reveal the anti-VWR, and DAPI at a 1:1,500 dilution for nuclei detection.
- Mount the insert pieces onto microscope slides using an antifade mounting solution (one drop per insert fragment), then close the sample with a cover slip. Analyze by confocal microscopy using an oil immersion lens at 40X magnification, 1.5 zoom and 1,024 x 1,024 pixel resolution.
Representative Results
During the establishment of the BBB model, cell attachment and growth on the inserts can be monitored using a light microscope thanks to the transparent nature of the PET membranes. RCAs, seeded at a density of 35,000 cells/cm2, attach efficiently to the bottom side of the insert after 4 hr of incubation at RT (Figure 2A) and grow to cover the membrane surface in 3 days, taking a spindle-shaped morphology (Figure 2B). RBMECs, seeded at a density of 60,000 cells/cm2, are visibly attached to the upper face of the PET membrane after about 3 hr of incubation at 37 °C (Figure 2C). The developing RBMEC layer can be difficult to visualize over the following days with an optical microscope, because of the overlap with the underlying RCA layer (Figure 2D).
A correct validation of the BBB model always requires TEER measurement and this can be confirmed by evaluating the trans-BBB Papp of a low permeability tracer, such as FD40.
The TEER values, recorded over the co-culture period, represent the first clear indication of the correct formation of the endothelial barrier. Three days after RBMEC seeding, the recorded TEER, subtracted from the TEER of the astrocytes-bearing inserts, is mainly due to the contribution of the non-electrogenic layer of RCAs and the developing layer of RBMECs. At this time point, our BBB gives values ranging between 20 and 40 Ω x cm2. During the following days, usually between the 4th and the 5th day of co-culture, the TEER values increase because of the formation of tight junctions between adjacent endothelial cells14, reaching values usually between 55 and 110 Ω x cm2, and in exceptional cases higher values14. The values measured at the 4th/5th day of co-culture remain stable at least until the 7th – 8th day before they start to decrease; therefore, there is a very narrow time window available for undertaking the trans-BBB flux experiments.
Between the 5th and the 7th day of co-culture, the integrity of the experimental models can be confirmed by evaluating the FD40 trans-BBB permeability. Figure 3 shows an example of the trans-BBB flux of FD40 (1 mg/ml), compared to the flux across empty inserts, over 3 hr of incubation; the BBBs are at the 6th day of co-culture and the recorded TEER is 55.6 ± 15.8 Ω cm2 (mean ± SE, n = 3). The flux is linear between 1 and 3 hr of incubation and the mean Papp calculated between 1 and 2 hr, and between 2 and 3 hr of incubation is 0.12 ± 0.01 x 10–6 cm sec–1 (± SE, n = 6).
Once at least three consecutive successful BBBs, in terms of TEER and FD40 Papp, have been obtained in independent experiments the measurement of tracer permeability can be avoided; however, the TEER always needs to be recorded for each experiment.
The trans-BBB permeability of fluorescent molecules and the effect of the nanocomplex on their delivery can be investigated using the rat BBB models described above. Figure 4 shows the permeation of the model dye FITC upon encapsulation in FnN across BBBs with TEER of 100.4 ± 3.5 Ω cm2 (n = 16) at the 7th day of co-culture. The histograms, representing FITC concentration in the lower chamber after 7 and 24 hr from the addition of free or nanoformulated dye in the upper compartment, indicate that FnN is able to significantly increase the delivery of FITC across the BBB.
Confocal microscope images of the upper side of the insert after 7 and 24 hr of incubation with FITC-FnN (Figure 5A, C) or FITC (Figure 5B, D) show that while free FITC is not internalized by the RBMECs, its loading into the FnN allows it to enter the cells.
Before processing the inserts for confocal microscopy, a further check of the TEER is necessary to ensure there are no FnN-mediated effects upon BBB integrity.
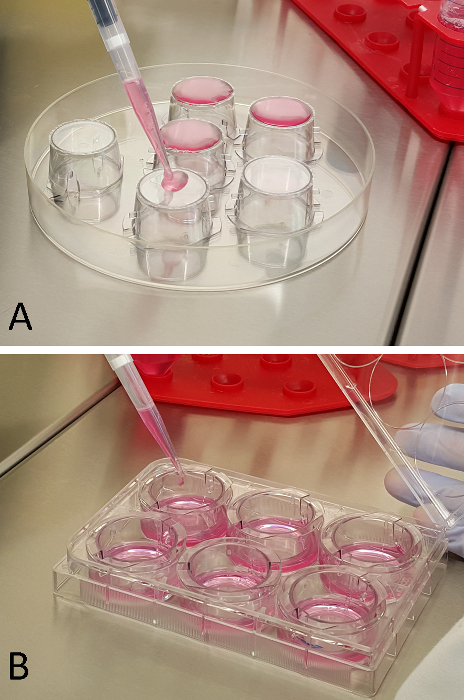
Figure 1: Cell Seeding onto Inserts. RCA (A) and RBMEC (B) seeding procedure onto the two opposite sides of the multi-well plate inserts. Please click here to view a larger version of this figure.

Figure 2: Optical Microscope Images of RCAs and RBMVECs on Inserts. Inserts seeded with RCAs and RBMECs are observed with a light microscope (20X optical zoom). (A) Four hr after seeding, round and translucent RCAs are visibly attached to the bottom surface of the insert; (B) Spindle-shaped RCAs are visible on the 3rd day of culture; (C) Round and translucent RBMECs are attached on the upper side of the insert after 3 hr of incubation; (D) On the 4th day of co-culture both the surfaces of the insert are completely covered with cells, and the two layers are not easily distinguishable. Scale bar = 100 µm. Please click here to view a larger version of this figure.
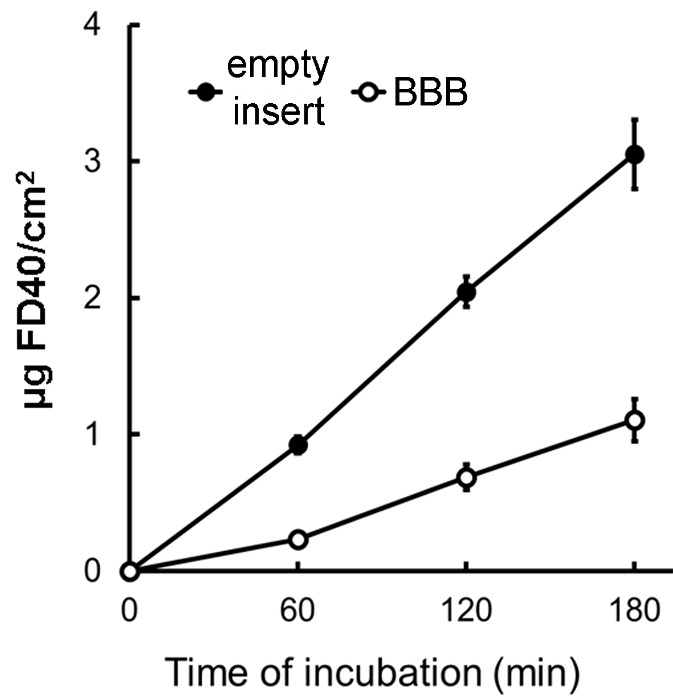
Figure 3: FD40 Flux across the BBB. Flux of FD40 (1 mg/ml) from the upper to the lower side of the BBB in vitro system, compared to that across the empty insert. The amount of FD40 in lower chamber has been measured at 60, 120 and 180 min post the addition of the dye to the upper chamber. Means ± SE; n° inserts = 3. Please click here to view a larger version of this figure.
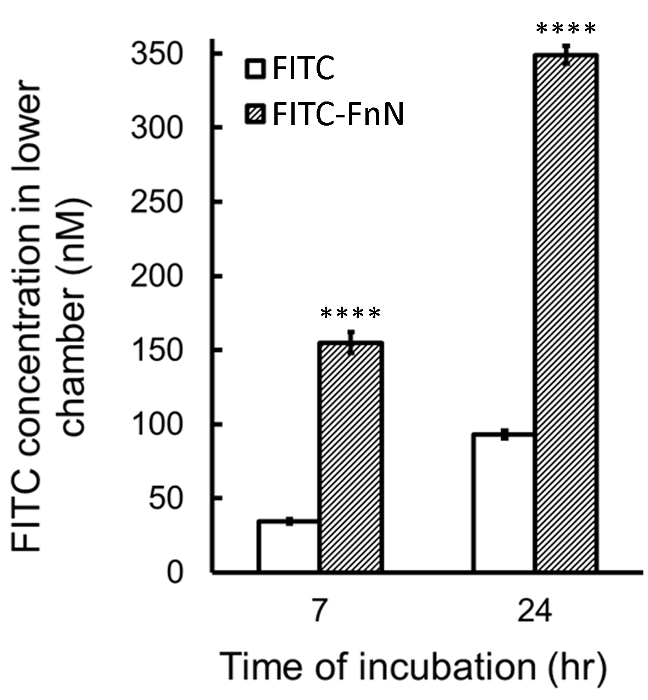
Figure 4: Effect of FnN Encapsulation on FITC Permeation Across the BBB. Concentration of FITC in the lower chamber of the BBB in vitro system calculated at 7 and 24 hr after the addition of FITC or FITC-FnN into the upper chamber. Mean ± SE of 4-5 replicates; ****P <0.0005, FITC-FnN vs. FITC (Student's t-test). Please click here to view a larger version of this figure.
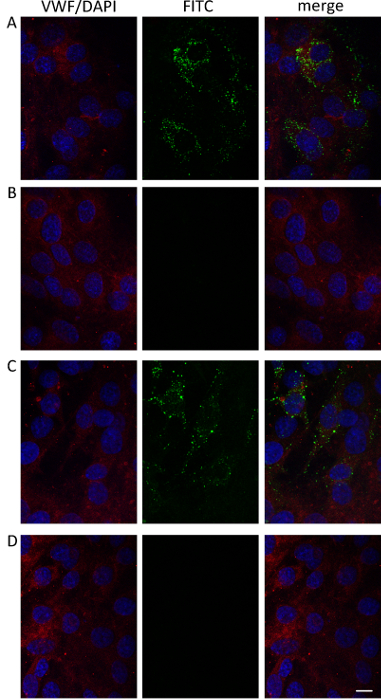
Figure 5: Confocal Microscopy of RBMECs on Inserts. Confocal laser-scanning micrographs (single optical sections) of RBMECs after 7 hr (A, B) or 24 hr (C, D) of incubation with free FITC (B, C) or FITC-FnN (A, C). FITC is green; endothelial cells are immunodecorated with anti-VWF (red) and DAPI (blue). Panels represent, from left to right, merged images of blue and red channels, green channel images, and merged images of all channels. Scale bar = 10 µm. Please click here to view a larger version of this figure.
Discussion
The in vitro method described here represents a useful validated approach to study the trans-BBB delivery of fluorescent molecules upon nanoformulation with nanoparticles. Here we use FnN, which represents a good candidate to study the translocation of cargo molecules across the BBB. FnN is considered the gold nanovector for trans-BBB delivery of drug/agents since it is specifically recognized by the TfR1 receptor, which is expressed on the luminal membrane of BMECs and mediates the nanoparticle uptake using a receptor-mediated internalization pathway. Moreover, FnN is a natural nanoparticle and it has therefore a good biocompatibility profile. Finally, FnN is able to encapsulate low dimension hydrosoluble molecules by a simple and efficient mechanism. The trans-BBB permeation of other different types of organic or inorganic nanoparticles could be also assessed with this protocol, according to few considerations regarding nanoparticles features. First prerequisite to study the permeation of nanoformulated drugs/agents is the safety of the void nanoparticle. Another crucial issue is the interaction of the investigated nanoparticle with the PET filter. This aspect, has to be evaluated preliminarily in order to exclude a significant retention into the membrane pores and avoid alterations of their permeation across the BBB. Our group has recently employed the proposed strategy to study a polymer-coated iron oxide nanoparticle as a trans-BBB delivery system for fluorescence-labeled antiretroviral drugs14. In that case, we associated electron microscopy localization of nanoformulations in RBMECs to the fluorescence detection. Moreover, other highly sensitive diagnostic methods, such as Inductively Coupled Plasma (ICP)-MS could be employed to evaluate the trans-BBB delivery of inorganic nanosystems.
The BBB in vitro model used in this protocol is based on co-culture of rat BMECs and astrocytes. In the wide scenario of the BBB models10,11,12, some of which have been accurately described in literature with a well-detailed protocol15, our model represents a good quality alternative. Indeed, even though the recorded TEER was below the best standards suggested in the literature (150-200 Ω cm2)10, the trans-BBB permeability of the high dimension tracer Dextran 40 was in total agreement with the formation of a tight barrier.
This in vitro model, obtained with commercially available rat BMECs and astrocytes, has a few advantages: (1) optimal growth efficiency of the endothelial cells, (2) a suitable period of time for the production of the final BBB model (no longer than 13 days), (3) the compliance with ethical issues, avoiding the use of cells from human brain tissue (autopsy materials, surgical specimens, and fetal tissue) and 4) saving of time and costs related to animal housing, and primary cells extraction. Nevertheless, a limitation of the present model is related to the high costs of non-immortalized primary RBMECs, which must be purchased (frozen at passage one) for each experimental setting. Indeed, our preliminary studies of BBB production have demonstrated that the ability of endothelial cells to produce a tight BBB is strictly associated with the number of passages in culture of the RBMECs after the first post-purchase thawing. Further implementation of the BBB model described here with endothelial supplements, such as cAMP and hydrocortisone, could be considered in order to improve endothelial tightness and increase TEER values.10,11,16
The present technique is related to the possibility to take advantage of a single BBB model for different assays, thus providing several data sets from each experimental setting. With a single BBB system exposed to free or nanocomplexed fluorescent molecules, it is possible: (1) to measure the trans-barrier flux of the molecule by analyzing the fluorescence intensity of sECM aliquots collected from the lower chamber at different time points of incubation; (2) to investigate the nano-mediated internalization of the molecules in RBMECs and their intracellular trafficking by confocal microscopy analysis of the cells on inserts; (3) to get an indication of the status of the BBB cells upon exposure to the nanoformulations, by measuring TEER at the end of the experiment or by analyzing endothelium integrity by electron microscopy.
In conclusion, here we described a protocol for the study of the permeation of nanocomplexed fluorescent molecules across a high-quality BBB in vitro model. We consider this methodology a useful tool for investigating the effect of nanoformulation on drug delivery across the BBB.
Divulgations
The authors have nothing to disclose.
Acknowledgements
The authors acknowledge Assessorato alla Sanità, Regione Lombardia and Sacco Hospital (NanoMeDia Project) for research funding.
Materials
| Rat Brain Microvascular Endothelial Cells | Innoprot | P10308 | isolated from Sprague Dawley rat brain tissue, cryopreserved at passage one and delivered frozen |
| Cortical Astrocytes | Innoprot | P10202 | isolated from 2 days rat brain tissue, cryopreserved at passage one and delivered frozen. |
| Endothelial Cell Medium kit | Innoprot | P60104 | ECM (500 ml) and fetal bovin serum (25 ml), endothelial cell growth supplement (5 ml) and penicillin/streptomycin (5 ml). Warm in 37 °C water bath before use and protect from light |
| Trypsin-EDTA without Phenol Red | EuroClone | ECM0920D | Warm in 37 °C water bath before use |
| Fluorescein isothiocyanate-dextran 40000 | Sigma | FD40S | protect from light |
| paraformaldehyde | Sigma | 158127 | diluition in chemical hood |
| Dulbecco's phosphate buffer saline w/o Ca and Mg | EuroClone | ECB4004L | |
| Triton X-100 | Sigma | T8787 | |
| bovine serum albumin | Sigma | A7906 | |
| goat serum | EuroClone | ECS0200D | |
| mouse monoclonal anti-Von Willebrand Factor | Dako | M0616 | |
| AlexaFluor 546-conjugated antibody against mouse IgGs | ThermoFischer Scientific | A-11003 | protect from light |
| DAPI (4’ ,6-diamidino-2-phenylindole) | ThermoFischer Scientific | D1306 | protect from light |
| ProLong Gold Antifade Mountant | ThermoFischer Scientific | P36934 | |
| Poly-L-lysine Hydrobromide | Sigma | P1274 | the same solution can be used several times |
| fibronectin from bovine plasma | Sigma | F1141 | the same solution can be used several times |
| Polyethylene terephthalate (PET) inserts | Falcon | F3090 | Transparent Polyethylene terephthalate (PET) membranes; surface area: 4.2 cm2; pore size 0.4 µm/surface area |
| T75 Primo TC flask | EuroClone | ET7076 | |
| T175 Primo TC flask | EuroClone | ET7181 | |
| EVOM2 Epithelial Tissue Volt/Ohmmeter | World Precision Instruments Germany | EVOM2 | |
| Endohm- 24SNAP cup | World Precision Instruments Germany | ENDOHM-24SNAP | |
| Light/fluorescence microscope with camera | Leica Microsystems | DM IL LED Fluo/ ICC50 W Camera Module | inverted microscope for live cells with camera |
| Confocal Microscope | Leica Microsystems | TCS SPE |
References
- Banks, W. A. Characteristics of compounds that cross the blood-brain barrier. BMC Neurol. 9 (1), 3 (2009).
- Löscher, W., Potschka, H. Drug resistance in brain disease and the role of efflux transporters. Nat Rev Neurosci. 6, 591-602 (2005).
- Xu, G., Mahajan, S., Roy, I., Yong, K. -. T. Theranostic quantum dots for crossing blood-brain barrier in vitro and providing therapy of HIV-associated encephalopathy. Front Pharmacol. 15 (4), 140 (2013).
- Masserini, M. Nanoparticles for Brain Drug Delivery. ISRN Biochem. , (2013).
- Sagar, V., Pilakka-Kanthikeel, S., Pottathil, R., Saxena, S. K., Nair, M. Towards nanomedicines for neuroAIDS. Rev. Med. Virol. 24 (2), 103-124 (2014).
- Kreuter, J. Drug delivery to the central nervous system by polymeric nanoparticles: What do we know. Adv. Drug Deliver. Rev. 71, 2-14 (2014).
- Arosio, P., Ingrassia, R., Cavadini, P. Ferritins: a family of molecules for iron storage, antioxidation and more. Biochim. Biophys. Acta. 1790 (7), 589-599 (2009).
- Jääskeläinen, A., Soukka, T., Lamminmäki, U., Korpimäki, T., Virta, M. Development of a denaturation/renaturation-based production process for ferritin nanoparticles. Biotechnol. Bioeng. 102 (4), 1012-1024 (2009).
- Jefferies, W. A., Brandon, M. R., Hunt, S. V., Williams, A. F., Gatter, K. C., Mason, D. Y. Transferrin receptor on endothelium of brain capillaries. Nature. 312, 162-163 (1984).
- Deli, M. A., Abraham, C. S., Kataoka, Y., Niwa, M. Permeability studies on in vitro blood-brain barrier models: physiology, pathology, and pharmacology. Cell. Mol. Neurobiol. 25 (1), 59-127 (2005).
- Wilhelm, I., Fazakas, C., Krizbai, I. A. In vitro models of the blood-brain barrier. Acta Neurobiol. Exp. (Wars). 71, 113-128 (2011).
- Wilhelm, I., Krizbai, I. A. In vitro models of the blood-brain barrier for the study of drug delivery to the brain. Mol Pharm. 11 (7), 1949-1963 (2014).
- Bellini, M., et al. Protein nanocages for self-triggered nuclear delivery of DNA-targeted chemotherapeutics in Cancer Cells. J. Control. Release. 196, 184-196 (2014).
- Fiandra, L., et al. Nanoformulation of antiretroviral drugs enhances their penetration across the blood brain barrier in mice. Nanomedicine. 11 (6), 1387-1397 (2015).
- Molino, Y., Jabès, F., Lacassagne, E., Gaudin, N., Khrestchatisky, M. Setting-up an in vitro model of rat blood-brain barrier (BBB): a focus on BBB impermeability and receptor-mediated transport. J. Vis. Exp. , e51278 (2014).
- Perrière, N., et al. A functional in vitro model of rat blood-brain barrier for molecular analysis of efflux transporters. Brain Res. 1150, 1-13 (2007).

