Method to Visualize and Analyze Membrane Interacting Proteins by Transmission Electron Microscopy
Summary
Many proteins perform their function when attached to membrane surfaces. The binding of extrinsic proteins on nanodisc membranes can be indirectly imaged by transmission electron microscopy. We show that the characteristic stacking (rouleau) of nanodiscs induced by the negative stain sodium phosphotungstate is prevented by the binding of extrinsic protein.
Abstract
Monotopic proteins exert their function when attached to a membrane surface, and such interactions depend on the specific lipid composition and on the availability of enough area to perform the function. Nanodiscs are used to provide a membrane surface of controlled size and lipid content. In the absence of bound extrinsic proteins, sodium phosphotungstate-stained nanodiscs appear as stacks of coins when viewed from the side by transmission electron microscopy (TEM). This protocol is therefore designed to intentionally promote stacking; consequently, the prevention of stacking can be interpreted as the binding of the membrane-binding protein to the nanodisc. In a further step, the TEM images of the protein-nanodisc complexes can be processed with standard single-particle methods to yield low-resolution structures as a basis for higher resolution cryoEM work. Furthermore, the nanodiscs provide samples suitable for either TEM or non-denaturing gel electrophoresis. To illustrate the method, Ca2+-induced binding of 5-lipoxygenase on nanodiscs is presented.
Introduction
In medical research, much attention is focused on membrane proteins, either intrinsic or extrinsic, involved in a variety of lipid interactions. Working with lipid-interacting proteins includes either selecting a substitute to the lipids, such as detergents, amphipols1, or small proteins2, or finding a membrane substitute that keeps the protein soluble and active. Lipoic membrane substitutes include liposomes and nanodiscs (ND)3,4.
Nanodiscs are near-native membrane platforms developed by engineering the protein part, ApoA-1, of the high-density lipoprotein (HDL) naturally occurring in blood. ApoA-1 is a 243 residue-long chain of short amphipathic α-helices and has a lipid-free soluble conformation. In vitro when in the presence of lipids, two copies of the protein ApoA-1 spontaneously rearrange to encircle the hydrophobic acyl chain portion of a lipid bilayer patch5. Engineered versions of ApoA-1 are generally called membrane scaffolding proteins (MSP), and an increasing number are commercially available as plasmids or as purified proteins. Repetitions or deletions of the α-helices in ApoA-1 result in longer6 or shorter7 membrane scaffolding proteins. This in turn makes it possible to form discs around 6 nm7 to 17 nm8 in diameter. There are different types of applications for the nanodiscs3,9. The most commonly used application is to provide a near-native membrane environment for the stabilization of an integral membrane protein8, reviewed previously3,9. A less-explored use is to provide a nanoscale membrane surface for the study of peripheral membrane proteins10,11,12,13,14,15,16,17. Section 1 of the protocol below visualizes the procedure for making nanodiscs composed of phospholipids and membrane scaffolding protein.
Sample preparation is a bottleneck in most methods. Method-specific samples may add particular information, but they also make comparisons of results difficult. Therefore, it is simpler when samples are multimodal and can be used directly in several different methods. One advantage with the use of nanodiscs is the small size of the nanodisc in comparison to liposomes (e.g., the samples can be directly used for both TEM and non-denaturing gel electrophoresis, as in the present protocol).
Vesicles and liposomes have long been used to understand the function of membrane-interacting proteins. For structural studies and visualization, an example of the structural determination of a transmembrane protein in liposomes is available18. However, no high-resolution 3D structure of a monotopic membrane protein embedded on a liposome membrane has been published yet, as far as we know. Gold nanoparticles or antibodies can be used to visualize proteins binding to liposomes or vesicles using TEM19. Even though these probes are very specific, they might interfere with membrane-binding proteins by veiling the membrane binding site or by masking areas of interest with the flexible parts. Gold-labeled or antibody-complexed proteins could probably be analyzed on a gel, but this would increase the cost of the experiment.
Though liposomes are an excellent platform, one cannot be certain that the population has a particular ratio of protein per liposome, a feature that can be explored by the use of nanodiscs20. In a liposome, cofactors and substrates can be trapped in the soluble interior. Substances that are membrane-soluble will share the same fate for both types of membrane mimetics. Nevertheless, as the bilayer area is smaller in nanodiscs, a smaller amount of substance is required to saturate the nanodisc membranes.
Understanding protein function through the determination of the atomic structure has been essential for many fields of research. Methods for protein structure determination include X-ray21; nuclear magnetic resonance (NMR)22,23; and transmission electron microscopy (TEM)24 at cryogenic temperatures, cryoEM. The resolution by cryoEM has lately been greatly improved, mainly due to the use of direct electron detectors25,26. The macromolecules are imaged in thin, vitreous ice27 in a near-native state. However, due to the low contrast of biological molecules, they become hard to detect in the size range of 100 – 200 kDa. For suitably sized samples, data collection can be made and the method of single particle reconstruction can be applied to obtain a structure28.
However, the determination of protein structure by TEM is a multistep process. It usually starts with the evaluation of sample monodispersity by negative-stain TEM29 using salts of heavy metals like phosphotungsten (PT)30 or uranium31. Reconstruction of a low-resolution model of the negatively stained macromolecule is usually made and may yield important information on the molecular structure29. In parallel, data collection using cryoEM may start. Care should be taken when evaluating negative-stain TEM data to avoid the misinterpretation of artefact formation. One particular artefact is the effect of the PT stain on phospholipids and liposomes32, resulting in the formation of long rods resembling stacks of coins viewed from the side33. Such "rouleau" or "stacks" (hereafter denoted as "stacks") were observed early on for HDL34, and later also for nanodiscs35.
The stacking and reshaping of membranes may occur for many reasons. For example, it can be induced by co-factors like copper, shown by TEM imaging in an ammonium molybdate stain36. A fraction of the membrane lipids in liposomes contained an iminodiacetic acid head group mimicking metal complexation by EDTA, thus stacking liposomes after the addition of copper ions36. Stacking could also be due to a protein-protein interaction by a protein in or on the lipid bilayers (the stain used is not mentioned)37. The stack formation of phospholipids by PT was observed early on; however, later work has focused on removing or abolishing this artifact formation38.
Here, we propose a method to take advantage of the NaPT-induced nanodisc stacking for the study of membrane-binding proteins by TEM. In short, protein binding on the nanodiscs would prevent the nanodiscs from stacking. Though the reasons for the stacking are not clear, it was proposed39 that there is an electrostatic interaction between the phospholipids and the phosphoryl group of PT, causing the discs to stick to each other (Figure 1A). The hypothesis behind our protocol is that when a protein binds to a nanodisc, most of the phospholipid surface is not available for the interaction with the PT due to steric hindrance by the protein. This would prevent stack formation (Figure 1B). Two conclusions can be drawn. First, the prevention of stacking means that the protein of interest has bound to the membrane. Secondly, the protein-ND complex can be treated with standard single-particle processing methods24,40 to get a rough morphology of the complex. Furthermore, analyses by methods like non-denaturing gel electrophoresis or dynamic light scattering can be performed.
To demonstrate this hypothesis, we used the membrane-binding protein 5-lipoxygenase (5LO), which is involved in many inflammatory diseases41,42. This 78-kDa protein requires calcium ions to bind to its membrane43. Though this membrane association has been studied extensively using liposomes44,45,46 and membrane fractions47, these cannot be used for TEM analysis and structure determination.
The preparation of nanodiscs starts by mixing MSP with lipid resuspended in the detergent sodium cholate. After incubation on ice for 1 h, the detergent is slowly removed from the reconstitution mixture using an adsorbent resin. This kind of material is frequently made of polystyrene shaped into small beads. They are relatively hydrophobic and have a strong preference for binding detergent compared to lipids48. After removing the hydrophobic beads and performing clarification using centrifugation, the nanodiscs are purified by size exclusion chromatography (SEC). The purified nanodiscs are mixed with a monotopic membrane protein (and possible cofactors) in an equimolar ratio (or several ratios for a titration) and are left to react (15 min). Analysis by TEM is carried out by applying µL-amounts of sample onto glow-discharged, carbon-coated grids and then by performing negative staining with NaPT. The same sample from when the aliquots were applied to the TEM grids can be used for analysis by non-denaturing or SDS PAGE gel-electrophoresis, as well as by various kinds of activity measurements, with no major changes.
Protocol
1. Preparation of Nanodiscs
- Expression and purification of the membrane scaffolding protein8,35
- Express the His-tagged MSP1E3D1 in the E. coli BL21 (DE3) T1R pRARE2 strain in flasks. Prepare a 50-mL overnight starter culture with LB medium supplemented with 50 µg/mL Kanamycin at 37 °C. Dilute the overnight starter culture in 2 L of terrific broth medium supplemented with 50 µg/mL kanamycin.
- Grow the cells at 37 °C until the optical density at 600 nm (OD600) reaches approximately 3. Induce the protein expression with 0.5 mM isopropyl β-D-1-thiogalactopyranoside (IPTG) for 3 h at 18 °C.
- Prepare the lysis buffer (100 mM Tris-HCl, pH 8.0; 100 mM NaCl; and 10% glycerol). Add 1 mM TCEP immediately before use.
- After 3 h of induction at 18 °C, harvest the cells by centrifugation for 10 min at 4,500 x g and 4 °C. Discard the supernatant, weigh the harvested cells, and re-suspend them in lysis buffer at a ratio of 2 mL of lysis buffer per g of cells.
- Lyse the cells by pulsed sonication (repetition rate: 4 s ON, 4 s OFF) for 3 min at 80% amplitude.
- Centrifuge the lysates for 20 min at 49,000 x g and 4 °C. Discard the pellet from this centrifugation.
- Inject the supernatant into a 5-mL Ni+ chelating column connected to an automated liquid chromatography system preferably kept at 4 °C.
- Before the elution step (step 1.1.9), wash the column with buffers 1 – 3 below to remove all proteins except the His-tagged MSP. Monitor the protein content at 280 nm to follow the purification process; the UV recording at 280 nm should go back to baseline after each wash.
- Wash with wash buffer 1: 40 mM Tris-HCl, 300 mM NaCl, 10 mM imidazole, and 1% Triton, pH 8.0.
- Wash with wash buffer 2: 40 mM Tris-HCl, 300 mM NaCl, 20 mM imidazole, and 50 mM sodium cholate, pH 8.0.
- Wash with wash buffer 3: 40 mM Tris-HCl, 300 mM NaCl, and 50 mM imidazole, pH 8.0.
- Elute the MSP with 40 mM Tris-HCl, 300 mM NaCl, and 500 mM imidazole, pH 8.0.
- Change the buffer to MSP standard buffer (20 mM Tris-HCl, pH 7.5; 100 mM NaCl; and 0.5 mM EDTA) by gel filtration8,35; the yield per batch should be around 7 mg L-1.
- Preparation of phospholipid stock solution
- Add 305 µL of 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine (POPC) dissolved in chloroform at a concentration of 25 mg/mL to a glass beaker and evaporate the chloroform by purging it with a gentle stream of nitrogen. Dry the lipid overnight in a vacuum desiccator. NOTE: This step is performed to remove the solvent from the lipid, which leaves a thin film of lipid at the bottom of the glass beaker.
- Resuspend the lipid cake in 200 µL of MSP standard buffer containing 100 mM sodium cholate by vortexing the tube until the solution becomes transparent; this will provide a solution with a POPC concentration of 50 mM.
NOTE: In general, the molar ratio of lipid to detergent at this point should be 1:2 (lipid:detergent)8. This lipid-detergent mix can be stored at -80 °C for almost 2 months.
- Preparation of hydrophobic beads for detergent removal
- Place 5 g of beads (dry-weight) in a 50-mL tube.
- Wash the beads with 30 mL of 100% methanol and then with 40 mL of ultrapure water.
- Wash the beads with 10 mL of MSP standard buffer. Finally, store the beads with 15 mL of MSP standard buffer at 4 °C.
- Nanodisc reconstitution
- Dispense 190 µL of MSP1E3D1 (0.124 mM) into a microfuge tube and add 61.5 µL of phospholipid stock solution (50 mM POPC) to the same tube. Incubate the reconstitution mixture on wet ice for 1 h. NOTE: This equals a molar ratio of 1:130 (MSP1E3D1: POPC).
- Add hydrophobic beads at a concentration of 0.5 g of beads per mL of reconstitution mixture to initiate the self-assembly process. Incubate in a rotary incubator for 16 h at 4 °C.
- After incubation, centrifuge for 10 min at 13,000 x g and 4 °C to remove the precipitates and aggregates. Discard the pellet and save the supernatant.
- Equilibrate the size exclusion chromatography column (mounted on an automated liquid chromatography system) with MSP standard buffer until the absorbance at 280 nm is stable. Inject the supernatant into the column and collect the peak fractions.
- Measure the concentrations of nanodiscs in the peak fractions at 280 nm. Use the molar extinction coefficient of MSP1E3D1 (ε = 29,910 cm-1 M-1) for the calculation of the nanodisc concentration. NOTE: The number of lipids per nanodisc can be measured by a combination of radiolabeled lipids and phosphate analysis4 or by phosphate analysis alone.
2. Preparation of the Monotopic Protein 5-Lipoxygenase35
- Prepare an overnight starter culture. Supplement 50 mL of LB medium with 100 µg/mL ampicillin. Inoculate the medium with E. coli BL21 (DE3) containing the plasmid of the 5-lipoxygenase gene (ALOX5). Dilute the overnight starter culture in expression medium containing 42 mM Na2HPO4, 24 mM KH2PO4, 9 mM NaCl, 19 mM NH4Cl, 1 mM MgSO4, 0.1 mM CaCl2, 0.2% D-glucose, 0.1%, 5 µM FeSO4, and 100 µg/mL ampicillin. Grow the cells until the OD600 is ~ 0.5 at 25 °C. Induce protein expression with 0.2 mM IPTG for 16 h at 20 °C35.
- Harvest by centrifugation for 10 min at 7,000 x g and 4 °C and discard the supernatant. Weigh the pellet found at the bottom of the tube containing the harvested cells and resuspend the harvested cells in lysis buffer (100 mM Tris-HCl, pH 8.0; 100 mM NaCl; 10% glycerol; and 1 mM TCEP) containing protease inhibitor and 0.5 mg/mL35 lysozyme at a ratio of 2 mL of lysis buffer per g of cells.
- Lyse the cells by sonication for 5 x 15 s at 80% amplitude. Remove the cell debris by centrifugation for 10 min at 7,000 x g and 4 °C. Perform ammonium sulfate precipitation to 30 – 60% saturation to precipitate the proteins in the solution35. Centrifuge for 15 min at 16,000 x g and 4 °C. NOTE: The 5LO pellet can be rapidly frozen and stored at -80 °C for up to 6 months.
- Resuspend the pellet with 20 mL of lysis buffer and centrifuge for 15 min at 40,000 x g and 4 °C.
- Incubate the supernatant on an ATP agarose column at 4 °C for 30 min. Wash the column once with one column-volume of lysis buffer containing 0.5 M NaCl. Elute the 5LO with 20 mM ATP in lysis buffer containing 10 µM FeSO4 and 20 µg/mL catalase. Perform gel filtration chromatography to remove the ATP35.
NOTE: The 5LO is unstable and should be used immediately after purification. Otherwise, it is recommended to halt at the step of ammonium sulphate precipitation (see the note after step 2.1.3).
3. Preparation of the Nanodisc-protein Complex
- Prepare a total volume of 100 µL of complex. Mix 0.8 µM ND and 0.8 µM 5LO with the 1 mM Ca2+ present in the MSP standard buffer (20 mM Tris-HCl, pH 7.5; 100 mM NaCl; 0.5 mM EDTA; and 1.5 mM CaCl2) and incubate for 10 min on ice. NOTE: The sample can be stored for up to one month at 4 °C35.
4. Analysis of the Samples
- Gel electrophoresis
- Non-denaturing electrophoresis
- Mix 15 µL of the sample with 5 µL of loading buffer (50 mM BisTris, 6 N HCl, 50 mM NaCl, 10% (w/v) glycerol, and 0.001% Ponceau S, pH 7.2)49 and load it on a 4 – 16% Bis-Tris gel to perform electrophoresis.
- Fill the cathode tank with light cathode buffer (50 mM Bis Tris; 50 mM Tricine, pH 6.8; and 0.002% Coomassie G-250) and the anode tank with running buffer (50 mM Bis Tris and 50 mM Tricine, pH 6.8). Start the separation by using a constant voltage at 150 V.
- Stop the electrophoretic separation when the Coomassie front reaches the end of the gel.
- Stain the gel with a standard Coomassie blue protocol.
- Denaturing electrophoresis
- Mix 40 µL of the sample with 10 µL of loading buffer containing SDS (0.05% (w/v) Bromophenol blue; 0.2 M Tris-HCl, pH 6.8; 20% (v/v) glycerol; 10% (w/v) SDS; and 10 mM 2-mercaptoethanol) and load it on a 4 – 20% Tris glycine gel to perform electrophoresis.
- Fill the cathode and anode tanks with running buffer (25 mM Tris-HCl, pH 6.8; 200 mM glycine; and 0.1% (w/v) SDS). Start the separation by using a constant voltage at 150 V.
- Stop the electrophoretic separation when the loading dye front reaches the end of the gel.
- Stain the gel with a standard Coomassie blue protocol.
- Non-denaturing electrophoresis
- Preparation of the NaPT solution
- Dissolve 1 g of phosphotungstate sodium salt in 50 mL of water by agitation at room temperature to give a 2% (w/v) acidic solution.
- Adjust the pH to 7.4 with 1 M NaOH. Remove the particles using a 0.22-µm syringe filter. Store the solution at room temperature or at 4 °C.
- Preparation of the sample for TEM analysis
- Glow-discharge carbon-coated copper grids (400 mesh) for 20 s at 30 mA to render the grids hydrophilic before the adsorption of the sample. Place 3.5 µL of the sample (0.8 µM with respect to the nanodiscs) on the grid and incubate for 30 s. NOTE: The volume applied may vary between 2.5 and 5 µL, depending on the sample concentration. A suitable concentration range for nanodiscs is 0.5 – 1 µM.
- Blot off surplus solution using filter paper.
- Immediately stain the grid with a drop of 2% NaPT for 30 s. Blot off excess solution and leave the grid to air dry.
- Evaluate the grids by TEM. A microscope with 120-200 keV accelerating voltage suffices to estimate the extent of stacking. NOTE: For the present protocol, a calibrated transmission electron microscope was used, equipped with a 200-keV field emission gun.
- Record the TEM images. For the images showing long stacks, do not process further; images show the complex of nanodiscs, with extrinsic protein that may contain a few short stacks, but most particles are in the complex. Record several images and process these according to standard methods to obtain class-averages and a low-resolution, 3 dimensional model of the complex.
NOTE: For the collection of negative-stain data, grids were made on at least three different days. For each day, fresh sample incubations (as in step 3.1) were made before the negative stain was applied.
Representative Results
The method we propose depends upon the preparation of nanodiscs to provide the membrane surface for monotopic membrane-protein binding. As there is no transmembrane protein embedded into the nanodisc lipid bilayer, the nanodiscs are here denoted as "empty nanodiscs" (Figure 2A). These have a calculated molecular weight of 256 kDa for a composition of two MSP1E3D1 scaffolding proteins and around 260 molecules of POPC8. Using this protein:lipid ratio for reconstitution, one major peak (Figure 2A) was eluted during gel filtration. The peak fractions around 9 mL (Figure 2A) were examined by blue native gel electrophoresis49, showing one band (Figure 2B, lane 1). Although the band corresponds to a size somewhat larger than the calculated 256 kDa, the nanodisc diameter measured in the TEM images corresponds to the expected 13 nm ± 0.5 nm.
The monotopic membrane protein 5-lipoxygenase (5LO) was purified as described above (Protocol step 2) and in more detail earlier35. The homogeneity of the 78-kDa 5LO was analyzed by SDS PAGE electrophoresis (Figure 2C, lane 1) and was fully functional35 (not shown).
In this protocol, conditions are intentionally created that promote the stacking of the nanodiscs. However, it should be noted that this stacking is induced only in the samples made for TEM imaging (so-called specimens), specifically by the addition of the negative stain sodium phosphotungstate as the last step after all other treatments and additions had been made. In fact, the images of NaPT-stained nanodiscs show the expected stacking (Figure 3A) very early on for HDL34 and other phospholipid entities5,50. The "long stacks of coins viewed from the side" (Figure 3A) are the nanodiscs aggregated with phosphotungstate intercalated between the bilayers, as described schematically in Figure 1A and proposed by Zhang et al.32,39.
It is important to check whether various additions in the buffers affect, prevent, or even induce stacking, and as will become evident below, the effects of calcium ions required investigation. In Figure 3B, the stacking is still present in a specimen where calcium ions were added to the nanodisc solution before staining with NaPT.
The essential step in the protocol is shown in Figure 4, where the membrane-binding protein is present in the solutions. The stacking could not be induced by the NaPT stain when 5LO was bound to the membrane surface of the nanodiscs (Figure 4A). For this specimen, the molar ratio of 5LO to nanodiscs was 1:1, as schematically shown in Figure 1 (Figure 1B, bottom right). Moreover, as the lipid bilayer of the nanodiscs is accessible from both sides, in contrast to liposomes, specimens with the ratio of two 5LO to one nanodisc were prepared, while the TEM images show even less stacking (Figure 4B).
For binding, some extrinsic membrane proteins could depend on the presence of a specific lipid in the membrane, such as phosphoinositols10,11,12 or phosphatidylserine10. In the case of 5LO, the presence of Ca2+ is necessary43. In Figure 4A and B, calcium ions were present in the solutions with nanodiscs and 5LO. In other words, the lack of stacking not only shows that the monotopic protein has bound but also that the binding depends on Ca2+ ions. In order to show this, a solution containing nanodiscs and 5LO but without calcium was stained with NaPT and imaged by TEM (Figure 4C). As expected, 5LO cannot bind to the nanodiscs without Ca2+, leaving the 5LO free in the solution, so the nanodiscs stack instead (Figure 4C).
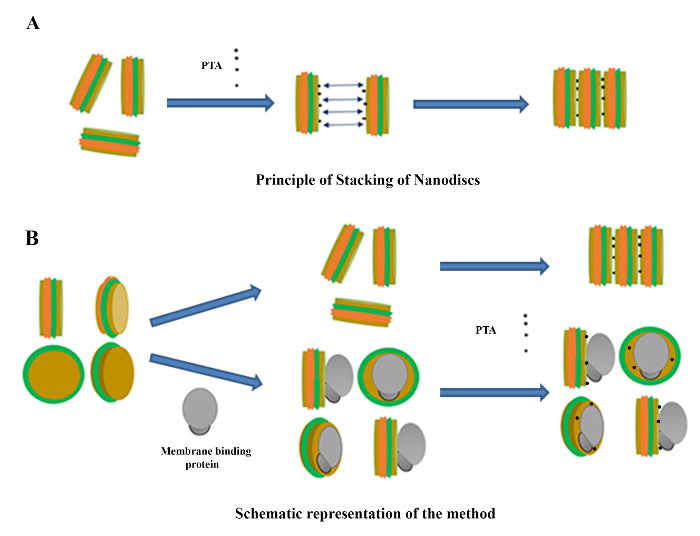
Figure 1. Principle of Nanodisc Stacking and Overview of the Method. A. Principle of nanodisc stacking. The addition of the negative stain sodium phosphotungstate to a solution of nanodiscs (left) leads to the stacking of the nanodiscs (right) due to electrostatic forces (middle, arrows) between the phospholipid bilayer and the PT molecules (middle, black dots). B. Schematic representation of the protocol. Empty nanodiscs (left) bind to an extrinsic membrane protein (gray) to form a complex, depicted from four different angles (lower middle). The presence of the monotopic protein sterically prevents the nanodiscs from stacking when stained with the phosphotungstate salt (lower right), in contrast to the stacking induced in the absence of any large objects (upper right). The green- and orange-colored rings represent the two MSP wrapping around the phospholipid core, which is represented in light brown. Please click here to view a larger version of this figure.
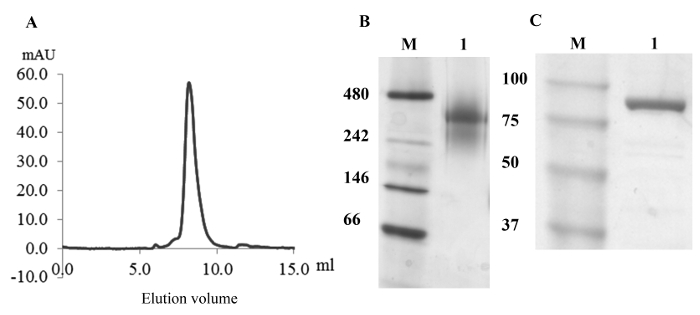
Figure 2. Homogeneity Analysis of Nanodiscs and 5LO. A. Size exclusion chromatography of reconstituted empty nanodiscs. The elution of the nanodiscs peaks at 9 mL. B. Non-denaturing gel electrophoretic analysis of the peak fraction from the SEC shown in (A). Lane M contains the marker. A single band is present in lane 1, where the peak fraction at 9 mL was loaded. C. Denaturing gel electrophoretic analysis of purified 5LO. The 78 kDa 5LO shows as an intense band in lane 1. Please click here to view a larger version of this figure.

Figure 3. Stack Formation of Nanodiscs in the Absence of Protein Analyzed by TEM. A. Nanodisc stacking was induced when stained with NaPT. The "stacks of coins" are the stacks of nanodiscs when viewed from the side. Each nanodisc appears as a white band. B. Stacking was not disturbed by the presence of calcium ions in the nanodisc solution when stained with NaPT. The scale bars are 100 nm. Please click here to view a larger version of this figure.
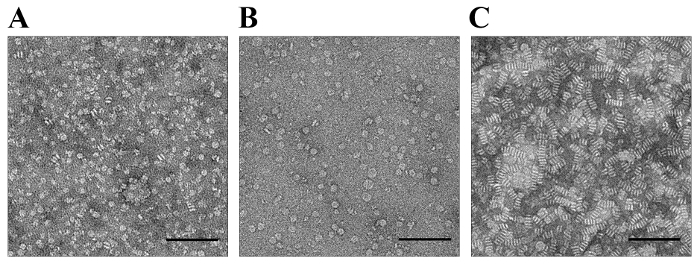
Figure 4. Preventing Stack Formation by the Binding of Protein in the Presence of a Cofactor Analyzed by TEM. A. and B. Calcium ions were present in mixtures of 5LO and nanodiscs at molar ratios of 1:1 (A) or 2:1 (B). The more 5LO binding to the nanodiscs, the fewer nanodiscs are available for stacking, as shown in (B). The single-particle complexes in both (A) and (B) are suitable for further processing35. C. In the absence of calcium ions, 5LO cannot bind. NaPT staining of this sample shows typical stacking of nanodiscs, leaving the 5LO unbound. The scale bars are 100 nm. The images were acquired by a 4K x 4K CCD camera at a magnification of 69,500x. The pixel size of the CCD camera is 15 µm, which gives a corresponding value of 2.16 Å on the specimen level for the EM images. Please click here to view a larger version of this figure.
Discussion
The method can be separated into three parts: the reconstitution of empty nanodiscs, the preparation of protein-nanodisc complexes, and the negative staining for the TEM of these complexes. Each part will be addressed separately regarding limitations of the technique, critical steps, and useful modifications.
Reconstitution of empty nanodiscs. Critical steps and limitations in the production and use of nanodiscs.
For the preparation of the empty nanodiscs, it is essential to optimize the MSP-to-lipid ratio. For the most common MSP and lipids (using cholate as a detergent), suggestions for the optimal ratios can be found in the literature (e.g., 125 – 130 POPC per MSP1E3D18,51 used in the present protocol). Differences obtained compared to the published ratios may depend on the method for protein concentration determination, the efficiency of lipid solvation in the detergent, and the method of detergent removal from the solution.
We propose to perform detergent removal using hydrophobic beads48 (step 1.3), while possible alternatives are dialysis or the addition of cyclodextrin52,53. Initially, dialysis was used for the reconstitution of HDL5,34 and is still preferred54. Regardless of the method for detergent removal, speed is essential. If the removal is too fast (the volume of hydrophobic beads is too large), the spontaneous rearrangement of MSP with the lipids may not have time to occur. Instead, large amounts of liposomes and protein aggregates may form. The results from size exclusion chromatography would show large amounts of liposomes in the void volume, a suboptimal peak of reconstituted nanodiscs, and a larger peak of unused MSP4,6,51. For too-small a volume of hydrophobic beads, the detergent removal may be incomplete, and the nanodiscs that form may show a variation in sizes, where most are smaller than expected. One could test two additions of smaller volumes of hydrophobic beads instead of one large addition to slow down the speed of reconstitution.
The choice of lipids should be carefully considered8,9,45,46,55, depending on the purpose for the nanodisc preparation. In general, for all methods aimed at the formation of membranes (reconstitution of nanodiscs or liposomes, as well as for 2D crystallization experiments), the lipids must be in a "fluid" state56. Below a certain temperature, the lipid is rigid. Above this so-called transition temperature (Tm), the lipid is fluid. Note that some commonly-used saturated lipids have a Tm well above room temperature. Furthermore, for lipid mixtures, the miscibility of lipids muse be considered57. The optimal temperature for Nanodisc formation is around the phase transition (Tm) of the lipid.
The detergent in the present protocol is sodium cholate8. If another detergent must be used, the same concentrations as those used for cholate cannot be directly used. Certain mild detergents dissolve lipids less efficiently, and a higher concentration may be needed. The lipid cake obtained after the evaporation of the chloroform (step 1.2.2 in the protocol) must be dissolved into a clear solution by the detergent. Vortexing, freeze-thawing cycles, ultrasound baths, or sonication could be used to accelerate the solubilization.
For combinations of lipid (-mixtures), MSP, and detergents other than those used here, titrations to minimize large aggregates and liposomes or excess MSP are necessary4,6,51,58. For some combinations of MSP and phospholipids, the optimization may be more difficult than for others4,6,51, and it becomes essential to determine the content of the peak fractions. The most precise method for the analysis of nanodisc composition and size is to determine the number of phospholipids per MSP by phosphate analysis4 and calculate the nanodisc molecular weight. Nanodisc size can be estimated by well-resolved, non-denaturing gel electrophoresis; dynamic light scattering (DLS)59; or negative-stain TEM, to name only a few methods. As for the negative-stain-induced stacking protocol presented here, the measurement of the width of the stacks present in the TEM images yields the diameter of the nanodiscs.
As for the expression and purification of the membrane scaffolding protein, all usual precautions and concerns for a soluble protein should be taken. The plasmids available may contain different tags and protease cleavage sites in case the tag must be removed before using the MSP.
5LO, the monotopic protein used in the protocol, inactivates rapidly unless precautions are taken, described elsewhere35 and in references therein. In short, the catalytic iron is easily lost, and oxidizing conditions may cause dimerization of the 5LO. As stated in the note (protocol step 2.1.3), the purification may be halted after the precipitation step and aliquots stored until later use. After the purification of an aliquot, the protein must be used within a few hours due to the rapid inactivation35.
Critical steps in the preparation of the protein-nanodisc complexes.
The size of the monotopic protein versus the nanodisc area is of importance. The length of the MSP1E3D1 protein chain corresponds to a nanodisc with a 10.5-nm inner diameter and about a 8,900-Å2 bilayer area8. This corresponds well to the area reported for the membrane-embedded part of 5LO45. Thus, our expected binding ratio was 1:1, or at most 2:1 5LO per nanodisc. This proved to be correct, as reported in a titration experiment35. However, the maximal binding of two 5LO per nanodisc was not obtained, as discussed elsewhere35. A somewhat larger nanodisc could possibly allow for a full 2:1 binding for experiments where this is desirable, but in this case, the 1:1 ratio is the main aim.
The choice of optimal lipids for protein-nanodisc complex formation may be aided by previous knowledge. This was the case in the present protocol, where the choice of lipid was synthetic POPC35. Both variations in the lipid head group46 and variations in the saturation of the acyl chains45 were tested in liposome-5LO binding studies. 5LO activity was increased by binding on liposomes containing choline phospholipids unsaturated in the sn-2 acyl chain position45,46. If specific lipid requirements are not known, but the identity of the original membrane is known, natural lipid extracts could be tested first55.
A cofactor dependence was known for 5LO, which depends on Ca2+ ions for membrane binding43. It is challenging to completely remove calcium from solutions. Here, the complexing agent EDTA is present in the buffers in amounts adjusted to leave a controlled amount of free Ca2+ ions. Calculations of free and bound molecules in the buffers were made by the BAD60 software for buffers containing more than one type of complexing agent35. The effect on concentration of a cofactor partly soluble in the membrane is not accounted for by this software.
Critical steps in the negative staining of nanodiscs to promote stack formation.
For the protocol at hand, where maximal stacking is desired, the stain should be the sodium salt of phosphotungsten. Different salts of tungsten are available, and we observed that phosphotungstic acid was less efficient at stacking (not shown). For unclear reasons, it seems that high amounts of sodium during PT staining promotes stacking. Also, for the sample buffer, it was shown that the lower the sodium content, the lower the amount of induced stacking32. For the same reason, after the sample has been applied to the EM grid, water washing before staining should not be performed, as this also reduces stacking. On the other hand, some stacking was always induced, although never to the extent needed for the best performance of the protocol.
Of course, cofactors like the Ca2+ ions here must be checked for interference with the negative stain or with the nanodiscs. For the present protocol, calcium ions would have precipitated a phosphate buffer, so a Tris buffer was used. The nanodiscs were not stacked by Ca2+ ions per se, as checked using another negative stain, uranyl formate35, nor did calcium prevent NaPT-induced stacking (Figure 3B).
For a small monotopic protein, the extra size added by binding to a nanodisc may render it detectable in cryoEM. A very large nanodisc compared to the protein may not be optimal, as a small protein on a large nanodisc may not prevent stacking. However, the shape of the extrinsic protein could have an influence. Although no precise ratio can be provided as of now, a nanodisc bilayer surface only slightly larger than the estimated binding surface of the protein is recommended. With this restriction, the maximal protein binding would be one protein on each side of the nanodisc.
General limitations and advantages of the method.
The protocol depends upon the availability of a TEM and the related equipment, which are not available in all laboratories. However, a 120- to 200-kV electron microscope equipped with a traditional CCD camera would be quite sufficient for the detection of stacking. The NaPT negative staining is performed at room temperature, and the grids can be stored at this temperature for later viewing.
TEM imaging would quickly show whether the stacking has been prevented or not. For the images showing protein-nanodisc complexes (e.g., Figure 4A and B), further processing to obtain structural information could be performed. Structural details of a negatively-stained sample can reach 1 nm and can provide useful structural information29. In laboratories dedicated to structure determination by cryoEM, the negative staining of samples is standard procedure. The use of our protocol would be a quick and simple addition to regular procedures.
Some monotopic proteins are anchored to the membrane by myristoylation, palmitoylation, or hydrophobic patches on the protein surface, making it necessary to form the nanodisc-protein complex by the same procedure as for the incorporation of a transmembrane protein into nanodiscs13. Here, one of the subunits in the protein complex contains a myristoylation on a flexible N-terminal tail for membrane binding. For uranyl acetate-stained TEM images, it was stated that the protein complex was bound perpendicularly to the membrane, and not on the side of the nanodisc. This and other results confirmed a rigid structure for the membrane attachment13. For this binding, NaPT staining according to our proposed protocol would show the same evenly spread single particles. However, if the attachment had been via a flexible chain, the uranyl acetate staining might not have given a clear result. We speculate that, for flexible chain membrane-attached protein complexes, the NaPT staining may induce the nanodiscs to stack, demonstrating the typical "stack of coins," but now decorated by the attached protein on the sides.
Why use nanodiscs if liposomes can be used? The optimization of lipids, cofactors, or other parameters could very well be tested in liposomes for the binding of peripheral proteins (cf. 5LO). NaPT-induced stacking of liposomes has been demonstrated32, so a monotopic protein could prevent liposome stacking. However, the sizes of liposomes in comparison to monotopic proteins and to nanodiscs also comes into play. A large extrinsic protein may be able to prevent the stacking of liposomes, whereas a small protein, like the 78-kDa 5LO, might not unless present in high numbers per liposome. Small liposomes can be prepared, yet the smaller the vesicle, the stronger the curvature of its membrane. The nanodisc bilayer is flat. Testing various parameters using nanodiscs could be less cost-effective due to the MSP. On the other hand, for liposomes, certain expensive cofactors or substrates that partially partition into the membrane and further, into the interior of the liposomes, may be required in larger amounts. In conclusion, the nanodisc platform provides the possibility of choosing a defined area for binding a precise number of proteins. The bilayer area is accessible from two sides and is flat.
Significance of the method.
Determining the 3D structure of proteins or complex macromolecules visualizes the inner workings of cells. In this crowded environment, specific proteins are active on membrane surfaces, alone or in complex with other proteins, but usually so that the physical interaction with components in the lipid bilayer is part of their mechanistic function. For some proteins, the conformation in solution is changed upon membrane embedding, whereas others are tethered to the membrane but nevertheless change conformation or orientation on the membrane due to some kind of trigger. A hydrophobic substrate entry path may then open, or a conformation that allows binding to a transmembrane protein may occur. To obtain the structure, or at least structural information, of such peripheral proteins bound to the membrane is challenging.
Nanodiscs were used in some studies regarding orientation and function on membranes10,11,12,13,14,15,16,17. Cytochrome P450, central in detoxification, is anchored to the membrane by an α-helix and contains a heme group in its globular part, allowing for linear dichroism measurements15. The results indicated an orientation of the heme with respect to the membrane in close agreement with simulations made, thereby strengthening other conclusions drawn from simulations of the membrane-embedded cytochrome P450. For a GTPase anchored to the membrane in a small nanodisc, NMR studies could identify two different areas on the protein with a membrane interface each and further, how nucleotide binding induced a switch from one interface to the other17. Mutations and effector binding, as well as GTP binding, were studied in another GTPase in the Ras-superfamily to find a number of effects on activity that depended on the orientation on the membrane, which would affect oncogenic signaling16. 5LO has a soluble conformation61 and requires calcium for binding on nuclear membranes43. Using giant unilamellar liposomes, polarized, attenuated, total-reflection infrared experiments were performed46. The result implied that the N-terminal β-barrel binds to membranes, with the symmetry axis of the β-barrel tilted approximately 45 degrees from the membrane normal. The 5LO C-terminal domain is a nearly cylindrical α-helical bundle, and this was proposed to bind with the long axis parallel to the membrane45,46. In our earlier work on 5LO embedded on nanodiscs, the area of the bilayer was chosen to allow one 5LO per side with the above-suggested orientation35. The TEM images confirmed this orientation, albeit at low resolution35. If, for example, only the N-terminal β-barrel was bound but not the C-terminal domain, a higher number of 5LO per nanodisc would have been obtained. Furthermore, another protein could change the membrane binding and orientation of 5LO, such as an activator, the transmembrane protein 5-lipoxygenase activating protein, FLAP. FLAP has been reconstituted into nanodiscs, and we use TEM and other methods to study the interaction between 5LO and FLAP. Their interaction results in endogenous arachidonic acid being converted to leukotriene A4, which is the first step in the synthesis of potent chemoattractants involved in many inflammatory diseases41,42,62.
The 5LO-nanodisc complex is calculated to have a molecular weight of 334 kDa, near the limit of around 200 kDa for structural investigation by cryoEM. Nonetheless, as the atomic structure of 5LO is available61, its docking into a medium-resolution, 3-dimensional electron density map would yield information on the interaction with the nanodisc membrane.
Innovations in cryoEM technology are not only pushing resolution to the atomic level63 but also pushing the size limit to smaller proteins. For structural determination by NMR22, the situation is the opposite, as the aim is to increase the size range, which is now approaching 200 kDa. Recently, the membrane binding of small GTPase proteins were investigated by NMR16,17. These proteins, of only around 20 kDa, were tethered to small nanodiscs of a 7.6-nm inner diameter, forming complexes of about 130 kDa. In the future, similarly sized complexes could be investigated by both NMR and cryoEM to elucidate different aspects of protein membrane binding.
An extension of the protocol could be to investigate nanodiscs containing a transmembrane protein (TMP) in the nanodisc bilayer. NaPT-induced stacking of the TMP-nanodiscs could be compared to empty nanodiscs containing the same lipid composition. Large extramembranous loops would prevent stacking, while short loops may not prevent stacking64. This could be turned into an advantage, as the protocol could be used to investigate proteins specifically binding to the transmembrane protein.
The advantage of this method is that the membrane interaction can be visualized immediately. The relative simplicity of making large amounts of empty nanodiscs implies that one can extend this method to screen for different conditions like pH, temperature, buffers, and cofactor dependence of monotopic protein binding. In fact, whereas the nanodisc platform9 was developed for the stabilization of transmembrane proteins and is increasingly used for this purpose, its potential for studies of the large numbers of more-or-less transiently-binding extrinsic proteins present inside cells should clearly be kept in mind.
Divulgations
The authors have nothing to disclose.
Acknowledgements
The authors thank the Swedish Research Council, Stockholm County Council, and KI funds for their support. The expression and purification of MSP was performed at the Karolinska Institutet/SciLifeLab Protein Science Core Facility (http://PSF.ki.se). The authors would also like to thank Dr. Pasi Purhonen and Dr. Mathilda Sjöberg for sharing their technical expertise and for their timely assistance.
Materials
| Transmission electron microscope: JEOL2100F | JEOL | ||
| CCD camera | Tiez Video and Imaging Processing System GmbH, Germany | ||
| Glow discharger | Baltec | ||
| TEM grid: 400 mesh | TAAB | GM016/C | |
| Size exclusion chromatography: Agilent SEC-5 | Agilent Technologies | 5190-2526 | |
| Superdex 200 HR 10/300 | GE Healthcare Life Sciences | 17-5172-01 | |
| Plasmid:MSP1E3D1 | Addgene | 20066 | |
| Bacteria: BL21DE3 | NEB | C2527H | |
| Bacteria: BL21 (DE3) T1R pRARE2 | Protein Science Facility, KI, Solna | ||
| Purification Matrix: ATP agarose | Sigma Aldrich | A2767 | |
| Purification Matrix: HisTrap HP-5 ml | GE Healthcare Life Sciences | 17-5247-01 | |
| Lipid:POPC | Avanti polar lipids | 850457C | 25 mg/ml in chloroform |
| Hydrophobic beads: Bio-Beads, SM-2 Resin | Bio-Rad | 1523920 | |
| 13 mm syringe filter: 0.2 μm | Pall life sciences | PN 4554T | |
| Stain: Sodium phosphotungstate tribasic hydrate | Sigma Aldrich | 31648 | |
| 2-mercaptoethanol | Sigma Aldrich | M3148-250ML | |
| Sodium Dodecyl Sulfate (SDS) | Bio-Rad | 161-0301 | |
| Protease inhibitor cocktail | Sigma Aldrich | 4693132001 | |
| TCEP | Sigma Aldrich | 646547 | |
| Detergent: Sodium cholate hydrate | Sigma Aldrich | C6445-10G | |
| Sodium Cholate | 500 mM Sodium cholate | Resuspend in miliQ water and store at -20°C | |
| Lipid Stock | 50 mM POPC, 100 mM sodium cholate, 20 mM Tris-HCl pH 7.5, 100 mM NaCl | Store at 4°C for a week or Store -80°C for a month, after purging the solution with nitrogen |
|
| MSP standard buffer | 20 mM Tris-HCl pH 7.5, 100 mM NaCl, 0.5 mMEDTA | Store at 4°C | |
| Non-Denaturaing Electrophoresis Anode Buffer | 50 mM Bis Tris 50 mM Tricine, pH 6.8 | BN2001 | Purchased from Thermofisher Scientific |
| Non-Denaturaing Electrophoresis Cathode Buffer | 50 mM Bis Tris 50 mM Tricine, pH 6.8 0.002% Coomassie G-250 | BN2002 | Purchased from Thermofisher Scientific |
| Non-Denaturaing Electrophoresis 4X Sample loading Buffer | 50 mM BisTrispH 7.2, 6N HCl, 50 mM NaCl, 10% (w/v) glycerol, 0.001% Ponceau S | BN2003 | Purchased from Thermofisher Scientific |
| Denaturaing Electrophoresis Running Buffer | 25 mM Tris-HCl pH 6.8, 200 mM Glycine, 0.1 % (w/v) SDS | Inhouse receipe | |
| Denaturaing Electrophoresis 5X Sample loading Buffer | 0.05 % (w/v) Bromophenolblue, 0.2 M Tris-HCl pH 6.8, 20 % (v/v) glycerol, 10% (w/v) SDS,10 mM 2-mercaptoethanol | Inhouse receipe | |
| Terrific broth | Tryptone – 12.0g Yeast Extract – 24.0g 100 mL 0.17M KH2PO4 and 0.72M K2HPO4 Glycerol – 4 mL |
Tryptone, yeast extract and glycerol were prepared to 900 ml and autoclaved seperately. KH2PO4 and K2HPO4 were prepared and autoclaved separately. Both were mixed before using the medium |
References
- Kleinschmidt, J. H., Popot, J. L. Folding and stability of integral membrane proteins in amphipols. Arch Biochem Biophys. 564, 327-343 (2014).
- Frauenfeld, J., et al. A saposin-lipoprotein nanoparticle system for membrane proteins. Nat Methods. 13 (4), 345-351 (2016).
- Denisov, I. G., Sligar, S. G. Nanodiscs for structural and functional studies of membrane proteins. Nat Struct Mol Biol. 23 (6), 481-486 (2016).
- Bayburt, T. H., Grinkova, Y. V., Sligar, S. G. Self-Assembly of Discoidal Phospholipid Bilayer Nanoparticles with Membrane Scaffold Proteins. Nano Letters. 2 (8), 853-856 (2002).
- Jonas, A., Steinmetz, A., Churgay, L. The number of amphipathic alpha-helical segments of apolipoproteins A-I, E, and A-IV determines the size and functional properties of their reconstituted lipoprotein particles. J Biol Chem. 268 (3), 1596-1602 (1993).
- Grinkova, Y. V., Denisov, I. G., Sligar, S. G. Engineering extended membrane scaffold proteins for self-assembly of soluble nanoscale lipid bilayers. Protein Eng Des Sel. 23 (11), 843-848 (2010).
- Hagn, F., Etzkorn, M., Raschle, T., Wagner, G. Optimized phospholipid bilayer nanodiscs facilitate high-resolution structure determination of membrane proteins. J Am Chem Soc. 135 (5), 1919-1925 (2013).
- Ritchie, T. K., Duzgunes, N. e. j. a. t., et al. . Methods in Enzymology. 464, 211-231 (2009).
- Schuler, M. A., Denisov, I. G., Sligar, S. G. Nanodiscs as a new tool to examine lipid-protein interactions. Methods Mol Biol. 974, 415-433 (2013).
- Nasr, M. L., et al. Membrane phospholipid bilayer as a determinant of monoacylglycerol lipase kinetic profile and conformational repertoire. Protein Sci. 22 (6), 774-787 (2013).
- Yokogawa, M., et al. NMR analyses of the interaction between the FYVE domain of early endosome antigen 1 (EEA1) and phosphoinositide embedded in a lipid bilayer. J Biol Chem. 287 (42), 34936-34945 (2012).
- Wan, C., et al. Insights into the molecular recognition of the granuphilin C2A domain with PI(4,5)P2. Chem Phys Lipids. 186, 61-67 (2015).
- Zhang, P., et al. An Isoform-Specific Myristylation Switch Targets Type II PKA Holoenzymes to Membranes. Structure. 23 (9), 1563-1572 (2015).
- Grushin, K., Miller, J., Dalm, D., Stoilova-McPhie, S. Factor VIII organisation on nanodiscs with different lipid composition. Thromb Haemost. 113 (4), 741-749 (2015).
- Baylon, J. L., Lenov, I. L., Sligar, S. G., Tajkhorshid, E. Characterizing the membrane-bound state of cytochrome P450 3A4: structure, depth of insertion, and orientation. J Am Chem Soc. 135 (23), 8542-8551 (2013).
- Mazhab-Jafari, M. T., et al. Oncogenic and RASopathy-associated K-RAS mutations relieve membrane-dependent occlusion of the effector-binding site. Proc Natl Acad Sci U S A. 112 (21), 6625-6630 (2015).
- Mazhab-Jafari, M. T., et al. Membrane-dependent modulation of the mTOR activator Rheb: NMR observations of a GTPase tethered to a lipid-bilayer nanodisc. J Am Chem Soc. 135 (9), 3367-3370 (2013).
- Wang, L., Sigworth, F. J. Structure of the BK potassium channel in a lipid membrane from electron cryomicroscopy. Nature. 461 (7261), 292-295 (2009).
- Ackerson, C. J., Powell, R. D., Hainfeld, J. F. Site-specific biomolecule labeling with gold clusters. Methods Enzymol. 481, 195-230 (2010).
- Boldog, T., Grimme, S., Li, M., Sligar, S. G., Hazelbauer, G. L. Nanodiscs separate chemoreceptor oligomeric states and reveal their signaling properties. Proc Natl Acad Sci U S A. 103 (31), 11509-11514 (2006).
- Moraes, I., Evans, G., Sanchez-Weatherby, J., Newstead, S., Stewart, P. D. Membrane protein structure determination – the next generation. Biochim Biophys Acta. 1838 (1 Pt A), 78-87 (2014).
- Dias, D. M., Ciulli, A. NMR approaches in structure-based lead discovery: recent developments and new frontiers for targeting multi-protein complexes. Prog Biophys Mol Biol. 116 (2-3), 101-112 (2014).
- Viegas, A., Viennet, T., Etzkorn, M. The power, pitfalls and potential of the nanodisc system for NMR-based studies. Biol Chem. , (2016).
- Cheng, Y., Grigorieff, N., Penczek, P. A., Walz, T. A primer to single-particle cryo-electron microscopy. Cell. 161 (3), 438-449 (2015).
- Wu, S., Armache, J. P., Cheng, Y. Single-particle cryo-EM data acquisition by using direct electron detection camera. Microscopy (Oxf). 65 (1), 35-41 (2016).
- Li, X., et al. Electron counting and beam-induced motion correction enable near-atomic-resolution single-particle cryo-EM. Nat Methods. 10 (6), 584-590 (2013).
- De Carlo, S., Adrian, M., Kalin, P., Mayer, J. M., Dubochet, J. Unexpected property of trehalose as observed by cryo-electron microscopy. J Microsc. 196 (1), 40-45 (1999).
- Nogales, E. The development of cryo-EM into a mainstream structural biology technique. Nat Methods. 13 (1), 24-27 (2016).
- Ohi, M., Li, Y., Cheng, Y., Walz, T. Negative Staining and Image Classification Powerful Tools in Modern Electron Microscopy. Biol Proced Online. 6, 23-34 (2004).
- Forte, T. M., Nordhausen, R. W. Electron microscopy of negatively stained lipoproteins. Methods Enzymol. 128, 442-457 (1986).
- Zhao, F. Q., Craig, R. Capturing time-resolved changes in molecular structure by negative staining. J Struct Biol. 141 (1), 43-52 (2003).
- Zhang, L., et al. Morphology and structure of lipoproteins revealed by an optimized negative-staining protocol of electron microscopy. J Lipid Res. 52 (1), 175-184 (2011).
- Zhang, L., et al. An optimized negative-staining protocol of electron microscopy for apoE4 POPC lipoprotein. J Lipid Res. 51 (5), 1228-1236 (2010).
- Matz, C. E., Jonas, A. Micellar complexes of human apolipoprotein A-I with phosphatidylcholines and cholesterol prepared from cholate-lipid dispersions. J Biol Chem. 257 (8), 4535-4540 (1982).
- Kumar, R. B., et al. Structural and Functional Analysis of Calcium Ion Mediated Binding of 5-Lipoxygenase to Nanodiscs. PLoS One. 11 (3), e0152116 (2016).
- Waggoner, T. A., Last, J. A., Kotula, P. G., Sasaki, D. Y. Self-assembled columns of stacked lipid bilayers mediated by metal ion recognition. J Am Chem Soc. 123 (3), 496-497 (2001).
- Kovacs, E., et al. Analysis of the Role of the C-Terminal Tail in the Regulation of the Epidermal Growth Factor Receptor. Mol Cell Biol. 35 (17), 3083-3102 (2015).
- Rames, M., Yu, Y., Ren, G. Optimized Negative Staining: a High-throughput Protocol for Examining Small and Asymmetric Protein Structure by Electron Microscopy. J Vis Exp. (90), e51087 (2014).
- Zhang, L., Tong, H., Garewal, M., Ren, G. Optimized negative-staining electron microscopy for lipoprotein studies. Biochim Biophys Acta. 1830 (1), 2150-2159 (2013).
- Cong, Y., Ludtke, S. J. Single particle analysis at high resolution. Methods Enzymol. 482, 211-235 (2010).
- Radmark, O., Werz, O., Steinhilber, D., Samuelsson, B. 5-Lipoxygenase, a key enzyme for leukotriene biosynthesis in health and disease. Biochim Biophys Acta. 1851 (4), 331-339 (2015).
- Anwar, Y., Sabir, J. S., Qureshi, M. I., Saini, K. S. 5-lipoxygenase: a promising drug target against inflammatory diseases-biochemical and pharmacological regulation. Curr Drug Targets. 15 (4), 410-422 (2014).
- Radmark, O., Samuelsson, B. Regulation of the activity of 5-lipoxygenase, a key enzyme in leukotriene biosynthesis. Biochem Biophys Res Commun. 396 (1), 105-110 (2010).
- Noguchi, M., Miyano, M., Matsumoto, T., Noma, M. Human 5-lipoxygenase associates with phosphatidylcholine liposomes and modulates LTA4 synthetase activity. Biochim Biophys Acta. 1215 (3), 300-306 (1994).
- Pande, A. H., Qin, S., Tatulian, S. A. Membrane fluidity is a key modulator of membrane binding, insertion, and activity of 5-lipoxygenase. Biophys J. 88 (6), 4084-4094 (2005).
- Pande, A. H., et al. Modulation of human 5-lipoxygenase activity by membrane lipids. Biochimie. 43 (46), 14653-14666 (2004).
- Wong, A., Hwang, S. M., Cook, M. N., Hogaboom, G. K., Crooke, S. T. Interactions of 5-lipoxygenase with membranes: studies on the association of soluble enzyme with membranes and alterations in enzyme activity. Biochimie. 27 (18), 6763-6769 (1988).
- Rigaud, J. L., Levy, D., Mosser, G., Lambert, O. Detergent removal by non-polar polystyrene beads. European Biophysics Journal. 27 (4), 305-319 (1998).
- Wittig, I., Braun, H. P., Schagger, H. Blue native PAGE. Nat Protoc. 1 (1), 418-428 (2006).
- Rames, M., Yu, Y., Ren, G. Optimized negative staining: a high-throughput protocol for examining small and asymmetric protein structure by electron microscopy. J Vis Exp. (90), e51087 (2014).
- Denisov, I. G., Grinkova, Y. V., Lazarides, A. A., Sligar, S. G. Directed self-assembly of monodisperse phospholipid bilayer Nanodiscs with controlled size. J Am Chem Soc. 126 (11), 3477-3487 (2004).
- Hornschemeyer, P., Liss, V., Heermann, R., Jung, K., Hunke, S. Interaction Analysis of a Two-Component System Using Nanodiscs. PLoS One. 11 (2), 0149187 (2016).
- Degrip, W. J., Vanoostrum, J., Bovee-Geurts, P. H. Selective detergent-extraction from mixed detergent/lipid/protein micelles, using cyclodextrin inclusion compounds: a novel generic approach for the preparation of proteoliposomes. Biochem J. 330 (Pt 2), 667-674 (1998).
- Martin, D. D., Budamagunta, M. S., Ryan, R. O., Voss, J. C., Oda, M. N. Apolipoprotein A-I assumes a "looped belt" conformation on reconstituted high density lipoprotein. J Biol Chem. 281 (29), 20418-20426 (2006).
- Cerione, R. A., Ross, E. M. Reconstitution of receptors and G proteins in phospholipid vesicles. Methods Enzymol. 195, 329-342 (1991).
- Shaw, A. W., McLean, M. A., Sligar, S. G. Phospholipid phase transitions in homogeneous nanometer scale bilayer discs. FEBS Lett. 556 (1-3), 260-264 (2004).
- Meer, G., Voelker, D. R., Feigenson, G. W. Membrane lipids: where they are and how they behave. Nat Rev Mol Cell Biol. 9 (2), 112-124 (2008).
- Civjan, N. R., Bayburt, T. H., Schuler, M. A., Sligar, S. G. Direct solubilization of heterologously expressed membrane proteins by incorporation into nanoscale lipid bilayers. Biotechniques. 35 (3), 553-562 (2003).
- Bao, H., Duong, F., Chan, C. S. A step-by-step method for the reconstitution of an ABC transporter into nanodisc lipid particles. J Vis Exp. (66), e3910 (2012).
- Brooks, S. P., Storey, K. B. Bound and determined: a computer program for making buffers of defined ion concentrations. Anal Biochem. 201 (1), 119-126 (1992).
- Gilbert, N. C., et al. The structure of human 5-lipoxygenase. Science. 331 (6014), 217-219 (2011).
- Radmark, O. 5-lipoxygenase-derived leukotrienes: mediators also of atherosclerotic inflammation. Arterioscler Thromb Vasc Biol. 23 (7), 1140-1142 (2003).
- Gao, Y., Cao, E., Julius, D., Cheng, Y. TRPV1 structures in nanodiscs reveal mechanisms of ligand and lipid action. Nature. 534 (7607), 347-351 (2016).
- Bayburt, T. H., Sligar, S. G. Self-assembly of single integral membrane proteins into soluble nanoscale phospholipid bilayers. Protein Sci. 12 (11), 2476-2481 (2003).

