Disposable Dosators for Pulmonary Insufflation of Therapeutic Agents to Small Animals
Summary
During development of drugs for pulmonary delivery, it is necessary to evaluate pharmacokinetics and efficacy in an animal model. We present a method to build a disposable aerosol dispersion system from of-the-shelf components that can be used to administer intrapulmonary dry powder aerosol to rodents.
Abstract
Development of new therapeutic products requires efficacy testing in an animal model. The pulmonary route of administration can be utilized to deliver drugs locally and systemically. Evaluation of dry powder aerosols necessitates an efficient dispersion mechanism to maintain high concentrations in an exposure chamber or for direct endotracheal administration. While solutions exist to expose animals by passive inhalation to dry powder aerosols, most require masses of powder in large excess of the dose delivered. This precludes conducting early feasibility studies as insufficient drug is available at the research or early development stage to support the dose delivery requirements for conventional aerosol delivery systems. When designing an aerosol drug product, aerodynamic performance can relate directly to delivery efficiency and efficacy. Dispersion of powder into an aerosol requires energy input sufficient to overcome interparticulate forces, and particle engineering approaches can substantially improve aerosol performance. We have developed a dispersion system (dosator) which can aerosolize engineered dry powder aerosols efficiently for the purpose of direct pulmonary insufflation, dispersion into an exposure system or generation for analytical purposes.
Introduction
Development of new therapeutic products requires efficacy testing in an animal model. The pulmonary route of administration can be utilized to deliver drugs locally and systemically 1. Evaluation of dry powder aerosols necessitates an efficient dispersion mechanism to maintain high concentrations in an exposure chamber or for direct endotracheal administration. While solutions exist to expose animals by passive inhalation to dry powder aerosols, most require masses of powder in large excess of the dose delivered 2.
This precludes conducting early feasibility studies as insufficient drug is available at the research or early development stage to support the dose delivery requirements for conventional aerosol delivery systems.
When designing an aerosol drug product, aerodynamic performance can relate directly to delivery efficiency and efficacy 3. Dispersion of powder into an aerosol requires energy input sufficient to overcome interparticulate forces, and particle engineering approaches can substantially improve aerosol performance 4,5,6. We have developed a dispersion system (dosator) which can aerosolize engineered dry powder aerosols efficiently for the purpose of direct pulmonary insufflation, dispersion into an exposure system or generation for analytical purposes.
Protocol
1. Preparation of Components of the Dosators
- Perforate the bottom of a 0.5 mL polypropylene microcentrifuge tube by drilling or by simply grinding or cutting off the bottom with a grinding wheel or a sharp pair of scissors. Ensure that the hole is in the center and is no more than 2 mm in diameter and no less than 1 mm in diameter (Figure 1).
- Perforate the top of the microcentrifuge tube. Using a #22 drill bit (approximately 4 mm in diameter), drill a hole in the center of the cap. This results in a hole that will fit a slip-tip syringe (Figure 1).
- Prepare the fine screens to be inserted into the microcentrifuge tubes. Make screens from 304 stainless steel wire cloth, 60 x 60 mesh with wire diameter of 0.0045". Cut the screens with tin snips or punch them using a die to an outer diameter of 5 mm (Figure 2).
2. Assembly of the Dosators
- Using either forceps or the end of a blunt tip needle, insert the screen into the microcentrifuge tube and seat it in the bottom so that it covers the opening. This will serve to support the powder before actuation.
- Press fit the narrow end of the microcentrifuge tip into the end of a blunt tip needle. Do this by hand or with a bench-top press and a guide. Gently wipe the top of the needle and bottom of the tube with ethanol to clean the parts to ensure a tight, resilient fit.
3. Filling the Dosators
- Using an analytical microbalance, tare the device and load the desired amount of powder into the dosator. A typical powder mass for dispersion ranges from 1 to 10 mg.
- Once filled, plug the top of the microcentrifuge tube with an allotment of cotton and a pair of forceps. This will prevent the powder from drawing into the syringe or falling out, while allowing airflow through the dosator to disperse the powder.
- Close the microcentrifuge tube cap.
- If not being used immediately, seal the top with a small amount of lab-film to minimize powder exposure to ambient moisture. NOTE: Generally, preloaded dosators should be stored according to storage requirements of the active pharmaceutical ingredient (API). Often aerosol powders are moisture sensitive and should be stored desiccated.
4. Device Actuation
- Draw back the syringe to the desired volume, which may be application specific. For intrapulmonary administration in guinea pigs, use 2 mL. Dispersion efficiency is related to the volume and velocity of air delivered during actuation, as well as the inherent dispersion properties of the powder 7. The top hole of the dosator accommodates a slip-tip syringe. A Luer-lock syringe may be used with an adapter.
- Insert the syringe into the back end of the dosator.
- Insert the needle end of the dosator into the dosing port on the exposure chamber. Depress the syringe forcefully, expelling powder out of the device and directly into the animal or into the exposure chamber or analytical instrument (Figure 3).
Representative Results
For easily dispersible powders such as those spray-dried with the intent for pulmonary delivery 5,8,9, the dosators deliver a bolus dose out of the device. There are many applications for the dosators, including in vitro particle characterization, direct intrapulmonary administration in live animals, and aerosol generation for passive inhalation systems.
To demonstrate the efficiency of powder dispersion from the dosators, a previously constructed nose-only exposure chamber was utilized. The key features of this chamber are a small central chamber and low airflow, allowing high aerosol concentrations to be achieved. The dosing chamber was assembled from acrylic and featured a central aerosol compartment 4" in diameter and a length of 3" with tapered nose cones for evaluation of aerosol exposure by passive inhalation (Figure 4).
A spray dried drug powder of mass median aerodynamic diameter (MMAD) 2.9 µm was evaluated in guinea pigs using this system (Figure 5). Dosing interval was determined by measuring chamber concentration in real time after delivering a single bolus dose of 5 mg using a 12 mL syringe. In vivo evaluation administering 5 doses of 10 mg at 3 minute intervals resulted in a maximum plasma concentration of 5 µg/mL (Figure 6). This compares to an endotracheally administered dose of 1 mg/kg delivered by a system widely used in the field 10. The observed plasma concentration as a result of passive inhalation is indicative of efficient dispersion to primary particle size.
As there is little to no airflow inside the chamber, the aerosol concentration will decrease as a function of particle aerodynamic diameter as particles fall out due to gravitational settling. As particles settle, and aerosol concentration decreases below a certain threshold, another dose can be administered through the port. Exposure times will depend on the objective but because high concentrations are achievable, a schedule can be determined to minimize exposure time. This again describes one application of using these disposable dosing systems, but another possible use would be direct intrapulmonary powder administration, which can have many analytical endpoints depending on the purpose of the evaluation.
Figure 1. Modified Centrifuge Tube. Modified 0.5 mL microcentrifuge tube with holes in the bottom and top. Please click here to view a larger version of this figure.

Figure 2. Mesh Screens. 5 mm diameter circles of 60 x 60 stainless steel mesh of wire diameter 0.0045". Please click here to view a larger version of this figure.

Figure 3. Actuation Schematic. Assembled dosator schematic depicting actuation of powder. Air is forced through the reservoir, dispersing the powder out of the dispensing end. Please click here to view a larger version of this figure.
Figure 4. Exposure Chamber Schematic. Animal exposure chamber central reservoir schematic with dimensions. Two animals can be exposed at once, with each animal being guided to the central chamber via the tapered nose cones. Please click here to view a larger version of this figure.

Figure 5. Dry Powder Aerosol. Representative engineered drug powder produced by spray drying. Powders can be designed for a variety of applications. For engineered dry powder aerosols, aerodynamic diameter and powder dispersion efficiency are critical engineering targets. Scale bar = 5 microns. Please click here to view a larger version of this figure.
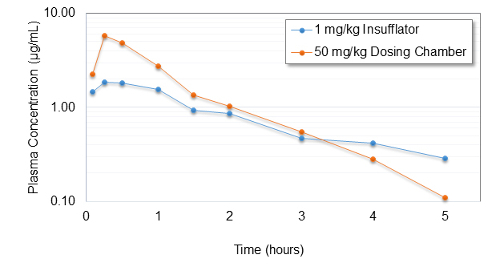
Figure 6. Pharmacokinetic Evaluation via Passive Inhalation. Pharmacokinetics of drug plasma concentration on a logarithmic scale over a period of 5 h dispersed by the dosators into the central aerosol reservoir compared to a benchmark dose of 1 mg/kg delivered by direct pulmonary administration. Please click here to view a larger version of this figure.
Figure 7. Filled and Bulk Dosators. (Left) Filled and actuation-ready dosator (10 mg). (Right) Depiction of the bulk preparation of disposable dosators. These can be preloaded for use in restricted environments. Please click here to view a larger version of this figure.
Discussion
The dispersion from the dosators was suitable for animals to receive a therapeutic dose via passive nose-only respiration, indicating a large proportion of aerosol dispersed to primary particle size. These dosators can be used for endotracheal administration, in vitro evaluation of powder performance, and general purpose dry powder aerosol dispersion for analytical or efficacious experiments. If used for intrapulmonary delivery, it is important to use an appropriate volume of air for the species chosen, as over-inflation of the lungs can cause tissue damage and distort pharmacokinetic results. A volume of 5 mL has been used in guinea pigs with a similar device 10, however other research has shown that lung damage can occur in rats with a single 2 mL administration 11.
Because of the cost of components and ease of preparation, these dosators can be used as single-use disposables. In contrast, multi-use devices require that between dosing the device has to be refilled 12, complicated the performance of the animal experiment and requiring the animal technician to perform a task that in most facilities is not easily accomplished. Filling can be performed in a dose preparation laboratory (with balances, hoods or glove boxes and supplies to support small mass weighing into the dosing chamber) and samples can be transferred to an animal procedures room rather having both activities performed in the same laboratory. This is particularly advantageous where any form of additional biological control is required for the animal experiments.
We demonstrate here the methods suitable for preparation of a dispersion system that can be easily manufactured and can efficiently facilitate early stage aerosol drug development with minimal drug usage. The low cost dosators can be manufactured in bulk and used as preloaded dosage forms for administering aerosol powder in restrictive environments where necessary personal protective equipment can be a hindrance (Figure 7).
Divulgations
The authors have nothing to disclose.
Acknowledgements
Authors would like to kindly thank the National Institute for Allergy and Infectious Disease for the funding to conduct this research.
Materials
| 0.5 mL microcentrifuge tube | VWR | 89000-026 | |
| High-Volume Particle-Filtering Stainless Steel Wire Cloth, Woven, 304 Stainless Steel, 60 x 60 Mesh, .0045" Wire Diameter | McMaster Carr | 9230T44 | |
| Stainless Steel Dispensing Needle, Straight, 18 Gauge, 1" Long | McMaster Carr | 75165A676 | Any luer-fit needle will suffice |
| Cotton balls | McMaster Carr | 54845T16 | |
| Parafilm M® Laboratory Film | VWR | 100229-550 | |
| Black-Oxide High-Speed Steel Jobbers' Drill Bit, Wire Gauge 22, 3-1/8" Overall Length, 1.8" Drill Depth, 135Deg Point | McMaster Carr | 2901A195 |
References
- Young, E. F., et al. Inhaled Pyrazinoic Acid Esters for the Treatment of Tuberculosis. Pharmaceutical Research. , 1-11 (2016).
- Hinds, W. C. . Aerosol Technology: Properties, Behavior, and Measurement of Airborne Particles. , (2012).
- Islam, N., Gladki, E. Dry powder inhalers (DPIs)-A review of device reliability and innovation. International Journal of Pharmaceutics. 360 (1-2), 1-11 (2008).
- Chan, H. K. Dry powder aerosol delivery systems: current and future research directions. J Aerosol Med. 19 (1), 21-27 (2006).
- Vehring, R. Pharmaceutical Particle Engineering via Spray Drying. Pharmaceutical Research. 25 (5), 999-1022 (2008).
- Bosquillon, C., Lombry, C., Préat, V., Vanbever, R. Influence of formulation excipients and physical characteristics of inhalation dry powders on their aerosolization performance. Journal of Controlled Release. 70 (3), 329-339 (2001).
- Hickey, A. J., Concessio, N. M., Van Oort, M. M., Platz, R. M. Factors influencing the dispersion of dry powders as aerosols. Pharmaceutical Technology. 18 (8), 58-64 (1994).
- Garcia-Contreras, L., et al. Inhaled large porous particles of capreomycin for treatment of tuberculosis in a guinea pig model. Antimicrob Agents Chemother. 51 (8), 2830-2836 (2007).
- Durham, P. G., et al. Spray Dried Aerosol Particles of Pyrazinoic Acid Salts for Tuberculosis Therapy. Molecular Pharmaceutics. 12 (8), 2574-2581 (2015).
- Sung, J. C., et al. Dry powder nitroimidazopyran antibiotic PA-824 aerosol for inhalation. Antimicrob Agents Chemother. 53 (4), 1338-1343 (2009).
- Guillon, A., et al. Pulmonary delivery of dry powders to rats: tolerability limits of an intra-tracheal administration model. Int J Pharm. 434 (1-2), 481-487 (2012).
- Morello, M., et al. Dry-powder pulmonary insufflation in the mouse for application to vaccine or drug studies. Tuberculosis. 89 (5), 371-377 (2009).

