Using a Whole-mount Immunohistochemical Method to Study the Innervation of the Biliary Tract in Suncus murinus
Summary
A whole-mount immunohistochemical approach, to visualize neurofilament protein expression in the extrahepatic biliary tract in Suncus murinus. is presented here. This protocol can be used to analyze the innervation of all visceral organs in S. murinus or other species.
Abstract
This work describes the whole-mount immunohistochemistry staining method in detail, using neurofilament protein antibody to label the innervation of the biliary tract in Suncus murinus (S. murinus ). First, the specimen was dissected from S. murinus and fixed in 4% paraformaldehyde (PFA). Second, an enzymatic treatment and potential endogenous peroxidase inactivation were performed. The specimen was then exposed to the primary antibody, anti-neurofilament protein antibody, for 3-6 days. It was then incubated with the secondary antibody conjugated with horseradish peroxidase. The color reaction was revealed by reacting the specimen with a 3,3'-diaminobenzidine (DAB) substrate. This method can be applied to analyze the innervation of all visceral organs. Furthermore, this protocol can also be adapted to test other neuronal antibodies, but optimization of the antibodies should be done first. This method was originally introduced by Kuratani and Tanaka1,2,3.
Introduction
The house musk shrew, Suncus murinus, belongs to the order Insectivora and the family Soricidae. This tiny mammal is distributed throughout Southeast Asia and inhabits houses and grassy areas that are situated near human habitations or cattle pens4. This species exhibits general morphological characteristics more similar to humans than other laboratory animals, such as the mouse, rat, and rabbit5. Previously, whole-mount immunostaining with a peripheral neuron marker for S. murinus neurofilament protein (NFP) was used to label peripheral nerves in the pancreas6, major duodenal papilla7, pylorus8, gallbladder5, and extrahepatic biliary tract9.
NFP is a macromolecular complex that is part of the mature neuronal cytoskeleton. Neurofilament-related proteins mediate interactions between NFP and the zygosome. Both enzyme function and the structure of linker proteins are regulated by NFP10. The NFP complex is made of three polypeptides: NF-L (70 kDa), NF-M (150-160 kDa), and NF-H (200 kDa). NFP can be found in neuronal axons in both the central and peripheral nervous system. Anti-human NFP antibody has been shown to label axons of the central and peripheral nervous system. Anatomical and electrophysiological investigations have demonstrated that the sphincter, gallbladder, and proximal gastrointestinal tract are connected11,12,13,14. However, the morphological features of their neuronal connections have not yet been defined.
This work demonstrates the use of a whole-mount immunohistochemical staining method to label the innervation of the S. murinus biliary tract with an NFP antibody.
Protocol
All experimental procedures were approved by the Tokyo Metropolitan University Institutional Animal Care and Use Committee (IACUC). The animals were housed and handled in accordance with the Guide for the Care and Use of Laboratory Animals and the Guide for the Care and Use of Experimental Animals of the Canadian Council on Animal Care. We used animals bred and maintained at the Functional Morphology Laboratory, Department of Frontier Health Sciences, Tokyo Metropolitan University, Japan.
1. Animal Care and Housing
- Obtain 7 S. murinus (3 females and 4 males weighing 70-90 g) from a closed breeding colony.
- Cage the S. murinus individually after weaning (20 d after birth) in plastic cages equipped with a wooden nest box containing paper strips. Keep them in a conventionally conditioned animal room: 23-27 °C, no humidity control, and a 12:12 h light:dark cycle. Supply commercial trout pellets and water ad libitum.
2. Tissue Preparation
- To prepare 4% (w/v) paraformaldehyde (PFA), dissolve 40 g of PFA in 1,000 mL of 0.01 M Phosphate-Buffered Saline (PBS, pH 7.4). In a fume cupboard, mix using a vortex until the solution is clear. Store the 4% PFA O/N at 4 °C.
Caution: Wear appropriate Personal Protective Equipment (PPE) when handling PFA. - To perfuse and fix the animal, anesthetize it with ether and then give it an intraperitoneal injection of somnopentyl (pentobarbital sodium, 0.6 mL/kg bodyweight).
- After the animal is completely narcotized, open the abdominal cavity with a scalpel, creating a midline abdominal incision 3 cm long. While incising the inferior vena cava for exsanguinating, insert a catheter retrogradely into the abdominal aorta at the level immediately above the bifurcation of this artery into the common iliac arteries.
- Inject somnopentyl (pentobarbital sodium, 1.0 mL/kg bodyweight). After complete euthanasia, perfuse with 0.01 M PBS (pH 7.4) and then with 4% PFA in a fume cupboard.
- After perfusion, inject 2-3 mL of white neoprene latex (dilute white neoprene latex 3:2 with Distilled Water (DW)) to label the blood vessels.
- Extract the abdominal organs, including the liver, gallbladder, common bile duct, duodenum, and pancreas en bloc, with the related nerves and vessels.
- Inject approximately 0.1 mL of blue neoprene latex (add a drop of blue ink to 10 mL of the diluted white neoprene latex) to the gallbladder to label the biliary system.
- Post-fix overnight with 4% PFA at 4 °C for whole-mount immunostaining.
Caution: PFA is toxic. Avoid handling PFA directly. PFA should be used in a fume cupboard.
3. Whole-mount Immunohistochemistry
Note: Throughout the protocol, including during washing, antibody incubation, and coloration, the specimen must remain on the nutator, gently rocking at RT or at 4°C.
- Day 1: Enzymatic treatment and inactivation of potential endogenous peroxidase
- Wash the prepared specimen with DW 4x for 1 h each at RT in an appropriately sized glass vial. Rock the specimen gently on the nutator to remove the PFA. Avoid damaging the specimen when exchanging solutions.
- To prepare 1% (w/v) orthoperiodic acid, dissolve 1 g of orthoperiodic acid in 100 mL of DW.
- Incubate the specimen in 1% orthoperiodic acid for 20 min at RT to prevent any intrinsic peroxidase reaction.
- Prepare 0.004% (w/v) papain by dissolving 0.004 g of papain in 100 mL of 0.025 mol/L Tris-HCl buffer (pH 7.6).
- Wash the specimen with DW for 10 min at RT.
- Incubate the specimen in freshly prepared 0.004% papain for 2 h at 37 °C in a constant-temperature bath with gentle rocking.
- Wash the specimen with DW for 50-60 min at RT.
- Store the specimen in 4% PFA at 4 °C O/N.
- Day 2: Enzymatic treatment
- Wash the stored specimen with DW 4 times for 1 h each at RT.
- Incubate the specimen in freshly prepared 0.004% papain for 2 h at 37 °C in a constant-temperature bath with gentle rocking.
- Wash the specimen with DW for 50-60 min at RT. Store the specimen in 4% PFA at 4 °C O/N.
- Day 3: Freezing treatment
- Wash the stored specimen with DW 4x for 1 h each at RT, as above.
- Prepare 2.5% (w/v), 5% (w/v), and 10% (w/v) sucrose by dissolving 2.5 g, 5 g, and 10 g of sucrose, respectively, in 100 mL of 0.01 M PBS (pH 7.4).
- Immerse the specimen in 2.5% (w/v), 5% (w/v), and 10% (w/v) sucrose for 30 min each.
- Freeze the specimen at -20 °C for 30 – 60 min and then completely thaw it at RT; repeat the cycle 3x.
- To prepare 2% Triton X-100 (v/v), add 2 mL of Triton X-100 to 100 mL of 0.01 M PBS (pH 7.4).
- Store the specimen in 2% Triton X-100 at 4 °C O/N.
- Day 4: Primary antibody incubation
- Prepare the dilution solution of the primary antibody by dissolving 0.2 g of Bovine Serum Albumin (BSA), 0.3 mL of Triton X-100, and 0.1 g of sodium azide in 100 mL of 0.01 M PBS (pH 7.4).
Caution: Sodium azide is acutely toxic. Inhalation and touching must be avoided. Wear appropriate PPE. - Dilute the primary antibody (NFP) 1:600 in the above dilution buffer (step 3.4.1). Incubate the specimen with NFP antibody at 4 °C for 3-6 days, keeping the specimen gently rocking on the nutator. Larger specimens will require increased incubation times.
- Prepare the dilution solution of the primary antibody by dissolving 0.2 g of Bovine Serum Albumin (BSA), 0.3 mL of Triton X-100, and 0.1 g of sodium azide in 100 mL of 0.01 M PBS (pH 7.4).
- Secondary antibody incubation
- Wash the specimen in PBS 4x for 1 h each at RT to remove unbound primary antibody.
- Prepare the dilution solution for the secondary antibody by dissolving 0.2 g of BSA and 0.3 mL of Triton X-100 in 100 mL of 0.01 M PBS (pH 7.4).
- Dilute the secondary antibody (peroxidase-conjugated, affinity-purified sheep anti-mouse IgG-HRP) 1:600 in the dilution buffer (step 3.5.2). Incubate the specimen with the secondary antibody at 4 °C for 3 d, and keep the specimen rocking gently on the nutator.
- Coloration
- Prepare DAB coloration solution by adding 0.002 g of 3,3'-diaminobenzidine tetrahydrochloride (DAB) to 100 mL of 0.05 mol/L Tris-HCl buffer (pH 7.6) under a fume cupboard, keeping the solution in the dark.
- Wash the specimens at RT in PBS 4x for 1 h each.
- Add 10 µL of 30% H2O2 in freshly prepared DAB coloration solution and incubate with the solution at 4 °C O/N or for 3 d while rocking gently on the nutator.
- Stop the reaction by placing the specimen in PBS with 0.04% sodium azide when the optimal staining intensity is reached.
NOTE: Avoid the use of sodium azide until this step, as it inhibits horseradish peroxidase.
- Imaging the stained tissue
- Carefully transfer the specimen to a petri glass dish (5 cm high, 10 cm in diameter) containing PBS. Capture whole-mount images using a camera mounted on a stereomicroscope.
NOTE: Whole-mount stained tissue should be imaged while immersed completely in PBS on a transparent glass dish.
- Carefully transfer the specimen to a petri glass dish (5 cm high, 10 cm in diameter) containing PBS. Capture whole-mount images using a camera mounted on a stereomicroscope.
Representative Results
Figures 1–4 show typical results for NFP-positive nerve fibers in the extrahepatic biliary tract in S. murinus. The antibody against NFP reproducibly labeled the innervation in the entire image of the extrahepatic biliary tract (Figure 1), gallbladder (Figure 2), upper bile duct (Figure 3), and duodenal papilla area (Figure 4), with high specificity and minimal background.
For all tissues of the specimen, regardless of shape and size, the tegmental nerves can be dyed at the same time. In addition to the innervation of the extrahepatic biliary tract, the running and distribution density of the vagus of the esophagus and the stomach were exhibited unambiguously (Figure 1).
As the blood vessels of the gallbladder were labeled with white neoprene latex, thin nervous fibroses were clearly demonstrated with high contrast. The density of innervation was higher in the neck than in the fundus of the gallbladder (Figure 2).
In the upper bile duct, two types of nerve bundles were labeled. The fine nerve bundles formed an irregular and dense network of nerves, ran adhesively, and resided on/in the biliary tract; the thicker neural bundles were distributed parallel to the surface of the biliary tract (Figure 3).
The common bile duct was labeled with blue neoprene latex and the vessels with white neoprene latex. The thin nervous fibers at the end of the common bile duct and the duodenal papilla area were clearly demonstrated with high contrast (Figure 4).
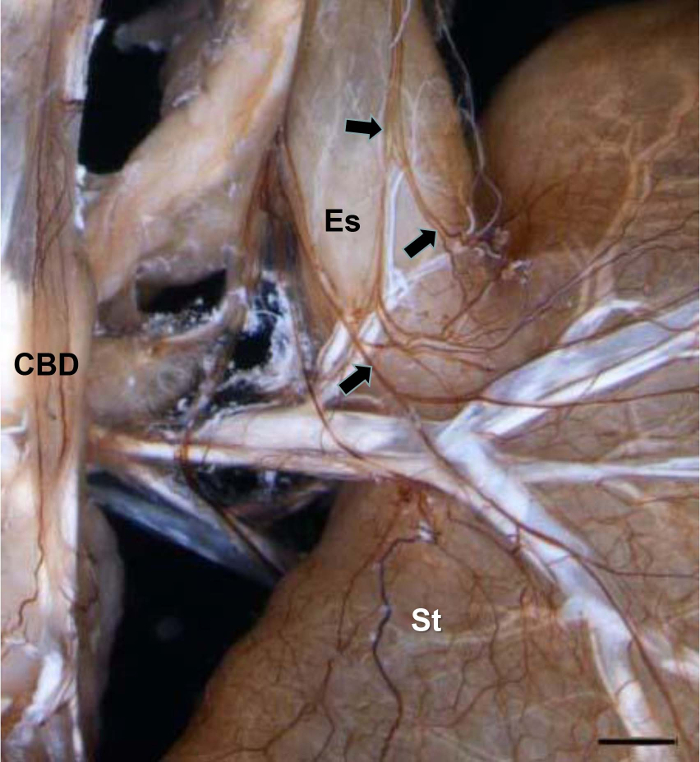
Figure 1: NFP-positive Nerve Fibers in the Biliary Tract, the Esophagus, and the Stomach. Arrows show the vagus running along the esophagus to the stomach. CBD, Common Bile Duct; Es, esophagus; St, stomach. Scale bar = 1,300 µm. Please click here to view a larger version of this figure.
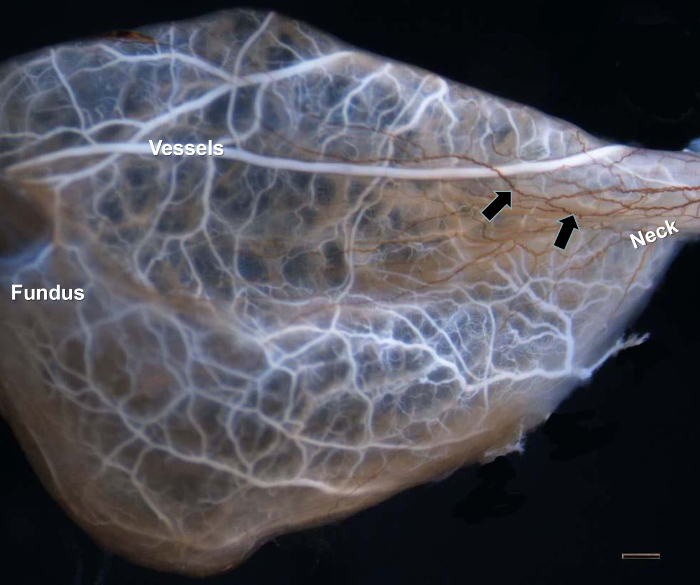
Figure 2: NFP-positive Nerve Fibers in the Gallbladder. The blood vessels of the gallbladder were labeled with white latex. Abundant innervation occurred in the neck of the gallbladder (arrows). Scale bar = 1,000 µm. Please click here to view a larger version of this figure.
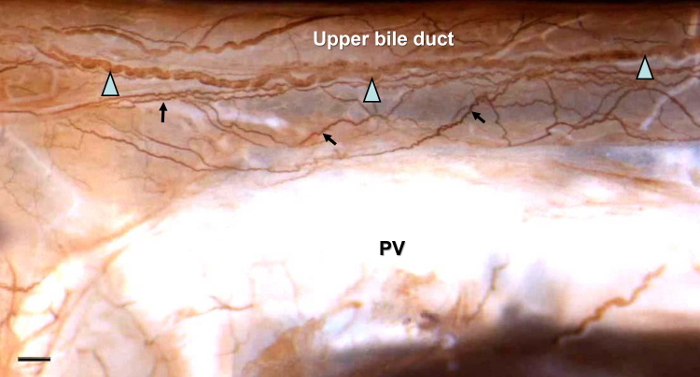
Figure 3: NFP-positive Nerve Fibers in the Upper Bile Duct. Two types of nerve bundles were observed. One type was the fine nerve bundles that ran adhesively and resided on/in the extrahepatic biliary tract (arrows); the other type was thicker neural bundles that were distributed parallel to the surface of the extrahepatic biliary tract and ran between the gallbladder and duodenum (triangles). PV, Portal Vein. Scale bar = 650 µm. Please click here to view a larger version of this figure.
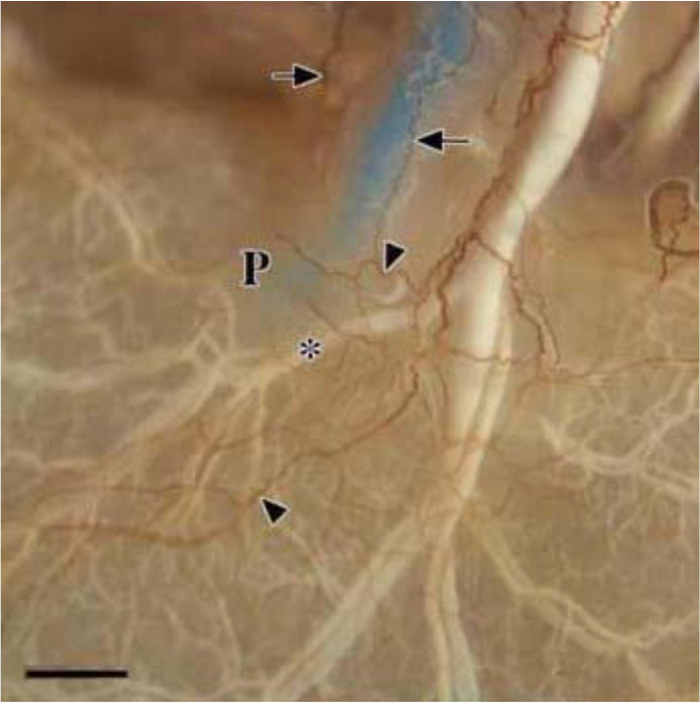
Figure 4: NFP-positive Nerve Fibers in the Duodenal Papilla Area. The common bile duct was labeled with blue latex and the vessels with white latex (*). Arrows show the innervation of the end of the common bile duct, and triangles show the innervation of the duodenal papilla area (P), which comes from the common bile duct and vessels. Scale bar = 1,000 µm. Please click here to view a larger version of this figure.
Discussion
This work describes the experimental procedure for the visualization of the innervation of the extrahepatic biliary tract using an antibody against neurofilament protein. This protocol was adapted from the protocol described by Kuratani and Tanaka1,2,3.
For this experiment, plan the timeline for each of the experiments, as the whole-mount staining approach continues for 2-3 weeks. The vessel containing the tissue must be rotated on a nutator at all times, except during the freeze/thaw process. Particularly during incubation with the primary and secondary antibodies, the specimen was set in a cold storage chamber while rotating on a nutator to ensure the uniform exposure of the specimen to the reaction solutions. Several solutions – 1% (w/v) orthoperiodic acid, 0.004% (w/v) papain, and the dilution buffer for the primary/secondary antibodies – must be made fresh for each experiment. Throughout the protocol, to avoid touching and damaging the specimen when changing solutions, the solutions should be poured out instead of being removed with forceps15.
After fixing with PFA, the samples should be washed sufficiently with DW or PBS. In addition, the enzymatic treatment with the papain incubation is important. The heat and the enzyme used for antigen retrieval help to prepare the tissue for antibody penetration. To enhance the penetration of the solutions, the outside membrane of the tissue should be disrupted by a freezing treatment at -20 °C for 30 min or -80 °C O/N15.
It is important to completely submerge the experimental specimen in adequate volumes of buffer solution to ensure that the solution can incubate the whole specimen. The incubation time of the primary antibody must be empirically determined for different tissues and sample sizes. Usually, it must be 3 days for a 2-3 cm sample. The optimal dilution must be empirically determined for each antibody. A dilution at 1:600 is recommended for both the primary and secondary antibodies when using the appropriate antibody buffer.
This work describes, in detail, the protocol of a versatile whole-mount immunohistochemical approach to reveal neurofilament protein expression in the biliary tract of S. murinus. This method can be used to analyze the innervation of all visceral organs in many species. Furthermore, this protocol can also be adapted to test other neuronal antibodies, but optimization of the antibodies should be performed.
However, the technique was limited in labeling shallow nervous tissue and in constructing a three-dimensional model of innervation. Also, it could not identify the characteristics of the nervous fibers (i.e. sympathetic or parasympathetic, motor or sensory, etc.).
Divulgations
The authors have nothing to disclose.
Acknowledgements
The authors have no acknowledgements.
Materials
| orthoperiodic acid | Wako | 162-00732 | |
| papain | Wako | 164-00172 | |
| bovine serum albumin (BSA) | Wako | 010-15131 | |
| sodium azide | Nacalai Tesque | 312-33 | |
| neurofilament protein (NFP) antibody | Dako | M0762, lot 089, clone: 2F11 | |
| peroxidase-conjugated affinity-purified sheep anti-mouse IgG-HRP | MBL | Code 330 | |
| 3,3'-diaminobenzidine tetrahydrochloride (DAB) | Wako | 349-00903 | |
| imidazole | Sigma | I-0125 |
References
- Kuratani, S., Tanaka, S., Ishikawa, Y., Zukeran, C. Early development of the hypoglossal nerve in the chick embryo as observed by the whole-mount nerve staining method. Am. J. Anat. 182 (2), 155-168 (1988).
- Kuratani, S., Tanaka, S. Peripheral development of the avian vagus nerve with special reference to the morphological innervation of heart and lung. Anat. Embryol. (Berl). 182 (5), 435-445 (1990).
- Kuratani, S., Tanaka, S. Peripheral development of avian trigeminal nerves. Am. J. Anat. 187 (1), 65-80 (1990).
- Yi, S. Q., et al. House musk shrew, Suncus murinus: a novel and natural obesity-resistant animal model. Obes. Metab. 6, 22-28 (2010).
- Yi, S. Q., et al. Surgical anatomy of innervation of the gallbladder in humans and Suncus murinus with special reference to morphological understanding of gallstone formation after gastrectomy. World J. Gastroenterol. 13 (14), 2066-2071 (2007).
- Yi, S. Q., et al. Anatomical study of the pancreas in the house musk shrew, Suncus murinus, with special reference to the blood supply and innervation. Anat. Rec. A Discov. Mol. Cell Evol. Biol. 273 (1), 630-635 (2003).
- Yi, S. Q., et al. Surgical anatomy of the innervation of the major duodenal papilla in humans and Suncus murinus, from the perspective of preserving innervation in organ-saving procedures. Pancreas. 30 (3), 211-217 (2005).
- Yi, S. Q., et al. Surgical anatomy of the innervation of the pylorus in humans and Suncus murinus, in relation to the surgical technique for pylorus-preserving pancreatoduodenectomy. World J. Gastroenterol. 12 (14), 2209-2216 (2006).
- Yi, S. Q., Ren, K., Kinoshita, M., Takano, N., Itoh, M., Ozaki, N. Innervation of Extrahepatic Biliary Tract, With Special Reference to the Direct Bidirectional Neural Connections of the Gall Bladder, Sphincter of Oddi and Duodenum in Suncus murinus, in Whole-Mount Immunohistochemical Study. Anat. Histol. Embryol. 45 (3), 184-188 (2016).
- Wang, H., et al. Neurofilament proteins in axonal regeneration and neurodegenerative diseases. Neural Regen. Res. 7 (8), 620-626 (2012).
- Wyatt, A. P. The relationship of the sphincter of Oddi to the stomach, duodenum and gall-bladder. J. Physiol. 193 (2), 225-243 (1967).
- Mawe, G. M., Kennedy, A. L. Duodenal neurons provide nicotinic fast synaptic input to sphincter of Oddi neurons in guinea pig. Am. J. Physiol. 277, G226-G234 (1999).
- Seo, J. H., Cho, S. S., Lee, I. S., Lee, H. S. Anatomical and neuropeptidergic properties of the duodenal neurons projecting to the gallbladder in the golden hamster. Arch. Histol. Cytol. 65 (4), 317-321 (2002).
- Padbury, R. T., Furness, J. B., Baker, R. A., Toouli, J., Messenger, J. P. Projections of nerve cells from the duodenum to the sphincter of Oddi and gallbladder of the Australian possum. Gastroenterology. 104 (1), 130-136 (1993).
- White, J. J., Reeber, S. L., Hawkes, R., Sillitoe, R. V. Wholemount immunohistochemistry for revealing complex brain topography. J. Vis. Exp. (62), e4042 (2012).

