Phenotypic Analysis of Rodent Malaria Parasite Asexual and Sexual Blood Stages and Mosquito Stages
Summary
Due to the striking similarities of the life cycle and biology of rodent malaria parasites to human malaria parasites, rodent malaria models have become indispensable for malaria research. Herein, we standardized some of the most important techniques used in the phenotypic analysis of wild-type and transgenic rodent malaria species.
Abstract
Recent advances in genetics and systems biology technologies have promoted our understanding of the biology of malaria parasites on the molecular level. However, effective malaria parasite targets for vaccine and chemotherapy development are still limited. This is largely due to the unavailability of relevant and practical in vivo infection models for human Plasmodium species, most notably for P. falciparum and P. vivax. Therefore, rodent malaria species have been extensively used as practical alternative in vivo models for malaria vaccine, drug targeting, immune response, and functional characterization studies of conserved Plasmodiumspp. genes. Indeed, rodent malaria models have proven to be invaluable, especially for exploring mosquito transmission and liver stage biology, and were indispensable for immunological studies. However, there are discrepancies in the methods used to evaluate the phenotypes of transgenic and wild-type asexual and sexual blood-stage parasites. Examples of these discrepancies are the choice of an intravenous vs. intraperitoneal infection of rodents with blood-stage parasites and the evaluation of male gamete exflagellation. Herein, we detail standardized experimental methods to evaluate the phenotypes of asexual and sexual blood stages in transgenic parasites expressing reporter-gene or wild-type rodent malaria parasite species. We also detail the methods to evaluate the phenotypes of malaria parasite mosquito stages (gametes, ookinetes, oocysts, and sporozoites) inside Anopheles mosquito vectors. These methods are detailed and simplified here for the lethal and non-lethal strains of P. berghei and P. yoelii but can also be applied with some adjustments to P. chabaudi and P. vinckei rodent malaria species.
Introduction
Malaria parasites cause hundreds of millions of malaria infections in humans worldwide, with more than 600,000 deaths every year1. Human infections are caused by five malaria parasite species, namely P. falciparum, P. vivax, P. ovale, P. malariae, and P. knowlesi. Most clinical malaria mortalities are caused by P. falciparum in sub-Saharan Africa1. Another human malaria parasite species that causes extensive worldwide morbidities outside of sub-Saharan Africa is P. vivax2. The other three species are all more geographically restricted and cause benign malaria infections, except the lethal P. knowlesi3. The unavailability of relevant and practical non-human in vivo models of infections has always been and still is an obstacle to malaria vaccine and drug development. Earlier malaria drug targeting and metabolic studies have relied extensively on avian malaria models like P. gallinaceum and P. lophurae, infecting chickens and ducks, respectively4. Thereafter, rodent malaria species were gradually introduced in various vaccines and drug targeting studies as in vivo models. Over the years, evidence of similarities of the biology and host-parasite interactions of life cycle stages of rodent malaria models to human malaria species have accumulated.
In particular, rodent malaria models were extremely important to explore and characterize the biology of mosquito and pre-erythrocytic stages5. However, there are four rodent malaria species (P. berghei, P. yoelii, P. chabaudi, and P. vinckei) that have different biological features, the most notable of which are in the blood stages6. Rodent malaria species differ in the synchronicity of blood stages, where blood stages of P. chabaudi and P. vinckei strains are mostly synchronous, while the blood stages of P. berghei and P. yoelii are not6,7. Another notable difference is the self-clearance of blood stages that occurs in some strains (e.g., P. yoelii 17X-NL, P. berghei NK65, and P. vinckei lentum), whereas the blood infection of other strains of the same species could be lethal if left untreated (P. yoelii 17X-L, P. berghei ANKA, and P. chabaudi AS). Moreover, P. yoelii 17X-NL strain and P. berghei ANKA strain preferentially invade reticulocytes8,9,10,11, although these features of P. yoelii and P. berghei strains are not a strict growth requirement12,13,14. Therefore, mice are treated with phenylhydrazine prior to an infection with the blood stages of those parasites to increase the parasitemia and gametocytemia needed for a mosquito infection for the P. berghei ANKA strain and for P. yoelii 17X-NL15,16,17,18,19.
Differences in mosquito stages development also exist among different rodent malaria species, the most notable being the temperature and time required for optimal mosquito stages development and the sporozoite length5,6,20.In pre-erythrocytic stages of rodent malaria species, differences include the rodent species and strain that are most susceptible to infectious sporozoite inoculation, the number of sporozoites needed for inoculation in a susceptible rodent strain, the mammalian cell types needed for in vitro liver stage development assays, and the time to complete liver stage development5,21,22,23,24,25,26,27,28,29,30.
Despite these variabilities, rodent malaria parasites were the favorable models early on for the application of reverse genetic approaches, because they were less time- and resource-consuming with a high probability of success31. In fact, rodent malaria models were the best models, and in many instances the only models, available for numerous years to functionally characterize genes expressed in mosquito and liver stages.
In light of the popularity and amenability of reverse genetic approaches in rodent malaria models, a number of different methodologies have been utilized to analyze the phenotypes of transgenic parasite life cycle stages, especially blood stages. However, some of these methodologies are inconsistent; for instance, comparing infections of blood-stage parasites following an IP injection (which are possibly drained to the peritoneal lymph nodes and, from there, can enter the bloodstream; therefore, the injected parasites do not end up equally in the bloodstream), comparing the mosquito transmission of clones with a different number of serial blood-stage transfers or G number (which could affect gametocytogenesis32,33), or comparing transgenic parasites directly to naive wild-type (WT) parasites that were never subjected to electroporation and positive drug selection and the various unstandardized evaluations of male gamete exflagellation. Therefore, it is crucial to standardize protocols that are simple to follow for the phenotypic analysis of any type of transgenic or WT rodent malaria parasites in the blood and in the mosquito to accommodate for the biological variabilities of rodent malaria parasite species.
Herein, we report on a standardized, detailed experimental protocol for the phenotypic analysis of the blood and mosquito life cycle stages of transgenic or wild-type P. yoelii and P. berghei parasites. These protocols are also applicable to P. chabaudi and P. vinckei parasites.
Protocol
All animal experiments described here were conducted according to the approved protocols of the Institutional Animal Care and Use Committee (IACUC) of Tulane University and the animals ethics committee of Bezmialem Vakif University. All other experimental protocols and the use of recombinant DNA were conducted according to the approved protocols of the Institutional Biosafety Committee (IBC) of Tulane University.
1. Infection of Mice with Blood-stage Parasites for Parasitemia Analysis and Mosquito Infection Assays
- Day -3: optional injection of phenylhydrazine into recipient mice
- Inject outbred SW or CD1 recipient mice (mice that will be used for mosquito infection and mosquito infection assays) intraperitoneally (IP) with 100 µL of phenylhydrazine (50 mg of phenylhydrazine in 5 mL of 1x phosphate-buffered saline (PBS)) using a 26 G needle syringe.
NOTE: For robust mosquito infections of P. berghei and P. yoelii, phenylhydrazine is used to induce reticulocytosis in the recipient mice, which increases the parasitemia and, subsequently, the percentage of gametocytes, and significantly increases the exflagellation of male gametes (Figure 5), which will lead to a better mosquito stages infection for both P. berghei and P. yoelii strains15,16,17,18.
- Inject outbred SW or CD1 recipient mice (mice that will be used for mosquito infection and mosquito infection assays) intraperitoneally (IP) with 100 µL of phenylhydrazine (50 mg of phenylhydrazine in 5 mL of 1x phosphate-buffered saline (PBS)) using a 26 G needle syringe.
- Day -2: injection of frozen parasite stocks into donor mice
- Quickly thaw cryopreserved frozen stocks and immediately inject each donor mouse (typically two mice per genotype) IP with ~200 µL of stock using a 26 G needle syringe.
NOTE: No more than five mice are housed per cage within controlled vivarium settings. - Start checking parasitemia under a 100x microscope objective on Giemsa-stained thin blood smear after 24 h postinfection.Follow section 2.1 of the protocol for Giemsa-stained thin blood smears.
NOTE: Due to the inability to precisely count the surviving viable blood stages after quickly thawing and injecting cryopreserved infected blood, donor mice receive the cryopreserved blood first. The infected erythrocytes of the donor mouse can be precisely quantified and used subsequently.
- Quickly thaw cryopreserved frozen stocks and immediately inject each donor mouse (typically two mice per genotype) IP with ~200 µL of stock using a 26 G needle syringe.
- Day 0: bleeding of the donor mice
- Bleed the donor mice (see section 3.1) when their parasitemia is between 0.1% and 1%, which is usually obtained 2-3 days post-frozen-stock-infection in the case of P. yoelii 17X-NL and P. berghei ANKA strains.
- Use the facial vein puncture (to get between 200 and 500 µL), or terminally bleed the animals by heart puncture (to get between 600 µL and 1.2 mL) of infected blood. Terminally bleed the mice by heart puncture as described in detail in section 3.1.
NOTE: Facial vein bleeding is not a routine method of obtaining blood as it is much more difficult to apply and is very distressful to mice, compared to terminal bleeding. The optimal parasitemia for the bleeding of donor mice is between 0.1%-1% because, at this parasitemia range, there are very few gametocytes (which cannot replicate to produce asexual blood stages) and there are less double- or triple-infected erythrocytes (which makes the quantification less accurate).
- Day 0: quantification of infected blood to prepare doses of infected erythrocytes by serial dilutions
- Prepare serial dilutions of donor blood by adding 100 µL of blood to a microcentrifuge tube (dilution tube 1). Add 900 µL of RPMI medium to tube 1 and mix well by pipetting up and down with the same pipette tip, creating a 1:10 dilution.
- Take 100 µL of the diluted blood from tube 1 and add it to a new microcentrifuge tube (dilution tube 2). Add 900 µL of RPMI medium to tube 2 and mix well by pipetting up and down with the same pipette tip, creating a 1:100 dilution.
- Repeat steps 1.4.1 and 1.4.2 to make two more serial dilutions, creating 1:1,000 (dilution tube 3) and 1:10,000 dilutions (dilution tube 4).
- Add 12 µL of diluted blood from tube 3 and 12 µL of diluted blood from tube 4 to different sides of a hemocytometer and determine the average number of erythrocytes on each side of the hemocytometer. Multiply these numbers by 10. Multiply these numbers by the dilution factor, 1,000 or 10,000, respectively, to determine the number of erythrocytes in 1 µL of each dilution.
- Divide the desired number of infected erythrocytes to be injected by the number of erythrocytes in 1 µL of each dilution to determine the volume of diluted donor blood necessary for injection. Choose the dilution with the most appropriate volume for injection, add the volume to a new microcentrifuge tube, and complete it to 120 µL with RPMI medium.
- Day 0: intravenous injection of the recipient mice
- Place the recipient mice under a heat red lamp to dilate their veins for injection. Load the parasite doses in a 27 G insulin syringe and place the recipient mouse in a restrainer.
- Inject 1 million or 10,000 infected erythrocytes intravenously (IV) using the 27 G insulin syringe in each recipient mouse for mosquito infection assays or for asexual and sexual blood-stage growth assays, respectively. Continue to maintain the mice under standard housing conditions.
NOTE: An IP injection is an indirect method of introducing parasites into the bloodstream, as parasitized erythrocytes have to pass through the draining lymph nodes of the peritoneal cavity to reach the blood. This makes an IP injection an indirect delivery route for injecting a precise number of parasites into the bloodstream. Therefore, the IV route is preferred, as it delivers accurate parasite doses directly into the bloodstream, as shown in Figure 3 and described in the Representative Results.
2. Determination of the Blood-stage Parasite Load for Sexual and Asexual Stages
NOTE: In this section, standardized phenotypic evaluation methods of malaria parasite blood stages are listed. These methods are useful in the evaluation of novel antimalarial or vaccine candidates or even gene knockout on sexual and asexual stages development in the same experimental mice. Of note, P. chabaudi and P. vinckei are also very rational and important alternative options for these types of assays, especially in drug screening.
- Determination of the parasitemia of asexual stages and male vs. female gametocytes by Giemsa-stained thin blood smears for parasite growth assays
- Using a 26 G or 27-G needle, prick the mouse's tail and collect the blood droplet on a microscope slide. Use the short edge of another microscope slide to quickly smear the blood across the slide.
- Fix the slide with 100% methanol for at least 1 min, only after the blood has completely dried on the slide. Stain in 10% Giemsa for 10-15 min. Wash the slide with single- or double-distilled water and allow it to dry.
- Read the slide using a 100x objective and oil immersion under a light microscope. For an accurate parasitemia measurement, check at least a total of 6,000 erythrocytes, which can be obtained by counting at least 30 or 40 microscopic grids with an average number of 200 or 150 erythrocytes (RBCs) per grid, respectively. Count the total number of RBCs in the first and last grids to determine the average number of red blood cells for all grids.
- Count the total number of infected erythrocytes per grid. Male and female gametocytes can be counted separately to determine gametocytemia but should also be included in the total parasitemia count.
- Determine the total parasitemia percentage by dividing the total number of parasitized erythrocytes by the total number of RBCs observed. Multiply this number by 100 to determine the parasitemia percentage.
- Determine the gametocytemia by dividing the total number of gametocytes counted by the total number of RBCs observed. Multiply this number by 100 to determine the gametocytemia percentage per mouse.
NOTE: The only reliable method of differentiation between different asexual and sexual blood stages is the use of a microscope to evaluate Giemsa-stained thin blood smears, as each of the stages has different morphology, with the exception of immature gametocytes, which are mostly indistinguishable from asexual blood stages.
- Determination of the blood-stage parasitemia by flow cytometry for fluorescent reporter protein-expressing WT-like parasite strains
- Label two microcentrifuge tubes for each mouse sample, designating one tube for a 1:1,000 dilution and the other for a 1:2,000 dilution. Add 1.5 µL of 1x heparin to the 1:1,000 dilution tube.
- Using a 26 G or 27 G needle, prick the mouse's tail and transfer 1.5 µL of blood to the tube containing 1.5 µL of 1x heparin (the 1:1,000 dilution tube). Mix the blood and the 1x heparin gently together by pipetting up and down.
- Add 1,497 µL of RPMI medium to the microcentrifuge tube, and gently pipette up and down to mix well.
- Transfer 500 µL of the mixture from the 1:1,000 dilution tube to the 1:2,000 dilution tube. Add 500 µL of RPMI medium to the 1:2,000 tube and gently mix by pipetting up and down.
- Follow the manufacturer's appropriate start-up protocol for the flow cytometer. Run the samples at a slow flow rate and set the detection for at least 20,000 events.
NOTE: There is an alternative practical assay to measure parasitemia via tricolor FACS sorting, whether parasites express fluorescent markers or not34. However, the assay described here offers an accurate measure of parasitemia in live parasites without the addition of any DNA-binding dyes and the use of FACS sorting.
- Determination of the exflagellation rate of male gametes per microliter of infected mouse blood
- Prepare a 1:10 dilution microcentrifuge tube with 40 µL of incomplete ookinete medium (RPMI + sodium bicarbonate + hypoxanthine + xanthurenic acid) and 5 µL of 1x heparin.
- Using a 26 G or 27 G needle, prick the mouse's tail and transfer 5 µL of blood (using a micropipette) quickly to the tube containing the incomplete ookinete media + heparin. Mix gently and load 12 µL of the 1:10 blood dilution tube onto a hemocytometer and leave it at room temperature (20-24 °C).
- Start counting the exflagellation events 9-10 min after preparing the 1:10 dilution. Use a phase contrast light microscope with 40x magnification.
- Multiply the average number of exflagellation events counted by 10 and then multiply by the dilution factor (10) to determine the average number of exflagellation per microliter of infected blood.
NOTE: This assay measures the number of male gamete exflagellation events in a 1 µL volume of blood, which can be quantified using a hemocytometer. An incomplete ookinete medium18 is mixed with the blood to closely mimic the conditions during which exflagellation occurs inside the mosquito midgut shortly after a blood meal.
3. Isolation and Processing of Blood-stage Parasites from Infected Erythrocytes and Frozen Stock Preparation
- Terminal bleeding of infected rodents by heart puncture
- Prepare a 26 G needle syringe that contains approximately ≤20 µL of 1x heparin (200 units/mL) to collect the blood of the donor mouse by heart puncture.
- Use a slow flow of CO2 to euthanize the mouse before bleeding. Alternatively, anesthetize the mouse with isoflurane (which has to be maintained before and during the skin incision to expose the heart).
- Lay the mouse on its back and pin down its appendages. Make a small incision in the skin near the base of the sternum by pulling up the skin with forceps and cutting it with scissors. Pull the skin apart at the site of the incision to expose the diaphragm and the top of the abdominal cavity.
- Hold the base of the rib cage with forceps and cut through the diaphragm to enter the thoracic cavity; be careful not to damage the heart or lungs. Pin back the rib cage to expose the heart.
- Gently puncture the heart and slowly pull up on the syringe plunger to collect ~1 mL of blood and transfer it to a microcentrifuge tube.
- Preparation of frozen stocks from infected blood
- Bleed the mice (see section 3.1) with blood parasitemias that are between about 2% and 5%, with a significant amount of ring stages and not as many gametocytes.
- Mix the desired portion of blood with freezing solution (10% glycerol in Alsever's solution) in a 1:2 ratio and store in cryogenic vials. Freeze in liquid nitrogen.
- Isolation and purification of blood-stage parasites for the extraction of DNA, RNA, and proteins
- Bleed mice (see section 3.1) with blood parasitemias that are higher than 0.5%.
- Prepare a 10 mL syringe by removing the plunger and inserting a small cotton swab. Pack down the cotton to the bottom of the syringe. Fill it with cellulose until the level of cellulose reaches the 3 mL mark but does not exceed the 4 mL mark on the syringe.
- Secure the syringe into a clip on a ring stand and place a waste collection tube below it. Add 5 mL of PBS to the syringe and allow it to flow through into the waste tube.
- Prepare frozen stocks if needed, using only 100-300 µL of blood. Add the rest of the infected blood (400-700 µL) to the column. Collect flow through with a waste tube until the droplets start to turn red. Once the droplets start to turn red, place a new 14-mL collection tube under the syringe to collect the flow through.
- Once the flow starts to slow, add PBS to the syringe and collect the flow through in the 15 mL tube until a total volume of 14 mL has been reached. Collect the remaining flow through with a waste tube.
- Centrifuge the 14 mL tube with blood/PBS mixture for 8 min at 250 x g with no brakes. Remove the supernatant and add 14 mL of chilled saponin (50 mg of saponin in 50 mL of PBS). To dislodge the pellet, invert the tube multiple times and vortex if necessary.
- Centrifuge the tube for 8 min at 1,217 x g with no brakes. Remove the supernatant, leaving about 0.5 mL of the solution above the pellet.
- Resuspend the pellet in the solution using a micropipette and transfer it to a microcentrifuge tube. Add 500 µL of PBS to the 14 mL tube to wash any residual parasites that are left in the tube. Add this to the same microcentrifuge tube.
- Centrifuge the tube for 2 min at 6,010 x g. Remove the supernatant and resuspend 1 mL of PBS. Centrifuge for 2 min at 9,391 x g.
- Proceed to extract genomic DNA, total RNA, and protein.
- For genomic DNA extraction, remove the supernatant and resuspend the pellet in 200 µL of PBS, and then follow any suitable protocol of gDNA extraction.
- For total RNA extraction, remove the supernatant, resuspend and vortex the pellet in 1 mL of any phenol- and guanidine isothiocyanate-based solution or any other suitable reagent and, then, follow any suitable protocol of total RNA extraction.
- For total protein extraction, remove the supernatant, resuspend the pellet in an appropriate volume of 6x sodium dodecyl sulfate (SDS)-loading dye or any other suitable reagent and, then, follow any suitable protocol of total protein processing.
4. Mosquito Infection Assays
NOTE: The mosquito is the primary host of the malaria parasites where sexual reproduction takes place. The infection of mice to transmit malaria parasites to mosquitoes is conducted by an IV injection of at least 1 million blood stages, followed by feeding an infected mouse (from each genotype that displays the highest male gamete exflagellation rate) to Anopheles mosquitoes in a cage at day 3 post-mouse-infection. The IV injection with 1 million blood-stage parasites in phenylhydrazine-treated mice will ensure the development of male and female gametocyte at a faster and higher rate. Mosquitoes infected with P. yoelii and P. berghei are incubated at 24 °C and 20-21 °C, respectively, to allow for the best possible mosquito stages development6.
- Mosquito feeding and determination of the number of ookinetes per mosquito
- Starve adult female Anopheles stephensi or A. gambiae mosquitoes (4-7 days old) for 8-12 h prior to feeding. Allow adult mosquitoes to feed for at least 15 min on the infected anesthetized mice (injected with an appropriate dose of ketamine/xylazine) with the highest exflagellation rate (measured in section 2.3).
NOTE: Ketamine/xylazine working solution is prepared by the 1:5 dilution of the stock solution in saline and IP injecting 100 µL per mouse. For a 10 mL stock solution, 1 mL of xylazine (100 mg/mL) is added to 9 mL of ketamine (100 mg/mL). - Remove unfed female mosquitoes and male mosquitoes with a mouth aspirator.
- At 18-20 h postfeeding, collect 20-30 mosquitos and place them in the freezer for not less than 10 min to ensure death. Using a binocular dissection scope, dissect out 20-30 blood-filled midguts with two 26 G or 27 G needles or a needle and forceps in RPMI or PBS dissection medium and transfer the midguts to a microcentrifuge tube containing 200 µL of RPMI or PBS (For dissection method please see section 4.3).
- Centrifuge the collection tube for 1 min at 700 x g. Grind the pelleted midguts using a pestle. Repeat this step.
- Transfer 50 µL from the tube of ground midguts to a new microcentrifuge tube. Add 200 µL of RPMI or PBS to create a 1:5 dilution.
- Transfer 12 µL to a hemocytometer and incubate it at room temperature for 5 min to allow the contents to settle.
- Determine the average number of ookinetes by using a light microscope with a phase contrast and 40x magnification.
- Multiply the average number of mature ookinetes by 10 and then by the dilution factor of 5 to determine the average total number of ookinetes.
- Divide the average number of ookinetes by the number of blood-fed mosquitoes dissected to determine the number of ookinetes per blood-fed mosquito.
- Starve adult female Anopheles stephensi or A. gambiae mosquitoes (4-7 days old) for 8-12 h prior to feeding. Allow adult mosquitoes to feed for at least 15 min on the infected anesthetized mice (injected with an appropriate dose of ketamine/xylazine) with the highest exflagellation rate (measured in section 2.3).
- Determination of the number of early oocysts for fluorescent reporter protein-expressing WT-like parasite strains
NOTE: The aim of this assay is to determine the number of live parasites expressing green fluorescent protein (GFP) that established a full infection of the mosquito midguts and formed early spherical oocysts. This assay determines if the ookinetes (their development estimated in the previous assay) completed their functions by the traversal through the mosquito midgut epithelial cells and the transformation to early oocysts on the basal lamina side of the midgut epithelia or not. This is another assay which makes great use of the parasites expressing GFP, as counting early oocysts at this stage will be nearly impossible without tedious immunostaining.- Dissect the midguts (see section 4.1) of 20-30 blood-fed mosquitoes, at day 3 or 4 post-mosquito-feeding (pmf) for P. yoelii 17X-NL, and at day 6 or 7 pmf for P. berghei ANKA, with two 26 G or 27 G needles or a needle and forceps in RPMI or PBS dissection medium (For dissection method please see section 4.3).
- Spread 40-50 µL of dissection medium on the horizontal midline of the longer edge of the glass slide. Transfer the midguts to this line, one at a time, during dissection, and add more medium to the midguts, if needed, to avoid drying out.
- Place a coverslip gently on the dissected midguts, and seal it with nail polish.
- Using the 10x or 20x objective of the fluorescence microscope with the green fluorescence filter, count the number of early oocysts on each midgut, in at least 20 midguts.
- Determination of the number of oocyst sporozoites per mosquito
- Dissect the midguts of at least 20-30 mosquitoes with two 26-G or 27-G needles or a needle and forceps in RPMI or PBS dissection medium and collect the dissected midguts in 200 µL of RPMI.
- Hold the lower abdomen segments firmly in place with one needle (or forceps) and lightly push the thorax in an upward direction with the other needle at the junction between the thorax and the abdomen. Do carefully short pushes until the thorax separates from the abdomen. As soon as the separation starts the white midgut will appear stretched, and then the midgut will be cut from the esophagus end using the same needle that was used to separate the thorax. If still attached to the abdomen, the same needle can be used to pull the midgut away from the abdomen. Transfer the midguts of all mosquitoes to the collection tube by collecting them from the medium with a needle, one at a time.
- Centrifuge the collection tube for 1 min at 700x g to settle the midguts to the bottom of the collection tube and grind them with a pestle. Repeat this step.
- Transfer 12 µL to a hemocytometer and incubate it at room temperature for 5 min to allow its contents to settle.
- Count the oocyst sporozoites using a 40X objective of a light microscope. Use 1:5 and/or 1:10 dilutions if mosquito debris prevents the precise counting of the sporozoites or if the number of sporozoites is too high to count accurately.
- Calculate the average total number of oocyst sporozoites by multiplying the average number of sporozoites by 10 and, then, multiplying that number by the dilution factor, if any, and by the total volume (200 µL).
- Divide the average total number of sporozoites by the number of mosquitoes dissected to calculate the average number of oocyst sporozoites per mosquito.
NOTE: A common mistake for measuring the success or productivity of mosquito stages infection is the counting of the number of oocysts at later stages of oocyst development. This is due to some oocysts being vacuolated at later time points of oocyst development, and others fail to develop sporozoites, even on the same midgut. The best and most reliable method to determine the productivity of mosquito stages infection is the counting of the average number of midgut oocyst sporozoites at days 10-12 post-mosquito-infection for P. yoelii and at days 12-14 post-mosquito-infection for P. berghei.
- Determination of the number of salivary gland sporozoites per mosquito
NOTE: The ultimate evaluation of the full completion of mosquito stages development is accomplished by estimating the number of sporozoites that invaded the salivary gland, which are the transmissible and infectious stages to vertebrates. The invasion of the salivary gland is established after the completion of oocyst sporozoite development, egress into the hemolymph, attachment to the basal lamina of the salivary gland acinar cells, and traversal of the acinar cells to reach the salivary glands ducts5. Therefore, this assay is also an evaluation of the success of all of these processes or not. Nonetheless, an estimation of hemolymph sporozoite numbers would allow the differentiation between defects in the egress from oocysts and defects in the invasion of salivary glands35. The purification of salivary gland sporozoites allows conducting multiple functional assays on sporozoite motility and invasion phenotypes. The salivary gland sporozoites can also be used for the in vivo infection of mice and in immunization studies36,37. Moreover, the most reproducible stages available to establish an in vitro liver stage invasion or a development assay is by the use of salivary gland sporozoites.- Dissect the salivary glands of 50-100 female mosquitoes, at days 14-16 pmf for P. yoelii and at days 17-20 pmf for P. berghei, preferably with two 26 G or 27 G needles, in RPMI or DMEM medium kept on ice and supplemented with 5% fetal bovine serum (FBS) or/bovine serum albumin (BSA) for P. yoelii sporozoites or 3% FBS/BSA for P. berghei sporozoites. If the sporozoites are to be used in hepatoma in vitro assays, add 1% penicillin/streptomycin to the dissection medium. There are generally two methods for the dissection of the salivary glands. The first method is time-consuming but leads to the dissection of salivary glands with very little or no contaminating tissues. The second method is less time consuming but leads to the collection of a significant amount of contaminating tissues. The first method is detailed here.
- Hold the upper abdomen segments in place with the beveled side of one needle and carefully push the head very lightly (in very short pushes) at the junction between the head and thorax in an upward direction until the head is carefully separated from the thorax without tearing up the glassy salivary glands, the exposed salivary glands are then separated from the head with the same needle that pushed the head upward.
- The dissected salivary gland will be raised from the dissection medium using a short glass Pasteur pipette.
- When all salivary glands have been dissected and harvested, talent will place the tube containing the midguts into the centrifuge (1min, 700 x g).
- Count sporozoites using a hemocytometer under a light microscope set to Phase 2 contrast and 40x magnification. If the number of the mosquitos dissected ≥40, dilute the salivary glands to 1:5 or 1:10, depending on the expected infectivity yield (determined in earlier assays).
- Multiply the average number of sporozoites by 10 and the dilution factor (if any). Multiply by the total volume to determine the average number of salivary gland sporozoites. Divide by the number of dissected female mosquitoes to determine the average salivary gland sporozoites per mosquito.
NOTE: The problem with the dissection of the salivary glands is their small size and glassy transparent appearance. Thus, it is very difficult to isolate the salivary glands and, therefore, they are usually dissected out as a lump sum with other tissues from the frontal part of the thorax, which is also important to protect the salivary glands from rupture during the collection.
Representative Results
The success of applying reverse genetic tools and techniques to malaria parasites has revolutionized the field of malaria research, with the ability to add, delete, or modify specific genomic segments of several Plasmodium species39. Importantly, dispensable genomic loci have been identified and used successfully to introduce fluorescence protein markers in rodent and human malaria parasites by double homologous recombination, to ensure a stable expression in all life cycle stages40,41,42. An example of these WT-like transgenic parasites is Py230p(-) parasites, which have been generated in our lab, and showed no apparent defect in the development of blood and mosquito life cycle stages15,16,17. These transgenic reporter parasites expressed eGFP, under the control of the strong and constitutive promoter of PyHSP70, in blood stages (Figure 1A) ookinetes (Figure 1B), young oocysts (Figure 1C) on Anopheles stephensi midguts, and in sporozoites isolated from the salivary glands of Anopheles stephensi females (Figure 1D). Thus, the eGFP-expressing blood parasites made it much easier and less time-consuming to evaluate blood-stage parasitemia using flow cytometry between different genotypes of transgenic parasites or between drug-treated and -untreated in drug-targeting assays using transgenic reporter parasites.
In order to confirm that there is no quantitative difference between the use of flow cytometry and the more tedious parasitemia estimation by microscopy, the eGFP-expressing Pyp230p(-) was used to estimate the percentage of parasitized erythrocytes by flow cytometry and by Giemsa-stained thin blood smear in a group of Swiss Webster mice IV-infected with 10,000 parasitized erythrocytes. The flow cytometry parasitemia% values corresponded directly to the estimated parasitemia% by monitoring Giemsa-stained thin blood smears, which were estimated by two expert scientists (Figure 2). This represents a more accurate alternative to the tedious and prone-to-human-error method of microscopy in the determination of the growth rate of blood-stage parasites.
An important discrepancy associated with the infection of rodents with malaria parasites blood stages is the choice of route of infection, with a strong preference in the literature for the IP compared to IV route of infection, as it is less time-consuming. In order to compare these two routes of infection, two groups of five BALB/c mice were infected with 1,000 eGFP-expressing Pyp230p(-) parasitized erythrocytes per mouse, either through IV or IP routes. The parasitemia was monitored daily using flow cytometry for a period of 4 days. A statistically significant decrease in the IP-infected group parasitemia% compared to the IV-infected group was noted on all days tested (Figure 3). This provides evidence that the IV infection route is a more quantitatively accurate route of infection for assays with the malaria parasite blood stages.
Nonetheless, one limitation to the use of flow cytometry to evaluate blood-stage parasitemia is the differentiation between sexual and asexual stages and between male and female gametocytes. Therefore, the estimation of the percentages of each of the different asexual and sexual stages (Figure 4) has to depend on a morphology evaluation of Giemsa-stained thin blood smear. Despite the apparent different morphology of mature male and female gametocytes (Figure 4), immature sexual stages are often indistinguishable from asexual stages.
One essential function of the male gametes upon emergence from male gametocyte in the mosquito midgut is the male gamete exflagellation, which is a very critical step in the transmission that must happen within a very short period of time. Variable methods used to evaluate this in many different systems have been described. Herein, we show a standardized method that can be repeated in any simple lab setting. We evaluated male gamete exflagellation with or without the injection of phenylhydrazine into recipient mice (Figure 5). We could show that the phenylhydrazine treatment significantly (four fold) increased the rate of male gamete exflagellation, which in turn will increase the fertilization rate and the number of all subsequent mosquito stages.
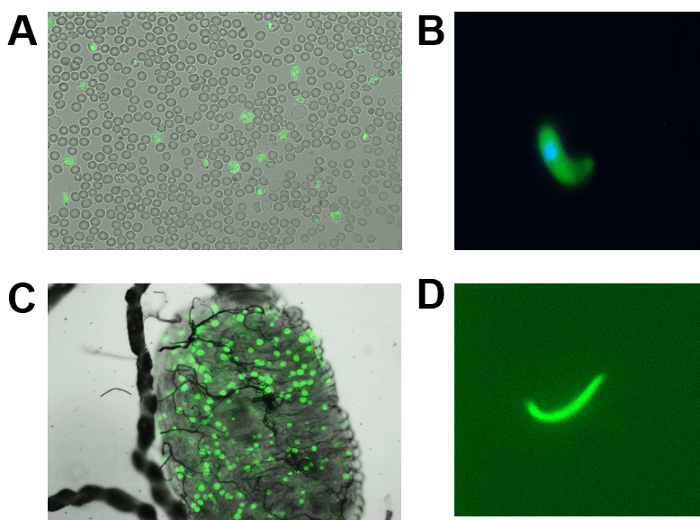
Figure 1: The development of P. yoelii p230p(-) parasites constitutively expressing eGFP in blood and mosquito stages. (A) Image of mixed blood-stage parasites (1,000X magnification). (B) Image of ookinete (400X magnification). (C) Image of an Anopheles stephensi mosquito midgut infected with day 4 pmf early oocysts of live p230p(-) parasites expressing eGFP (100X magnification). (D) This panel shows a live image of a P. yoelii p230p(-) salivary gland sporozoite, dissected out at day 15 pmf, expressing eGFP (400X magnification). Please click here to view a larger version of this figure.
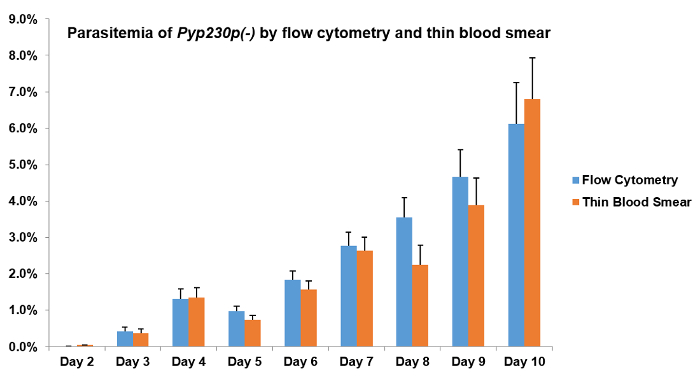
Figure 2: Flow cytometry and microscopy evaluations of average blood-stage parasitemia of P. yoelii p230p(-) parasites are not significantly different. A group of four Swiss Webster mice was intravenously infected with 10,000 parasitized erythrocytes of Pyp230p(-) per mouse and the average blood-stage parasitemias% were recorded daily for 7 days by flow cytometry and by the microscopic evaluation of Giemsa-stained thin blood smears from a total of 20,000 and ~6,000 erythrocytes, respectively. The microscopic examination results shown are the average of two readings by two expert scientists per slide, and the time for evaluating the parasitemia of each slide was at least 10 minutes by each scientist. No significant differences could be detected on any of the days shown here. The mean values for all parasite strains were analyzed with the two-tailed t-test. The error bars represent the standard deviation. Please click here to view a larger version of this figure.
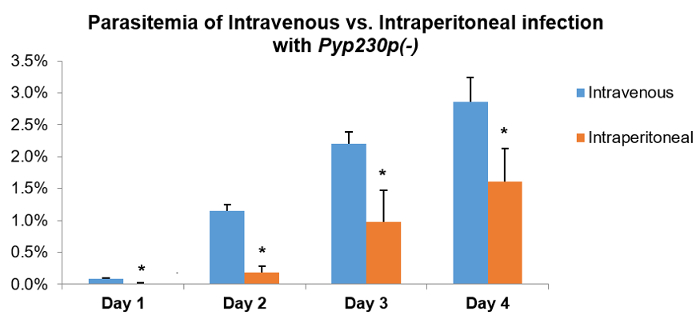
Figure 3: An intravenous injection of P. yoelii p230p(-) parasites yields significantly different blood-stage parasitemia from an intraperitoneal injection. Two groups of five BALB/c mice were infected with 1,000 parasitized erythrocytes of Pyp230p(-) per mouse, either through the intravenous or intraperitoneal route, and the average blood-stage parasitemias% were recorded daily for 4 days by flow cytometry from a total of 20,000 erythrocytes. A statistically significant reduction (denoted by an asterisk) of blood-stage parasitemia could be detected for all days tested in the intraperitoneal route group compared to the intravenous route group. The mean values for all parasite strains were analyzed with the two-tailed t-test. The error bars represent the standard deviation. Please click here to view a larger version of this figure.
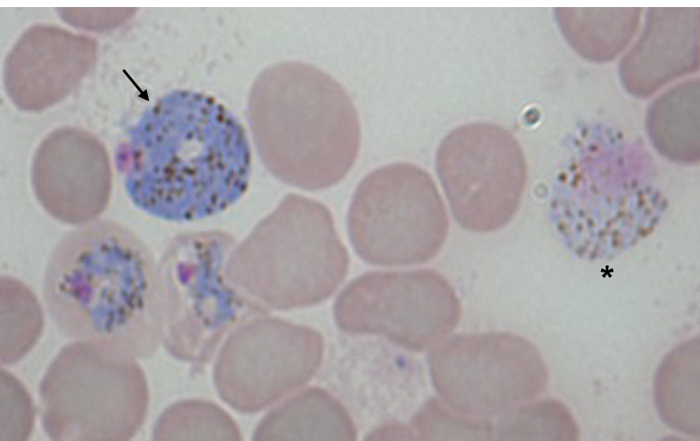
Figure 4: Morphology of P. yoelii gametocytes in a Giemsa-stained thin blood smear. An image of a Giemsa-stained thin blood smear (1,000X magnification) of a Swiss Webster mouse infected with WT P. yoelii 17X-NL strain shows the typical blueish colored female gametocyte on the left side (denoted by an arrow) and the pinkish colored male (denoted by an asterisk) gametocyte on the right side of the image. The other stages shown are asexual blood stages. Please click here to view a larger version of this figure.
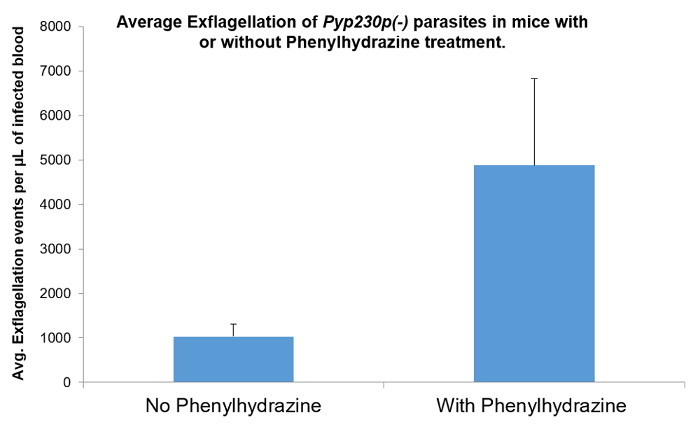
Figure 5: The effect of phenylhydrazine on male gamete exflagellation. The effect of phenylhydrazine injected in recipient mice 5 days prior to the male gamete exflagellation rate estimation of P. yoelii. Phenylhydrazine significantly increases the rate of male gamete exflagellation, which leads to a higher mosquito stages infection post-mosquito-feeding. The mean values for all parasite strains were analyzed with the two-tailed t-test. The error bars represent the standard deviation. Please click here to view a larger version of this figure.
Discussion
In spite of the similarity in the general biology of their life cycles to that of human malaria parasites, mouse malaria models also have many dissimilarities to human Plasmodium species that would limit their use as reliable in vivo models. For instance, with the exception of live-attenuated parasites as vaccines, all vaccine studies with subunit and DNA and other vaccines gave excellent results in the mouse model, but in humans living in endemic areas, the results were far from satisfactory.
Another problem is the difference of life cycle stage infectivity from one mouse strain to another, and sometimes from one animal vendor to another for the same mouse strain. Moreover, the main two rodent malaria species that are widely used as preferred in vivo malaria models, P. berghei and P. yoelii, do not exhibit a synchronous blood-stage cycle, which is completely different from any human malaria parasite. However, the benefits of using mouse malaria parasites as in vivo models outweigh by far these dissimilarities, which can be also overcome by more in-depth analyses of the molecular drives of these limitations. Nevertheless, these limitations are mostly displayed by the rodent malaria blood-stage parasites, but not as much for the other life cycle stages of the malaria parasite.
Although blood stages are important for various vaccine, drug targeting, immunology, and functional genomic studies, there is a scarcity of standardized methods and protocols that concentrate on the phenotypical analysis and functional assays that involve rodent malaria parasite blood-stage parasites, with more focus on mosquito transmission and transfection protocols. Therefore, the methods in this article will help to provide standardized and simplified protocols for studying the pathogenic stages of the rodent malaria parasites.
Divulgations
The authors have nothing to disclose.
Acknowledgements
Ahmed Aly is supported by funding to Bezmialem Vakif University from the Turkish Ministry of Development grant 2015BSV036, and by funding provided by the Tulane University School of Public Health and Tropical Medicine, and by funding from NIH-NIAID for R21Grant 1R21AI111058-01A1.
Materials
| Heparin | Sigma | 375095-100KU | |
| Xanthurenic acid | Sigma | D120804-5G | |
| Hypoxanthine | Sigma | H9377-25G | |
| Alsever's solution | Sigma | A3551-500ML | |
| Sodium Bicarbonate | Sigma | S5761-500G | |
| Phenylhydrazine | Sigma | P26252-5G | |
| Glycerol | Sigma | G5516-500ML | |
| Giemsa | Sigma | GS1L-1L | |
| 26G x 3/8 Precision Glide Needle, | Becton Dickinson | 305110 | |
| 1 ml TB Syringe, 26G x 3/8 | Becton Dickinson | 309625 | |
| 1 cc Insulin Syringe, U-100 27G | Becton Dickinson | 329412 | |
| Isoflurane, USB | Piramal | 2667- 46- 7 | |
| PBS, pH 7.4 | Gibco | 10010049 | |
| RPMI | Gibco | 22400105 | |
| DMEM | Gibco | 11995065 | |
| Pencillin/ Streptomycin | Gibco | 10378016 | |
| Fetal Bovine Serum | Gibco | 10082147 | |
| Fiber Glass Wool | Corning | 3950 |
References
- Who/Unicef Report. Malaria Mdg Target Achieved Amid Sharp Drop in Cases and Mortality, but 3 Billion People Remain at Risk. Neurosciences (Riyadh). 21, 87-88 (2016).
- Naing, C., Whittaker, M. A., Nyunt Wai, V., Mak, W. J. Is Plasmodium vivax malaria a severe malaria?: a systematic review and meta-analysis. PLoS Neglected Tropical Diseases. 8, e3071 (2014).
- Millar, S. B., Cox-Singh, J. Human infections with Plasmodium knowlesi–zoonotic malaria. Clinical Microbiology and Infection: The Official Publication of the European Society of Clinical Microbiology and Infectious Diseases. 21, 640-648 (2015).
- Spry, C., Kirk, K., Saliba, K. J. Coenzyme A biosynthesis: an antimicrobial drug target. FEMS Microbiology Reviews. 32, 56-106 (2008).
- Aly, A. S., Vaughan, A. M., Kappe, S. H. Malaria parasite development in the mosquito and infection of the mammalian host. Annual Review of Microbiology. 63, 195-221 (2009).
- Stephens, R., Culleton, R. L., Lamb, T. J. The contribution of Plasmodium chabaudi to our understanding of malaria. Trends in Parasitology. 28, 73-82 (2012).
- Bagnaresi, P., et al. Unlike the synchronous Plasmodium falciparum and P. chabaudi infection, the P. berghei and P. yoelii asynchronous infections are not affected by melatonin. International Journal of General Medicine. 2, 47-55 (2009).
- Cromer, D., Evans, K. J., Schofield, L., Davenport, M. P. Preferential invasion of reticulocytes during late-stage Plasmodium berghei infection accounts for reduced circulating reticulocyte levels. International Journal for Parasitology. 36, 1389-1397 (2006).
- Jayawardena, A. N., Mogil, R., Murphy, D. B., Burger, D., Gershon, R. K. Enhanced expression of H-2K and H-2D antigens on reticulocytes infected with Plasmodium yoelii. Nature. 302, 623-626 (1983).
- Okada, H., et al. A transient resistance to blood-stage malaria in interferon-gamma-deficient mice through impaired production of the host cells preferred by malaria parasites. Frontiers in Microbiology. 6, 600 (2015).
- Walliker, D., Sanderson, A., Yoeli, M., Hargreaves, B. J. A genetic investigation of virulence in a rodent malaria parasite. Parasitology. 72, 183-194 (1976).
- Deharo, E., Coquelin, F., Chabaud, A. G., Landau, I. The erythrocytic schizogony of two synchronized strains of plasmodium berghei, NK65 and ANKA, in normocytes and reticulocytes. Parasitology Research. 82, 178-182 (1996).
- Fahey, J. R., Spitalny, G. L. Virulent and nonvirulent forms of Plasmodium yoelii are not restricted to growth within a single erythrocyte type. Infection and Immunity. 44, 151-156 (1984).
- Srivastava, A., et al. Host reticulocytes provide metabolic reservoirs that can be exploited by malaria parasites. PLoS Pathogens. 11, e1004882 (2015).
- Hart, R. J., et al. Genetic Characterization of Plasmodium Putative Pantothenate Kinase Genes Reveals Their Essential Role in Malaria Parasite Transmission to the Mosquito. Scientific Reports. 6, 33518 (2016).
- Hart, R. J., Ghaffar, A., Abdalal, S., Perrin, B., Aly, A. S. Plasmodium AdoMetDC/ODC bifunctional enzyme is essential for male sexual stage development and mosquito transmission. Biology Open. 5, 1022-1029 (2016).
- Hart, R. J., Lawres, L., Fritzen, E., Ben Mamoun, C., Aly, A. S. Plasmodium yoelii vitamin B5 pantothenate transporter candidate is essential for parasite transmission to the mosquito. Scientific Reports. 4, 5665 (2014).
- Ramakrishnan, C., et al. Laboratory maintenance of rodent malaria parasites. Methods in Molecular Biology. 923, 51-72 (2013).
- Hart, R. J., Abraham, A., Aly, A. S. I. Genetic Characterization of Coenzyme A Biosynthesis Reveals Essential Distinctive Functions during Malaria Parasite Development in Blood and Mosquito. Frontiers in Cellular and Infection Microbiology. 7, 260 (2017).
- Vanderberg, J. P., Yoeli, M. Effects of temperature on sporogonic development of Plasmodium berghei. The Journal of Parasitology. 52, 559-564 (1966).
- Vaughan, A. M., Aly, A. S., Kappe, S. H. Malaria parasite pre-erythrocytic stage infection: gliding and hiding. Cell Host & Microbe. 4, 209-218 (2008).
- Briones, M. R., Tsuji, M., Nussenzweig, V. The large difference in infectivity for mice of Plasmodium berghei and Plasmodium yoelii sporozoites cannot be correlated with their ability to enter into hepatocytes. Molecular and Biochemical Parasitology. 77, 7-17 (1996).
- Hollingdale, M. R., Leland, P., Leef, J. L., Beaudoin, R. L. The influence of cell type and culture medium on the in vitro cultivation of exoerythrocytic stages of Plasmodium berghei. The Journal of Parasitology. 69, 346-352 (1983).
- House, B. L., Hollingdale, M. R., Sacci, J. B., Richie, T. L. Functional immunoassays using an in vitro malaria liver-stage infection model: where do we go from here?. Trends in Parasitology. 25, 525-533 (2009).
- Khan, Z. M., Vanderberg, J. P. Role of host cellular response in differential susceptibility of nonimmunized BALB/c mice to Plasmodium berghei and Plasmodium yoelii sporozoites. Infection and Immunity. 59, 2529-2534 (1991).
- Most, H., Nussenzweig, R. S., Vanderberg, J., Herman, R., Yoeli, M. Susceptibility of genetically standardized (JAX) mouse strains to sporozoite- and blood-induced Plasmodium berghei infections. Military Medicine. 131 (Suppl), 915-918 (1966).
- Nussenzweig, R., Herman, R., Vanderberg, J., Yoeli, M., Most, H. Studies on sporozoite-induced infections of rodent malaria. 3. The course of sporozoite-induced Plasmodium berghei in different hosts. The American Journal of Tropical Medicine and Hygiene. 15, 684-689 (1966).
- Silvie, O., Franetich, J. F., Boucheix, C., Rubinstein, E., Mazier, D. Alternative invasion pathways for Plasmodium berghei sporozoites. International Journal for Parasitology. 37, 173-182 (2007).
- Tarun, A. S., et al. Protracted sterile protection with Plasmodium yoelii pre-erythrocytic genetically attenuated parasite malaria vaccines is independent of significant liver-stage persistence and is mediated by CD8+ T cells. The Journal of Infectious Diseases. 196, 608-616 (2007).
- Weiss, W. R., Good, M. F., Hollingdale, M. R., Miller, L. H., Berzofsky, J. A. Genetic control of immunity to Plasmodium yoelii sporozoites. The Journal of Immunology. 143, 4263-4266 (1989).
- Philip, N., Orr, R., Waters, A. P. Transfection of rodent malaria parasites. Methods in Molecular Biology. 923, 99-125 (2013).
- Janse, C. J., Ponzi, M., Sinden, R. E., Waters, A. P. Chromosomes and sexual development of rodent malaria parasites. Memorias do Instituto Oswaldo Cruz. 89 (Suppl), 43-46 (1994).
- Sinha, A., et al. A cascade of DNA-binding proteins for sexual commitment and development in Plasmodium. Nature. 507, 253-257 (2014).
- Malleret, B., et al. A rapid and robust tri-color flow cytometry assay for monitoring malaria parasite development. Scientific Reports. 1, 118 (2011).
- Aly, A. S., Matuschewski, K. A malarial cysteine protease is necessary for Plasmodium sporozoite egress from oocysts. The Journal of Experimental Medicine. 202, 225-230 (2005).
- Aly, A. S., Lindner, S. E., MacKellar, D. C., Peng, X., Kappe, S. H. SAP1 is a critical post-transcriptional regulator of infectivity in malaria parasite sporozoite stages. Molecular Microbiology. 79, 929-939 (2011).
- Aly, A. S., et al. Targeted deletion of SAP1 abolishes the expression of infectivity factors necessary for successful malaria parasite liver infection. Molecular Microbiology. 69, 152-163 (2008).
- Ozaki, L. S., Gwadz, R. W., Godson, G. N. Simple centrifugation method for rapid separation of sporozoites from mosquitoes. The Journal of Parasitology. 70, 831-833 (1984).
- de Koning-Ward, T. F., Gilson, P. R., Crabb, B. S. Advances in molecular genetic systems in malaria. Nature Reviews. Microbiology. 13, 373-387 (2015).
- Janse, C. J., Ramesar, J., Waters, A. P. High-efficiency transfection and drug selection of genetically transformed blood stages of the rodent malaria parasite Plasmodium berghei. Nature Protocols. 1, 346-356 (2006).
- Lin, J. W., et al. A novel ‘gene insertion/marker out’ (GIMO) method for transgene expression and gene complementation in rodent malaria parasites. PLoS One. 6, e29289 (2011).
- Manzoni, G., et al. A rapid and robust selection procedure for generating drug-selectable marker-free recombinant malaria parasites. Scientific Reports. 4, 4760 (2014).

