Rat Model of Adhesive Capsulitis of the Shoulder
Summary
This protocol presents an in vivo rat model of adhesive capsulitis. The model includes an internal fixation of the glenohumeral joint with extra-articular suture fixation for an extended time, resulting in a decreased rotational range of motion (ROM) and increased joint stiffness.
Abstract
This proposal aims to create an in vivo rat model of adhesive capsulitis for researching potential treatment options for this condition and other etiologies of comparable arthrofibrosis. The model includes extra-articular fixation of the shoulder in rats via scapular to humeral suturing, resulting in a secondary contracture without invading the intra-articular space and resulting in decreased rotational ROM and increased joint stiffness.
We used 10 Sprague-Dawley rats for the purpose of this study. Baseline ROM measurements were taken before glenohumeral immobilization. The rats were subjected to 8 weeks of immobilization before the fixation sutures were removed and changes in ROM and joint stiffness were evaluated. To evaluate whether immobilization resulted in a significant reduction in ROM, changes in kinematics were calculated. ROM was measured at each time point in the follow-up period and was compared to the baseline internal and external ROM measurements. In order to evaluate the stiffness, joint kinetics were calculated by determining the differences in torque (text and tint ) needed to reach the initial external rotation of 60° and initial internal rotation of 80°.
After the removal of the extra-articular suture fixation on follow-up day 0, we found a 63% decrease in total ROM compared to baseline. We observed continuous improvement until week 5 of follow-up, with the progress slowing down around a 19% restriction. On week 8 of follow-up, there was still an 18% restriction of ROM. Additionally, on follow-up day 0, we found the torque increased by 13.3 Nmm when compared to baseline. On week 8, the total torque was measured to be 1.4 ± 0.2 Nmm higher than initial measurements. This work introduces a rat model of shoulder adhesive capsulitis with lasting reduced ROM and increased stiffness.
Introduction
Adhesive capsulitis of the shoulder is frequently referred to as frozen shoulder or shoulder contracture. It is characterized by restricted glenohumeral motion and pain, presumably as a result of advanced fibrosis and joint contracture1,2,3. The condition involves fibroblast and myofibroblast cell recruitment with a resultant dense collagen matrix (types I and III) in the joint capsule2,3. There are many possible risk factors for developing a joint contracture, including gender, diabetes mellitus, hyperthyroidism, traumatic injury, and prolonged immobilization4,5,6.
Effective treatment options are lacking and mostly include physical therapy, with intervention in the form of surgical release in extreme cases that have not improved with conservative care. The best treatment method remains undetermined and has been a subject of great interest for years in the medical field7,8. Development of novel therapeutic options will require a reproducible animal model for the condition that does not rely on intra-articular induced trauma. The optimal adhesive capsulitis model should involve the two main characteristics of the disease: contracture of the shoulder capsule and a prolonged reduction in range of motion (ROM). Schollmeier et al.9 described one of the first joint contracture models by using a cast to develop shoulder contracture in canines. They also reported that changes in ROM and intra-articular pressure returned to normal levels after cessation of immobilization9. However, an important limitation mentioned in the study is the variation in limb position between animals because of the use of a cast technique.In order to obtain a more reproducible model, Kanno et al.10 later presented an adhesive capsulitis rat model using rigid internal fixation of the shoulder. However, although they achieved a significant reduction in ROM with their model, they did not state whether these changes were temporary or long lasting. The aim of our study was to create a suitable in vivo shoulder contracture rat model by investigating the effect of prolonged extra-articular glenohumeral joint immobilization on ROM and joint stiffness.
Protocol
The study was approved by the Institutional Animal Care and Use Committee at Beth Israel Deaconess Medical Center. Care was taken to avoid unnecessary prolonged anesthesia and also to avoid hypothermia. Animals were weighted at each ROM measurement session and monitored for weight loss.
1. Study Subjects
- Use 10 Sprague-Dawley rats that are 13 weeks old at the time of surgery and that range between 250–300 g of body weight.
2. Surgical Procedure
- Under anesthesia and before surgical immobilization, measure the baseline torque as a function of rotation angle between 60° of external rotation and 80° of internal rotation (see Step 5).
- Induce anesthesia with 5% isoflurane through inhalation in an induction chamber, and then sustain with 2% isoflurane through a nose cone throughout the surgery.
- Use a water-based heating element underneath the animal to maintain body temperature during anesthesia.
- For the purpose of pain control, administer sustained-release buprenorphine subcutaneously at a dose of 1.2 mg/kg depending on the rat’s body weight.
- Immobilize the left glenohumeral joints at an angle of 60° of abduction (the angle between the humeral shaft and the scapular spine)10,11.
- Start the surgical procedure with a posterior longitudinal incision, parallel to the humeral shaft. Make the skin incision just below the glenohumeral joint and extend for about 3 cm.
- Use 2 sutures (braided polyester) to immobilize the glenohumeral joint, by piercing through the lateral edge of the scapula and around the distal two thirds of the humeral shaft, and subsequently tighten as illustrated in Figure 1A. Take extra effort to avoid constricting critical structures such as the brachial artery.
Note: The surgical technique has an advantage of being able to limit the glenohumeral joint at 60° of abduction (Z) without affecting other plans (X & Y). By placing sutures around the humerus and through the lateral border of the scapula, an approximation of the two structures is achieved and maintained at the scapular blade plane. After tightening the sutures, the arm is fixed at the resting position when considering X & Y planes and at 60° of abduction when Z plane is considered. This is thought to limit variability between the animals which might be generated from failure of fine tuning the joint position in the three plans.
3. Closing the Incision
- After proper hemostasis, close the skin incision using skin clips.
- Discontinue the anesthesia and allow the animal to recover under supervision in a warm environment. After the animal regains sufficient consciousness to maintain sternal recumbency, return it back to its cage.
- Immediately after the procedure, allow the animals to return to normal activity. Allow the animals to move without restriction, relying mainly on internal sutures to fix the glenohumeral joint.
- To monitor for possible infection, inspect the incision sites daily during the first post-operative week.
- Remove wound clips on the 10th day after surgery.
NOTE: There was no manipulation of muscles during the procedure, and the technique did not involve any intra-articular trauma, thus preserving capsular and articular continuity and anatomic integrity. Neither external restrain nor activity limitation was followed in our protocol. - Provide analgesia using sustained-release buprenorphine, injected subcutaneously at a dose of 1.2 mg/kg starting at anesthesia induction and repeated every 72 h if needed.
4. Suture Removal 8 Weeks After Immobilization
- Induce anesthesia with 5% isoflurane through inhalation in an induction chamber, then sustained with 2% isoflurane through a nose cone throughout the surgery. For the purpose of pain control, administer sustained-release buprenorphine subcutaneously at a dose of 1.2 mg/kg depending on the rat’s body weight.
- Make an incision on top of the scar from the previous procedure.
- Cut the sutures and remove from the humerus and the scapula.
- Close the incision with wound clips.
- Examine the incision site daily during the first week to detect any signs of infection. Monitor pain and distress as well.
- Remove wound clips on the 10th day after surgery.
5. Range of Motion and Joint Stiffness Measurements
- Measure the ROM and passive shoulder mechanics before and after immobilization using a customized device consisting of an arm clamp, a sensor assembly and a pivoting axle11. This is performed pre-operatively (baseline) and continuously after suture removal.
- Measure ROM immediately after suture removal (follow-up day 0), and subsequently twice a week.
- Reduce measurements to once weekly, upon recording less than 10% change from the previous time point.
Note: After testing ROM for weeks, the ROM of each animal did not change drastically between testing time points (less than 10% change). The ROM seemed to plateau at this point, therefore, we felt that decreasing testing frequency from twice a week to once a week was sufficient.
- Carry out measurements under anesthesia using isoflurane via precision vaporizer at 5% for induction and 2% for maintenance for the full length of the procedure (approximately 5 minutes), in order to facilitate measurements efficiently. Use a water-based heating element underneath the animal to maintain body temperature during anesthesia.
- Position the animal properly for ROM measurement with the aid of a laser guide. Position the forelimb on the arm clamp in 90° forward flexion, with the sensing axis aligned with the long axis of the humerus. Secure the forelimb by the wrist and elbow as illustrated in Figure 1B.
- Control passive forelimb rotation by a stepper motor to assess ROM and torque in a consistent manner. Control the stepper motor with a microcontroller. Use inputs from the inclinometer, in conjunction with those from a torque sensor to indicate the start and end of the measurements.
- Connect the microcontroller to a computer and control using an in-house developed MATLAB code.
- For the baseline measurements only, cycle the sensor assembly 3 times between 60° of external rotation and 80° of internal rotation to obtain initial torque measurements (external: text and internal: tint) for later comparison.
- For ROM measurements, use the torque values of each animal at their own baseline measurements (text and tint) as preset input variables in the program to detect changes in rotation ROM. Use the baseline measurements as a comparative stopping point for subsequent ROM measurements. Changes were detected with a 0.2° resolution.
- For stiffness measurements, use the original rotation angles of 60° external rotation and 80° internal rotation as preset input in the program in order to detect changes in torque. Changes were detected with a resolution of 0.01 Nmm.
- Weigh animals on the same day as each ROM assessment. Place the animals on scales and record their masses. These data were used as one of the tools to assess animal health status during the study.
- Following recovery, return rats to their cages and monitor for signs of pain or distress. Throughout this testing, no animal is left unattended until it has regained sufficient consciousness to maintain sternal recumbency.
NOTE: ROM and torque measurements were achieved with a customized device, previously reported by our group {Villa-Camacho 2015}. The device is a custom-made jig that consists of a shaft rotated by stepper motor and controlled by a custom MATLAB script. A torque sensor and inertial measurement unit are used to capture torque and position data during articulation of the specimen.
6. Post-mortem Immunohistologic Analysis
- At the end of the 8-week ROM measurement period, euthanize rats with CO2 exposure.
- Dissect both left (immobilized) and right (healthy control) shoulders by disarticulating the humerus from the ulna, and by sectioning the scapula from the clavicle and thoracic cavity.
- Fix the excised shoulders in a solution of 10% neutral buffered formalin for 3 days, followed by decalcification in a solution of ethylenediaminetetraacetic acid (EDTA) 10% at pH 7.4 for another 2 months.
- During this process, place samples on a shaker in a gentle agitation cycle and store at 4 °C. Monitor decalcification of the glenohumeral joint with weekly microcomputed tomography (uCT) scans.
NOTE: EDTA is a chelating agent that binds calcium ions from the apatite crystal outer surface, progressively reducing the crystal size12,13,14. This process is very slow and gentle, and it is used to detect specific tissue elements that must be preserved for techniques such as immunohistochemistry (IHC). The rate at which the EDTA decalcifies the specimen is dependent on the pH and concentration of the solution. In terms of pH, it can range from 7 to 7.4, with more alkaline solutions accelerating the decalcification speed. However, solutions with higher pH levels can damage key tissue elements. In addition, the usual EDTA concentration for such experiments lies between 10% to 14%, but it is very important to remember that the active agent becomes depleted once it bonds to calcium, so this requires replacement of the decalcification fluid at least 3 to 4 times per week15. - Once the decalcification process was concluded, mount the glenohumeral joints in paraffin stacks for histological sectioning. Orient the samples to allow for coronal slices. Slices obtained at 50% depth of the humeral head (at the center, mid-coronally) are shown in Figure 1. Perform immunohistochemical staining using the peroxidase-anti-peroxidase method to denote the presence of fibrotic tissue in the joint.4,9,10,16
- Perform antigen retrieval by microwave irradiation by submerging the slides in a plastic Coplin jar in a sodium citrate buffer solution (10 mM sodium citrate, 0.05% Tween 20, pH 6.0, preheated for 5 min at 95–100 °C) and placing in a regular microwave oven for 10 minutes at medium power. Allow slides to cool for 30 min at room temperature.
- Perform blocking with goat serum for 30 min, and incubate the specimens with a primary mouse mono-clonal antibody (1:400 dilution) to fibronectin overnight at 4 °C.
- After incubation with the primary antibody, wash specimens twice with PBS (0.5 ug/mL) for 10 minutes; and incubate with a secondary antibody, goat antimouse IgG-peroxidase conjugate (1:400 dilution) for 30 minutes.
- Wash the specimens twice with PBS (2.5 µg/mL) on a shaker for 10 minutes and expose to 3,30-diaminobenzidine tetrahydro-chloride and 30% hydrogen peroxide in the dark for 10 min. Counterstain with Carazzi hematoxylin.
Representative Results
Range of motion
On follow-up day 0, we found a 63% decrease in total ROM compared to baseline (P < .001). We observed a gradual improvement of ROM until week 5 of follow-up, when progression stopped at 19% restriction (P <0.001). The remaining restriction, 18% of total ROM, was still apparent at 8 weeks of follow-up (P <0.001).
Stiffness
On follow-up day 0, we found an increase of 13.3 Nmm in total torque compared with baseline (P <0.001); 8.9 Nmm externally (P = .002) and 4.4 Nmm internally (P <0.001), resulting in a 138.8% increase in external rotation torque, 159.6% increase in internal rotation torque, and a total of 149.2% increased torque overall. On week 8 of follow-up, we found the total measured torque to be 1.4 ± 0.2 Nmm higher than baseline (P = 0.115), with a 0.6 ± 0.1 Nmm increase of external torque (P = 0.369) and 0.7 ± 0.2 Nmm increase of internal torque (P = 0.036). This indicates torque increases 10% externally and 25.7% internally, for an overall 17.9% increase. At the beginning of post-operative week 3, the improvement of stiffness plateaued.
Histologic Results
As seen in Figure 2A, the intact group displayed proper separation between the capsule and articular surface of the femoral head and normal cellular organization. In addition, normal cellular organization was also observed in the synovial tissue and articular cartilage. However, the surgically immobilized group shows evidence of capsular adhesions in the inferior aspect of the glenohumeral joint. Moreover, the surrounding tissue appears to be denser when compared to the intact shoulders, leading to a tighter capsule with a decreased joint space (Figure 2B). Slices stained for fibronectin show an increased capsular thickness in the contracted surgical group (Figure 2B). when compared to healthy controls (Figure 2A). These findings are in agreement with previously reported literature on animal models of joint immobilization10 and support the creation of a proper contracture model.
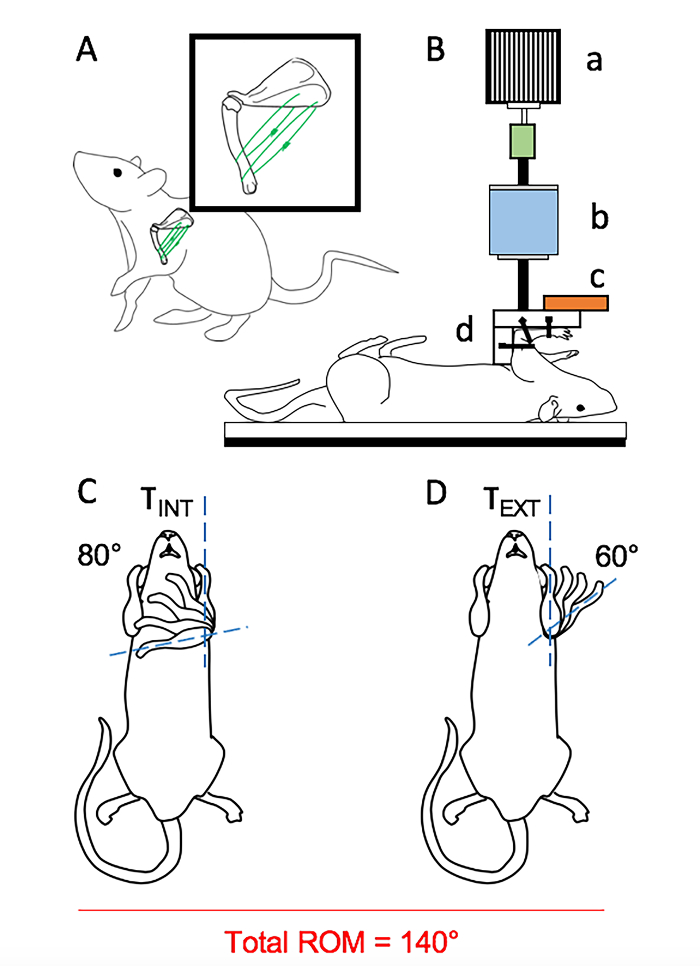
Figure 1: Testing Apparatus. (A) Immobilization of the glenohumeral joint with 2 braided polyester sutures, passed firmly between the lateral edge of scapular and the humerus; (B) Customized device for measurement of ROM and passive shoulder mechanics; a) A stepper monitor. A sensor assembly consisting of b) a reaction torque sensor, and c) an orientation sensor. d) an arm clamp. (C) Internal or (D) external rotation of the glenohumeral joint11. Please click here to view a larger version of this figure.
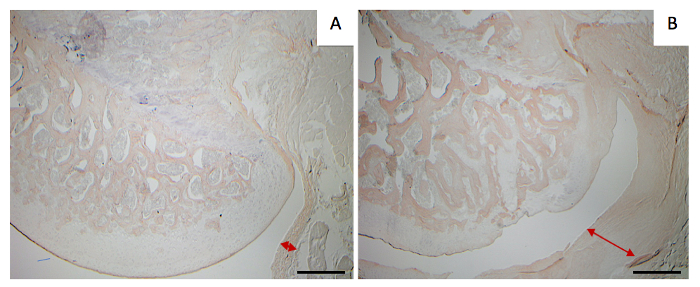
Figure 2: Coronal slices of the humeral head. Image stained for fibronectin (IHC) obtained at 40X magnification. Scale bar = 200 µm. (A) Healthy control. (B) Surgical control. Red arrows outline joint capsular thickness. Please click here to view a larger version of this figure.
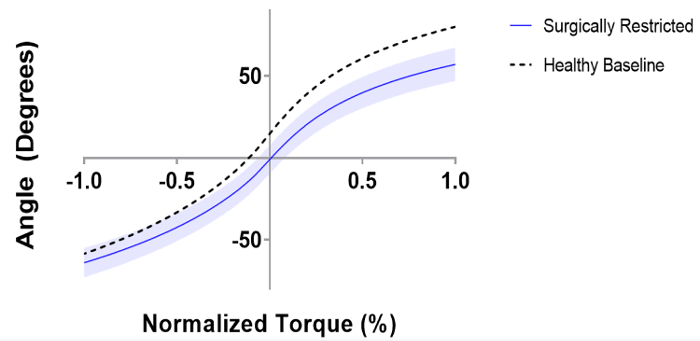
Figure 3: Range of motion versus normalized torque for both healthy and surgically restricted rat glenohumeral joints. Internal Rotation is denoted as positive, external rotation is negative. The shaded region shows the 95% Confidence Interval (CI). Please click here to view a larger version of this figure.
Discussion
This study presents a rat model of adhesive capsulitis of the shoulder through internal fixation of the glenohumeral joint. Furthermore, it shows an extended reduction of total ROM for at least 8 weeks after removal of the fixation. In order to calculate the alterations in ROM at different time points, measurements were compared to animal specific baselines. Conversely, Kanno et al.10 used a standardized torque for all of the animals in order to determine ex vivo ROM changes.
In 2008, Sarver et al.17 reported on joint stiffness of the shoulder resulting from non-surgical external fixation. Their study showed a transient increase in joint stiffness after immobilization of injured and treated shoulders, which was resolved by week 8 of follow-up. Yet, in the present study, we did not find a linear relationship between torque and angle, rather a polynomial fit (Figure 3). Furthermore, we only found a statistically significant difference in joint stiffness compared to baseline during assessment of internal rotation, where a 25.7% increase in stiffness persisted after 8 weeks of immobilization.
We used baseline ROM and stiffness measurements for each animal as their own internal control. Given the possible variation between the animals18, using the contralateral shoulder of the same animal as internal control increases internal validity and helps reduce the number of animals required.
One of the limitations of our study is that the apparatus used to measure ROM does not stabilize the scapula. However, a rat's scapula is more obliquely oriented with even more upward rotation than in humans. Having the rats in supine position should theoretically control for scapular tilting as the scapula rests against the firm plate of the testing jig. A further limitation of the study is that we only assess internal-rotation and external-rotation of the glenohumeral joint. This is partly due to the fact that abduction, flexion, and overhead activities require thorough external fixation or restriction of the scapulothoracic joint during ROM testing, which requires a different system than ours.
The pathogenesis and treatment of adhesive capsulitis continues to be inadequately understood. Regardless of the etiology, it has been shown that it is the contracture of the capsule that causes pain and limits glenohumeral movement1,3,11. Furthermore, it has been suggested that there are inflammatory triggers that result in joint fibrosis, thereby causing contracture10. Although our rat model cannot imitate the initial inflammatory insult of a primary contracture, nevertheless, it adequately replicates the characteristic kinetics of adhesive capsulitis and its pathologic changes10,19. This model invokes a lasting reduction in ROM and increased joint stiffness, allowing for a comprehensive assessment of current and potential therapeutic treatments for shoulder contracture.
Divulgations
The authors have nothing to disclose.
Acknowledgements
The Authors would like to acknowledge Mr. and Mrs. Tom and Phyllis Froeschle for providing financial support towards this project.
Materials
| Sprague-Dawley rats | Charles River Laboratories, Wilmington, MA, USA | 250-300 g | |
| Surgical tool: | |||
| Injection needle | BD 1' 30 guage | ||
| Needle holder | |||
| 5% isoflurane | |||
| 2% isoflurane | |||
| Nose cone | |||
| Skalpel and skalpel holder | No. 11 scalpel | ||
| Curved hemostat forceps | |||
| Staright hemostat forceps | |||
| Tissue retractor | |||
| Toothed tissue forceps | |||
| Plain tissue forceps | |||
| Dissecting scissors | |||
| Suture scissors | |||
| Skin clip applicator | Any standard staples for wound closure | ||
| Immobilization material | Ethicon | No. 2-0 braided polyester ethibond suture was used for immobilization | |
| Other materials: | |||
| Costumized device for ROM: 1)Sensor assembly, 2)pivoting axle, 3)arm clamp | Assembly that is described in relaxin paper and adhesive capsulitis paper | ||
| Orientation sensor (part of sensor assembly) | MicroStrain Inc., Williston, VT, USA | 3DM-GX3-15 | |
| Reaction torque sensor (part of sensor assembly) | Futek Inc., Irvine, CA, USA | TFF400 | |
| Stepper Motor | SparkFun Electronics, Niwot, CO 80503 | https://www.sparkfun.com/products/13656 | |
| Microcontroller | Torino, Italy). | Arduino UNO, R3 | |
| MATLAB code | MATLAB 7.13.0.564, Natick, Ma, USA | ||
| Weight Scale | Ohaus |
References
- Bunker, T. D. Time for a new name for ‘frozen shoulder’. British medical journal. 290 (6477), 1233-1234 (1985).
- Bunker, T. D., Anthony, P. P. The pathology of frozen shoulder. A Dupuytren-like disease. The Journal of bone and joint surgery. British volume. 77 (5), 677-683 (1995).
- Kilian, O., et al. The frozen shoulder. Arthroscopy, histological findings and transmission electron microscopy imaging. Chirurg. 72 (11), 1303-1308 (2001).
- Wang, K., et al. Risk factors in idiopathic adhesive capsulitis: a case control study. Journal of Shoulder and Elbow Surgery. 22 (7), e24-e29 (2013).
- Milgrom, C., et al. Risk factors for idiopathic frozen shoulder. The Israel Medical Association Journal. 10 (5), 361-364 (2008).
- Huang, S. W., et al. Hyperthyroidism is a risk factor for developing adhesive capsulitis of the shoulder: a nationwide longitudinal population-based study. Scientific Reports. 4, 4183 (2014).
- Struyf, F., Meeus, M. Current evidence on physical therapy in patients with adhesive capsulitis: what are we missing. Clinical Rheumatology. 33 (5), 593-600 (2014).
- Song, A., Higgins, L. D., Newman, J., Jain, N. B. Glenohumeral corticosteroid injections in adhesive capsulitis: a systematic search and review. Journal Of Physical Medicine And Rehabilitation. 6 (12), 1143-1156 (2014).
- Schollmeier, G., Sarkar, K., Fukuhara, K., Uhthoff, H. K. Structural and functional changes in the canine shoulder after cessation of immobilization. Clinical Orthopaedics and Related Research. 323 (323), 310-315 (1996).
- Kanno, A., Sano, H., Itoi, E. Development of a shoulder contracture model in rats. Journal of Shoulder and Elbow Surgery. 19 (5), 700-708 (2010).
- Villa-Camacho, J. C., et al. In vivo kinetic evaluation of an adhesive capsulitis model in rats. Journal of Shoulder and Elbow Surgery. 24 (11), 1809-1816 (2015).
- Liu, H., et al. Evaluation of Decalcification Techniques for Rat Femurs Using HE and Immunohistochemical Staining. BioMed Research International. 2017, 9050754 (2017).
- Gonzalez-Chavez, S. A., Pacheco-Tena, C., Macias-Vazquez, C. E., Luevano-Flores, E. Assessment of different decalcifying protocols on Osteopontin and Osteocalcin immunostaining in whole bone specimens of arthritis rat model by confocal immunofluorescence. International Journal of Clinical and Experimental Pathology. 6 (10), 1972-1983 (2013).
- Sanjai, K., et al. Evaluation and comparison of decalcification agents on the human teeth. Journal of Oral and Maxillofacial Pathology. 16 (2), 222-227 (2012).
- Rolls, G. . An Introduction to Decalcification. , (2013).
- Burry, R. W. Controls for immunocytochemistry: an update. Journal of Histochemistry & Cytochemistry. 59 (1), 6-12 (2011).
- Sarver, J. J., et al. After rotator cuff repair, stiffness–but not the loss in range of motion–increased transiently for immobilized shoulders in a rat model. Journal of Shoulder and Elbow Surgery. 17 (1 Suppl), 108S-113S (2008).
- . Variations within Outbred Strains: Know Your Strains and Stocks | Taconic Biosciences Available from: https://www.taconic.com/taconic-insights/quality/variations-within-outbred-strains-know-your-strains-and-stocks.html (2016)
- Liu, Y. L., Ao, Y. F., Cui, G. Q., Zhu, J. X. Changes of histology and capsular collagen in a rat shoulder immobilization model. Chinese Medical Journal. 124 (23), 3939-3944 (2011).

