Evaluation of Photosynthetic Behaviors by Simultaneous Measurements of Leaf Reflectance and Chlorophyll Fluorescence Analyses
Summary
We describe a new technical approach to study photosynthetic responses in higher plants involving simultaneous measurements of chlorophyll a fluorescence and leaf reflectance using a PAM and a spectral radiometer for the detection of signals from the same leaf area in Arabidopsis.
Abstract
Chlorophyll a fluorescence analysis is widely used to measure photosynthetic behaviors in intact plants, and has resulted in the development of many parameters that efficiently measure photosynthesis. Leaf reflectance analysis provides several vegetation indices in ecology and agriculture, including the photochemical reflectance index (PRI), which can be used as an indicator of thermal energy dissipation during photosynthesis because it correlates with non-photochemical quenching (NPQ). However, since NPQ is a composite parameter, its validation is required to understand the nature of the PRI parameter. To obtain physiological evidence for evaluation of the PRI parameter, we simultaneously measured chlorophyll fluorescence and leaf reflectance in xanthophyll cycle defective mutant (npq1) and wild-type Arabidopsis plants. Additionally, the qZ parameter, which likely reflects the xanthophyll cycle, was extracted from the results of chlorophyll fluorescence analysis by monitoring relaxation kinetics of NPQ after switching the light off. These simultaneous measurements were carried out using a pulse-amplitude modulation (PAM) chlorophyll fluorometer and a spectral radiometer. The fiber probes from both instruments were positioned close to each other to detect signals from the same leaf position. An external light source was used to activate photosynthesis, and the measuring lights and saturated light were provided from the PAM instrument. This experimental system enabled us to monitor light-dependent PRI in the intact plant and revealed that light-dependent changes in PRI differ significantly between the wild type and npq1 mutant. Furthermore, PRI was strongly correlated with qZ, meaning that qZ reflects the xanthophyll cycle. Together, these measurements demonstrated that simultaneous measurement of leaf reflectance and chlorophyll fluorescence is a valid approach for parameter evaluation.
Introduction
Leaf reflectance is used to remotely sense vegetation indices that reflect photosynthesis or traits in plants1,2. The normalized difference vegetation index (NDVI), which is based on infrared reflection signals, is one of the most widely known vegetation indices for the detection of chlorophyll-related properties, and it is used in the ecology and agricultural sciences as an indicator of environmental responses in trees or crops3. In field studies, although many parameters (e.g., chlorophyll index (CI), water index (WI), etc.) have been developed and used, few detailed verifications of what these parameters directly (or indirectly) detect have been performed using mutants.
Pulse-amplitude modulation (PAM) analysis of chlorophyll fluorescence is an effective method to measure photosynthetic reactions and processes involved in photosystem II (PSII)4. Chlorophyll fluorescence can be detected with a camera and used for screening photosynthesis mutants5. However, camera detection of chlorophyll fluorescence requires complex protocols such as dark treatment or light saturation pulses, which are difficult to implement in field studies.
Leaf absorbed solar light energy is mainly consumed by photosynthetic reactions. By contrast, the absorption of excess light energy can generate reactive oxygen species, which causes damage to photosynthetic molecules. The excess light energy must be dissipated as heat through non-photochemical quenching (NPQ) mechanisms6. The photochemical reflectance index (PRI), which reflects light-dependent changes in leaf reflectance parameters, is derived from narrow-band reflectance at 531 and 570 nm (reference wavelength)7,8. It is reported to correlate with NPQ in chlorophyll fluorescence analysis9. However, since NPQ is a composite parameter that includes the xanthophyll cycle, state tradition, and photoinhibition, detailed validation is required to understand what the PRI parameter measures. We have focused on the xanthophyll cycle, a thermal dissipation system involving the de-epoxidation of xanthophyll pigments (violaxanthin to antheraxanthin and zeaxanthin) and a main component of NPQ because correlations between PRI and conversion of these pigments has been reported in previous studies8.
Many photosynthesis-related mutants have been isolated and identified in Arabidopsis. The npq1 mutant does not accumulate zeaxanthin because it carries a mutation in violaxanthin de-epoxidase (VDE), which catalyzes the conversion of violaxanthin to zeaxanthin10. To establish whether PRI only detects changes in xanthophyll pigments, we simultaneously measured PRI and chlorophyll fluorescence in the same leaf area in npq1 and the wild-type and then dissected NPQ at varying time scales of dark relaxation to extract the xanthophyll-related component11. These simultaneous measurements provide a valuable technique for the assignment of vegetation indices. Furthermore, since PRI correlates with gross primary productivity (GPP), the ability to assign PRI precisely to one component has important applications in ecology12.
Protocol
1. Cultivation of Arabidopsis plants
- Soak Arabidopsis thaliana seeds in sterilized deionized water in a microtube, and incubate for 2 days at 4 °C in the dark.
- Place approximately four of the imbibed, cold-treated seeds onto the soil surface using a micropipette. Incubate the planted pots in a growth chamber with a 16 h light (120 μmol photons m–2 s–1) and 8 h dark period at 22 °C and 20 °C, respectively.
- Grow one plant per pot by thinning other seedlings after germination. Prepare at least five pots. Incubate the plants in the growth chamber for an additional 4 weeks. Three plants are used for the experiments.
- Use the youngest, fully-opened mature leaf for photosynthesis measurements.
2. Setting up the sample stage, photosynthetic instruments, and light source
NOTE: For this protocol, a custom-built sample stage was used for fixing leaves and detection probes (Figure 1).
- Attach a 10 cm2 steel plate with a 1 cm diameter hole onto the custom-made sample stage. The hole size in this plate can be changed to accommodate different leaf samples or plant species. The stage has a clip for fixing the detection probes and an adjuster to adjust the distance between the probes and the leaf sample.
- Prepare thin fiber probes for measuring chlorophyll fluorescence and leaf reflectance. These thin fiber probes will be positioned closely so that they measure signals from the same leaf position.
NOTE: A PAM chlorophyll fluorometer and a spectral radiometer were adapted for signal detection of chlorophyll a fluorescence and leaf reflectance, respectively. Both instruments use thin fiber probes with diameters of 1 mm and 2 mm, respectively. - Fit these two probes tightly together and wrap them with plastic tape.
- Clip the taped probes onto the sample stage using a coaxial lens holder (see Figure 1), and position them vertical to the leaf surface.
NOTE: The field of view of the fiber optic cable in the spectral radiometer is α = 25°. In this method, the distance between the fiber probe tips and leaf surface is shorter than 1 cm. Therefore, the measuring leaf area is almost the same as that of the fiber. - Attach a biforked light guide made of glass fibers to the halogen light source and irradiate the sample stage from both directions at angles of approximately 45°.
NOTE: A halogen lamp, which is close to the wavelength distribution of natural sunlight, is used as actinic light for inducing photosynthesis. The halogen light source was adapted with a built-in cold filter, which removes long wavelengths from near infrared, to prevent increases in leaf surface temperature (λ = 400 to 800 nm). - Adjust the light source so that light uniformly illuminates the sample stage without casting shadows.
3. Setting up simultaneous measurements of leaf reflectance and chlorophyll fluorescence
NOTE: All steps are performed in the dark room to avoid the detection of light other than actinic light. A weak-green light (e.g., green-cellophaned light) should be turned off before the actual measurements.
- Measuring the distance between the leaf sample and the probes on the sample stage.
- Place a test leaf on the leaf holder of the sample stage in the dark. Press the leaf against a steel plate on the stage (black square in Figure 1).
- Turn on the PAM and irradiate the leaf sample with a measuring light. The values of the chlorophyll fluorescence intensities are confirmed using PAM controlling software (see Table of Materials).
- Move the adjuster so that the fluorescence intensity measures approximately 100. Measure the distance between the probe and the leaf. Fix the adjuster, and record the value of the distance on the adjuster.
- Turn off the measuring light. Remove the test leaf.
- Measuring the irradiance intensities of the actinic light
NOTE: To observe light-dependent photosynthetic behaviors, actinic light of varying intensity is used to irradiate the leaf sample.- Set a light quantum meter at the position where the sample leaf would be placed.
- Irradiate light from the halogen light source and measure the intensity.
- Determine which positions of the light source dial would generate intensities of 30, 60, 120, 240, and 480 μmol photons m–2 s–1.
NOTE: Arabidopsis plants are grown under 120 μmol photons m–2 s–1; therefore, the irradiance intensities of the actinic light are selected to provide a range of small and large intensities. - Mark each irradiance intensity on the dial.
- Measure a reflection standard.
NOTE: A reflection standard is required to calculate the leaf reflectance ratio at each irradiance intensity.- Place a white plate as a reflectance standard at the position of the leaf sample.
- Turn on a spectral radiometer. The reflectance signal is shown by the spectral radiometer controlling software. At this time, there are no spectral data because there is no irradiating light.
- Turn on the halogen lamp to irradiate with 480 μmol photons m–2 s–1, the highest irradiance intensity in this test.
- Adjust the detection strength of the radiometer to avoid saturation.
- Record spectral reflectance between 450–850 nm at 1 nm intervals under illumination with 30, 60, 120, 240, and 480 μmol photons m–2 s–1.
NOTE: A baseline electrical signal (dark current) is corrected and subtracted at every spectral measurement.
4. Simultaneous measurements of leaf reflectance and chlorophyll a fluorescence, and calculation of photosynthetic parameters
- Set a plant at the leaf sample position.
- Transfer the Arabidopsis plant from the grown chamber to the controlled dark room with the same temperature and humidity as that of the growth chamber.
- Incubate the plant for 1 h in the dark at 22 °C to dissipate electrons from the PSII reaction center and to relax of non-photochemical quenching.
- Place the dark-adapted whole plant on a lab jack under the sample stage (Figure 1).
- Fix the sample leaf to the leaf holder so that the leaf surface is perpendicular to the detection probes.
- Measurement of the maximum quantum yield of PSII.
- Turn on the PAM and start recording the curve. This value is called 0.
- Turn on the measuring light, and wait approximately 30 s for the curve to respond. This value is called F0.
- Give a saturated pulse of 4000 μmol photons m–2 s–1 for 0.8 s from the PAM.
- Obtain the highest value of the spike in the curve with increased fluorescence intensity. This value is called FM.
- Calculate the maximum quantum yield of PSII in the dark (FV/FM), using the following equation.
FV/FM = (FM – F0) / FM
- Measurement of photosynthetic behaviors at steady state.
- Turn on the halogen lamp as the external light source with the measuring light on after recording FM (see 4.2.4). First, irradiate the leaf sample with the weakest light (30 μmol photons m–2 s–1).
- Turn on the spectral radiometer at the same time to monitor the leaf reflectance.
- Wait for 20 min or longer for the photosynthetic reaction to reach steady state under the light conditions. The fluorescence intensity of the steady state is called Fs.
- Supply a saturating pulse at intervals of 1 min during illumination with the actinic light. The maximum fluorescence value achieved under the pulsed light is called FM′.
- Record the data of FM′ at 20 min after turning on the actinic light.
- Take leaf reflectance data by averaging 10 scans at an optimized integration time, with a dark current subtraction.
- Calculation of photosynthetic parameters at steady state.
- Calculate the quantum yields of PSII photochemistry (ΦPSII), which can be estimated by irradiating with saturated pulses under actinic light, using the following equation.
ΦPSII = (FM′- FS) / FM′ - Estimate the linear electron flux (LEF) from the PSII reaction center as follows 4.
LEF = The irradiance intensity of the actinic light × ΦPSII × 0.5 × 0.84 - Calculate the NPQ, which can be expressed the thermal dissipation, using the following equation.
NPQ = (FM – FM′) / FM′
NOTE: Light energy is primarily consumed by photosynthesis reactions. However, when plants absorb more light energy than energy consumed by photosynthesis, the mechanisms for the thermal dissipation are induced to avoid the excess energy. - Using the spectral data acquired with the radiometer under the same light conditions, calculate the leaf reflectance ratio as follows.
Reflectance ratio = Rleaf / Rstandard - Calculate PRI from 531 nm and 570 nm as follows. These two wavelengths are extracted from the reflectance ratio.
PRI = (R531–R570) / (R531+R570)
NOTE: R is a reflectance.
- Calculate the quantum yields of PSII photochemistry (ΦPSII), which can be estimated by irradiating with saturated pulses under actinic light, using the following equation.
- Measuring of the relaxation kinetics of non-photochemical quenching.
- Turn off the actinic light after acquiring Fs and leaf reflectance.
- Monitor chlorophyll fluorescence by PAM for 10 min after turning off light.
- Provide a saturating pulse at intervals of 1 min during the dark relaxation. The maximum fluorescence value induced by the saturation pulse under the dark is called FM′′. Obtain ten of FM′′ in one test.
- Save the data of FM′′ at 2 min and 10 min after turning off the actinic light.
- Turn the actinic light on set to the next irradiance intensity, 60 μ mol photons m–2 s–1.
- Repeat a light adaptation for 20 min and a dark relaxation for 10 min with pulsing saturation light at intervals of 1 min. Record all data as described above. Repeat all steps and measurements using irradiation at 120, 240, and 480 μ mol photons m–2 s–1.
- Calculation of parameters of the non-photochemical quenching from the relaxation kinetics.
NOTE: Light-dependent induction of NPQ is relaxed by turning off the light source13. It is possible to fractionate each NPQ function by adjusting the relaxation timescales.- Estimate the qE (energy-dependent quenching) fraction using FM′′ after 2-min dark adaptation.
qE = (FM2m′′–FM′) / FM′
NOTE: The qE fraction is rapidly reversed within 1–2 min. This fraction includes mainly PsbS protonation and part of xanthophyll conversion, which depend on light-induced ΔpH across the thylakoid membrane13. Both are reversible on breakdown of the gradient. - Calculate the qZ (zeaxanthin dependent quenching) fraction using FM′′ after 10-min dark adaptation.
qZ = (FM10m′′–FM′) / FM′
NOTE: A relaxation kinetics of NPQ at approximately 10 min after actinic light off reflects a xanthophyll cycle14. Most of the xanthophyll conversion is reversed at longer timescales of approximately 10 min (qZ) because the conversion requires a VDE (violaxanthin de-epoxidase) enzymatic reaction. The fraction is also relaxed by breakdown of the ΔpH across the thylakoid membrane. - Calculate the qI (photoinhibitory state) as follows.
qI = (FM–FM10m′′) / FM′
NOTE: The slowest recovery among NPQ fractions is thought to be photodamage of PSII (indicating D1 turnover). This fraction of the photoinhibitory state (qI), which does not recover by 10 min15.
- Estimate the qE (energy-dependent quenching) fraction using FM′′ after 2-min dark adaptation.
Representative Results
Figure 1 presents a schematic diagram of the experimental set up for simultaneously measuring chlorophyll fluorescence and leaf reflectance. The fiber probes of the PAM and spectral radiometer were set perpendicularly to the leaf surface at the leaf holder on the custom-made sample stage, and a halogen lamp was used for actinic light irradiation from both left and right directions without casting any shadows. The PAM and leaf reflectance signals were detected using the software of the separate systems. This experimental system was used to compare Arabidopsis wild-type (Col) and npq1 mutant (lack zeaxanthin) plants (Figure 2). The ΔPRI calculated from the leaf reflectance was plotted against light-dependent linear electron flow from PSII estimated by the PAM (Figure 2A). PRI is reported to be affected not only by xanthophyll but also by carotenoids16. The PRI was corrected by being PRI at each light intensity minus PRI at the lowest light intensity (ΔPRI) to observe only light-dependent PRI changes11. The results showed that ΔPRI was negatively correlated with LEF in wild-type plants, but not in npq1. We also dissected qZ, which represents the xanthophyll cycle, from the dark relaxation kinetics of NPQ and plotted it as ΔPRI in Figure 2B. The results show that qZ is strongly correlated with ΔPRI (r2 = -0.87, p-value < 0.001), implying that PRI reflects the xanthophyll cycle.
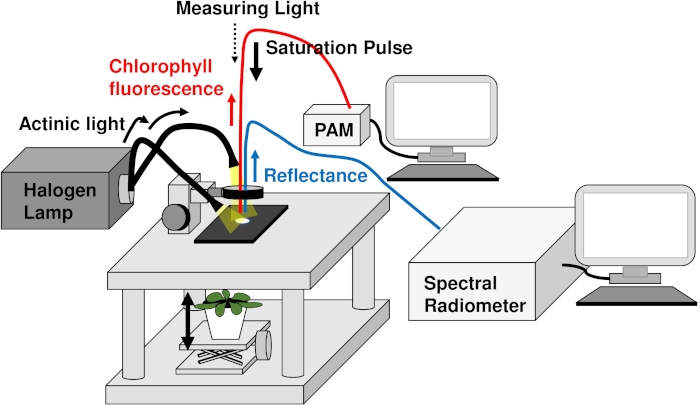
Figure 1: Schematic diagram of the experimental system for simultaneous measurement of chlorophyll a fluorescence and leaf reflectance. Details are described in the Protocol section. A plant pot was positioned by a lab jack (solid double-headed arrow). A halogen lamp was used to irradiate various light intensities to activate photosynthesis (thin solid arrow). Chlorophyll a fluorescence signals were detected using a system of pulse amplitude modulation (PAM); the red line indicates the fiber probe from the PAM chlorophyll fluorometer. The leaf reflectance was detected by a spectral radiometer under the light illumination; the blue line indicates the fiber probe from the spectral radiometer. The measuring light (dotted arrow) and the short-saturated light (thick solid arrow) also were provided by the PAM chlorophyll fluorometer. The saturated lights were pulsed with 1 min intervals during light adaptation for 20 min and dark relaxation for 10 min. Please click here to view a larger version of this figure.
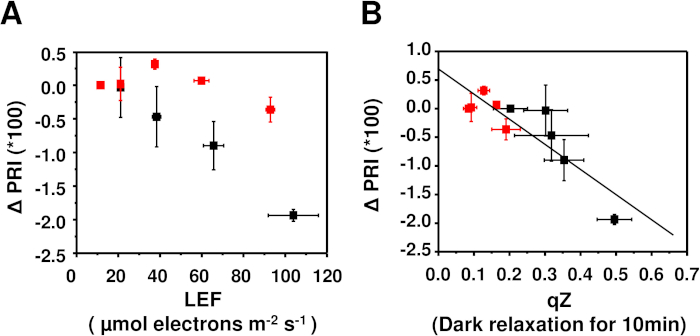
Figure 2: Changes in photosynthetic parameters in wild-type Columbia (black squares), and npq1 mutant (red squares) Arabidopsis plants. ΔPRI (PRI at each irradiance intensity minus PRI at the lowest intensity of 30 µmol photons m-2 s-1) was plotted against (A) the rate of linear electron flux (LEF), and (B) qZ after dark relaxation for 10 min. The irradiance intensity of the actinic light was 30, 60, 120, 240, and 480 µmol photons m-2 s-1. Data points and error bars represent means ± SD for n=3. The line in B is a regression curve that applies for all data points. Please click here to view a larger version of this figure.
Discussion
In this study, we obtained additional evidence to show that PRI represents xanthophyll pigments by simultaneously measuring chlorophyll fluorescence and leaf reflectance.
A halogen light, which has wavelengths similar to sunlight, was adapted for use as an actinic light source to activate photosynthesis. We initially used a white LED light source to avoid thermal damage of the leaf surface, but this produced slow dark relaxation kinetics and exceptionally high qI (photoinhibitory quenching), possibly by photodamaging PSII. We therefore adapted the halogen lamp with a built-in cold filter to reduce heat production. This light source did not cause any abnormalities in dark recovery or qI.
The most important variable in our method is the positional relationship between the leaf, the light source, and the detection probes. We have tested measuring the chlorophyll fluorescence and leaf reflectance from various diagonal angles with light irradiating from directly above to the leaf. However, the intensity of the detection signals differed depending on the angle. To avoid this variability, the probes were fixed vertically above the leaf sample (Figure 1). The light source was delivered using bifurcated fibers that irradiated the leaf surface from both the left and right sides to generate a uniform irradiating light (Figure 1).
Studies of leaf reflectance have been primarily used in ecology to determine various plant vegetation indices in field settings, such as differences between plant species, nutritional conditions, or seasonal changes. However, few studies have tested and verified these vegetation indices in model plants such as Arabidopsis and tobacco, whose mutants could possess a wealth of genetic information and omics analyses data. Verifying and developing vegetation indices for these plants could identify novel photosynthetic parameters represented in innovative vegetation indices, which would contribute to the discipline of ecology.
This study focused on the dark relaxation kinetics of NPQ to verify the xanthophyll cycle behavior. New photosynthesis-related parameters are currently under development for chlorophyll fluorescence analysis (e.g., estimations of the redox state of the plastoquinone pool (qL) or the activity of cyclic electron flow around PSI17,18). The simultaneous measurement of chlorophyll fluorescence and leaf reflectance in related Arabidopsis mutants will advance research into the molecular mechanisms of photosynthesis and help to utilize this knowledge in field studies. A recent study reported that chlorophyll fluorescence in plants can be remotely sensed from leaf spectral reflectance. The parameter calls solar-induced chlorophyll fluorescence (SIF) is measured utilizing a Fraunhofer line, dark lines absorbed by Oxygen, under solar light 12,19. If the currently developed vegetation indices were reassigned using these techniques, it would be possible to remotely assess photosynthetic responses in plants without using special treatments such as saturated pulses or dark adaptation.
Divulgations
The authors have nothing to disclose.
Acknowledgements
We are grateful to Dr. Kouki Hikosaka (Tohoku University) for stimulating discussions, assistance with a work space, and instruments for experiments. The work was supported in part by KAKENHI [grant numbers 18K05592, 18J40098] and Naito Foundation.
Materials
| Halogen light source | OptoSigma | SHLA-150 | |
| Light quantum meter | LI-COR | LI-1000 | |
| PAM chlorophyll fluorometer | Walz | JUNIOR-PAM | |
| PAM controliing software | Walz | WinControl-3.27 | |
| Reflectance standard | Labsphere, Inc. | SRT-99-050 | |
| Spectral radiometer | ADS Inc. | Field Spec3 | |
| Spectral radiometer controlling software | ADS Inc. | RS3 |
References
- Xue, J., Su, B. Significant remote sensing vegetation indices: A review of developments and applications. Journal of Sensors. 1353691, (2017).
- Cotrozzi, L., Townsend, P. A., Pellegrini, E., Nali, C., Couture, J. J. Reflectance spectroscopy: a novel approach to better understand and monitor the impact of air pollution on Mediterranean plants. Environmental Science and Pollution Research. 25 (9), 8249-8267 (2018).
- Han, L., Yang, G., Yang, H., Xu, B., Li, Z., Yang, X. Clustering Field-Based Maize Phenotyping of Plant-Height Growth and Canopy Spectral Dynamics Using a UAV Remote-Sensing Approach. Frontiers in Plant Science. 9, 1638 (2018).
- Baker, N. R. Chlorophyll Fluorescence: A Probe of Photosynthesis In. Vivo. Annual Review of Plant Biology. 59 (1), 89-113 (2008).
- Cruz, J. A., et al. Dynamic Environmental Photosynthetic Imaging Reveals Emergent Phenotypes. Cell Systems. 2 (6), 365-377 (2016).
- Ruban, A. V. Quantifying the efficiency of photoprotection. Philosophical Transactions of the Royal Society B: Biological Sciences. 372 (1730), 20160393 (2017).
- Gamon, J. A., et al. Remote sensing of the xanthophyll cycle and chlorophyll fluorescence in sunflower leaves and canopies. Oecologia. 85 (1), 1-7 (1990).
- Gamon, J. A., Peñuelas, J., Field, C. B. A narrow-waveband spectral index that tracks diurnal changes in photosynthetic efficiency. Remote Sensing of Environment. 41 (1), 35-44 (1992).
- Rahimzadeh-Bajgiran, P., Munehiro, M., Omasa, K. Relationships between the photochemical reflectance index (PRI) and chlorophyll fluorescence parameters and plant pigment indices at different leaf growth stages. Photosynthesis Research. 113 (1-3), 261-271 (2012).
- Niyogi, K. K., Grossman, A. R., Björkman, O. Arabidopsis mutants define a central role for the xanthophyll cycle in the regulation of photosynthetic energy conversion. Plant Cell. 10 (7), 1121-1134 (1998).
- Kohzuma, K., Hikosaka, K. Physiological validation of photochemical reflectance index (PRI) as a photosynthetic parameter using Arabidopsis thaliana mutants. Biochemical and Biophysical Research Communications. 498, 52-57 (2018).
- Hikosaka, K., Noda, H. M. Modeling leaf CO2 assimilation and Photosystem II photochemistry from chlorophyll fluorescence and the photochemical reflectance index. Plant, Cell and Environment. 42 (2), 730-739 (2019).
- Brooks, M. D., Sylak-Glassman, E. J., Fleming, G. R., Niyogi, K. K. A thioredoxin-like/β-propeller protein maintains the efficiency of light harvesting in Arabidopsis. Proceedings of the National Academy of Sciences of the United States of America. 110 (29), E2733-E2740 (2013).
- Nilkens, M., et al. Identification of a slowly inducible zeaxanthin-dependent component of non-photochemical quenching of chlorophyll fluorescence generated under steady-state conditions in Arabidopsis. Biochimica et Biophysica Acta (BBA) – Bioenergetics. 1797 (4), 466-475 (2010).
- Davis, G. A., et al. Limitations to photosynthesis by proton motive force-induced photosystem II photodamage. Elife. 5, 16921 (2016).
- Wong, C. Y. S., Gamon, J. A. The photochemical reflectance index provides an optical indicator of spring photosynthetic activation in evergreen conifers. New Phytologist. 206 (1), 196-208 (2015).
- Miyake, C., Amako, K., Shiraishi, N., Sugimoto, T. Acclimation of Tobacco Leaves to High Light Intensity Drives the Plastoquinone Oxidation System—Relationship Among the Fraction of Open PSII Centers, Non-Photochemical Quenching of Chl Fluorescence and the Maximum Quantum Yield of PSII in the Dark. Plant and Cell Physiology. 50 (4), 730-743 (2009).
- Munekage, Y., et al. Cyclic electron flow around photosystem I is essential for photosynthesis. Nature. 429 (6991), 579-582 (2004).
- Tubuxin, B., Rahimzadeh-Bajgiran, P., Ginnan, Y., Hosoi, F., Omasa, K. Estimating chlorophyll content and photochemical yield of photosystem II (ΦPSII) using solar-induced chlorophyll fluorescence measurements at different growing stages of attached leaves. Journal of Experimental Botany. 66 (18), 5595-5603 (2015).

