A Spine Robotic-Assisted Navigation System for Pedicle Screw Placement
Summary
This article presents a standardized surgical technique for robotic-assisted pedicle screw placement by using robotic-assisted navigational systems. We present a step-by-step protocol and describe the workflow and precautions of this procedure.
Abstract
Pedicle screw implantation has excellent treatment effects and is often used by surgeons in spinal fusion surgery. However, due to the complexity of human body anatomy, this surgical procedure is difficult and challenging, especially in minimally invasive surgery or patients with congenital anomalies and kyphoscoliosis deformity. In addition to the abovementioned factors, the surgical experience and technique of the surgeon also affect the recovery rates and complications of the patients after the surgical operation. Therefore, accurately performing pedicle screw implantation has is a constant topic of common concern for surgeons and patients. In recent years, with the technological development, robot-assisted navigation systems have gradually become adopted. These robot-assisted navigation systems provide surgeons with complete preoperative planning before surgery. The system provides 3D reconstructed images of each vertebra, allowing surgeons to understand the patient's physiological characteristics more quickly. It also provides 2D images of sagittal, coronal, axial and oblique planes so that surgeons can accurately perform pedicle screw placement plan.
Previous studies have demonstrated the effectiveness of robot-assisted navigation systems for pedicle screw implantation procedures, including accuracy and safety assessments. This step-by-step protocol aims to outline a standardized surgical technique note for robotic-assisted pedicle screw placement.
Introduction
In the field of spinal surgery, spinal fusion surgery is a fundamental surgical procedure, especially posterior pedicle screw fixation, which can provide three-column support of the vertebrae and enhance the strength of biomechanics; thus, it has become one of the most commonly used surgical procedures1. In many early studies, the clinical effect of posterior pedicle screw implantation has been confirmed, and it has been widely used in surgery for many different spinal disorders, such as degenerative, traumatic, and complicated spinal conditions2.
However, although the posterior lumbar spinal fusion surgery can achieve excellent treatment effects, it is still risky due to the human body anatomy. There are many vital tissue structures close to the pedicle, such as the central nervous system, nerve roots, and main blood vessels. The damage of these tissues during the surgical procedure may cause serious complications, such as vascular injuries, neurological deficits, or screw loosening2,3. Moreover, the surgeons and staff are exposed to additional radiation, particularly in the case of minimally invasive spinal procedures4. Surgeons may experience fatigue and hand tremors after lengthy and tedious spinal surgery procedures, such as screw placements, bone osteotomy, and nerve decompression5.
The unsatisfactory rate of the pedicle screw placement procedure necessitated the proposal for a robotic-assisted navigation system to be applied in spinal surgeries to improve the surgery accuracy and patients’ safety. Several studies on robotic-assisted navigation systems have demonstrated improvements in the safety, accuracy, and precision of pedicle screw placement, as well as decreased radiation exposure and operative times6,7,8,9,10. However, thorough screw trajectory planning, pre-operative planning with images, comprehensive robotic system with fixation device, and robot control software still need to be addressed to achieve this goal. This study focuses on the description of the robotic structure and the workflow of a self-developed navigation system (i.e., the Point spine navigation system (PSNS)) for robotic-assisted pedicle screw placement surgeries.
System description and surgical protocol
The PSNS comprises a navigation workstation that includes the following. (1) There is a user interface software responsible for image reading through three-dimensional (3D) reconstruction, pre-operative planning, spatial kinematic relationship calculation, and registration. (2) The PSNS uses infrared optical guidance systems to track the spatial position of surgical robots and patients. The infrared optical guidance system contains the following components: (i) an optical tracker that actively emits infrared light and performs stereo positioning through a dual camera (Figure 1); (ii) a marker sphere whose surface has a reflective coating with reflective properties for precise tool tracking; and (iii) a tool with a dynamic reference frame (DRF) that comprises a base and four marker spheres. To avoid the identification failure of the tracking system, each device has a unique DRF design and cannot be shared with each other. The DRF used includes a base frame (BF) attached to the base of the handpiece to confirm the handpiece position, an end-effector frame (EF) attached to the end of the handpiece to confirm the handpiece position, a fiducial frame (FF) anchored on the patient’s bone to confirm the patient’s position, and a probe whose tip is used to confirm the target position in 3D space. (3) There is a handpiece comprising a six degrees of freedom (DOF) Stewart platform, with one end of the robot equipped with an operation tool used for drilling the screw path. The handpiece is a robotic-assisted navigation system that assists surgeons toward the accurate placement of implants, such as pedicle screws, or positioning of surgical tools during spinal surgery. The movement of the surgical target is tracked as the robot automatically compensates for the correct target. The robot is designed as a semi-active system that offers surgical tool guidance; however, the actual surgery is performed by surgeons. The operating principle and equipment are illustrated in Figure 2.
PSNS is indicated for procedures including but not limited to the following sample procedures: (i) open, minimally invasive, or percutaneous spinal surgery; (ii) spinal surgery site for thoracic, lumbar, or sacral vertebrae; (iii) posterior spinal fusion for trauma, degenerative stenosis disease, instability, spondylolisthesis, herniated disc, tumor, infection, or spinal deformity correction; (iv) placement of temporary or permanent devices, such as k-wires or needles, while performing vertebroplasty, or either transforaminal or interlaminar percutaneous endoscopic lumbar discectomy; and (iv) bone tumor excision, including the ablation of osteoid osteoma or tumor biopsy, in which the robot directed needles or guidewires to a given vertebral location. This procedure is contraindicated for those with an inability to tolerate anesthesia, surgical procedure, or when satisfactory navigation images have not been acquired.
Note that the operation staff, including neurosurgeons and orthopedic surgeons, must be licensed and trained in guiding courses. All procedures for operating the robot during surgery need to follow the recommended standardized procedures to avoid causing harm to the patient or surgeon. Surgeons must possess conventional surgical experience to ensure that it is possible to switch back to conventional surgical instruments and complete the surgery when it is determined that the navigation is inaccurate, based on the surgeons’ anatomical knowledge.
Protocol
All procedures followed were in accordance with the ethical standards of the National Taiwan University Hospital (NTUH) Research Ethics Committee (REC) and the Helsinki Declaration of 1975 (in its most recently amended version). Informed consent must be obtained from all patients if further clinical trial is prepared.
NOTE: The anesthesia procedure can be categorized into three steps: pre-operative evaluation of the patient, intraoperative management, and postoperative management. During pre-operative evaluation, all patient data, including the thorough history and physical examination, should be collected and the staff should recognize patient comorbidities and how they relate to the anesthetic care of the patient. A thorough airway exam should be performed, and the staff should be aware of the anesthetic options to formulate a basic anesthetic care plan. During intraoperative management, the anesthesiologist should check the basic functions of the anesthesia machine, and apply basic physiologic monitors recommended by the American Society of Anesthesiologists, which include a pulse oximeter, electrocardiography, a noninvasive blood pressure device, and a temperature monitor, airway management options, pharmacology of inductions agents, and indications during an anesthetic induction. Intraoperative events, such as hypotension, hypertension, hypoxia, and oliguria, must be recognized, evaluated, and managed. Additionally, the staff must recognize when the patient meets the extubation criteria.
1. Pre-operative setting and planning
NOTE: During surgery, sterile surgical drapes should be used to prevent contact with unprepared surfaces and to maintain surgical site sterility of the environmental surfaces, equipment, and patient’s surroundings. To reduce the risk of pathogen transmission to both the patients and the surgical team, sterile surgical gowns should be worn over the scrub suits by the operating team during surgery.
- Remove all components that can affect fluoroscopy from the surgical site; this depends on the surgical plan according to each individual patient.
- Place the patient in a prone position after administering anesthesia and prepare as per surgical requirements.
NOTE: All anesthesia procedures must be performed under the supervision of an anesthesiologist and each plan should be adjusted according to each individual patient. - Clean and sterilize the surgical site of the patient.
- Cover the OP-site at the surgical site of the patient.
- Place the sterile surgical drape on the patient, except at the surgical site.
- Anchor the FF to the patient; users can choose one of the following two methods according to their needs.
- Anchoring to the iliac bone (applicable surgical site: L5 or S1).
- Place two percutaneous wires (Φ = 1.5 mm) on the posterior iliac crest and check the entry point under fluoroscopy. Repeat the step if surgeon has a concern about the entry point. Mark the entry point by using a marker pen.
- Insert the first percutaneous pin (Φ = 5 mm, L = 140 mm) into the patient’s posterior iliac crest by using a power drill (1000 RPM).
- Place the FF along with the first percutaneous pin. Adjust the FF until it is recognized by the optical tracking camera. Fix the FF to the first percutaneous pin using a screwdriver.
- Insert the second percutaneous pin (Φ = 5 mm, L = 140 mm) along with a hole on the FF using a power drill (1000 RPM). Fix the screw on the FF to the second percutaneous pin using screwdriver.
NOTE: According to the manual of the optical tracking system, the marker sphere can be identified within 3 m from the optical tracker.
- Anchoring to the current or adjacent vertebral spinous process with a clamp applicable surgical site: thoracic, lumbar, or sacral vertebrae.
- Place a wire (Φ = 1.5 mm) on the patient’s back as a reference under fluoroscopy. Check the surgical field under fluoroscopy. Repeat the step if surgeon has a concern about the surgical field. Mark the surgical field by using a marker pen.
- Incise the skin tissue on surgical field using a surgical scalpel. Fix the FF to the spinous process using a screwdriver. Due to the difference of bone mineral density, have the surgeon determine if the FF is anchored on spinous process firmly.
- Anchoring to the iliac bone (applicable surgical site: L5 or S1).
- Check whether the equipment and components of PSNS have been prepared, including the handpiece, the optical tracking system, the robotic workstation, and the navigation toolkit (i.e., probe) (Figure 3 & Figure 4).
NOTE: Avoid interfering with the surgical staff; Avoid blocking the optical tracking camera; Ensure the tracker is stable and recognized by the optical tracking system; Sterilize the navigational toolkit and place it on the operating table.
2. Spatial labeling and registration
- Transfer the patient’s preoperative CT images to the system through DVD or USB and crop the image size to adjust the orientation based on surgical needs. The system provides virtual surgical guided images, including sagittal, coronal, axial, and oblique planes, and customized 3D reconstructions for each vertebra.
- As the PSNS software provides the labeling interface, ask the surgeon to label each vertebra with the anterior-posterior view and lateral view, differentiating the intervertebral disc for the subsequent steps to be identified.
- Select the optimal screw length and implant dimensions based on the device software.
- Plan the optimal positioning and trajectory of the screw based on the 3D and multi-planar image reconstruction of the preoperative CT scan.
- Confirm whether all the planned screws are correct and appropriate.
- Enter the DRF monitoring interface in the PSNS software that presents multiple planar views (include 3D volume and three cross-sectional planes on the side). All the DRFs should be inside the vision area of the optical tracking system (according to the user instructions, the recommended best recognition range is range B.) When the DRF vector arrow indicating the tracker is displayed on the user interface, it is stably recognized by the tracking system (Figure 5).
- Perform a subperiosteal dissection bilaterally along the spinous process, the laminae out to the tips of the transverse processes of all the levels. Remove the facet joint capsules to expose the joints. The use of self-retained retractors aid in vertebra exposure by holding the musculature off to the side.
- Perform registration procedures, including landmark registration and surface matching. Follow the sequence below to ensure the correctness of the registration result.
- Landmark registration
- Select at least four non-coplanar feature points (such as the spinous process, the laminar, and the transverse process) on the patient’s pre-operative 3D reconstruction CT images.
- Use the tip of probe to keep in contact with the first feature point selected in step 2.8.1.1 in the actual surgical area.
- Press the probe selection button on the software interface to confirm the access point.
- Repeat steps 2.8.1.2-2.8.1.3 until the four feature points selected in step 2.8.1.1 are confirmed.
- Press the calculation button on the software interface; the system will calculate the result of the landmark registration and present it in the software interface.
- The acceptance criteria for the registration accuracy must meet the needs of the clinical indications (<5 mm). If the result is not satisfactory, repeat steps 2.8.1.1-2.8.1.5 until the registration result meets the acceptance criteria.
NOTE: Ensure that the validity of using the probe to obtain the position information of the bone surface, such as clearing the soft tissue on the bone surface and avoiding the dangling of the probe tip when collecting points.
- Surface matching
- Use the probe tip to continuously contact any point on the bone surface in the actual surgical area.
- Press the probe selection button on the software interface to confirm the access point
- Move the probe (make probe different from previous pick point), and repeat steps 2.8.2.1-2.8.2.2 until at least 50 pick points are completed.
- Press the calculation button on the software interface; the system will calculate the surface matching result and present it on the software interface.
- The acceptance criteria for the registration accuracy must meet the needs of the clinical indications (<0.5 mm). If the result is not satisfactory, repeat steps 2.8.2.1-2.8.2.4 until the registration results meet the acceptance criteria.
- Landmark registration
- Use the probe to select obvious anatomical landmarks (such as spinous process, transverse processes, facet joint) of the actual surgical area for confirmation once the registration result is accepted (Figure 6).
NOTE: Appropriate reflection and reception of the infrared light must be maintained during surgery. If the optical tracking system cannot recognize the markers, the software interface will display a red light reminder. The camera should be adjusted such that the surgical field is at the center of the camera’s detection range, and the tracker should be protected from light and blood.
3. Robot assembly and motion
- Cover the handpiece with sterilization drapes and install the surgical instruments on the robot (e.g., trocar (Φ = 5 mm) and k-pin (Φ = 1.8 mm)).
- Adjust the angle and position of the handpiece in space according to the following instructions (steps 3.2.1-3.2.2) so that the handpiece is within the compensation range (within a distance of one centimeter and an included angle of 4 degrees from the planned path).
- Angle adjustment: Turn the angle of the handpiece in space so that the two circles representing the angle of the handpiece coincide on the software interface.
- Position adjustment: horizontally and vertically move the position of the handpiece in space, so that the dots representing the position of the handpiece on the software interface are aligned with the entry points of the planned path.
NOTE: When steps 3.2.1 & 3.2.2 are completed at the same time, the handpiece will automatically activate the active compensation function to maintain the angle and position of the instrument to conform to the pre-planned path (Figure 7).
- Determine the robot's operating status by judging the marker color of the robot displayed on the UI. If it is green, it can be operated, if it is red, it cannot be operated.
NOTE: If the handpiece comes in contact with the patient or the surrounding obstacles, an emergency stop button located above the navigation workstation housing can be pressed by the surgeon or technician. Regular maintenance of the robot should be performed. The platform must be recalibrated for the kinematics parameters after 250 uses. The trocar and k-pin must be discarded after a single use.
4. Pedicle preparation and screw insertion
- Activate the drill function of the handpiece, and drill the instruments mounted on the front end (including K-pin: Φ = 1.8 mm and trocar: Φ = 5 mm) into the patient's body along the planned path.
- Use the c-arm to confirm the position of k-pin and trocar.
- If the k-pin and trocar positions are not correct under fluoroscopy, remove the k-pin and trocar. Then, using the handpiece, drill into the pedicle again until the k-pin and trocar insert into prone positions under fluoroscopy (refer to 4.3.1-4.3.2).
- Under AP view, determine whether the instrument is located in the oval area formed by the pedicle in the perspective image.
- Under LAT view, determine whether the instrument is within the range of the pedicle and vertebra.
- Replace K-pin and trocar with guidewires (Φ = 1.5 mm, L = 400 mm) once the positions are appropriate.
- Insert the pedicle screw through the guidewires.
- Repeat steps 4.1–4.4 to complete all surgical planning paths.
NOTE: As for postoperative management, patients should be monitored in the post-anesthesia recovery unit (PACU) and the post-operative analgesia options should be selected. The basic PACU events, such as nausea, pain, hypotension, hypertension, and hypoxia, should be evaluated. Additionally, the staff should recognize when the patient meets the criteria for PACU discharge.
Representative Results
The safety and accuracy of robotic-assisted pedicle screw placements have been addressed in several studies6,11. We match the vertebrae with pre-operative planning images under an optical tracking system in the proposed method. After determining the planned surgical path, this information was transferred to the handpiece through the handpiece control unit. The navigation system integrates the tracking information and displays it on the monitor during the surgery. Furthermore, the screen displays the admission path on the spine and the positions of the instruments.
In our previous study12, a low overall screw malposition rate of 1.7% from a total of 59 screws were placed on 30 porcine vertebrae through the PSNS was demonstrated (Figure 8). Surgical procedures proceeded smoothly while using the PSNS and these 59 pedicle screws were assessed by postoperative CT scans. 51 screws (86.4%) fell into group A, 7 screws (11.9%) fell into group B, and 1 screw (1.7%) fell into group E according to the Gertzbein–Robbins classification12. No spinal canal perforations or injuries to any other major vessels were found and all pedicle screws were inserted within the safe zone. We recorded the tip position data at a frequency of 60 Hz and a linear regression curve was calculated with the optical tracking system during the surgery. Differences including the angle, shortest distance, and entry point between the actual pedicle screw position and preoperative planning path were also recorded12.
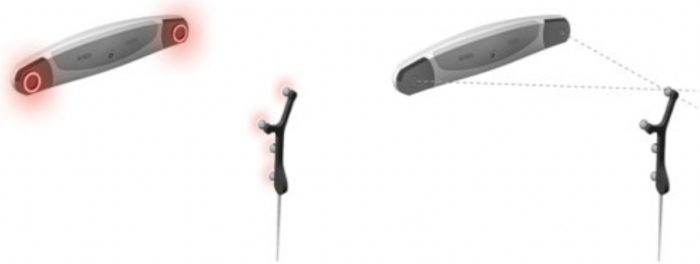
Figure 1: Working principle of optical tracking system13. The optical tracker will actively emit infrared light and perform stereo positioning through dual camera. Please click here to view a larger version of this figure.
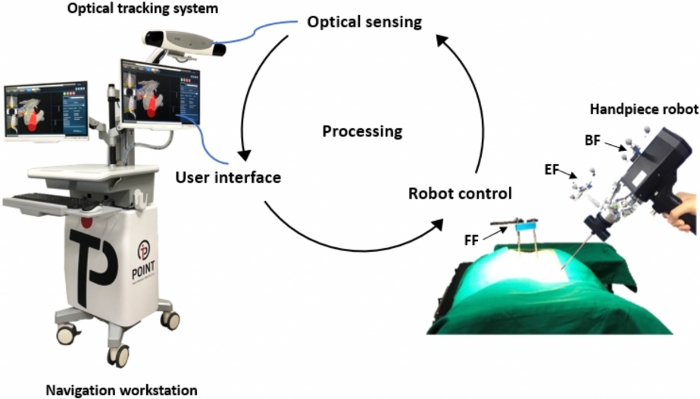
Figure 2: Working principles of spine navigation system. The application process of the system includes robot control, user interface, and optical sensing Please click here to view a larger version of this figure.
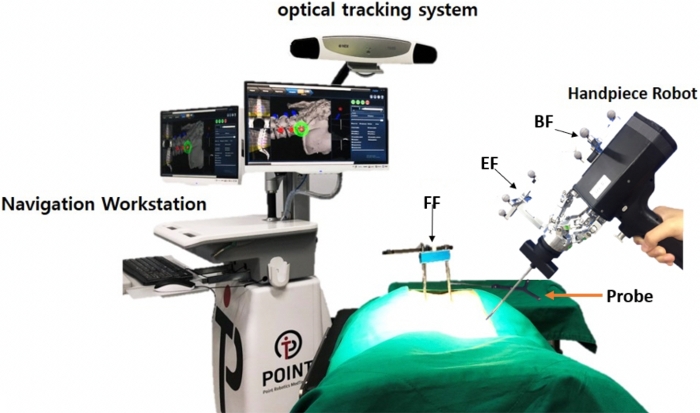
Figure 3: Spine navigation system, including handpiece, optical tracking system, robotic workstation, and navigation toolkit. (i.e., probe) Please click here to view a larger version of this figure.
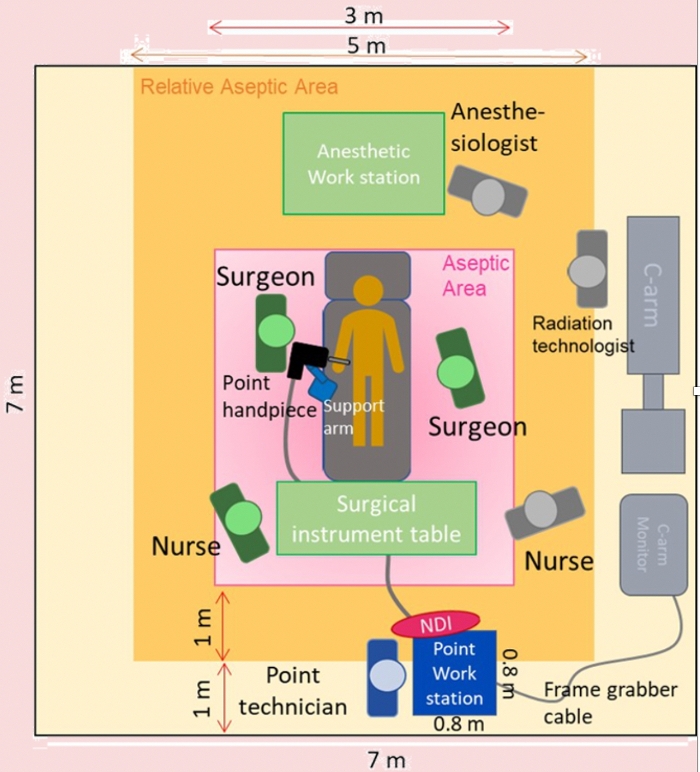
Figure 4: Schematic diagram of operation room configuration, users must refer to the schematic diagram to set up the PSNS in the operating room. Please click here to view a larger version of this figure.
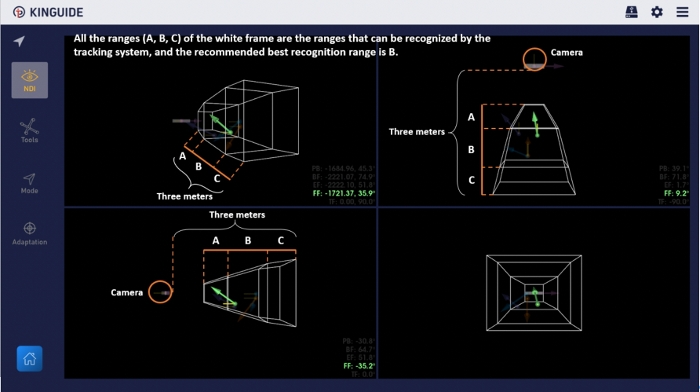
Figure 5: DRF monitoring interface in the software. Users can confirm the current status of all DRFs according to the display on the interface. Please click here to view a larger version of this figure.
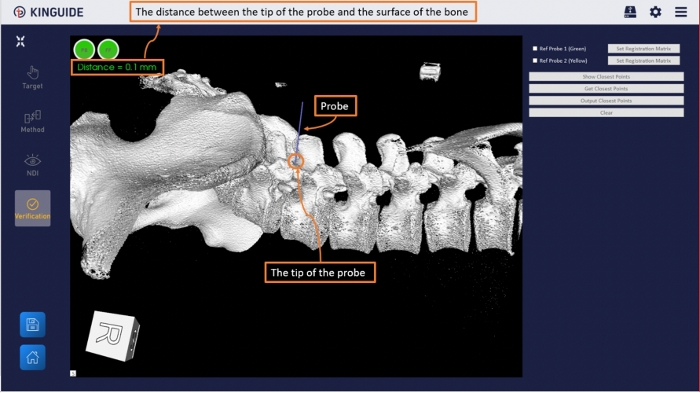
Figure 6: Registration accuracy verification interface in the software. Use probe to select a specific anatomical feature (such as spinous process, transverse processes, facet joint) in the actual surgical area, and the system will calculate the distance from the probe tip to the anatomical feature as a reference for accuracy. Please click here to view a larger version of this figure.
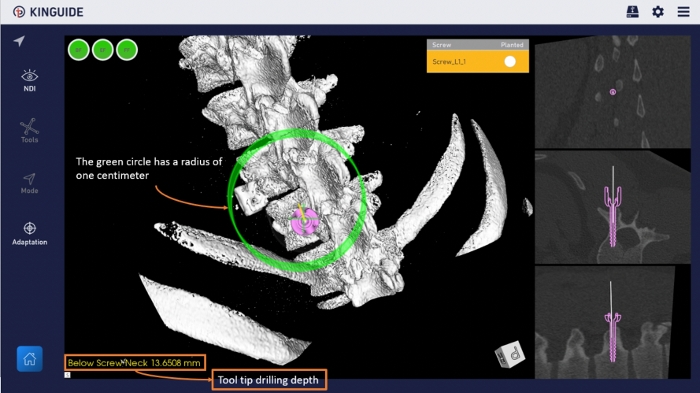
Figure 7: Navigation interface in the software. Using a 3D reconstructed bone model and virtualized pedicle screw to provide guidance for the surgical path. Please click here to view a larger version of this figure.
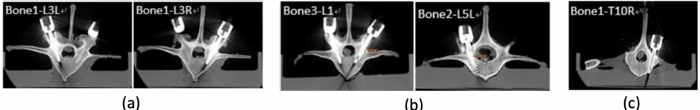
Figure 8: Postoperative CT scans assessed according to Gertzbein and Robbins classification with an example of grade A (a), grade B (b) and grade E (c)14. Please click here to view a larger version of this figure.
Discussion
Since 1990, there have been rapid developments in surgical applications involving the use of robots. The available robotic technologies have been optimized, resulting in improved accuracy, overcoming the tremor in human hands, and reduced matching and registration times of navigation systems15. The benefits of surgical robot assistance include: (1) immediate standardization without lengthy learning processes; (2) surgeons can precisely follow the pre-operative plan, which is superimposed on a CT-based image through the user-interface; (3) reduction of radiation exposure to surgeons and operating staff; and (4) improved accuracy, especially while facing complex anatomy or complicated revision surgery.
Despite the widely accepted use of pedicle screws, freehand pedicle placement techniques depend largely on anatomic landmarks, image guides, and the surgeons’ experience. Even with experienced surgeons, the implant malposition rates are in the range of 5.1–31%, as described in multiple review studies3,16. Many surgeons accept deviations between 2 and 3 mm while assessing the accuracy of screw positions, as this deviation rate rarely becomes symptomatic. Lonstein et al. reported that 5.1% of 4,790 screws breached the cortical bone in their meta-analysis study, and approximately 0.2% of these caused neurological symptoms17. Additionally, even minor screw deviations may result in symptoms and surgeons may be hesitant to operate again. Therefore, a great variety of systems offering spinal image guidance such as electromagnetic navigation, intra-operative 3D fluoroscopy and CT navigation, percutaneous reference frames, and robotic-guided surgery are under research or in clinical use. These technologies allow surgeons to determine precise pre-operative and intra-operative execution plans, including pedicle screw length and diameter, even in the presence of severe deformities and lack of anatomic landmarks.
The use of robotic-assisted pedicle screw placements is encouraging due to its accuracy of up to 98.3%12. Despite the overall high accuracy of pedicle screw placement under PSNS, the robot system failed to adequately register 10–20% of the conditions during our testing. In conditions of such as high-degree of curvature, obesity, osteoporosis, loosening of previously placed hardware during revision surgery, poor-quality intra-operative fluoroscopic imaging, physical limitations of the handpiece extensibility, device failure, mechanical movement, and technical issues, may result in difficulties with the registration and may require reverting to a freehand pedicle screw placement. Spine surgeons should possess traditional surgical experience to determine whether the navigation system is working appropriately and be able to switch to traditional surgery should the robotic system fail. Additionally, currently, PSNS is indicated for thoracolumbar pedicle screw implantation, and the accuracy of this system is 2 mm. In clinical surgery, the error tolerance of cervical pedicle screw implantation is approximately 0.2–0.5 mm; thus, this system is not suitable for cervical surgery at present.
PSNS comprising a handpiece can be used in combination with surgical tools to directly drill into the vertebra. The device footprint is small and occupies little space in the operating room. These features are different from other navigation robotic spinal surgery systems, making spinal navigation surgery more flexible and convenient for surgeons. The PSNS consists of image registration and matching, robotic and navigation technology, and precise equipment manufacturing. The system relies on these components working together appropriately as errors may occur if any one of these components fails. The spatial positioning of the anatomy at the surgical site will be relatively fixed after the images are acquired. Factors such as excessively soft tissue disturbance, decompression or osteotomy, long segment surgeries over 3 vertebrae, or the amount of respiratory tidal volume may cause navigational deviations. If the surgeon suspects a navigational deviation, the probe can be used to select the anatomical landmarks for confirmation (e.g., spinous process or facet joints). If the position is correct, the operation can continue. However, if the position is incorrect, some possible causes and solutions are as follows: (1) The dynamic reference frame-fiducial frame is moved during the operation. The surgeon should restrict the dynamic reference frame-fiducial frame and registration again. (2) There is relative displacement between the anatomical structures, such as after deformity correction, caused by the operation. The surgeon should re-scan the fluoroscopy to obtain new images for surgery. According to previously published research, robot-assisted navigation systems can reduce the time taken for each pedicle screw insertion; however, the operative time increases due to the robot setup and registration10.
Several limitations of robot-assisted surgery still exist, such as registration problems including difficulty in landmark accessing, incompatibility in minimally invasive surgery and time consuming, patients being exposed to additional radiation, tool skiving due to lack of live-intraoperative feedback, impact on traditional spine training, dependence on technology, and high costs. PSNS has certain limitations: first, the surgeon needs to spend time to learn the PSNS system thoroughly; second, it is heavy for the surgeons to hold it. Our team will focus on making the user learning curve easier and provide a supporting arm for reducing the weight of the handpiece. Nevertheless, we believe that there are continuous developments in robotic-assisted navigation systems, which have potential for improving surgical outcomes.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This study was partially supported by Point Robotics Medtech Incorporation, which provided the robot system. The funder provided support in the form of salaries for X.Y. Xiao, C.W. Chen, H.K. Chou, and C.Y. Sung, but did not have any additional role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Materials
| Dynamic reference frames | POINT | ||
| FF tool kit: 1.Connecting Rod 2.Combination clamps 3.Multi-pin clamps 4.Schanz screw 5.Spinous process clamp 6.Open wrench 7.Hexagonal wrench |
POINT | ||
| Handpiece | POINT | ||
| Handpiece holder | POINT | ||
| Handpiece stand | POINT | ||
| K-pin | POINT | ||
| Optical tracker | NDI | ||
| Passive spheres | NDI | ||
| Probe | POINT | ||
| Sterile box | POINT | ||
| Sterile drape | POINT | ||
| Trocar | POINT | ||
| Workstation cart | POINT |
References
- Verma, K., Boniello, A., Rihn, J. Emerging techniques for posterior fixation of the lumbar spine. Journal of the American Academy of Orthopaedic Surgery. 24 (6), 357-364 (2016).
- Gaines, R. W. The use of pedicle-screw internal fixation for the operative treatment of spinal disorders. The Journal of Bone and Joint Surgery-American. 82 (10), 1458-1476 (2000).
- Dede, O., Ward, W., Bosch, P., Bowles, A., Roach, J. Using the freehand pedicle screw placement technique in adolescent idiopathic scoliosis surgery: what is the incidence of neurological symptoms secondary to misplaced screws. Spine. 39 (4), 286-290 (2014).
- Costa, F. Erratum: Radiation exposure in spine surgery using an image-guided system based on intraoperative cone-beam computed tomography: analysis of 107 consecutive cases. Journal of Neurosurgery: Spine SPI. 26 (4), 542 (2017).
- Stuer, C., et al. Robotic technology in spine surgery: Current applications and future developments. Intraoperative Imaging. 109, 241-245 (2011).
- Devito, D. P., et al. Clinical acceptance and accuracy assessment of spinal implants guided with SpineAssist surgical robot: retrospective study. Spine. 35 (24), 2109-2115 (2010).
- Fan, Y., et al. Radiological and clinical differences among three assisted technologies in pedicle screw fixation of adult degenerative scoliosis. Scientific Reports. 8 (1), 890 (2018).
- Kantelhardt, S. R., et al. Perioperative course and accuracy of screw positioning in conventional, open robotic-guided and percutaneous robotic-guided, pedicle screw placement. European Spine Joutnal. 20 (6), 860-868 (2011).
- Verma, R., Krishnan, S., Haendlmayer, K., Mohsen, A. Functional outcome of computer-assisted spinal pedicle screw placement: a systematic review and meta-analysis of 23 studies including 5,992 pedicle screws. European Spine Journal. 19 (3), 370-375 (2010).
- Ghasem, A., Sharma, A., Greif, D., Alam, M., Maaieh, M. The Arrival of Robotics in Spine Surgery: A Review of the Literature. Spine. 43 (23), 1670-1677 (2018).
- Roser, F., Tatagiba, M., Maier, G. Spinal robotics: current applications and future perspectives. Neurosurgery. 72 (1), 12-18 (2013).
- Chen, H. Y., et al. Results of using robotic-assisted navigational system in pedicle screw placement. PLoS One. 14 (8), 0220851 (2019).
- . NDI Medical Available from: https://www.ndigital.com/medical/products/polaris-vega (2020)
- Gertzbein, S. D., Robbins, S. E. Accuracy of pedicular screw placement in vivo. Spine. 15 (1), 11-14 (1990).
- Kim, T. T., Johnson, J. P., Pashman, R., Drazin, D. Minimally Invasive Spinal Surgery with Intraoperative Image-Guided Navigation. Biomed Research International. 2016, 5716235 (2016).
- Bailey, S. I., et al. The BWM spinal fixator system. A preliminary report of a 2-year prospective, international multicenter study in a range of indications requiring surgical intervention for bone grafting and pedicle screw fixation. Spine. 21 (17), 2006-2015 (1996).
- Lonstein, J. E., et al. Complications associated with pedicle screws. The Journal of Bone and Joint Surgery-American Volume. 81 (11), 1519-1528 (1999).

