Mitigation of Blood Borne Cell Attachment to Metal Implants through CD47-Derived Peptide Immobilization
Summary
Presented here is a protocol for appending peptide CD47 (pepCD47) to metal stents using polybisphosphonate chemistry. Functionalization of metal stents using pepCD47 prevents the attachment and activation of inflammatory cells thus improving their biocompatibility.
Abstract
The key complications associated with bare metal stents and drug eluting stents are in-stent restenosis and late stent thrombosis, respectively. Thus, improving the biocompatibility of metal stents remains a significant challenge. The goal of this protocol is to describe a robust technique of metal surface modification by biologically active peptides to increase biocompatibility of blood contacting medical implants, including endovascular stents. CD47 is an immunological species-specific marker of self and has anti-inflammatory properties. Studies have shown that a 22 amino acid peptide corresponding to the Ig domain of CD47 in the extracellular region (pepCD47), has anti-inflammatory properties like the full-length protein. In vivo studies in rats, and ex vivo studies in rabbit and human blood experimental systems from our lab have demonstrated that pepCD47 immobilization on metals improves their biocompatibility by preventing inflammatory cell attachment and activation. This paper describes the step-by step protocol for the functionalization of metal surfaces and peptide attachment. The metal surfaces are modified using polyallylamine bisphosphate with latent thiol groups (PABT) followed by deprotection of thiols and amplification of thiol-reactive sites via reaction with polyethyleneimine installed with pyridyldithio groups (PEI-PDT). Finally, pepCD47, incorporating terminal cysteine residues connected to the core peptide sequence through a dual 8-amino-3,6-dioxa-octanoyl spacer, are attached to the metal surface via disulfide bonds. This methodology of peptide attachment to metal surface is efficient and relatively inexpensive and thus can be applied to improve biocompatibility of several metallic biomaterials.
Introduction
Percutaneous coronary intervention is the first line of therapy to treat coronary artery diseases (CAD) and primarily involves stenting the diseased arteries. However, in-stent restenosis (ISR) and stent thrombosis are common complications associated with stent deployment1. Blood interaction at the blood-stent interface is characterized by an almost immediate adsorption of plasma proteins on the metal surface, followed by platelet and inflammatory cell attachment and activation2. The release of the inflammatory cytokines and chemokines from activated inflammatory cells leads to the phenotypic modification of the vascular smooth muscle cells (VSMCs) in the tunica media and triggers their centripetal migration to the intimal compartment. Proliferation of activated VSMC in the intima results in intimal layer thickening, lumen narrowing and in-stent restenosis3. Drug eluting stents (DES) were developed to prevent VSMC proliferation; however, these drugs have an off-target cytotoxic effect on the endothelial cells4,5. Therefore, late stent thrombosis is a common complication associated with DES6,7. Stents made of biodegradable polymers, such as poly-L-lactide have shown much promise in the animal experiments and initial clinical trials, but were eventually recalled when the “real-life” clinical use demonstrated their inferiority to the 3rd generation DES8. Therefore, there is a need to improve the biocompatibility of bare metal stents for better patient outcomes.
CD47 is a ubiquitously expressed transmembrane protein that inhibits the innate immune response when bound to its cognate receptor Signal Regulatory Protein alpha (SIRPα)9. The SIRPα receptor has an immune cell tyrosine inhibitory motif (ITIM) domain and the signaling events upon SIRPα – CD47 interaction ultimately result in the downregulation of inflammatory cell activation10,11,12,13. Research in our lab has shown that recombinant CD47 or its peptide derivative, corresponding to the 22 amino acid Ig domain of the extracellular region of CD47 (pepCD47), can reduce the host immune response to a range of clinically relevant biomaterials14,15,16. Recently, we have demonstrated that pepCD47 can be immobilized to stainless steel stent surfaces and significantly reduce the pathophysiological response associated with restenosis. Of note, the pepCD47 modified surfaces are amenable to relevant usage conditions such as long term storage and ethylene oxide sterilization17. To that end, pepCD47 may be a useful therapeutic target to address the clinical limitations of endovascular stents.
The strategy for the covalent attachment of pepCD47 to a metal surface involves a series of novel chemical modifications of the metal surface. The metal surfaces are first coated with polyallylamine bisphosphonate with latent thiol groups (PABT) followed by the deprotection of the thiols and attachment of polyethyleneimine (PEI) with installed pyridyldithio groups (PDT). PDT groups of PEI unconsumed in the reaction with deprotected PABT thiols are then reacted with pepCD47 incorporating thiols in the terminal cysteine residues, resulting in binding pepCD47 to the metal surface via a disulfide bond14,17,18. We used a fluorophore conjugated pepCD47 (TAMRA-pepCD47) to determine the input concentration of peptide that results in the maximum surface immobilization of the peptide. Finally, we evaluated the acute and chronic anti-inflammatory capacity of the pepCD47 coated metal surfaces, ex vivo, using the Chandler loop apparatus, and monocyte attachment/macrophage expansion assay, respectively.
This paper provides a systematic protocol for the attachment of thiolated peptides to the metal surface; determining the maximum immobilization density of the peptide; and assessing the anti-inflammatory properties of pepCD47 coated metal surfaces exposed to whole blood and isolated monocytes.
Protocol
All human samples for this experiment were obtained in accordance with the IRB of the Children’s Hospital of Philadelphia. All animal experiments were performed upon approval from IACUC of the Children’s Hospital of Philadelphia.
1. Coating bare metal surfaces with PEI-PDT
- Wash the stainless steel foil coupons (1 cm x 1 cm or 0.65 cm x 1 cm) or stainless steel mesh disks with 2-isopropanol in a shaker (60 °C, speed of 200 rpm) for 5 min. Perform this step 2x. Then wash 2x with chloroform (60 °C, speed of 200 rpm) for 10 min each.
- Place the cleansed stainless-steel samples in an oven at 220 °C for 30 min.
- Prepare 5 mL of 0.5% of PABT solution by dissolving 25 mg of polyallylamine bisphosphonate with latent thiol groups (PABT) and 5 mg of potassium bicarbonate (KHCO3) in 5 mL of DDW and incubate at 72 °C in a shaker at 200 rpm for 30 min.
NOTE: For PABT synthesis refer to previously published literature18. - Immerse the baked foils or mesh disks in 0.5% aqueous solution of PABT and incubate in a shaker (72 °C, speed of 200 rpm) for 1 h.
- Wash the PABT-modified samples with deionized distilled water (DDW) for 5x, transfer the specimens in a new vial and wash again with DDW for 5x.
- Prepare a total of 5 mL of TCEP solution (12 mg/mL) by dissolving 60 mg of Tris (2-carboxyethyl) phosphine hydrochloride (TCEP) in 5 mL of 0.1 M acetic buffer (0.57 mL of glacial acetic acid, 820 mg of sodium acetate in 100 mL of DDW).
- Treat the PABT-modified samples with TCEP for 15 min at room temperature (RT) on a shaker.
NOTE: TCEP is used to deprotect the thiol groups. - Degas DDW in a round bottom flask by using a vacuum generating device, such as a lyophilizer and wash the TCEP treated foils or mesh disks with degassed DDW for 5x. Transfer the samples in a new vial, and additionally wash 5x with degassed DDW.
NOTE: It is paramount to work fast to prevent oxidation of thiols on the metal surface by atmospheric oxygen. - Prepare 5 mL of 1% PEI-PDT solution by diluting 212.5 µL of stock PEI-PDT and 125 µL of 0.4 M sodium acetate in degassed DDW. Carry out step 1.9 simultaneously with step 1.8 to minimize exposure of the samples to atmospheric air.
NOTE: The synthesis of PEI-PDT is described in previously published literature18. - Incubate the washed stainless-steel specimens with 1% PEI-PDT. Replace air with argon gas, seal the vials air-tight and mix on a shaker at RT for 1 h. Either proceed immediately with the peptide conjugation or store at 4 °C up to 1 week.
2. Attachment and qualitative/quantitative assessment of fluorophore conjugated pepCD47 retention on metal surface using fluorescence microscopy and fluorimetry
- Wash foil coupons or mesh disks prepared as described in the section 1, steps 1.1-1.10 above with DDW 5x. Transfer the samples to a new vial and wash with DDW for additional 5x. Finally wash 2x with degassed ethanol and 2x with degassed dimethyl formamide (DMF).
- Prepare tetramethylrhodamine (TAMRA)-conjugated pepCD47 stock solution by dissolving TAMRA-conjugated pepCD47 powder in degassed dimethylformamide (DMF) to a final concentration of 1 mg/mL. Aliquot the stock solution in 1 mL allotments. Store in well-sealed tubes under argon atmosphere at -20 °C.
- Dilute 1 mg/mL of the stock solution of TAMRA-conjugated pepCD47 using degassed DMF to prepare the following concentrations of fluorophore conjugated pepCD47 – 10, 30, 100, and 200 µg/mL.
NOTE: If TAMRA-conjugated pepCD47 appears to be precipitated, reduce the stock TAMRA-conjugated pepCD47 solution using TCEP beads in ratio 1:1, at RT for 20 min. Note that before adding the TAMRA-conjugated pepCD47 to the TCEP beads, spin the TCEP beads solution, remove the supernatant and then proceed with addition of the TAMRA-conjugated pepCD47. - Incubate the PEI-PDT modified foil coupons with 10, 30, 100 or 200 µg/mL of TAMRA-conjugated pepCD47 in triplicates for each condition on a shaker at RT under argon atmosphere for 1 hour. Incubate PEI-PDT modified mesh disks, as well as mesh disk not modified beyond step 1.2 (bare metal controls) with 100 µg/mL of TAMRA-conjugated pepCD47 in triplicates at RT under argon atmosphere on a shaker for 1 h.
NOTE: From this step onwards, the vials are wrapped in aluminum foil to protect the contents from light. - Wash the fluorophore conjugated pepCD47-coated surfaces to remove the non-covalently attached peptide in the following order: DMF (3x), DMF/DDW at 1:1, DDW (3x), 0.3% SDS in 20 mM Tris pH 7.4 (3x, 5 min each at 70 °C on a shaker), DDW (3x), change vials, and a final DDW wash.
- Place control and covalently conjugated mesh disks on a microscope glass, add 50 µL of PBS and place a coverslip. Image mesh disks using an inverted fluorescence microscope equipped with a rhodamine filter set. Take representative images at 100x magnification.
- Prepare 15 mL of 12 mg/mL TCEP solution by dissolving 180 mg of TCEP in 1:1 v/v mixture of methanol and 0.1 M acetic buffer.
- Incubate each washed foil with 1 mL of TCEP solution on a shaker at RT for 15 min.
- Prepare the following standards by serially diluting the TAMRA-conjugated pepCD47 stock (1 mg/mL) – 100 µg/mL, 10 µg/mL, 1 µg/mL, 0.1 µg/mL, and 0.01 µg/mL. Use the TCEP solution as the diluent.
- Analyze the TAMRA-conjugated pepCD47 released from the metal surface against the calibration curve generated using the standards by fluorimetry at 544/590 nm excitation and emission wavelengths.
3. Attaching human pepCD47 to PEI-PDT modified surfaces
- Wash PEI-PDT coated samples formulated as described in the section 1, steps 1.1-1.10 above, with degassed DDW 5x, change the vial and wash with degassed DDW 5x.
- Prepare human pepCD47 stock solution (1 mg/mL) by dissolving human pepCD47 powder in degassed 50% acetic acid to achieve a concentration 1 mg/mL.
- Prepare working concentration of human pepCD47 (100 µg/mL) by dissolving 500 µL of the stock of human pepCD47 in 4,500 µL of degassed 1x phosphate buffered saline (PBS).
- Incubate the washed PEI-PDT coated samples with 100 µg/mL of pepCD47 at RT with shaking for 1 h.
- Wash the human pepCD47-coated samples to remove excess peptide in the following order PBS (3x), DDW (3x), 0.2% Tween-20 (3x, 5 min each), DDW (3x), change vials and final DDW wash.
NOTE: Human pepCD47 coated surfaces can be stored dry at 4 °C for up to 6 months.
4. Coating the PEI-PDT modified surfaces with scrambled sequence (Scr)
- Dissolve the scrambled sequence powder in degassed 0.1% acetic acid to prepare a stock solution of 1 mg/mL.
- Prepare a solution of 100 µg/mL of scrambled peptide by dissolving 500 µL of the in 4,500 µL of degassed 0.1% acetic acid.
- Coat the washed PEI-PDT specimens with 100 µg/mL of scrambled peptide at RT with shaking for 1 h.
- To remove unattached scrambled peptide, wash the surfaces in the following order 0.01% acetic acid (3x), DDW (3x), 0.2% Tween-20 (3x, 5 min), DDW (3x), change vials and one DDW wash.
5. Chandler loop for analyzing cellular attachment to metal surfaces
- Coat the metal foils (0.65 cm x 1cm) with either human pepCD47 or scrambled peptide as per the description on section 1 followed by 3 or 4.
- Cut ¼” PVC tubes into three 38 cm long pieces.
- Insert up to 8 unmodified, scrambled peptide or pepCD47 modified metal foils in three different tubes.
- Collect 30 mL of blood from healthy human donors free of any anti-platelet medications as per institutional IRB protocol. Preload syringe with 1 mL of 4% sodium citrate to prevent coagulation of collected blood.
- Put 10 mL of blood into each tube using a 10 mL syringe and connect the ends with metal adapters. Place the blood-filled tubes on the wheels of the Chandler loop apparatus.
- Pass the blood along the metal foils by wheel rotation at 37 °C at the speed calculated to produce the shear of 25 dyns/cm2 for 4 h.
- Drain the blood from the tubes and dispose of the blood according to the IRB requirements.
- Cut the tubes using the scalpel to retrieve the foils from each tube.
- Prepare 2% glutaraldehyde solution by diluting the 10 ml of 4% glutaraldehyde solution using 10 mL of sodium cacodylate buffer with 0.1 M sodium chloride.
- Incubate the foils in 2% glutaraldehyde solution for 15 min and store at 4 °C overnight. Before analyzing, wash the metal foils 3x with PBS.
6. Analyzing cellular attachment to metal surfaces using CFDA dye
- Warm 8 mL of PBS in 15 mL tube in a water bath set to 37 °C.
- Prepare the CFDA (carboxy-fluorescein diacetate, succinimidyl ester) dye solution as follows – add 90 µL of DMSO to one CFDA dye vial to achieve a stock concentration of 10 mM. Next, prepare the working concentration of 93.75 µM by adding 75 µL of the stock CFDA to 8 mL of warm PBS. Mix by inverting the tubes a few times and cover the tube with aluminium foil.
NOTE: It is advisable to freshly prepare the CFDA dye before every use. - Incubate each foil with 1 mL of CFDA dye in a 24 well plate. Cover the plate with aluminum foil and incubate the plate at 37 °C for 15 min.
- Wash the metal foils 3x with PBS to remove excess dye. Image using the inverted fluorescence microscope.
7. Monocyte attachment and macrophage expansion on the pepCD47-modified and bare metal surfaces
- Collect 10 mL of peripheral blood via the vena cava access during the sacrifice of a 400-450 g male Sprague-Dawley rat. Mix immediately with 1,000 IU of sodium heparin to prevent coagulation.
- Pipette 10 mL of density gradient medium into a 50 mL conical tube. Mix the blood with 5 mL of PBS, and carefully layer diluted blood over the density gradient medium using a Pasteur pipette. Centrifuge in a swinging bucket rotor at 800 x g and 18-25 °C for 20 min with minimal settings of acceleration and deceleration.
- Using a Pasteur pipette, collect an opaque layer of buffy coat on the interface between plasma and Ficoll. Dilute buffy coat 1:3 with PBS and centrifuge at 550 x g and 4 °C for 10 min to buffy coat cells.
- Re-suspend buffy coat cells in 3 mL of ACK lysis buffer to lyse contaminating erythrocytes. Incubate on ice for 4 min. Add 12 mL of cell separation buffer (CSB; 0.5% BSA, 0.5% FBS, 2 mM EDTA/PBS).
- Centrifuge at 550 x g and 4 °C for 10 min. Re-suspend the pellet in 10 mL of CSB.
- Centrifuge at 200 x g and 4 °C for 10 min to eliminate platelets. Repeat twice.
- Re-suspend the pellet in 500 µL of CSB. Add 10 µg of each of the following mouse anti-rat antibodies: CD8a (clone OX-8), anti-CD5 (clone OX-19), anti-CD45RA (clone OX-33), and anti-CD6 (clone OX-52).
- Incubate at 4 °C on a vertical tube rotator for 1 h. Add 9.5 mL of CSB. Centrifuge at 300 x g and 4 °C for 10 min. Discard supernatant, re-suspend the pellet in 10 mL of CSB and repeat centrifugation.
- Re-suspend the pellet in 500 µL of CSB. Add 150 µL of goat anti-mouse IgG microbeads. Incubate at 4 °C on a vertical tube rotator for 20 min. Add 9.5 mL of CSB. Centrifuge at 300 x g and 4 °C for 10 min.
- Discard the supernatant. Re-suspend the pellet in 1 mL of CSB.
- Place a LS column in a magnetic separator. Prime the LS column with 3 mL of CSB. Discard the column throughput. Add 1 mL of re-suspended cell pellet from the step 7.10. Start collecting the throughput. After flow stops, add 5 mL of CSB and keep collecting the column throughput until the flow stops.
- Centrifuge the column throughput (6 mL) containing negatively selected monocytes at 300 x g and 4 °C for 10 min. Re-suspend the resulting small pellet in 2 mL of RPMI-1640 medium supplemented with 10% FCS, 1% pen/strep and 100 ng/mL rat macrophage colony stimulating factor (M-CSF).
- Count the monocytes using hemocytometer. Adjust monocyte concentration to 5 x 105 cells/mL.
- Add 1 mL volumes of monocyte suspension to the individual wells of a 12 well plate with the bare stainless steel foil samples (N=3) or stainless steel samples modified with rat pepCD47 (N=3) as per 1.1-1.10 and 3.1-3.6.
- Change the medium on days 3 and 5 post-seeding. On day 6 post-seeding wash the cells with PBS and fix with 4% paraformaldehyde at room temperature for 15 min. Wash twice with PBS for 5 min.
- Remove the stainless-steel foils and place them individually into a new 12 well plate. Do not flip the foils.
- Incubate in 0.5% Tween-20/PBS for 15 min to permeabilize the cells. Wash twice with PBS for 5 min.
- Block in 10% goat serum/PBS for 20 min. Aspirate the serum. Do not wash. Add mouse anti-rat CD68 antibody (diluted 1:100 in 1%BSA/PBS). Incubate at room temperature for 1 h. Wash 3x in PBS for 5 min each.
- Add goat anti-mouse IgG Alexa Fluor 546 (diluted 1:200 in 1% BSA/PBS). Incubate in dark at room temperature for 45 min. Wash in PBS for 5 min. Counterstain with 1 µg/mL Hoechst 33342 dye in dark at room temperature for 10 min. Wash 3x in PBS for 5 min each.
- Flip the foils and image using a fluorescent microscope with the inverted optics. Capture images at 200x magnification with blue and red filter settings.
- Count the attached monocytes in the individual images and calculated the group averages and standard deviations.
Representative Results
The metal surfaces are rendered thiol-reactive for peptide attachment via a series of chemical modifications, as illustrated in Figure 1. PABT incubation followed by PEI-PDT treatment makes the metal surface amenable for peptide attachment. Peptide CD47 (pepCD47) containing cysteine residue at C-terminus joined to the core pepCD47 sequence through a flexible dual AEEAc bridge is covalently attached to the thiol-reactive surfaces via disulfide bonds. Using this protocol, we have demonstrated that pepCD47 remains stably attached to the metal surface for up to six months and can withstand normal physiological shear stress and sterilization procedures17.
The maximum peptide retention was determined by appending TAMRA-conjugated pepCD47 to the metal surface followed by the extensive washing to eliminate non-covalently bound peptide, cleavage of TAMRA-pepCD47 by reduction of disulfide bridges tethering the peptide to the surface, and analytical quantification using a fluorimetric assay. Specifically, increasing input amounts of TAMRA-conjugated pepD47 (1 mL of 10 – 200 µg/mL solution) were appended to the PEI-PDT coated surfaces and washed several times to remove the non-covalently bound peptide. The concentration of covalently bound TAMRA-conjugated pepCD47 was determined by using a reducing agent TCEP to release the covalently attached peptide and assessing its fluorescence against the standards. The input concentration of 10, 30, 100 and 200 µg/mL demonstrated peptide retention of 28 ± 2, 78 ± 2, 182 ± 14 and 157 ± 25 ng/cm2 respectively (Figure 2). Thus, the maximal immobilization density of pepCD47 on the metal surface was found to be approximately 180 ng/cm2 which was achieved with an input concentration of 100 µg/mL. The proper immobilization of TAMRA-conjugated pepCD47 on the PABT/PEI-PDT-modified metal surfaces was further corroborated by fluorescence microscopy (Figure 3) that demonstrated a uniform fluorescence emitted from the surface of TAMRA-conjugated pepCD47-treated mesh disks (Figure 3A). Only minimal fluorescence was detected on the surface of the control meshes that lacked PABT/PEI-PDT modification (Figure 3B), thereby excluding a non-specifically bound TAMRA-conjugated pepCD47 as the main source of fluorescence.
Next, we evaluated the ability of the pepCD47 coated surfaces to prevent acute bloodborne cell attachment as compared to the unmodified and scrambled sequence peptide modified surfaces. The scramble peptide has the same amino acid composition as pepCD47 but in a different order14,17. The bloodborne cell attachment was evaluated by rotation of blood from healthy human volunteers across unmodified, scrambled, and pepCD47 modified surfaces in the Chandler loop apparatus followed by washing to remove unattached cells, fixation, and staining with CFDA dye. The surfaces were visualized by fluorescence microscopy. Consistent with our previously published data14,17 pepCD47 coated surfaces show a drastic reduction in bloodborne cell attachment as compared to the scrambled modified and unmodified controls (Figure 4).
To expand on the effects of pepCD47 surface modification on inflammatory cell attachment and proliferation, we used negative immunoselection of rat buffy coat cells to isolate monocytes19, and cultured the isolated monocytes on bare metal and pepCD47-modified stainless steel foils for 6 days in the presence of M-CSF. The results of this study show a combined 58% attenuation of acute monocyte attachment, their phenotypic conversion to macrophages, and macrophage proliferation on the pepCD47 functionalized surfaces (Figure 5A) compared to the bare metal controls (Figure 5 B,C).
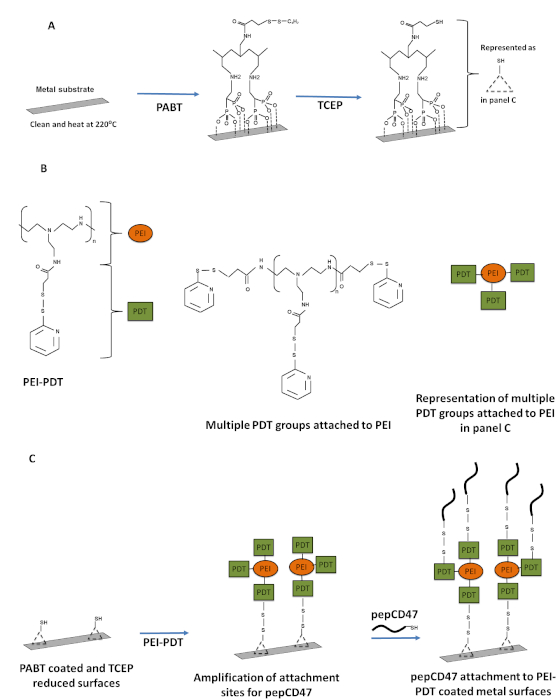
Figure 1: Schematic representation of the steps involved in appending pepCD47 to metal surfaces. (A) The clean metal samples were baked at 220 °C to oxidize the metal surface. The bisphosphonate groups of the polallylamine bisphosphonate with latent thiol groups (PABT) formed co-ordinate bonds with the metal oxides and coat the metal surfaces to form a functionalized monolayer. The PABT coated metal surfaces were further treated with TCEP for deprotection of the thiol groups. (B) The structure of PEI-PDT and symbolic representation. (C) The PABT coated and TCEP reduced surfaces were treated with PEI-PDT which amplified the total number of thiol-reactive groups available for attachment of the thiolated peptide. Finally, PEI-PDT coated surfaces were reacted with terminal cysteine groups of pepCD47, and the peptide was attached to the surface via disulfide bonds. Please click here to view a larger version of this figure.
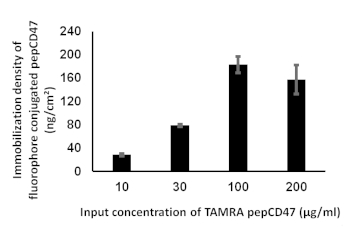
Figure 2: Determining the immobilization density of pepCD47 on metal surface. 1 cm x 1cm metal foils were modified using increasing concentrations (10, 30, 100 and 200 µg/mL) of fluorophore conjugated pepCD47. The excess peptide was removed using several washing steps then treated with 1 mL TCEP solution to cleave the fluorophore conjugated fluorophore. The concentration of the peptide covalently attached to the metal surface was analyzed fluorimetrically using a standard curve prepared with defined concentrations of fluorophore conjugated pepCD47. The immobilization density was represented as ng/cm² of peptide attached to the metal surface. Data is expressed as mean ± SEM and is representative of at least three independent experiments. Please click here to view a larger version of this figure.
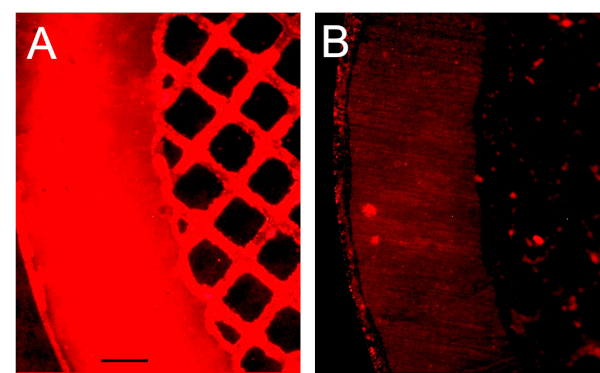
Figure 3: Fluorescence microscopy imaging of the stainless-steel surface modified with TAMRA-conjugated pepCD47. Stainless steel mesh disks, consecutively modified with PABT, TCEP, PEI-PDT (A) or unmodified (B) were reacted with TAMRA-conjugated pepCD47. The properly conjugated and control bare metal meshes were extensively washed and imaged at 100x magnification. The scale bar length is 100 µm. Please click here to view a larger version of this figure.
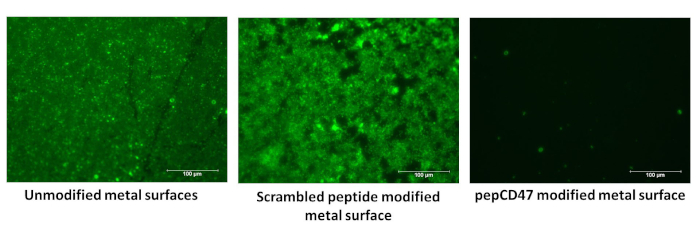
Figure 4: Evaluating acute anti-inflammatory and anti-thrombotic functions of pepCD47. 0.65 cm x 1cm metal foils were coated with 100 µg/mL of either human pepCD47 or scrambled peptide and exposed to blood in the Chandler loop apparatus. The unbound cells were removed by washing with PBS and the foils were fixed in 2% glutaraldehyde. The unmodified, scrambled modified and human pepCD47 modified surfaces were then incubated with the CFDA dye for 15 mins at 37 °C, washed with PBS and analyzed using a fluorescence microscope. Please click here to view a larger version of this figure.
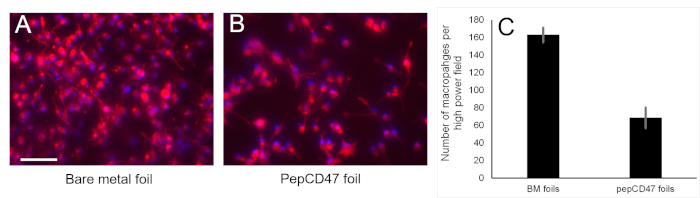
Figure 5: A prevalence of CD68-positive macrophages on the bare and pepCD47-functionalized metal surfaces. Rat peripheral blood-derived monocytes were isolated by gradient density centrifugation followed by negative immunoselection with magnetic microbeads. 5 x 105 monocytes were added into the wells of a 12 well plate with individually placed bare metal foil samples (N=3) or the samples derivatized with rat pepCD47. Macrophage differentiation was stimulated by 100 ng/ml M-CSF. Six days after seeding the cells were fixed, and immunostained with anti-rat CD68 antibody, secondary Alexa Fluor-546 (red) conjugated antibody and counterstained with Hoechst 33342 nuclear dye (blue). Representative images of the bare metal (A) and pepCD47-functionalized (B) surfaces were captured at 200x magnification and merged. The scale bar length is 100 µm. Please click here to view a larger version of this figure.
Discussion
We demonstrate and describe a relatively novel chemical strategy to append therapeutic peptide moieties to a stainless-steel surface with the overarching goal of reducing the surface’s reactivity with inflammatory cells found in blood. The bisphosphonate chemistry described herein involves co-ordinate bond formation between the metal oxides and bisphosphonate groups of PABT. The thickness of polybisphosphonate monolayer formed on the metal surface does not exceed 5 nm18, and, therefore, is inconsequential for the potential proinflammatory effects of the thick polymer coatings20. Deprotection of latent thiol group in the aliphatic side chains of PABT primes the metal surfaces for further chemical modification with thiol-reactive compounds. PEI-PDT is branched polyethyleneimine (average Mw=25,000) in which ~20% of ethyleneimine links are modified with thiol-reactive pyridyldithio groups. Since only a minority of PDT groups in PEI-PDT are consumed in the reaction with the PABT-derived thiols, the surface chemistry after PEI-PDT tethering is changed to thiol-reactive to enable attachment of thiolated peptides18. This chemical strategy was developed and widely used in our lab for attachment of several biomolecules, such as recombinant proteins14, peptides14,17, and viral gene vectors18 to the metal surface. Although, for most of our work we have used stainless steel surfaces, PABT can interact with other metal alloys21 and thus has the potential to improve the biocompatibility and therapeutic potential of a wide range of metallic biomaterials.
Our current methodology indicates that the maximum immobilization density of fluorophore conjugated pepCD47 on metal surfaces is 180 ng/cm2, which is less as compared to our previously published data17. We attribute this discrepancy to the different washing strategies used in both studies. In our current protocol we have used SDS which completely removes non-covalently bound peptides as compared to Tween-20 which was used in our initial studies. However, 180 ng/cm2 corresponds to approximately 4 x 106 molecules/µm2 of pepCD47. This immobilization density is much higher than the physiological levels of CD47 on cell surfaces which are approximately 390 molecules/µm2,22. Thus, we predict that stringent washing conditions would not significantly affect the anti-inflammatory properties of pepCD47 modified metal surfaces.
This methodology of attachment of pepCD47 to metal is highly reproducible, however there are several steps in the protocol which need careful attention. Firstly, after coating the metal surface with PABT and reducing it using TCEP, the thiols are prone to oxidation when exposed to air. Thus, it is of utmost importance that the surfaces are always submerged in degassed water. For the same reason the PEI-PDT solution is made in degassed water and argon is added to the vials during the incubation of foils coated with PEI-PDT. Secondly, one must consider the potential for the precipitation of solubilized peptides. PepCD47 has a terminal cysteine residue, as well as cysteine residues in the sequence. Thus, when improperly stored, there is a high possibility that the peptides polymerize and precipitate out of the solution. To address this potential problem, it is recommended that the peptides should be reduced using TCEP beads for 20 min to 1 h prior to incubation with PEI-PDT coated surfaces. TCEP would help to reduce the peptides and increase their solubility in their respective diluent. It is also recommended to displace ambient air with argon in the vials while incubating the PEI-PDT coated samples with thiolated peptide.
If the above-mentioned precautions are followed, the coating technique is reproducible and the only potential limitation for this approach is the commercial non availability of reagents PABT and PEI-PDT.
We used a Chandler loop apparatus to provide ex vivo proof of concept analysis to understand the interaction of pepCD47 modified metal surfaces with blood cells and it has been successfully used by our lab to show the anti-inflammatory properties of pepCD47 coated surfaces14,15,17,23. In this study, we used CFDA dye that fluoresces only after entering the cells when the acetate groups of the dye are cleaved by the intracellular esterase. The benefits of this staining procedure are that it stains the anucleated platelets and red blood cells as wells as the nucleated cells such as leukocytes. Thus, utilizing CFDA dye provides an assessment of both acute anti-thrombotic and anti-adhesive properties of pepCD47 coated surfaces. More detailed assessment can be acquired through scanning electron microscopy and/or immunostaining, both of which we detailed previously24. Results of the current study validate that the human pepCD47 coated metal surfaces are anti-thrombotic and anti-adhesive as compared to the unmodified and scrambled sequence controls.
To further investigate whether pepCD47-modified surfaces alter growth characteristics of attached inflammatory cells, bare metal stainless steel foils or foils formulated with pepCD47 were exposed to isolated rat monocytes in the presence of syngeneic M-CSF. The cells were cultured under stationary conditions for 6 days to allow the phenotypic conversion and expansion of macrophages. In accordance with our previous results observed in the different experimental settings14, a profound decrease of inflammatory cell number on pepCD47 functionalized surfaces was demonstrated in the current study.
Thus, our studies ultimately prove that the novel polybisphosphonate chemistry for coating metals with pepCD47 is an effective way of improving biocompatibility of metal surfaces and can be applied in other biomedical applications, such as artificial joints.
Divulgations
The authors have nothing to disclose.
Acknowledgements
Protocol development and studies presented in this paper were supported by NIH (NBIB) R01 funding (# EB023921) to IF and SJS, and NIH (NHLBI) R01 funding (# HL137762) to IF and RJL.
Materials
| 1 M Tris-HCL | Invitrogen | 15567-027 | pH – 7.5 |
| 4% Glutaraldehyde | Electron Microscopy Sciences | 16539-07 | |
| 4% Sodium Citrate | Sigma | S5770 | |
| ACK lysing buffer | Quality Biologicals | 118-156-721 | |
| anti-CD45RA Ab (mouse anti-rat; clone OX-19) | Biolegend | 202301 | |
| anti-CD5 Ab (mouse anti-rat; clone OX-19) | Biolegend | 203501 | |
| anti-CD6 Ab (mouse anti-rat; clone OX-52) | BD Biosciences | 550979 | |
| anti-CD68 Ab (mouse anti-rat; clone ED-1) | BioRad | MCA341 | |
| anti-CD8a Ab (mouse anti-rat; clone OX-8) | Biolegend | 201701 | |
| Chloroform Certified ACS | Fisher Chemical | C298-500 | |
| Dimethyl Formammide (DMF) | Alfa Aesar | 39117 | |
| Embra stainless steel grid | Electron Microscopy Sciences | E200-SS | stainless steel mesh mesh disks |
| Ficoll Hypaque | GE Healthcare | 17-1440-02 | |
| Glacial acetic acid | ACROS organic | 148930025 | |
| goat anti-mouse IgG Alexa Fluor | ThermoFisher | A11030 | |
| Heparin sodium | Sagent Pharmaceuticals | 402-01 | |
| Human pepCD47 | Bachem | 4099101 | |
| Isopropanol | Fisher Chemical | A426P-4 | |
| Metal adapters | Leur Fitting | 6515IND | 1 way adapter 316 ss 1/4"-5/16" hoes end |
| Methanol | RICCA chemical company | 4829-32 | |
| Microscope | Nikon Eclipse | TE300 | |
| Phosphate buffered saline (PBS) | Gibco | 14190-136 | |
| Pottasium Bicarbonate (KHCO3) | Fisher Chemical | P184-500 | |
| PVC tubes | Terumo-CVS | 60050 | 1/4" X 1/16 8' |
| sodium cacodylate buffer with 0.1M sodium chloride | Electron Microscopy Sciences | 11653 | |
| Sodium Dodecyl Sulfate (SDS) | Bio-Rad laboratories | 161-0302 | |
| Sodum actetate (C2H3NaO2) | Alfa Aesar | A13184 | |
| Src peptide | Bachem | 4092599 | |
| Stainless steel (AISI 304) cylinder-shaped samples with a lumen | Microgroup, Medway, MA | 20097328 | 1 cm X 6 mm OD |
| Stainless steel foils (AISI 316L) | Goodfellow, Coraopolis, PA | 100 mm X 100 mm X 0.05 mm | |
| Tetramethylrhodamine-conjugated pepCD47 (TAMRA-pepCD47) | Bachem | 4100277 | |
| TMB (3,3’ ,5,5’ -tetramethylbenzidine) substrate and tris (2-carboxyethyl) phosphine hydrochloride (TCEP) | Thermo Scientific | PG82089 | |
| Tween-20 | Bio-Rad laboratories | 170-6531 | |
| Vybrant CFDA SE Cell Tracer Kit | Invitrogen | V12883 |
References
- Buccheri, D., Piraino, D., Andolina, G., Cortese, B. Understanding and managing in-stent restenosis: a review of clinical data, from pathogenesis to treatment. Journal Thoracic Disease. 8 (10), 1150-1162 (2016).
- van Oeveren, W. Obstacles in haemocompatibility testing. Scientifica. 2013, 392584 (2013).
- Mitra, A. K., Agrawal, D. K. In stent restenosis: bane of the stent era. Journal of Clinical Pathology. 59 (3), 232-239 (2006).
- Iqbal, J., Gunn, J., Serruys, P. W. Coronary stents: historical development, current status and future directions. British Medical Bulletin. 106, 193-211 (2013).
- Hoffmann, R., et al. Patterns and mechanisms of in-stent restenosis. A serial intravascular ultrasound study. Circulation. 94 (6), 1247-1254 (1996).
- Stefanini, G. G., Windecker, S. Stent thrombosis: no longer an issue with newer-generation drug-eluting stents. Circulation: Cardiovascular Interventions. 5 (3), 332-335 (2012).
- Palmerini, T., et al. Clinical outcomes with bioabsorbable polymer- versus durable polymer-based drug-eluting and bare-metal stents: evidence from a comprehensive network meta-analysis. Journal of the American College of Cardiology. 63 (4), 299-307 (2014).
- Omar, W. A., Kumbhani, D. J. The Current Literature on Bioabsorbable Stents: a Review. Current Atherosclerosis Reports. 21 (12), 54 (2019).
- Slee, J. B., Christian, A. J., Levy, R. J., Stachelek, S. J. Addressing the Inflammatory Response to Clinically Relevant Polymers by Manipulating the Host Response Using ITIM Domain-Containing Receptors. Polymers (Basel). 6 (10), 2526-2551 (2014).
- Oldenborg, P. A., et al. Role of CD47 as a marker of self on red blood cells. Science. 288 (5473), 2051-2054 (2000).
- vanden Berg, T. K., vander Schoot, C. E. Innate immune ‘self’ recognition: a role for CD47-SIRPalpha interactions in hematopoietic stem cell transplantation. Trends in Immunology. 29 (5), 203-206 (2008).
- Tengood, J. E., Levy, R. J., Stachelek, S. J. The use of CD47-modified biomaterials to mitigate the immune response. Experimental Biology Medicine (Maywood). 241 (10), 1033-1041 (2016).
- Tsai, R. K., Rodriguez, P. L., Discher, D. E. Self inhibition of phagocytosis: the affinity of ‘marker of self’ CD47 for SIRPalpha dictates potency of inhibition but only at low expression levels. Blood Cells, Molecules and Diseases. 45 (1), 67-74 (2010).
- Slee, J. B., et al. Enhanced biocompatibility of CD47-functionalized vascular stents. Biomaterials. 87, 82-92 (2016).
- Finley, M. J., et al. Diminished adhesion and activation of platelets and neutrophils with CD47 functionalized blood contacting surfaces. Biomaterials. 33 (24), 5803-5811 (2012).
- Stachelek, S. J., et al. The effect of CD47 modified polymer surfaces on inflammatory cell attachment and activation. Biomaterials. 32 (19), 4317-4326 (2011).
- Inamdar, V. V., et al. Stability and bioactivity of pepCD47 attachment on stainless steel surfaces. Acta Biomaterialia. 104, 231-240 (2020).
- Fishbein, I., et al. Local delivery of gene vectors from bare-metal stents by use of a biodegradable synthetic complex inhibits in-stent restenosis in rat carotid arteries. Circulation. 117 (16), 2096-2103 (2008).
- Moser, K. V., Humpel, C. Primary rat monocytes migrate through a BCEC-monolayer and express microglia-markers at the basolateral side. Brain Research Bulletin. 74 (5), 336-343 (2007).
- vander Giessen, W. J., et al. Marked inflammatory sequelae to implantation of biodegradable and nonbiodegradable polymers in porcine coronary arteries. Circulation. 94 (7), 1690-1697 (1996).
- Fishbein, I., et al. Bisphosphonate-mediated gene vector delivery from the metal surfaces of stents. Proceedings of the National Academy of Sciences of the United States of America. 103 (1), 159-164 (2006).
- Mouro-Chanteloup, I., et al. Evidence that the red cell skeleton protein 4.2 interacts with the Rh membrane complex member CD47. Blood. 101 (1), 338-344 (2003).
- Finley, M. J., Clark, K. A., Alferiev, I. S., Levy, R. J., Stachelek, S. J. Intracellular signaling mechanisms associated with CD47 modified surfaces. Biomaterials. 34 (34), 8640-8649 (2013).
- Slee, J. B., Alferiev, I. S., Levy, R. J., Stachelek, S. J. The use of the ex vivo Chandler Loop Apparatus to assess the biocompatibility of modified polymeric blood conduits. Journal of Visualized Experiments. (90), e51871 (2014).

