In Vitro 3D Cell-Cultured Arterial Models for Studying Vascular Drug Targeting Under Flow
Summary
Here, we present a new protocol to study and map the targeted deposition of drug carriers to endothelial cells in fabricated, real-sized, three-dimensional human artery models under physiological flow. The presented method may serve as a new platform for targeting drug carriers within the vascular system.
Abstract
The use of three-dimensional (3D) models of human arteries, which are designed with the correct dimensions and anatomy, enables the proper modeling of various important processes in the cardiovascular system. Recently, although several biological studies have been performed using such 3D models of human arteries, they have not been applied to study vascular targeting. This paper presents a new method to fabricate real-sized, reconstructed human arterial models using a 3D printing technique, line them with human endothelial cells (ECs), and study particle targeting under physiological flow. These models have the advantage of replicating the physiological size and conditions of blood vessels in the human body using low-cost components. This technique may serve as a new platform for studying and understanding drug targeting in the cardiovascular system and may improve the design of new injectable nanomedicines. Moreover, the presented approach may provide significant tools for the study of targeted delivery of different agents for cardiovascular diseases under patient-specific flow and physiological conditions.
Introduction
Several approaches have recently been applied utilizing 3D models of human arteries1,2,3,4,5. These models replicate the physiological anatomy and environment of different arteries in the human body in vitro. However, they have been mainly used in cell biology studies. Current studies on vascular targeting of particles to the endothelium include in silico computational simulations6,7,8, in vitro microfluidic models9,10,11, and in vivo animal models12. Despite the insights they have provided, these experimental models have failed to accurately simulate the targeting process that occurs in human arteries, wherein blood flow and hemodynamics constitute dominant factors. For example, the study of particle targeting to atherosclerotic regions in the carotid artery bifurcation, which are known for their complex recirculation flow pattern and wall shear stress gradient, may impact the journey taken by the particles before they reach the endothelium13,14,15,16. Therefore, these studies must be performed under conditions that replicate the physiological environment, i.e., size, dimension, anatomy, and flow profile.
Recently, this research group fabricated 3D-reconstructed human arterial models to study the deposition and targeting of particles to the vasculature17. The models were based on geometrical 3D replicas of human blood vessels, which were then cultured with human ECs that subsequently lined their inner walls. In addition, when subjected to a perfusion system that produces physiological flow, the models accurately replicated physiological conditions. The perfusion system was designed to perfuse fluids at a constant flow rate, using a peristaltic pump in both closed and open-circuit configurations (Figure 1). The system can be used as a closed-circuit to map particle deposition and targeting to the cells seeded inside the carotid model. In addition, it can be used as an open circuit to wash out non-adherent particles at the end of the experiments and to clean and maintain the system. This paper presents protocols for the fabrication of 3D models of the human carotid bifurcation, design of the perfusion system, and mapping of the deposition of targeted particles inside the models.
Protocol
NOTE: This protocol describes the fabrication of a 3D model of the carotid artery and can be applied to generate any other artery of interest by simply modifying the geometric parameters.
1. Design and fabrication of a 3D bifurcation of the human carotid artery model
- Choose images from patients or previously studied geometries of the human carotid artery bifurcation, and create a computer-aided design model of the mold that needs to be printed.
NOTE: The carotid artery bifurcation has one inlet and two outlets. It is important to design a 3D mold frame around the artery and temporary printing supports between the frame and the artery mold (Figure 2A-C). - Print the geometries using a 3D printer. Cut the temporary printing supports, and use sandpaper to polish and smooth the molds, especially in the areas where the supports were cut. Rinse the sanded model with isopropyl alcohol to remove the plastic dust, and allow to completely dry in a chemical hood for 2-3 h.
NOTE: Here, the printed molds were made of clear resin v4 (Figure 2D,E). - To easily dissolve the plastic, spray the molds with transparent lacquer inside a chemical hood, and allow to air-dry for 1 h. Repeat this 3x.
NOTE: Here, 2X Ultra Cover Clear Spray was used, but any other kind should be suitable as long as it is not wood lacquer. Confirm that there is no exposed plastic left because the plastic may react with the silicone and prevent it from properly solidifying. The quality of the sprayed surface will determine the quality of the surface of the final silicone model. - Cut transparent rectangular slides/strips of smooth plastic of the same dimensions as the mold frame, and glue them using the lacquer to the frame on all sides and from one side of the frame, such that it will be sealed at the bottom and open at the top. Apply the lacquer using a paint brush inside a chemical hood, and allow the slides to completely dry for at least 24 h (Figure 2F).
- To prepare the silicone rubber mixture, add liquid silicone with its curing agent (mass ratio 1:10) in a plastic plate, and stir thoroughly to ensure complete mixing. For the carotid model, add 54 g of the silicone and 6 g of its curing agent.
- Cool the mixture for 15 min at 4 °C, then degas it in a vacuum desiccator until all the air bubbles have been eliminated. Place the mold in the desiccator (with the open side facing up), slowly pour the silicone mixture into the mold, and again remove the air bubbles until the mixture is clear.
- Let the mold stand with the silicone overnight at room temperature (if possible, leave it in the desiccator without vacuum). If the mixture has not fully dried, incubate it at 60 °C for another few hours.
- Once the mixture is fully dry, remove the transparent slides, and immerse the model in absolute acetone for 48 h in a chemical hood until the plastic is fully dissolved. Remove any plastic leftovers with a wooden stick. To evaporate the acetone trapped inside the model, incubate it at 60 °C for at least 4 days before cell seeding.
2. Cell culture and seeding in models
- Prepare three connectors for the carotid model: one for the inlet and two for the outlets (see Table of Materials). Sterilize the model and the connectors by ultraviolet irradiation in a biological hood for 20 min.
- Coat the models with 4 mL of 100 µg/mL fibronectin (in 1x phosphate-buffered saline, (PBS)) for 2 h at 37 °C or overnight at 4 °C. Inject the fibronectin solution into the model through the inlet using a 5 or 10 mL plastic syringe. Remove the fibronectin through the outlet, and wash the model with EC medium.
- Suspend 2.5 × 106 cells/mL human umbilical vein endothelial cells (HUVECs, passage < 6), and fill the model with 4 mL of the cell suspension using a 5 or 10 mL plastic syringe (Figure 3A). Place the model on a rotator inside an incubator (37 °C) at a speed of 1 rpm for 48 h to ensure homogeneous seeding. Make sure the model is well-attached to the rotator (Figure 3B). Change the medium after 24 h inside a biological hood, and return to the rotator inside the incubator for another 24 h.
NOTE: After 24 h, the cells are seeded and can be imaged using a microscope. - Remove the model from the rotator, and wash with 1x PBS using a 10 mL plastic syringe. To fix the cells, incubate the cells with 4 mL of 4% paraformaldehyde (PFA) to the model for 15 min inside a chemical hood, and then rinse 3x with PBS. Add 4 mL of PBS, and store at 4 °C until the experiment (Figure 3C). Stain the cells inside the model using standard staining protocols (e.g., nuclear staining with 4′,6-diamidino-2-phenylindole (DAPI), Figure 3D).
3. Design of the perfusion system
- The perfusion system has two inlets and two outlet tubes. Merge the two inlets into a single 4 mm ID tube and again into two 6 mm ID tubes, which connect to the peristaltic pump. Merge the two 6 mm ID tubes coming out of the peristaltic pump into a single 4 mm tube, and connect it to an oscillation damper to eliminate any oscillations from the pump. Use a 250 mL narrow-mouthed bottle with a three-orifice lid as the damper.
- Connect one orifice to the inlet from the pump, close the second with a cork that is used for pressure venting in emergencies, and extend the third orifice (which is the outlet) to the bottom of the bottle.
- Connect the damper to the inlet of the cultured carotid model using the outlet tubing. Merge the two outlets of the model to one tubing, which will be the outlet of the system (All tubing are 4 mm ID).
- Split the outlet tubing to two outlet tubes (one for the closed circuit and the other for the waste container in the open circuit). Attach a plastic clamp to each tube.
NOTE: The combination of open/closed clamps will determine whether the system is in a closed or open circuit configuration. As shown in Figure 1, if clamps a and d are close while b and c are open, the system is a closed circuit; the reverse brings the system to open circuit configuration. - Prepare three containers: one that can hold 300 mL fluid (for closed-circuit) and two others of 1 L each: one for washing and the other for waste (for open circuit).
4. Closed-circuit configuration: perfusion experiment and imaging
- Add 300 mL of PBS to the closed-circuit container, which is sufficient to fill the entire system, including the tubing and the model. Place one inlet tube and one outlet tube (open clamps b and c) inside the container.
- Fill the 1 L washing container with distilled water (for washing at the end of the experiment), and leave the other 1 L waste container empty. Place the other inlet and outlet tubing (close clamps a and d) in the washing and waste containers, respectively.
- Take the fixed cell cultured carotid model out of 4 °C storage, and empty the PBS. Connect the inlet and outlets of the carotid as described in steps 3.3-3.4. Do not leave the model dry for a long time. Once the model is connected, activate the pump to perfuse fluid.
- Place the carotid model under the microscope. Open the tubing before the inlet and after the outlet of the carotid model. Set the peristaltic pump at 10 rpm, and turn it on. Increase the speed in increments of 5 rpm, every 4-5 min. Make sure there are no leakages.
- At 100 rpm, which equals the maximum flow rate of the physiological waveform of the human carotid artery (~400 mL/min), add fluorescent carboxylated polystyrene (PS) particles (2 µm, at a concentration of 1.6 µg/mL) to the 300 mL of PBS in the closed-circuit container. Image the region of interest every 10 s for 1.5 h (still single images or video as needed).
5. Open-circuit configuration: the washing step
- Open the clamps of the washing and waste tubes in the 1 L containers (clamps a and d), and immediately close the clamps of both the inlet and outlet tubes in the 300 mL container (clamps b and c) to change the system from a closed to open circuit configuration.
- Let most of the water flow from the washing container to the waste container at 100 rpm. Before it is completely transferred, press stop on the peristaltic pump, and close the tube clamps before the inlet and after the outlet of the carotid model.
- Using the appropriate filters, capture images of the model at the region of interest to show the deposition and adhesion of particles to the cells. Disconnect the carotid model. Carefully and slowly add 4 mL of PBS with a 10 mL syringe through the model's inlet.
- Connect a "dummy model", (which is also a silicone rubber 3D carotid model used only for washing, without cultured cells) instead of the carotid model, and rinse the system. Add another 1 L of water, and wash the system again until all the water is transferred from the washing container to the waste. Turn off the peristaltic pump.
6. Data acquisition and analysis
- Acquire a digital movie of particle deposition at the region of interest with the images taken during the experiment, using a customized software code (see the Table of Materials).
- For mapping of the deposition of the particles along the model, tile multiple images to cover the examined region of interest (Figure 4A,B).
NOTE: A customized software code can be written to quantify the number of adhered particles at a site of interest (a sample file has been provided as Supplemental Information)17.
Representative Results
This paper presents a new protocol to map the deposition of particles inside real-sized 3D human artery models, which may provide a new platform for drug delivery research. Using a 3D printing technique, a model of the human carotid bifurcation artery was fabricated (Figure 2). The model was made of silicone rubber and seeded with human ECs (Figure 3). Importantly, this protocol enabled the replication of physiological conditions, especially with respect to fluid dynamics. A perfusion system was designed to infuse particles to the carotid bifurcation under constant flow at the magnitude of the physiological waveform characteristic of the carotid. Figure 1 presents the perfusion system, which consists of the peristaltic pump, an oscillation damper, the cultured bifurcation model, tubing, and fluid containers.
To map the deposition and adhesion of the perfused particles, the arterial model was imaged under a stereomicroscope, both at the end of the experiment and after washing (step 5.3). The images were captured using 2x objective and tiled together to form a whole image of the model. Then, the number of adhered particles was calculated using a customized software code. To examine the formation of the recirculation pattern at the bifurcation, 10 µm fluorescent glass beads were infused into the model. Figure 4A shows the recirculation, which suggests that the conditions inside the model mimic physiological conditions.
To map the deposition of particles inside the model, 2 µm fluorescent carboxylated PS particles were infused, and their adhesion to the ECs was imaged (Figure 4B,C). These particles adhered differently at various regions along the model-more adhesion was observed out of the recirculation area, where wall shear stress is high. These results have been previously discussed to show that the adhesion of particles is a function of the model's geometry, particle surface characteristics, and shear stress17. These deposition maps are relatively simple and may be quickly obtained for screening drug carriers' affinity and targeting under physiological conditions in patient-specific models.

Figure 1: The perfusion system. A perfusion system was designed to perfuse fluids under constant flow. It is comprised of (1) a peristaltic pump, (2) an oscillation damper, (3) the cultured 3D arterial model, and three glass containers: two with a 1 L capacity (4 and 6) and a third that can hold 300 mL fluid (5). The system can operate in two configurations: (i) an open circuit, in which clamps a+d are open and b+c are closed, or (ii) a closed circuit, in which clamps a+d are closed and b+c are open. Please click here to view a larger version of this figure.

Figure 2: Fabrication process of a 3D carotid artery bifurcation model. (A–C) Human carotid bifurcation, the mold frame around the artery, and temporary printing supports were designed. (D, E) The geometries were printed using a 3D printer. (F) The temporary printing supports were cut, and the model was sanded and sprayed with lacquer. Then, transparent rectangular slides were glued to the frame from all sides. Silicone rubber was cast when the glue was dry. Abbreviation: 3D = three-dimensional. Please click here to view a larger version of this figure.
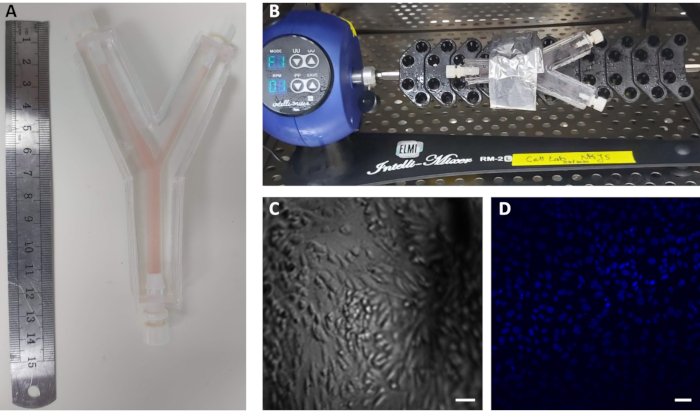
Figure 3: Seeding of ECs inside 3D models of the carotid artery. (A) Real-sized 3D model of the human carotid bifurcation made of silicone rubber. The model was cultured with human ECs and filled with cell medium. (B) The cultured model was placed on a rotator at 37 °C for 48 h. (C) Images of the cultured ECs inside the 3D model in brightfield and (D) with DAPI for nuclear staining in blue. Scale bars = 10 µm. Abbreviations: ECs = endothelial cells; DAPI = 4′,6-diamidino-2-phenylindole; 3D = three-dimensional. Please click here to view a larger version of this figure.
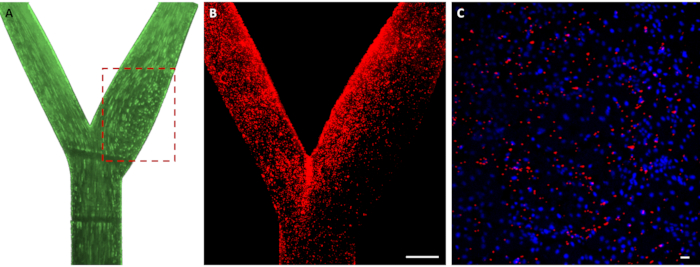
Figure 4: Perfusing and mapping the adhesion of particles. (A) Streak-line image of streamlines and recirculation (dashed rectangle) generated upon perfusion of 10 µm fluorescent glass particles at a constant flow of 400 mL/min through the model. (B) Deposition map of the 2 µm fluorescent carboxylated PS particles (in red) inside the 3D-cultured model. Scale bar = 2 mm. (C) Adhesion of the particles (in red) to the cultured ECs (in blue-DAPI) inside the model at a 10x magnification. Scale bar = 10 µm. Abbreviations: PS = polystyrene; 3D = three-dimensional; ECs = endothelial cells; DAPI = 4′,6-diamidino-2-phenylindole. Please click here to view a larger version of this figure.
Supplemental Information: Please click here to download this File.
Discussion
Current approaches to study vascular targeting of particles fall short in replicating the physiological conditions present in the human body. Presented here is a protocol to fabricate 3D-reconstructed models of human arteries to study particle targeting to the ECs lining the artery under physiological flow applied using a customized perfusion system. When choosing the material for 3D printing, it is best to use a clear plastic to avoid pigment transfer to the silicone model, which should be as transparent as possible. In addition, it is important to choose a material that does not dissolve in acetone, but instead becomes soft and brittle and can subsequently easily be removed from the model.
The presented 3D models are made of silicone rubber, a transparent silicone, mixed with its curing agent. It is important to ensure that the mixture is always at room temperature or below, otherwise the crosslinking between the silicone and the curing agent will begin before the degassing and casting onto the molds. Although polydimethylsiloxane can also be used to fabricate such models (1:10 ratio with its crosslinker), silicone rubber is more durable. After immersing the model in acetone to dissolve the plastic, it is crucial to incubate it at 60 °C for at least 4 days to ensure full evaporation of any acetone residue. If any acetone remains trapped inside the model, the cells will not grow properly. Changing the medium after 24 h and fixation of the cells after 48 h are the two steps that involve manual injection of fluid using a 10 mL syringe. It is therefore important to perfuse slowly, otherwise the cells might be washed out.
The perfusion system has two inlets and two outlet tubes. Each tube has a plastic tube clamp for flow control. Most of the tubing system is comprised of 4 mm inner dimeter (ID) tubes, except for the tubes that are clamped in the pump, which are 6 mm ID tubes. The ID of the tubes clamped in the pump will determine the maximum flow rate that can be achieved in the system. This perfusion system can also generate a pulsatile waveform by superposing oscillations on the constant mean flow17 by connecting the outlet of the damper to an oscillator assembly, which superposes the oscillatory part of a desired waveform on the constant flow rate produced by the peristaltic pump. This configuration enables operation under oscillatory flow or under constant flow conditions when the oscillator is turned off.
In this paper, the perfusion system was customized based on experiments with the 3D human carotid artery bifurcation. Therefore, if other arterial models or other tubing are used, the amounts of fluids and flow rate may require adjustments. In such cases, the system and flow rate will have to be calibrated, while ensuring no cell detachment from the model walls. It is very important to gradually increase the flow rate in the peristaltic pump to guarantee that the cells are not washed away with the flow. Moreover, it is crucial to ensure that the entire system, including the tubing, the model, as well as the container are filled with fluids (e.g., in this case, it was filled with a total volume of 300 mL of fluid).In addition, before and after each experiment, the system should be washed with distilled water in an open-circuit configuration.
Blood can also be perfused into the models using the perfusion system17. In this case, extra caution must be exercised to prevent any leakage, especially if human blood is used. Moreover, the washing step is crucial as bleach must be perfused at the end of the experiment to ensure full wash out of the blood. After the bleach, water should be perfused as mentioned in the protocol. It is important to note that in this paper, carboxylated PS particles were used, which have a uniform composition and a narrow size distribution. Moreover, these particles adhere to the cells primarily through electrostatic interactions. However, other drug nanocarriers may be used, and specific targeting should be examined as well with ligand-labeled particles, e.g., to anti-intercellular adhesion molecule 1 and anti-platelet endothelial cell adhesion molecule 1, which will increase particle accumulation to ECs at the site of interest.
In addition, in this protocol, ECs were fixed prior to connecting the models to the perfusion system and injection of the particles. The adhesion of particles to fixed cells represents the first stage in the binding process and therefore, experiments with live cells need to be performed, where internalization of particles may occur during later stages of the adhesion process. This protocol can be used to fabricate 3D arterial models for the study of drug carriers under physiological conditions. The outlined approach may assist in the study of delivery of agents under patient-specific conditions.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This work was supported by the Israel Science Foundation (ISF grant # 902/18). Maria Khoury's scholarship was supported by The Baroness Ariane de Rothschild Women Doctoral Program.
Materials
| 3D printer | FormLabs | PKG-F2-REFURB | |
| Acetone, absolute (AR grade) | |||
| Connectors | Nordson Medical | FTLL013-1 | Female Luer |
| FTLL230-1 | Female Luer | ||
| FTLL360-1 | Female Luer | ||
| LP4-1 | Male Luer Integral Lock | ||
| Damper | Thermo-Fisher Scientific | DS2127-0250 | Nalgene Polycarbonate, Validation Bottle |
| Damper Cover | Thermo-Fisher Scientific | 2162-0531 | Nalgene Filling/Venting Closures |
| Elastosil Elastosil RT 601 A | Wacker | 60003805 | |
| Elastosil RT 601 B | Wacker | 60003817 | The crosslinker |
| Endothelial Cell Media | ScienCell | 1001 | |
| Fibrontectin | Sigma Aldrich | F0895-5mg | |
| HUVEC | Lonza | CC-2519 | |
| Isopropyl alcohol, AR grade 99.5% | Remove plastic dust from the sanded model | ||
| Lacquer | Rust-Oleum | 2X-Ultra cover Gloss Clear | |
| Matlab | Mathworks | https://www.mathworks.com/products/matlab.html | |
| Microscope | Nikon | SMZ25 | |
| Microscope Camera | Nikon | DS-Qi2 | |
| Peristaltic pump | Watson Marlow | 530U IP31 | With 2 pumpheads: 313D |
| Plastic tube clamp | Quickun | 1-2240-stopvalve-2pcs | |
| Polystyrene Particles | Thermo-Fisher Scientific | F8827 | Diameter = 2 µm |
| Printer resin | FormLabs | RS-F2-GPCL-04 | |
| Rotator | ELMI Ltd. | Intelli-Mixer RM-2 | |
| Solidworks | SolidWorks Corp., Dassault Systèmes | https://www.solidworks.com/ | |
| Tubing | Watson Marlow | 933.0064.016 | Tubing for the pump: 6.4 mm ID |
| All the other tubing: Silicon tubing: 4 mm ID |
References
- Chiu, J. J., et al. Analysis of the effect of disturbed flow on monocytic adhesion to endothelial cells. Journal of Biomechanics. 36 (12), 1883-1895 (2003).
- Martorell, J., et al. Extent of flow recirculation governs expression of atherosclerotic and thrombotic biomarkers in arterial bifurcations. Cardiovascular Research. 103 (1), 37-46 (2014).
- Karino, T., Goldsmith, H. L. Flow behaviour of blood cells and rigid spheres in an annular vortex. Philosophical transactions of the Royal Society of London. Series B, Biological. 279 (967), 413-445 (1977).
- Goldsmith, H. L., Karino, T. Platelets in a region of disturbed flow. Transactions – American Society for Artificial Internal Organs. 23, 632-638 (1977).
- Farcas, M. A., Rouleau, L., Fraser, R., Leask, R. L. The development of 3-D, in vitro, endothelial culture models for the study of coronary artery disease. Biomedical Engineering Online. 8, 30 (2009).
- Peng, B., et al. Modeling nanoparticle targeting to a vascular surface in shear flow through diffusive particle dynamics. Nanoscale Research Letters. 10 (1), 942 (2015).
- Shah, S., Liu, Y., Hu, W., Gao, J. Modeling particle shape-dependent dynamics in nanomedicine. Journal of Nanoscience and Nanotechnology. 11 (2), 919-928 (2011).
- Hossain, S. S., Hughes, T. J., Decuzzi, P. Vascular deposition patterns for nanoparticles in an inflamed patient-specific arterial tree. Biomechanics and Modeling in Mechanobiology. 13 (3), 585-597 (2014).
- Charoenphol, P., Huang, R. B., Eniola-Adefeso, O. Potential role of size and hemodynamics in the efficacy of vascular-targeted spherical drug carriers. Biomaterials. 31 (6), 1392-1402 (2010).
- Ta, H. T., Truong, N. P., Whittaker, A. K., Davis, T. P., Peter, K. The effects of particle size, shape, density and flow characteristics on particle margination to vascular walls in cardiovascular diseases. Expert Opinion on Drug Delivery. 15 (1), 33-45 (2018).
- Cooley, M., et al. Influence of particle size and shape on their margination and wall-adhesion: implications in drug delivery vehicle design across nano-to-micro scale. Nanoscale. 10 (32), 15350-15364 (2018).
- Jiang, X. Y., et al. Quantum dot interactions and flow effects in angiogenic zebrafish (Danio rerio) vessels and human endothelial cells. Nanomedicine: Nanotechnology, Biology, and Medicine. 13 (3), 999-1010 (2017).
- Zarins, C. K., et al. Carotid bifurcation atherosclerosis. Quantitative correlation of plaque localization with flow velocity profiles and wall shear stress. Circulation Research. 53 (4), 502-514 (1983).
- Chien, S. Effects of disturbed flow on endothelial cells. Annals of Biomedical Engineering. 36 (4), 554-562 (2008).
- Malek, A. M., Alper, S. L., Izumo, S. Hemodynamic shear stress and its role in atherosclerosis. JAMA. 282 (21), 2035-2042 (1999).
- Glagov, S., Zarins, C., Giddens, D. P., Ku, D. N. Hemodynamics and atherosclerosis. Insights and perspectives gained from studies of human arteries. Archives of Pathology & Laboratory Medicine. 112 (10), 1018-1031 (1988).
- Khoury, M., Epshtein, M., Zidan, H., Zukerman, H., Korin, N. Mapping deposition of particles in reconstructed models of human arteries. Journal of Controlled Release. 318, 78-85 (2020).

