Staining the Cytoplasmic Ca2+ with Fluo-4/AM in Apple Pulp
Summary
Isolated protoplasts of apple pulp cells were loaded with a calcium fluorescent reagent to detect cytoplasmic Ca2+ concentration.
Abstract
Cytosolic Ca2+ plays a key role in plant development. Calcium imaging is the most versatile method to detect dynamic changes in Ca2+ in the cytoplasm. In this study, we obtained viable protoplasts of pulp cells by enzymatic hydrolysis. Isolated protoplasts were incubated with the small-molecule fluorescent reagent (Fluo-4/AM) for 30 min at 37 °C. The fluorescent probes successfully stained cytosolic Ca2+ but did not accumulate in vacuoles. La3+, a Ca2+ channel blocker, decreased cytoplasmic fluorescence intensity. These results suggest that Fluo-4/AM can be used to detect changes in cytosolic Ca2+ in the fruit flesh. In summary, we present a method to effectively isolate protoplasts from flesh cells of the fruit and detect Ca2+ by loading a small-molecule calcium fluorescent reagent in the cytoplasm of pulp cells.
Introduction
Ca2+ plays an important role in plant signal transduction and metabolism1,2. Further, it regulates fruit quality traits3,4, including hardness, sugar content, and susceptibility to physiological disorders during storage5,6. Cytoplasmic Ca2+ plays an important role in signal transduction and regulates plant growth and development7. Disturbance of cellular calcium homeostasis can induce bitter pit in apples8, brown spot disease in pears9, and umbilical rot in tomatoes10, affecting fruit quality and causing severe economic losses3,11. Calcium imaging has sufficient spatial and temporal resolution and is an important method for observing Ca2+ dynamics in living cells12,13.
At present, there are two main methods for intracellular calcium imaging in live cells: one employs chemical small-molecular fluorescent probes14, and the other is the gene encoding sensor (GECI)15,16. Given the difficulty of establishing a stable transgenic system in fruit trees and longer fruit development, GECIS is unsuitable for fruit Ca2+ fluorescence imaging.
Small-molecule fluorescent probes such as Fluo-4/AM have a particular advantage: their AM ester form (cell-permeable acetoxymethyl ester derivative) can be readily bulk-loaded into living cells without the need for transfection, which makes it flexible, rapid, and non-cytotoxic17. Fluo-4/AM could successfully be loaded into the pollen tube of Pyrus pyrifolia18 and Petunia,19 as well as into guard cells20 and root hair of Arabidopsis21.
At present, there are few reports on the calcium fluorescence staining of pulp cells22. As an important mineral element, calcium plays a key role in the growth and quality control of tree fruits such as apples. Apple trees are globally recognized as an important economic species, and apples are considered a healthy food23. In this study, we obtained viable protoplasts from apple fruit pulp through enzymatic hydrolysis and then loaded small-molecule fluorescent reagents into the cytoplasm to detect Ca2+.
Protocol
1. Protoplast extraction
- Prepare the basic solution: 20 mM CaCl2, 5 mM 2-(N-morpholino)ethanesulfonic acid, and 0.4 M D-sorbitol.
NOTE: The pH of the basic solution was adjusted to 5.8 with 0.1 M Tris buffer, filtered through 0.22 µm water-soluble filters, and stored at 4 °C. - Prepare the enzymatic solution: Mix 0.3%(w/v) Macerozyme R-10 and 0.5%(w/v) cellulase R-10 with the basic solution.
- Add 0.5 mL of enzymatic solution into a 1.5 mL centrifuge tube. Pick a healthy and ripe apple. Then slice the pulp into 10 x 5 x 1 mm3 size (Figure 1A-1C).
- Place the apple fruit pulp pieces into a 1.5 mL centrifuge tube containing enzymatic solution and then close the tube (Figure 1D).
- Incubate the tube at 28 °C for 1 h, shaking at 70 rpm/min in a shaker in the dark.
- Wash the pulp pieces. Aspirate all the enzymatic solution and then add 0.5 mL of the basic solution.
- Centrifuge at 300 x g for 2 min at room temperature.
- To obtain a protoplast suspension, aspirate the solution from the bottom of the centrifuge tube (Figure 1E).
2. Small-molecule calcium ion fluorescence staining
- Prepare the Fluo-4/AM loading solution with 2 mM Fluo-4/AM, 20% F-127, and 10x phosphate-buffered saline (PBS:80 mM Na2HPO4, 1.36 M NaCI, 20 mM KH2PO4, and 26 mM KCI) in a 1:1:2 ratio.
- Add 1 µL of Fluo-4/AM loading solution to 99 µL of the protoplast suspension present in the 1.5 mL centrifuge tubes. Ensure that the final concentration of the fluorescent dye is 5 µM. Mix the solution and then close the tube.
- La3+ treatment: Prepare 100 µM La3+ solution with 98 µL of the protoplast suspension, 1 µL of 10 mM La3+, and 1 µL of Fluo-4/AM loading solution. Mix the solution and close the tube.
- Incubate for 30 min at 37 °C in the dark.
- Wash the protoplasts by centrifugation at 300 x g for 2 min at room temperature. Aspirate 70 µL of the solution and add 70 µL of the basic solution.
- Incubate the protoplast suspension at 37 °C for 30 min to completely de-esterify.
- Aspirate 15 µL of the protoplast suspension and drip onto a slide.
- Observe under a fluorescence microscope (Supplementary Figure S1).
NOTE: Use a color camera with high sensitivity, i.e., 3.2 MP (2048 x 1536) CMOS sensor with 3.45 µm pixel resolution. - Select the GFP channel for imaging (20x). Set the brightness to 0.5.
NOTE:Illumination is adjustable-intensity LED light cubes with an integrated hard-coated filter set. The excitation wavelength of Fluo-4/AM is 490 nm.
3. Protoplast viability assay
- Prepare the Fluorescein diacetate (FDA) stock solution: Dissolve FDA in acetone until the final concentration is 1 mg/mL.
- Prepare the FDA working solution with 1 µL of the stock solution and 99 µL of acetone.
- Add 1 µL of FDA working solution to 99 µL of protoplast suspension. Mix the solution by pipetting up and down and then close the tube.
NOTE: The final concentration of the FDA is 100 µg/L. - Stain at room temperature for 5 min in the dark.
- Prepare the slides and observe under a fluorescence microscope.
- Select the GFP channel for imaging (Figure 1F).
4. Image analysis
- Analyze the acquired images using image analysis and spreadsheet software (e.g., Image-Pro Plus and Excel 2010).
- Select two vertical diameters from the protoplasts to calculate fluorescence intensity (Supplementary Figure S2). Measure the fluorescence intensity of all protoplasts under different treatments was measured. For final processing, use photo editing software.
5. Statistical analysis
- Perform statistical analysis using statistical software (Supplementary Figure S3). Data are presented in Mean ± SD. The student's t-test was used to analyze the differences between the experimental groups.
Representative Results
Following the protocol described above, we used the enzymatic method to obtain viable protoplasts from the pulp (Figure 1). Some protoplasts had vacuoles, while others did not. While the protoplasts exhibited no fluorescence when the Ca2+ fluorescent indicator was not loaded into them. When Fluo-4/AM was loaded into the protoplasts, the cytoplasm, but not the vacuole, became fluorescent (Figure 2). This result indicated that Fluo-4/AM successfully stained Ca2+ in the cytoplasm and that no compartmentalization was observed24. Protoplasts were stained with FDA for 5 min and showed cytoplasmic fluorescence. This indicated that high temperature (37 °C) does not affect protoplast viability.
La3+, a blocking agent of the Ca2+ channel25, was added when Fluo-4/AM was loaded into protoplasts. At 100 µM, La3+ decreased calcium fluorescence intensity (Figure 3).
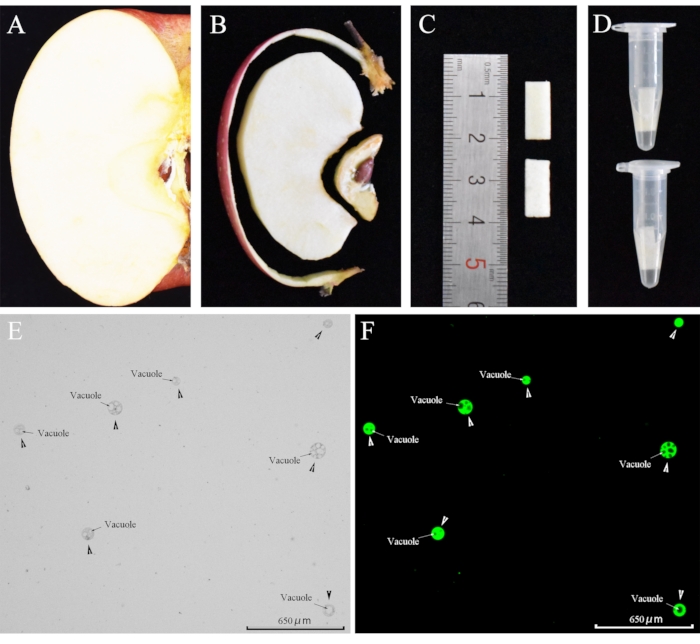
Figure 1: Protoplasts obtained by enzymatic hydrolysis. (A) A piece was cut from a mature apple. (B) Protoplasts were extracted from pulp pieces. (C) The pulp was cut into 10 × 5 × 1 mm3 pieces. (D) Pulp pieces were placed in centrifuge tubes containing 0.5 mL enzyme solution. (E) Protoplasts. Arrowheads point to the protoplast, and arrows indicate the vacuole in the protoplast. (F) Protoplasts were stained with FDA for 5 min at room temperature. (This figure has been modified from reference 22 [Horticulture research: Loading calcium fluorescent probes into protoplasts to detect calcium in the flesh tissue cells of Malus domestica]. Please click here to view a larger version of this figure.
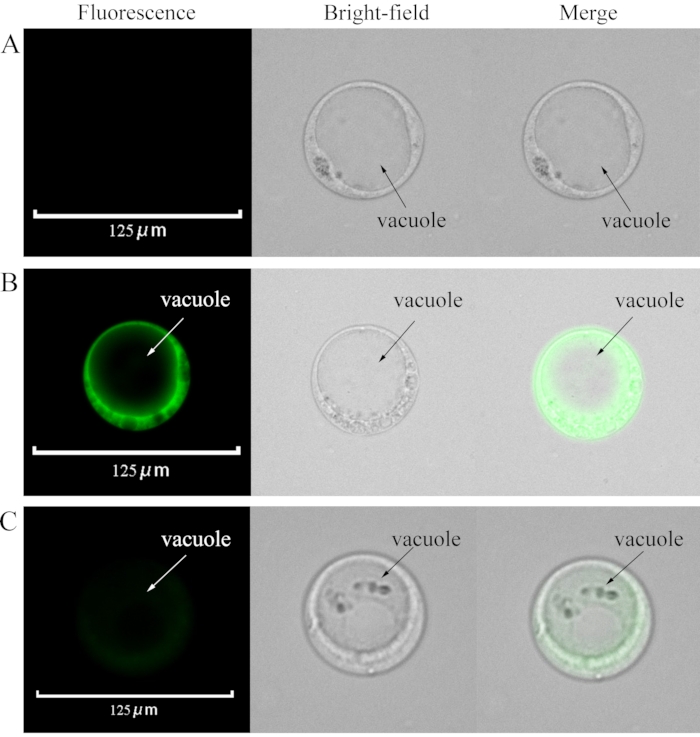
Figure 2: Loading Fluo-4/AM and La3+ into the protoplasts. (A) Control: Intact protoplast without any loaded fluorescent probe. (B) Protoplasts loaded with Fluo-4/AM. (C) La3+ was added when the protoplasts were loaded with Fluo-4/AM. The final concentration of La3+ was 100 µM. Please click here to view a larger version of this figure.
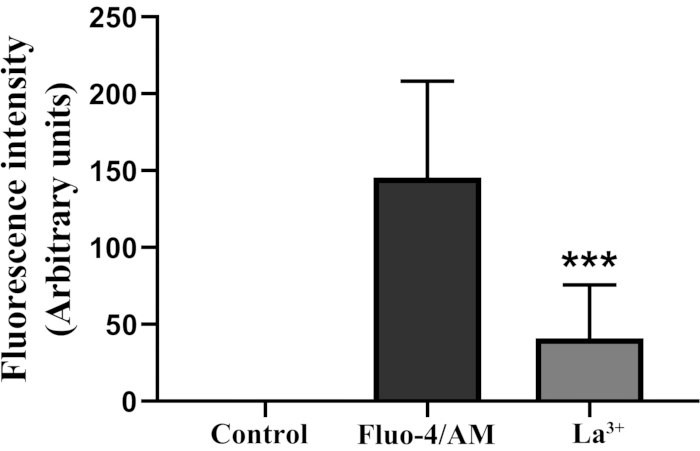
Figure 3: Statistical analysis of fluorescence intensity in the protoplasts. *** Indicate significant difference as per Student´s t-test (P <0.001). Vertical bars indicate ± SD. Each data point represents the mean of 20 protoplasts. Please click here to view a larger version of this figure.
Supplementary Figure S1: Fluorescence microscope. (A) The overall appearance of the fluorescence microscope. (B) Settings page. Select 20x objective, GFP channel, and uniformly adjust the brightness to 0.5. (C) Fluorescence excitation region. Please click here to download this File.
Supplementary Figure S2: Steps used to calculate the Ca2+ fluorescence intensity at protoplast of fruit cell. Fluorescence intensity units used in this study are based on previous reports26,27,28,29. The calculation process is as follows: (A) Open the protoplast fluorescence image in the software. Click on the Measure tool. From the dropdown menu select Profile Line. (B) Select Circle in the Line Profile window. Using this draw an ellipse at the protoplast. (C) In the Line Profile window now select File | Export data (D) Ensure that the blank spreadsheet is opened and import data by clicking Data Export. Use the 'average' function to calculate the average fluorescence intensity. Each treatment was repeated three times with more than 20 protoplasts each. Please click here to download this File.
Supplementary Figure S3: Data statistics were performed using statistical analysis software. Paste the data into the table and click Analyze for data analysis. Please click here to download this File.
Supplementary Figure S4: Extraction of protoplasts from the pulp of other varieties of apples. (A) Dounan. (B) Honey Crisp. Please click here to download this File.
Discussion
In this study, viable protoplasts were obtained by enzymatic hydrolysis. Note that this method requires fresh apples. The present protocol allows for the rapid isolation of a large number of protoplasts from fruit pulp for use in research studies. The applicability of this method is not limited to 'Fuji'; the protoplasts of the apple pulp of 'Dounan' and 'Honey Crisp' can also be extracted through the same protocol (Supplementary Figure S4). The protoplast solution after enzymolysis contains cell debris, which was somewhat improved compared to previous methods. As an essential cell material, apple pulp protoplasts can be used for cell protein expression technology, single-cell sequencing, and other research.
There are many methods regarding chemical fluorescent reagent staining in plant cells30. For example, Fluo-3/AM was loaded at a low temperature (4 °C), such that it would enter the root tip cells31. Fluo-3/AM can also be loaded at a high temperature (37 °C) into the pollen tube32. Fluo-4/AM has successfully loaded into the pollen tube of Pyrus pyrifolia (25 °C for 15 min)18 and the root hair of Arabidopsis (4 °C for 30 min)33. However, these methods are not suitable for staining apple pulp protoplasts. In this study, we successfully loaded fluorescent probes into protoplasts at a high temperature (37 °C). In addition, the present method is simple, fast, accurate, and does not affect protoplast vitality. A critical step in the proposed method is to wash the stained protoplasts and incubate them for 30 min. After staining, the protoplasts should be washed. The washing will reduce the background fluorescence during observation so that the fluorescence intensity of the protoplasts can be counted more accurately. Incubate for 30 min to allow more probes to be loaded into the protoplasts. Note that it is essential to avoid light exposure during the loading.
La3+ is an important tool for studying Ca2+. It binds to the Mg2+ catalytic site on the cytoplasmic membrane Ca2+-ATPase, thereby inhibiting the steady-state turnover of Ca2+-ATPase and blocking the Ca2+ transmembrane functioning34. La3+ treatment reduced cytoplasmic Ca2+, and Fluo-4/AM could reflect this change, excluding the interference of other divalent cations and verifying the feasibility of chemical fluorescence reagents.
Although this loading method offers more advantages than other methods, it still needs to be further improved in transforming data results. Here, we estimated the relative cytoplasmic Ca2+ content by detecting the fluorescence value of protoplasts, which is qualitative data rather than quantitative. Therefore, it is particularly important to translate the staining results into specific cytoplasmic Ca2+ content in future studies, which could provide a research basis for analyzing fruit Ca2+ signaling.
In summary, the method described herein can detect cytoplasmic Ca2+ in apple pulp cells, thereby providing technical support for the related studies of fruit pulp-cell calcium.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This work was supported by the Agricultural Variety Improvement Project of Shandong Province (2019LZGC007) and Fruit tree innovation team of Shandong modern agricultural industry technology system (SDAIT-06-05).
Materials
| 10× phosphate-buffered saline | Solarbio | P1022 | PBS (phosphate buffered solution) is a phosphate buffer solution, which can provide a relatively stable ionic environment and pH buffering capacity. It is a buffer salt solution often used in biology for molecular cloning and cell culture. The pH is 7.4. |
| 2-(N-morpholino)ethanesulfonic acid | Solarbio | M8010 | Biological buffer |
| CaCl2·2H2O | Solarbio | C8370 | Calcium chloride dihydrate is a white or gray chemical, mostly in granular form. |
| Cellulase R-10 | Yakult Honsha | MX7352 | Degrade plant cell walls. |
| D-sorbitol | Solarbio | S8090 | It has good moisturizing properties, prevents drying, and prevents sugar, salt, etc. from crystallizing. |
| F-127 | Thermo Fisher Scientific | P6867 | Pluronic F-127 is a non-ionic, surfactant polyol (molecular weight of approximately 12500 Daltons), which has been found to be beneficial to promote the dissolution of water-insoluble dyes and other materials in physiological media. |
| FDA | Thermo Fisher Scientific | F1303 | FDA is a cell-permeant esterase substrate that can serve as a viability probe that measures both enzymatic activity, which is require to activate its fluorescence, and cell-membrane integrity, which is required for intracellular retention of their fluorescent product. |
| Fluo-4/AM | Thermo Fisher Scientific | F14201 | The green fluorescent calcium indicator Fluo-4/AM is an improved version of the calcium indicator Fluo-3/AM. The Fluo-4/AM loads faster and is brighter at the same concentration. It can be well excited with a 488 nm argon ion laser. |
| Fluorescence microscope | Thermo Fisher | EVOS Auto 2 | Observe the fluorescence image. |
| Macerozyme R-10 | Yakult Honsha | MX7351 | Degrade plant tissue to separate single cells. |
| Tris | Solarbio | T8060 | It is widely used in the preparation of buffers in biochemistry and molecular biology experiments. |
References
- Hocking, B., Tyerman, S. D., Burton, R. A., Gilliham, M. Fruit calcium: Transport and physiology. Frontiers in Plant Science. 7, 569 (2016).
- Li, J., Yang, H. -. q., Yan, T. -. l., Shu, H. -. r. Effect of indole butyric acid on the transportation of stored calcium in Malus hupehensis rhed. Seedling. Agricultural Sciences in China. 5 (11), 834-838 (2006).
- Gao, Q., Xiong, T., Li, X., Chen, W., Zhu, X. Calcium and calcium sensors in fruit development and ripening. Scientia Horticulturae. 253, 412-421 (2019).
- Barrett, D. M., Beaulieu, J. C., Shewfelt, R. L. Color, flavor, texture, and nutritional quality of fresh-cut fruits and vegetables: desirable levels, instrumental and sensory measurement, and the effects of processing. Critical Reviews in Food Science and Nutrition. 50 (5), 369-389 (2010).
- Deell, J. R., Khanizadeh, S., Saad, F., Ferree, D. C. Factors affecting apple fruit firmness–a review. Journal- American Pomological Society. 55 (1), 8-27 (2001).
- Johnston, J., Hewett, E., Hertog, M. A. T. M. Postharvest softening of apple (Malus domestica) fruit: A review. New Zealand Journal of Experimental Agriculture. 30 (3), 145-160 (2002).
- Demidchik, V., Shabala, S., Isayenkov, S., Cuin, T. A., Pottosin, I. Calcium transport across plant membranes: mechanisms and functions. New Phytologist. 220 (1), 49-69 (2018).
- Miqueloto, A., et al. Mechanisms regulating fruit calcium content and susceptibility to bitter pit in cultivars of apple. Acta horticulturae. 1194 (1194), 469-474 (2018).
- Kou, X., et al. Effects of CaCl2 dipping and pullulan coating on the development of brown spot on ‘Huangguan’ pears during cold storage. Postharvest Biology and Technology. 99, 63-72 (2015).
- Vinh, T. D., et al. Comparative analysis on blossom-end rot incidence in two tomato cultivars in relation to calcium nutrition and fruit growth. The Horticulture Journal. 87 (1), 97-105 (2018).
- Yamane, T. Foliar calcium applications for controlling fruit disorders and storage life in deciduous fruit trees. Japan Agricultural Research Quarterly. 48 (1), 29-33 (2014).
- Grienberger, C., Konnerth, A. Imaging calcium in neurons. Neuron. 73 (5), 862-885 (2012).
- Bootman, M. D., Rietdorf, K., Collins, T. J., Walker, S., Sanderson, M. J. Ca2+-sensitive fluorescent dyes and intracellular Ca2+ imaging. CSH Protocols. 2013 (2), 83 (2013).
- Hirabayashi, K., et al. Development of practical red fluorescent probe for cytoplasmic calcium ions with greatly improved cell-membrane permeability. Cell Calcium. 60 (4), 256-265 (2016).
- Krebs, M., et al. FRET-based genetically encoded sensors allow high-resolution live cell imaging of Ca(2)(+) dynamics. Plant Journal. 69 (1), 181-192 (2012).
- Zhao, Y., et al. An expanded palette of genetically encoded Ca(2)(+) indicators. Science. 333 (2), 1888-1891 (2011).
- Gee, K. R., et al. Chemical and physiological characterization of fluo-4 Ca2+-indicator dyes. Cell Calcium. 27 (2), 97-106 (2000).
- Qu, H., Xing, W., Wu, F., Wang, Y. Rapid and inexpensive method of loading fluorescent dye into pollen tubes and root hairs. PLoS One. 11, 0152320 (2016).
- Suwińska, A., Wasąg, P., Zakrzewski, P., Lenartowska, M., Lenartowski, R. Calreticulin is required for calcium homeostasis and proper pollen tube tip growth in Petunia. Planta. 245 (5), 909-926 (2017).
- Sun, L., et al. NADK2 positively modulates abscisic acid-induced stomatal closure by affecting accumulation of H2O2, Ca2+ and nitric oxide in Arabidopsis guard cells. Plant Science. 262, 81-90 (2017).
- Niu, Y. F., et al. Magnesium availability regulates the development of root hairs in Arabidopsis thaliana (L.) Heynh. Plant Cell and Environment. 37 (12), 2795-2813 (2014).
- Qiu, L., Wang, Y., Qu, H. Loading calcium fluorescent probes into protoplasts to detect calcium in the flesh tissue cells of Malus domestica. Horticulture Research. 7, 91 (2020).
- Boyer, J., Liu, R. H. Apple phytochemicals and their health benefits. Nutrition Journal. 3 (1), 5 (2004).
- Takahashi, A., Camacho, P., Lechleiter, J. D., Herman, B. Measurement of intracellular calcium. Physiological Reviews. 79 (4), 1089-1125 (1999).
- Qu, H., Shang, Z., Zhang, S., Liu, L., Wu, J. Identification of hyperpolarization-activated calcium channels in apical pollen tubes of Pyrus pyrifolia. New Phytologist. 174 (3), 524-536 (2007).
- Hadjantonakis, A. K., Pisano, E., Papaioannou, V. E. Tbx6 regulates left/right patterning in mouse embryos through effects on nodal cilia and perinodal signaling. PLoS One. 3 (6), 2511 (2008).
- DeSimone, J. A., et al. Changes in taste receptor cell [Ca2+]i modulate chorda tympani responses to salty and sour taste stimuli. Journal of Neurophysiology. 108 (12), 3206-3220 (2012).
- Kao, J. P., Harootunian, A. T., Tsien, R. Y. Photochemically generated cytosolic calcium pulses and their detection by fluo-3. Journal of Biological Chemistry. 264 (14), 8179-8184 (1989).
- Merritt, J. E., Mccarthy, S. A., Davies, M., Moores, K. E. Use of fluo-3 to measure cytosolic Ca2+ in platelets and neutrophils. Loading cells with the dye, calibration of traces, measurements in the presence of plasma, and buffering of cytosolic Ca2. Biochemical Journal. 269 (2), 513-519 (1990).
- Li, W., et al. A comparative study on Ca content and distribution in two Gesneriaceae species reveals distinctive mechanisms to cope with high rhizospheric soluble calcium. Frontiers in Plant Science. 5 (5), 647 (2014).
- Zhang, W., Rengel, Z., Kuo, J. Determination of intracellular Ca2+ in cells of intact wheat roots: loading of acetoxymethyl ester of Fluo-3 under low temperature. Plant Journal. 15 (1), 147-151 (1998).
- Qu, H., Jiang, X., Shi, Z., Liu, L., Zhang, S. Fast loading ester fluorescent Ca2+ and pH indicators into pollen of Pyrus pyrifolia. Journal of Plant Research. 125 (1), 185-195 (2012).
- Wang, Y., et al. Disruption of actin filaments induces mitochondrial Ca2+ release to the cytoplasm and [Ca2+]c changes in Arabidopsis. root hairs. BMC Plant Biology. 10, 53 (2010).
- Fujimori, T., Jencks, W. P. Lanthanum inhibits steady-state turnover of the sarcoplasmic reticulum calcium ATPase by replacing magnesium as the catalytic ion. Journal of Biological Chemistry. 265 (27), 16262-16270 (1990).

