Construction of Model Lipid Membranes Incorporating G-protein Coupled Receptors (GPCRs)
Summary
This protocol utilizes agarose swelling as a powerful and generalizable technique for incorporating integral membrane proteins (IMPs) into giant unilamellar lipid vesicles (GUVs), as described here for the reconstitution of the human 1A serotonin receptor protein (5-HT1AR), one of the classes of pharmacologically important G protein-coupled receptors.
Abstract
Robust in vitro investigations of the structure and function of integral membrane proteins has been a challenge due to the complexities of the plasma membrane and the numerous factors that influence protein behavior in live cells. Giant unilamellar vesicles (GUVs) are a biomimetic and highly tunable in vitro model system for investigating protein-membrane interactions and probing protein behavior in a precise, stimulus-dependent manner. In this protocol, we present an inexpensive and effective method for fabricating GUVs with the human serotonin 1A receptor (5-HT1AR) stably integrated in the membrane. We fabricate GUVs using a modified hydrogel swelling method; by depositing a lipid film on top of a mixture of agarose and 5-HT1AR and then hydrating the entire system, vesicles can be formed with properly oriented and functional 5-HT1AR incorporated into the membrane. These GUVs can then be used to examine protein-membrane interactions and localization behavior via microscopy. Ultimately, this protocol can advance our understanding of the functionality of integral membrane proteins, providing profound physiological insight.
Introduction
Synthetic model membranes are powerful tools in the investigation of the fundamental properties and functions of biomembranes. Giant unilamellar vesicles (GUVs) are one of the most prominent platforms to study a variety of plasma membrane properties and can be engineered to mimic different physiological conditions1,2,3,4,5,6,7,8. It is well established that the plasma membrane and its organization play a key role in a multitude of cellular processes, such as signal transduction, adhesion, endocytosis, and transport9,10,11,12,13,14,15.
GUVs have been fabricated using various methods, including gentle hydration16, hydrogel swelling17, electroformation18, microfluidic techniques19,20,21,22, jetting23, and solvent exchange24,25,26. Due to challenges in handling integral membrane proteins (IMPs), in vitro platforms to study them have been limited. GUVs present a simplified platform for studying IMPs in an environment that mimics their native environment. Although there have been several approaches for protein reconstitution in GUVs, challenges arise from incorporating proteins with the correct orientation and maintaining protein functionality27.
Most successful protein-reconstitution in GUVs requires the detergent exchange method; which involves solubilizing the proteins from their native environment by detergents, followed by protein purification, and then replacing the detergent molecules with lipids through various methods28. While detergents serve to stabilize the tertiary structure of IMPs during purification, detergent micelles are a relatively unnatural environment for these proteins, which are better stabilized, particularly for functional studies, in lipid bilayers28,29,30. Moreover, incorporating functional transmembrane proteins into the lipid bilayer using traditional GUV fabrication techniques has been difficult due to the size, the delicacy of these proteins, and the additional detergent exchange steps that would be needed27,31,32,33. The use of organic solvent to remove detergents causes protein aggregation and denaturing34. An improved detergent-mediated method has been promising, however, caution is needed for the detergent removal step and optimization might be needed for specific proteins31,35. Additionally, methods that utilize electroformation could restrict the choice of protein and may not be suitable for all lipid compositions especially charged lipids31,36,37. Another technique that has been used is peptide-induced fusion of large unilamellar vesicles (LUVs) containing the desired protein with GUVs, though it was found to be laborious and can lead to the insertion of foreign molecules-the fusogenic peptides33,38,39. Giant plasma membrane vesicles (GPMVs), which are derived from living cells, can be used to overcome some of these issues, however they allow minimal control of the resultant lipid and protein composition14,40,41. Therefore, the integration of IMPs in the bilipid layer of GUVs using our modified agarose swelling method presents a reliable method to further examine these proteins in the membrane environment42,43,44,45.
Cellular signaling and communication involves a family of proteins known as G protein-coupled receptors (GPCRs); GPCRs are among the largest family of proteins and are associated with modulating mood, appetite, blood pressure, cardiovascular function, respiration, and sleep among many other physiological functions46. In this study, we used human serotonin 1A receptor (5-HT1AR) which is a prototypical member of the GPCR family. 5-HT1AR can be found in the central nervous system (CNS) and blood vessels; it influences numerous functions such as cardiovascular, gastrointestinal, endocrine functions, as well as participating in the regulation of mood47. A large barrier to GPCR research arises from their complex amphiphilic structure, and GUVs present a promising platform for the investigation of various properties of interest, ranging from protein functionality, lipid-protein interactions, and protein-protein interactions. Various approaches have been utilized to study lipid-protein interactions such as surface plasmon resonance (SPR)48,49, nuclear magnetic resonance spectroscopy (NMR)50,51, protein lipid overlay (PLO) assay51,52,53,54, native mass spectrometry55, isothermal titration calorimetry (ITC)56,57, and liposome sedimentation assay58,59. Our lab has used the simplified GUV approach to investigate the effect of lipid-protein interactions on protein functionality by incapsulating BODIPY-GTPγS, which binds with the Giα subunit in the active state of the receptor. Their binding unquenches the fluorophore producing a fluorescence signal that could be detected over time45. Moreover, various studies investigated Lipid-protein interactions and the role of proteins in sensing or stabilizing membrane curvature60,61, and utilizing a feasible GUV approach could be a key advantage.
This protocol demonstrates a straightforward method to incorporate GPCRs into the membrane of GUVs using a modified agarose hydrogel system17,42. Furthermore, based on our previous work, our method could be suitable for IMPs that can bear short-term exposure to 30-40 °C. Briefly, we spread a thin film of agarose combined with membrane fragments containing the GPCR of interest. Following gelation of this layer, we deposit a lipid solution atop the agarose and allow the solvent to evaporate. Rehydration of the system was then performed with an aqueous buffer, resulting in the formation of GUVs with protein incorporated in the lipid bilayer.
Protocol
1. Protein labeling
- Allow NHS-Rhodamine, 5-HT1A membrane fragments, and one 7 K MWCO spin desalting column to equilibrate at room temperature.
- Dissolve 1 mg of NHS-rhodamine in 100 µL of dimethyl sulfoxide (DMSO).
- Add 5 µL of 1 M sodium bicarbonate solution to increase the pH of 5-HT1AR solution to pH 8.
- Add 3.66 µL of the NHS-rhodamine solution to 50 µL of the 5-HT1AR solution and pipette gently up and down in a microcentrifuge tube.
NOTE: Ensure to have at least 10x molar excess of NHS-rhodamine. - Keep the mixture protected from light and put on rotator at room temperature for 1 h.
- Wash a 7 k MWCO spin column with 200 µL of 1x phosphate buffer saline (1x PBS) three times for 1.5 min at 1.5 RCF for each wash.
- Add the labeled protein to one column and balance the amount in another microcentrifuge tube.
- Spin down the labeled protein once for 5 min at 1.5 RCF.
- Take a UV-vis spectrum using a nanodrop spectrophotometer at 280 nm and 554 nm and calculate the labeling efficiency following the manufacturer's manual.
- Store the labeled protein covered at 5 °C until further use. The solution is stable for approximately a week after labeling.
2. GUVs with membrane-incorporated 5-HT 1A
- Preparation of materials and reagents
- Allow the protein, lipids and BSA (Bovine serum albumin) to equilibrate to room temperature.
- During this time, clean the coverslips by placing them in methanol and sonicating for 30 min at 40 °C. Ensure that the methanol completely covers the coverslips and the water level in the water bath is above the level of the methanol in the container.
NOTE: Methanol is toxic and should be handled in appropriate chemical hood. - Dry off the excess methanol on the coverslips with a gentle stream of air. Place the coverslip rack covered in a 40 °C oven for 15 min to ensure that the excess coverslips dry off.
- Begin the plasma cleaning process. First, place the coverslips into the plasma cleaner and close off the air intake valve to evacuate all the air inside the chamber.
- Once the chamber is under vacuum, clean the coverslips for 5 min using high RF power setting and a near complete vacuum, with only a slight air intake into the vacuum chamber. To ensure the proper level of plasma, adjust the opening of the vacuum chamber such that the resultant color of the plasma is a steady, bright pink.
NOTE: It is crucial when using air that the plasma remains a bright pink color for the duration of the plasma treatment step, as a darker purple color indicates that there is an improper amount of air in the chamber and will result in a suboptimal plasma treatment. - Once the 5 min have passed, shut off the RF power and release the vacuum.
NOTE: Upon removal from the plasma chamber, please ensure that the coverslips remain covered.
- Hydrogel preparation
- Combine 6 mg of ultra-low melting temperature agarose with 300 µL of ultrapure water (i.e., 2% (w/v) agarose).
NOTE: 2% agarose will be used to make protein-free GUVs. Agarose solution can be kept at 45 °C for two days. - Combine 9 mg of ultra-low temperature agarose with 300 µL of ultrapure water for 3 w/v% agarose by as prepared in step 3.1. 3% agarose will be used to make protein incorporated GUVs.
- Vortex the solution briefly before placing them on the 90 °C heat block for 10 min. Then, vortex the tube again before transferring it to a 45 °C heat block to keep it in the molten form until further use.
- Combine 6 mg of ultra-low melting temperature agarose with 300 µL of ultrapure water (i.e., 2% (w/v) agarose).
- Agarose and protein mixing
- Mix 21 µL of 3% agarose with the 7 µL of 5-HT1AR membrane fragments. Pipette up and down slowly many times to ensure adequate mixing. Then, incubate at 45 °C for 1 min.
- Hydrogel and lipid deposition
- For protein-free GUVs: Make a thin film on freshly plasma-cleaned coverslips using 20 µL of 2% agarose. Quickly drop another coverslip on top of the agarose droplet and gently slide the coverslips apart to make a thin film on both coverslips.
NOTE: This step is tricky in that the sliding of the droplet must occur while the agarose is still in the molten form. - For protein-incorporated GUVs: Pipette the protein/agarose mixture up and down one more time, and then deposit 20 µL of the 2% agarose on a plasma-cleaned coverslip. Follow the slip-casting directions as described above.
- Allow the agarose to gel protected from light for 30 min at room temperature.
- Deposit the lipids dropwise on top of the agarose layer. Use a total of 10 µL of 2 mg/mL of 1-palmitoyl-2-oleoyl-glycero-3-phosphocholine (POPC) with 0.4 Mol% 1,2-Dipalmitoyl-sn-glycero-3-phosphoethanolamine (DPPE) labeled with ATTO 488 (ATTO-488-DPPE) (or lipid mixture of interest) in chloroform on top of the agarose film. Deposit the droplets using a gas chromatography needle and spread one droplet at a time around via a gentle air stream.
NOTE: Caution is needed with this step to make a relatively uniform layer of lipids on top of the hydrogel. Also, chloroform is toxic and should be handled in appropriate chemical hood. - Assemble the Sykes-Moore (S-M) chambers by placing an O-ring on top of the coverslip, and then placing the top component of the chamber on top of the O-ring. Use the key provided by the manufacturer to assemble the chamber by screwing the chamber components together to seal the chamber and prevent any leakage.
NOTE: The top of the chamber should be tightened on the O-ring but caution is needed to ensure the coverslip stays intact as the coverslip can crack if the O-ring doesn't sit properly in the chamber. Also, ensure that the chamber is sealed tight enough such that the chamber does not leak when the swelling solution is added. Failure to tighten the chamber enough will result in leaks and loss of sample.
- For protein-free GUVs: Make a thin film on freshly plasma-cleaned coverslips using 20 µL of 2% agarose. Quickly drop another coverslip on top of the agarose droplet and gently slide the coverslips apart to make a thin film on both coverslips.
- Swelling and harvesting of vesicles
- Hydrate the entire system by gently pipetting 450 µL of 200 mM sucrose in 1x PBS and gently tapping the chambers to ensure adequate buffer coverage of the hydrogel-lipid layers.
NOTE: The sucrose solution can be replaced with a rehydration buffer containing biological probes of interest. - Place the chambers at 45 °C and cover the top part of the chamber with a coverslip to prevent evaporation. Allow the sample to swell, protected from debris and light for 1 h.
- Add 100 µL of 1 mg/mL BSA in ultrapure water into each well of a 96-well plate intended to be used. Incubate at room temperature for 1 h.
- Wash three times with ultra-pure water and once with 200 mM sucrose in 1x PBS.
- Finally, add 200 mM of glucose in 1x PBS until the addition of the GUV sample solution.
NOTE: BSA was used to block GUV adsorption. - After allowing the hydrogel to swell, gently shake and tap the chamber to dislodge any GUVs that may remain attached to the hydrogel surface. Then, carefully pipette up the GUV-sucrose solution.
NOTE: As an optional step to ensure all vesicles are detached from the surface, gently pipette some of the sucrose suspension back onto the hydrogel surface. - Move the suspension into a previously prepared microcentrifuge tube containing 700 µL of 200 mM glucose in 1x PBS.
NOTE: The density gradient will lead to settling of the vesicles to the bottom of the centrifuge tube. - Allow the vesicles to settle for another hour to ensure that the vesicles can sink to the bottom of the microcentrifuge tube, allowing for optimal collection.
- After the settling of GUVs in glucose, transfer 300 µL from the bottom of the centrifuge tube (the settled vesicles) into the previously prepared and BSA-treated 96-well plate to examine the vesicles under the confocal microscope.
NOTE: Be sure to avoid the very bottom of the microcentrifuge tube to minimize the amount of debris collected in the final sample.
- Hydrate the entire system by gently pipetting 450 µL of 200 mM sucrose in 1x PBS and gently tapping the chambers to ensure adequate buffer coverage of the hydrogel-lipid layers.
- Check the samples under the microscope.
- Shine a 488 nm laser on the sample (that allows us to visualize the membrane, as the bilayer has been labeled with ATTO-488-DPPE).
- Shine a 561 nm laser on the sample (that allows us to visualize the protein, since it has been labeled with NHS-Rhodamine).
NOTE: Caution is needed while imaging the sample as photooxidation can destabilize the vesicles. Vesicles were observed on the same day.
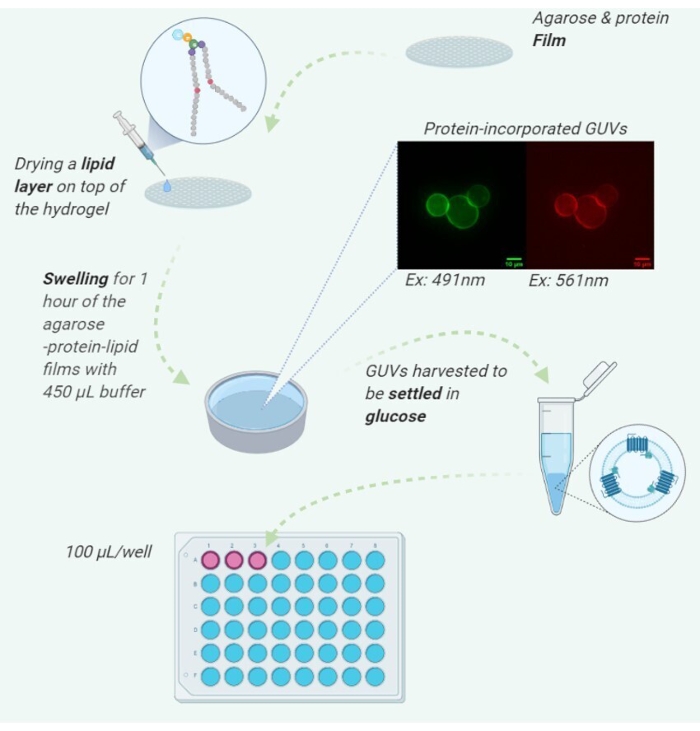
Figure 1: Illustration of the detailed protocol steps. Created with BioRender.com Please click here to view a larger version of this figure.
Representative Results
The concentration of protein was measured, and the degree of labeling was calculated as the molar ratio between the dye and the protein to be 1:1. By examining the GUVs using confocal microscopy, we were able to confirm successful formation and protein integration of the vesicles. The lipids were labeled with 0.4 mol% ATTO 488-DPPE, and the protein was covalently labeled via rhodamine NHS-ester modification of primary amines. Figure 2a and Figure 2b show a protein-incorporated vesicle in the ATTO 488 and rhodamine channels, respectively. All micrographs have been dark current and flatfield corrected. Figure 2c and Figure 2d show a negative control GUV with no protein incorporated. Figure 3a and Figure 3b show a protein incorporated GUV with line intensity profiles given by the dashed white line of the same vesicle in both channels. The line intensity profile shows a two-dimensional plot of the intensities of the pixels along the white drawn line within the image. The x-axis is the distance along the line and the y-axis is the pixel intensity. ImageJ software was used to plot the profile intensity of the indicated line.
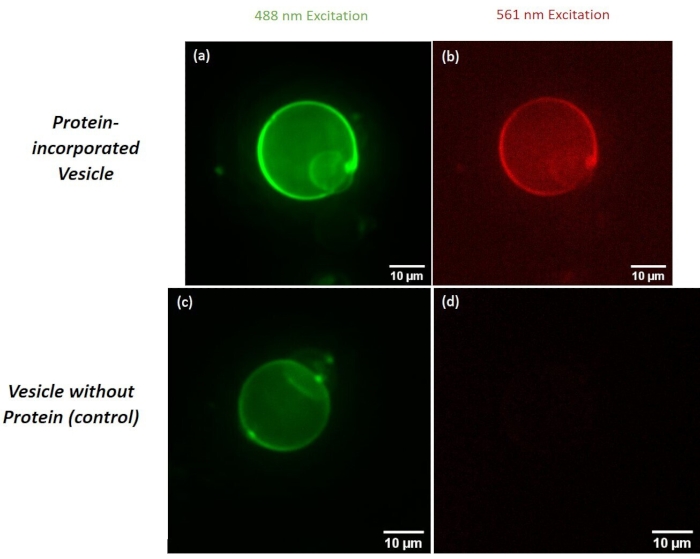
Figure 2: Micrographs comparing protein incorporated GUVs and GUVs without protein (control). Micrographs (a) and (b) show protein incorporated GUV fluorescence with the respective ATTO 488 and rhodamine channels, respectively. Micrographs (c) and (d) show a protein omitted GUV when excited with ATTO 488 and rhodamine channels, respectively. Please click here to view a larger version of this figure.
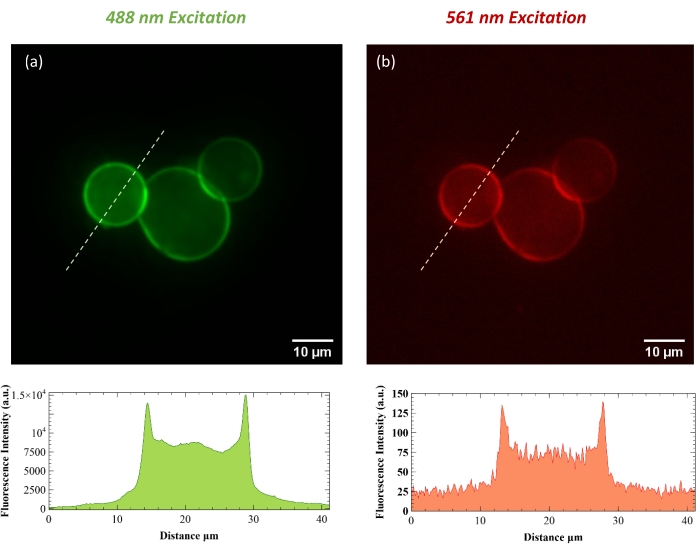
Figure 3: Top row shows micrographs of protein incorporated GUVs in ATTO 488 (a) and rhodamine (b) channels. Line intensity profiles for the indicated white-dashed lines are below. The analysis was performed using ImageJ software. Please click here to view a larger version of this figure.
Discussion
We have identified two steps that are critical to the success of the overall protocol: plasma treatment and lipid deposition. Plasma cleaning of the coverslips is essential in ensuring that there is adequate coverage and adhesion of the agarose hydrogel to the glass coverslip. Plasma cleaning accomplishes two things: first, it removes traces of organic matter from the glass surface; second, it activates the coverslip surface, allowing for an increase in wettability as the glass surface hydrophilicity increases62,63. Touching the coverslip surface post-plasma cleaning will inactivate and contaminate the ultraclean surface and is strongly advised against. Our recommendation is to only touch the very edges and undersides of the coverslip when handling the coverslips for the agarose slip casting step. The second critical step is the deposition of lipids onto the dry hydrogel surface. This method uses a dropwise lipid deposition, which requires a gas chromatography (GC) needle and an air stream to deposit a few microliters of lipid solution at a time, allowing for precise control of the amount of lipid added and the placement of the lipid film on the hydrogel surface. The drawback of this method is that if not done carefully, it can result in a few select areas with a thicker lipid film, resulting in reduced GUV yields. Thus, it is critical to ensure that there is as uniformly thin of a lipid layer as possible on the surface of the agarose.
One of the most significant benefits of this protocol is the flexibility of the platform itself; this method lends itself very well to changes in protein and lipid composition, as well as encapsulation and buffer modifications. This protocol can, in principle, include any transmembrane protein, as we have been able to successfully incorporate a number of different transmembrane proteins, ranging from the adenosine receptor (A2AR) to plant aquaporins without sacrificing functionality42,45,64. Traditionally, proteins have been incorporated into GUVs following solubilization by detergents or incorporation into proteo-liposomes or small unilamellar vesicles that can be subsequently integrated into a preformed GUV65. The advantage of our modified hydrogel swelling method is that it removes the dependency of detergents or intermediate vesicles and provides an intermediate hydrated scaffold. The benefits of this are twofold: we can stably incorporate functional GPCRs into the membrane in a more physiologically relevant buffer without relying on detergent exchange methods that require more preparation and care regarding the concentration of the said detergents, and that the process by which GUVs bud off the surface of the hydrogel allows for the correct orientation of the proteins in the bilayer66. We have shown that the GUV budding process involves the coalescence of many smaller nanometer-scale vesicles into larger, micron-scale vesicles, which encourages correct protein orientation from the beginning. We have shown this to be the case in our previous work; in short, we covalently labeled an antibody targeting a specific cytosolic loop of the Adenosine receptor and incubated the labeled antibody with the protein, and then incorporated the labeled protein into lipid-dye-labeled GUVs using the modified hydrogel swelling method. We then exposed the protein-incorporated vesicles to a charged quencher, which is unable to cross the bilayer. We subsequently see a 50% reduction in fluorescence of the lipid dye, but the fluorescence of the labeled protein remains unaffected by the quencher, demonstrating proper orientation44.
Previous work out of our lab has investigated the role in which lipid headgroup charge, zwitterionic and net-ionic charged lipids, as well as buffer and hydrogel properties such as pH, ionic strength, osmolarity, and hydrogel concentration have on the dynamics of GUV formation67. In short, lipid charge does not largely affect GUV formation, while buffer properties such as increases in sucrose concentration (e.g., 500 mM Sucrose in 185 mM ionic strength PBS buffer) negatively affect GUV formation, resulting in irregularly shaped vesicles that most likely will not readily lend themselves to harvesting. Acidic solutions (pH = 3) increase the rate of formation, while a more basic solution (pH = 8) suppresses the rate of GUV formation. GUVs still form at both the acidic and basic buffers, with only marginal differences in vesicle size. Low agarose concentrations (~0.1-1 w/v%) also negatively affect GUV formation due to a lack of homogenous surface coverage and a decrease in hydrogel swelling, a necessary force in the coalescence and budding of GUVs off the hydrogel surface. Thus, we have determined that a 2 w/v% final agarose concentration with a sucrose/glucose solution of 100-200 mM, combined with a buffer ionic strength of 185 mM PBS at pH 7.4 achieves a good balance of agarose swelling, GUV formation rate, and subsequent vesicle size. For vesicles that contain protein, increasing the initial agarose concentration to 3 w/v% allows for a final agarose concentration of 2 w/v% after the addition of the protein solution. In addition to formation dynamics, the sucrose/glucose buffer system also facilitates the sedimentation and subsequent collection of formed GUVs, as well as visualization under phase contrast microscopy65,68.
There are some points of caution regarding this protocol, specifically regarding the agarose and the selection of vesicles. For instance, while we use an ultralow melting temperature agarose, the agarose-water suspension needs to reach at least 60 °C to become molten, and the agarose-protein mixture is incubated at 45 °C. In our experience, this temperature does not eliminate the activity of 5-HT1AR, but caution is warranted for other proteins. In general, the agarose we use begins to gel at 20 °C and thus the swelling reaction can take place at temperatures above 20 °C, but this process cannot function below that temperature. It should also be noted that the closer the temperature gets to 20 °C, the less efficient the swelling step becomes, leading to subsequent decreases in GUV yields. The agarose can also present an issue during the settling and visualization steps, as it can persist at the bottom of the settling/collection tube as debris. Thus, caution is required for the temperature required to maintain the molten agarose and ensuring that the said temperature will not denature the protein of interest as well as aspirating the settling solution to avoid any excess suspended agarose from being included in the final sample. This method in its current state also results in a heterogeneous GUV population size, with some vesicles displaying multilamellarity and other flawed vesicle phenomena such as vesicles within vesicles. This is typical of common GUV formation methods and requires vigilance and discretion when selecting vesicles for microscopy and analysis. GUVs that display unusually high levels of fluorescence are also not recommended for analysis, as agarose can be found on the interior of some of these vesicles. Unpublished work out of our lab has been able to run micropipette aspiration experiments using vesicles made using this technique, illustrating that the agarose method produces vesicles without mechanics-altering agarose in the lumen.
Limitations aside, this protocol presents a robust and straightforward method for generating protein incorporated GUVs. It can generate high yields of GUVs in physiologically relevant conditions that incorporate properly oriented transmembrane proteins into the bilayer without compromising their functionality. This is a departure from other methods of vesicle formation, which involve electric currents or gentle hydration, that would significantly damage the structure of the protein and render it nonfunctional or require further detergent solubilization and removal steps. Given that GPCRs represent upwards of a third of all pharmaceutical targets, there is significant interest in being able to study this family of proteins in a highly tunable, high-throughput, biomimetic platform. More specifically, the applications of this work range from the study of protein-lipid interactions, how the lipid microenvironment influences protein functionality and localization, and other basic biophysical questions that can inform pharmaceutical drug development and discovery. An example of this can be found in the work completed within our lab, which has been able to discern variances in receptor functionality as a result of lipid oxidation.
Divulgations
The authors have nothing to disclose.
Acknowledgements
We thank Matthew Blosser for valuable discussion and advice. This work was supported by the Office of Naval Research (N00014-16-1-2382) and the National Science Foundation (PHY-1915017).
Materials
| 1,2-dioleoyl-sn-glycero-3-phosphocholine (DOPC) | Avanti Polar Lipids, Alabaster, AL | 850375C-25mg | |
| TI-Eclipse inverted microscope | Nikon, Melville, NY | Eclipse Ti | |
| 1,2-dipalmitoyl-sn-glycero-3-phosphatidylcholine (DPPC) | Avanti Polar Lipids, Alabaster, AL | 850355C-25mg | |
| 13/16″ ID, 1″ OD silicon O-rings | Sterling Seal & Supply, Neptune, IN | 5-003-8770 | |
| 16-bit Cascade II 512 electron-multiplied charge coupled device camera | Photometrics, Huntington Beach, CA | Cascade II 512 | |
| 1-palmitoyl-2-oleoyl-glycero-3-phosphocholine (POPC) | Avanti Polar Lipids, Alabaster, AL | 850457C-25mg | |
| 50 mW solid-state lasers at 488 nm and emission filter centered at 525 nm, and 561 nm with emission filter centered at 595 nm | Coherent, Santa Clara, CA | 488/561-50-LS | |
| 5-HT1AR membrane fragments | Perkin Elmer, Waltham, MA | RBHS1AM400UA | |
| ATTO-488-1,2-Dipalmitoyl-sn-glycero-3-phosphoethanolamine (DPPE) | ATTO-TEC, Siegen, Germany | AD 488-155 | |
| Bench top plasma cleaner | Harrick Plasma, Ithaca, NY | PDC-32G | |
| bovine serum albumin (BSA) | Sigma Aldrich, St. Louis, MO | A9418 | |
| chloroform (CHCl3) | Millipore Sigma, Burlington, MA | CX1055 | |
| Cholesterol (Chol) | Sigma Aldrich, St. Louis, MO | C8667-5G | |
| Corning 96-well Flat Clear Bottom | Corning, Corning, NY | 3904 | |
| Elmasonic E-Series E15H Ultrasonic | Elma, Singen, Germany | [no longer sold on main website] | |
| glucose | Sigma Aldrich, St. Louis, MO | G7528 | |
| methanol (MeOH) | Millipore Sigma, Burlington, MA | MX0485 | |
| NanoDrop ND-1000 | Thermo Fisher Scientific, Waltham, MA | ND-1000 | |
| NHS-Rhodamine | Thermo Fisher Scientific, Waltham, MA | 46406 | |
| phosphate buffered saline (PBS) (10x PBS) | Corning, Corning, NY | 21-040 | |
| spinning-disc CSUX confocal head | Yokogawa,Tokyo, Japan | CSU-X1 | |
| standard 25 mm no. 1 glass coverslips | ChemGlass, Vineland, NJ | CLS-1760 | |
| sucrose | Sigma Aldrich, St. Louis, MO | S7903 | |
| Sykes-Moore chambers | Bellco, Vineland, NJ | 1943-11111 | |
| Ultra-low melting temperature agarose | Sigma Aldrich, St. Louis, MO | A5030 | |
| VWR Analog Heatblock | VWR International, Radnor, PA | [no longer sold on main website] | |
| VWR Tube Rotator | VWR International, Radnor, PA | 10136-084 | |
| Zeba Spin Desalting Columns, 7K MWCO, 0.5 mL | Thermo Fisher Scientific, Waltham, MA | 89882 |
References
- Szoka, F., Papahadjopoulos, D. Comparative properties and methods of preparation of lipid vesicles (liposomes). Annual Review of Biophysics and Bioengineering. 9, 467-508 (1980).
- Mouritsen, O. G. Model answers to lipid membrane questions. Cold Spring Harbor Perspectives in Biology. 3 (9), 004622 (2011).
- Chan, Y. -. H. M., Boxer, S. G. Model membrane systems and their applications. Current Opinion in Chemical Biology. 11 (6), 581-587 (2007).
- Li, S., Hu, P., Malmstadt, N. Confocal imaging to quantify passive transport across biomimetic lipid membranes. Analytical Chemistry. 82 (18), 7766-7771 (2010).
- Lingwood, D., Simons, K. Lipid rafts as a membrane-organizing principle. Science. 327 (5961), 46-50 (2010).
- Elbaradei, A., Brown, S. L., Miller, J. B., May, S., Hobbie, E. K. Interaction of polymer-coated silicon nanocrystals with lipid bilayers and surfactant interfaces. Physical Review E. 94 (4), 042804 (2016).
- Veatch, S. L., Keller, S. L. Organization in lipid membranes containing cholesterol. Physical Review Letters. 89 (26), 268101 (2002).
- Plasencia, I., Norlén, L., Bagatolli, L. A. Direct visualization of lipid domains in human skin stratum corneum’s lipid membranes: Effect of pH and temperature. Biophysical Journal. 93 (9), 3142-3155 (2007).
- Dietrich, C., et al. Lipid rafts reconstituted in model membranes. Biophysical Journal. 80 (3), 1417-1428 (2001).
- Deans, J. P., Li, H., Polyak, M. J. CD20-mediated apoptosis: signalling through lipid rafts. Immunology. 107 (2), 176-182 (2002).
- Edidin, M. The state of lipid rafts: from model membranes to cells. Annual Review of Biophysics and Biomolecular Structure. 32, 257-283 (2003).
- Pike, L. J. Lipid rafts: bringing order to chaos. Journal of Lipid Research. 44 (4), 655-667 (2003).
- Tsui-Pierchala, B. A., Encinas, M., Milbrandt, J., Johnson, E. M. Lipid rafts in neuronal signaling and function. Trends in Neurosciences. 25 (8), 412-417 (2002).
- Sezgin, E., Levental, I., Mayor, S., Eggeling, C. The mystery of membrane organization: composition, regulation and roles of lipid rafts. Nature Reviews Molecular Cell Biology. 18 (6), 361-374 (2017).
- Scheve, C. S., Gonzales, P. A., Momin, N., Stachowiak, J. C. Steric pressure between membrane-bound proteins opposes lipid phase separation. Journal of the American Chemical Society. 135 (4), 1185-1188 (2013).
- Reeves, J. P., Dowben, R. M. Formation and properties of thin-walled phospholipid vesicles. Journal of Cellular Physiology. 73 (1), 49-60 (1969).
- Horger, K. S., Estes, D. J., Capone, R., Mayer, M. Films of agarose enable rapid formation of giant liposomes in solutions of physiologic ionic strength. Journal of the American Chemical Society. 131 (5), 1810-1819 (2009).
- Angelova, M. I., Dimitrov, D. S. Liposome electroformation. Faraday Discussions of the Chemical Society. 81, 303-311 (1986).
- Teh, S. -. Y., Khnouf, R., Fan, H., Lee, A. P. Stable, biocompatible lipid vesicle generation by solvent extraction-based droplet microfluidics. Biomicrofluidics. 5 (4), (2011).
- Hu, P. C., Li, S., Malmstadt, N. Microfluidic fabrication of asymmetric giant lipid vesicles. ACS Applied Materials & Interfaces. 3 (5), 1434-1440 (2011).
- Lu, L., Schertzer, J. W., Chiarot, P. R. Continuous microfluidic fabrication of synthetic asymmetric vesicles. Lab on a Chip. 15 (17), 3591-3599 (2015).
- Maktabi, S., Schertzer, J. W., Chiarot, P. R. Dewetting-induced formation and mechanical properties of synthetic bacterial outer membrane models (GUVs) with controlled inner-leaflet lipid composition. Soft Matter. 15 (19), 3938-3948 (2019).
- Stachowiak, J. C., et al. Unilamellar vesicle formation and encapsulation by microfluidic jetting. Proceedings of the National Academy of Sciences of the United States of America. 105 (12), 4697-4702 (2008).
- Kim, S., Martin, G. M. Preparation of cell-size unilamellar liposomes with high captured volume and defined size distribution. Biochimica et Biophysica Acta (BBA) – Biomembranes. 646 (1), 1-9 (1981).
- Moscho, A., Orwar, O., Chiu, D. T., Modi, B. P., Zare, R. N. Rapid preparation of giant unilamellar vesicles. Proceedings of the National Academy of Sciences of the United States of America. 93 (21), 11443-11447 (1996).
- Walde, P., Cosentino, K., Engel, H., Stano, P. Giant vesicles: Preparations and applications. ChemBioChem. 11 (7), 848-865 (2010).
- Seddon, A. M., Curnow, P., Booth, P. J. Membrane proteins, lipids and detergents: not just a soap opera. Biochimica et Biophysica Acta (BBA) – Biomembranes. 1666 (1), 105-117 (2004).
- le Maire, M., Champeil, P., Møller, J. V. Interaction of membrane proteins and lipids with solubilizing detergents. Biochimica et Biophysica Acta (BBA) – Biomembranes. 1508 (1), 86-111 (2000).
- Rigaud, J. -. L., Lévy, D. Reconstitution of membrane proteins into liposomes. Methods in Enzymology. 372, 65-86 (2003).
- Renthal, R. An unfolding story of helical transmembrane proteins. Biochimie. 45 (49), 14559-14566 (2006).
- Jørgensen, I. L., Kemmer, G. C., Pomorski, T. G. Membrane protein reconstitution into giant unilamellar vesicles: a review on current techniques. European Biophysics Journal. 46 (2), 103-119 (2017).
- Hansen, J. S., et al. Formation of giant protein vesicles by a lipid cosolvent method. ChemBioChem. 12 (18), 2856-2862 (2011).
- Kahya, N., Pécheur, E. -. I., de Boeij, W. P., Wiersma, D. A., Hoekstra, D. Reconstitution of membrane proteins into giant unilamellar vesicles via peptide-induced fusion. Biophysical Journal. 81 (3), 1464-1474 (2001).
- Kahya, N., Merkle, D., Schwille, P. Pushing the complexity of model bilayers: Novel prospects for membrane biophysics. Fluorescence of Supermolecules, Polymers, and Nanosystems. , 339-359 (2008).
- Dezi, M., Di Cicco, A., Bassereau, P., Lévy, D. Detergent-mediated incorporation of transmembrane proteins in giant unilamellar vesicles with controlled physiological contents. Proceedings of the National Academy of Sciences of the United States of America. 110 (18), 7276-7281 (2013).
- Shaklee, P. M., et al. Protein incorporation in giant lipid vesicles under physiological conditions. ChemBioChem. 11 (2), 175-179 (2010).
- Estes, D. J., Mayer, M. Giant liposomes in physiological buffer using electroformation in a flow chamber. Biochimica et Biophysica Acta (BBA) – Biomembranes. 1712 (2), 152-160 (2005).
- Girard, P., et al. A new method for the reconstitution of membrane proteins into giant unilamellar vesicles. Biophysical Journal. 87 (1), 419-429 (2004).
- Doeven, M. K., et al. lateral mobility and function of membrane proteins incorporated into giant unilamellar vesicles. Biophysical Journal. 88 (2), 1134-1142 (2005).
- Levental, I., et al. Cholesterol-dependent phase separation in cell-derived giant plasma-membrane vesicles. The Biochemical Journal. 424 (2), 163-167 (2009).
- López-Montero, I., Rodríguez-García, R., Monroy, F. Artificial spectrin shells reconstituted on giant vesicles. The Journal of Physical Chemistry Letters. 3 (12), 1583-1588 (2012).
- Hansen, J. S., Thompson, J. R., Hélix-Nielsen, C., Malmstadt, N. Lipid directed intrinsic membrane protein segregation. Journal of the American Chemical Society. 135 (46), 17294-17297 (2013).
- Gutierrez, M. G., Malmstadt, N. Human serotonin receptor 5-HT 1A preferentially segregates to the liquid disordered phase in synthetic lipid bilayers. Journal of the American Chemical Society. 136 (39), 13530-13533 (2014).
- Gutierrez, M. G., et al. The lipid phase preference of the adenosine A2A receptor depends on its ligand binding state. Chemical Communications. 55 (40), 5724-5727 (2019).
- Gutierrez, M. G., Mansfield, K. S., Malmstadt, N. The functional activity of the human serotonin 5-HT 1A receptor is controlled by lipid bilayer composition. Biophysical Journal. 110, 2486-2495 (2016).
- Sriram, K., Insel, P. A. G Protein-coupled receptors as targets for approved drugs: How many targets and how many drugs. Molecular Pharmacology. 93 (4), 251-258 (2018).
- Nichols, D. E., Nichols, C. D. Serotonin receptors. Chemical Reviews. 108 (5), 1614-1641 (2008).
- Del Vecchio, K., Stahelin, R. V. Using surface plasmon resonance to quantitatively assess lipid-protein interactions. Methods in Molecular Biology. 1376, 141-153 (2016).
- Place, J. F., Sutherland, R. M., Dähne, C. Opto-electronic immunosensors: a review of optical immunoassay at continuous surfaces. Biosensors. 1 (4), 321-353 (1985).
- Brown, M. F., Miljanich, G. P., Franklin, L. K., Dratz, E. A. H-NMR studies of protein-lipid interactions in retinal rod outer segment disc membranes. FEBS letters. 70 (1), 56-60 (1976).
- Sun, F., et al. Structural basis for interactions of the Phytophthora sojae RxLR effector Avh5 with phosphatidylinositol 3-phosphate and for host cell entry. Molecular Plant-Microbe Interactions: MPMI. 26 (3), 330-344 (2013).
- Kavran, J. M., et al. Specificity and promiscuity in phosphoinositide binding by pleckstrin homology domains. The Journal of Biological Chemistry. 273 (46), 30497-30508 (1998).
- Stevenson, J. M., Perera, I. Y., Boss, W. F. A phosphatidylinositol 4-Kinase pleckstrin homology domain that binds phosphatidylinositol 4-Monophosphate. Journal of Biological Chemistry. 273 (35), 22761-22767 (1998).
- Han, X., Yang, Y., Zhao, F., Zhang, T., Yu, X. An improved protein lipid overlay assay for studying lipid-protein interactions. Plant Methods. 16 (1), 33 (2020).
- Yen, H. -. Y., et al. PtdIns(4,5)P 2 stabilizes active states of GPCRs and enhances selectivity of G-protein coupling. Nature. 559 (7714), 423-427 (2018).
- Myers, M., Mayorga, O. L., Emtage, J., Freire, E. Thermodynamic characterization of interactions between ornithine transcarbamylase leader peptide and phospholipid bilayer membranes. Biochimie. 26 (14), 4309-4315 (1987).
- Swamy, M. J., Sankhala, R. S. Probing the thermodynamics of protein-lipid interactions by isothermal titration calorimetry. Lipid-Protein Interactions: Methods and Protocols. , 37-53 (2013).
- Han, X., Shi, Y., Liu, G., Guo, Y., Yang, Y. Activation of ROP6 GTPase by phosphatidylglycerol in arabidopsis. Frontiers in Plant Science. , (2018).
- Surolia, A., Bachhawat, B. K. The effect of lipid composition on liposome-lectin interaction. Biochemical and Biophysical Research Communications. 83 (3), 779-785 (1978).
- Sarkis, J., Vié, V. Biomimetic models to investigate membrane biophysics affecting lipid-protein interaction. Frontiers in Bioengineering and Biotechnology. 8, 270 (2020).
- McMahon, H. T., Boucrot, E. Membrane curvature at a glance. Journal of Cell Science. 128 (6), 1065-1070 (2015).
- Banerjee, K. K., Kumar, S., Bremmell, K. E., Griesser, H. J. Molecular-level removal of proteinaceous contamination from model surfaces and biomedical device materials by air plasma treatment. Journal of Hospital Infection. 76 (3), 234-242 (2010).
- Raiber, K., Terfort, A., Benndorf, C., Krings, N., Strehblow, H. -. H. Removal of self-assembled monolayers of alkanethiolates on gold by plasma cleaning. Surface Science. 595 (1), 56-63 (2005).
- Gutierrez, M. G., et al. The lipid phase preference of the adenosine A 2A receptor depends on its ligand binding state. Chemical Communications. 55 (40), 5724-5727 (2019).
- Garten, M., Levy, D., Bassereau, P. The giant vesicle book. The giant vesicle book. , 38-51 (2021).
- Gutierrez, M. G., et al. G Protein-coupled receptors incorporated into rehydrated diblock copolymer vesicles retain functionality. Small. 12 (38), 5256-5260 (2016).
- Peruzzi, J., Gutierrez, M. G., Mansfield, K., Malmstadt, N. Dynamics of hydrogel-assisted giant unilamellar vesicle formation from unsaturated lipid systems. Langmuir. 32 (48), 12702-12709 (2016).
- Shchelokovskyy, P., Tristram-Nagle, S., Dimova, R. Effect of the HIV-1 fusion peptide on the mechanical properties and leaflet coupling of lipid bilayers. New Journal of Physics. 13, 025004 (2011).

