Magnetic Isolation of Microglial Cells from Neonate Mouse for Primary Cell Cultures
Summary
Primary microglia cultures are commonly used to evaluate new anti-inflammatory molecules. The present protocol describes a reproducible and relevant method to magnetically isolate microglia from neonate pups.
Abstract
Microglia, as brain resident macrophages, are fundamental to several functions, including response to environmental stress and brain homeostasis. Microglia can adopt a large spectrum of activation phenotypes. Moreover, microglia that endorse pro-inflammatory phenotype is associated with both neurodevelopmental and neurodegenerative disorders. In vitro studies are widely used in research to evaluate potential therapeutic strategies in specific cell types. In this context studying microglial activation and neuroinflammation in vitro using primary microglial cultures is more relevant than microglial cell lines or stem-cell-derived microglia. However, the use of some primary cultures might suffer from a lack of reproducibility. This protocol proposes a reproducible and relevant method of magnetically isolating microglia from neonate pups. Microglial activation using several stimuli after 4 h and 24 h by mRNA expression quantification and a Cy3-bead phagocytic assay is demonstrated here. The current work is expected to provide an easily reproducible technique for isolating physiologically relevant microglia from juvenile developmental stages.
Introduction
Microglia are the central nervous system resident macrophage-like cells derived from erythropoietic precursors of the yolk sac that migrate to the neuroepithelium during early embryonic development1. Apart from their immunity functions, they also play a significant role during neurodevelopment, particularly for synaptogenesis, neuronal homeostasis, and myelination2. In adulthood, microglia develop long cellular processes to scan the environment continuously. In case of homeostasis ruptures such as brain injury or brain disease, microglia can change their morphological appearance to adopt an amoeboid shape, migrate to the injured area, increase and release many cytoprotective or cytotoxic factors. Microglia have heterogeneous activation states depending on their developmental stage and the type of injury sustained3,4,5. In this study, these activation states are broadly classified into three different phenotypes: pro-inflammatory/phagocytic, anti-inflammatory, and immuno-regulatory, keeping in mind that in reality, the situation is likely to be more complex6.
Studying in vivo microglial activation and screening for neuroprotective strategies at early stages of brain development can be challenging due to (1) the fragility of animals before weaning and (2) the low number of microglial cells. Therefore in vitro studies on microglia are widely used for toxicity7,8,9, neuroprotective strategies5,10,11,12,13,14, and co-cultures15,16,17,18,19,20,21. In vitro studies can use either microglial cell lines, stem-cell-derived microglia, or primary microglia culture. All these approaches have advantages and disadvantages, and the choice depends on the initial biological question. The benefits of using primary microglia cultures are the homogenous genetic background, pathogen-free history, and control of the time when the microglia are stimulated after animal death22.
Over the years, different methods (flow cytometry, shaking, or magnetic labeling) were developed for culturing primary microglia from rodents, both neonate and adult23,24,25,26,27,28,29. In the present work, microglia isolation from mouse neonate pups is performed using previously described magnetic-activated cell sorting technology using microbead-coated anti-mouse CD11b25,27,29. CD11b is an integrin-receptor expressed at the surface of myeloid cells, including microglia. When there is no inflammatory challenge within the brain, almost all CD11b+ cells are microglia30. Compared to other previously published methods23,24,25,26,27,28,29, the present protocol balances immediate ex vivo microglial activation analyses and common in vitro primary microglial culture. Thus, microglia are (1) isolated at postnatal day (P)8 without myelin removal, (2) cultured without serum, and (3) exposed either to siRNA, miRNA, pharmacological compound, and/or inflammatory stimuli only 48 h after brain isolation. Each of these three aspects makes the current protocol relevant and rapid. First of all, the use of pediatric microglia allows obtaining dynamic and reactive viable cells in culture without requiring an additional demyelination step that could potentially modify microglial reactivity in vitro. The present protocol aims to get as close as possible to the physiological environment of microglia. Indeed, microglia never encounter serum, and this protocol does not require the use of serum either. Moreover, exposing microglia as early as 48 h after culture prevents them from losing their physiological faculties.
Protocol
The protocol was approved, and all the animals were handled according to the institutional guidelines of Institut National de la Santé et de la Recherche Scientifique (Inserm, France). Magnetic isolation of microglia from the brains of 24 OF1 mouse pups (both male and female) at P8, divided into 6-well, 12-well, or 96-well plates, are presented. The experimental work was performed under a hood to maintain sterile conditions.
1. Preparation of sterile solutions for isolation and cell culture
- Prepare 50 mL of 1x Hanks' Balanced Salt Solution (HBSS) without Ca2+ and Mg2+ (HBSS-/-) from the commercially available 10x solution (see Table of Materials).
- Prepare dissociation mixture according to the composition provided in Table 1 using a commercially available tissue dissociation kit (see Table of Materials).
- Prepare 200 mL of 1x HBSS with Ca2+ and Mg2+ (HBSS+/+) from the commercially available 10x solution.
- Prepare 200 mL of 1x PBS + 0.5% BSA (referred to as cell sorting buffer).
- Prepare CD11b-microbeads according to Table 2.
- Prepare 500 mL of macrophage serum-free culture medium (SFM) + 1% of Penicillin-Streptomycin (P/S). Make aliquots of 50 mL tubes and store them at 4 °C. This is referred to as the microglia medium later in the text.
NOTE: All isolation solutions must be freshly prepared under sterile conditions on experimentation day and kept on ice. Microglia cell culture medium can be prepared, aliquoted in 50 mL tubes, and kept at 4 °C for future use. Filtration is not needed.
2. Brain dissection
- Decapitate the pup's head using large scissors without previous general anesthesia.
- Cut the skin from the neck to the nose following the sagittal suture (15-20 mm) with small scissors (Figure 1A-C).
- Insert a small scissor's tips within the Foramen magnum parallel to the skull. Cut from each side carefully to the eyes (Figure 1D,E).
- Cut between the eyes with small scissors to detach the skull and brain from the head (Figure 1F).
- With two forceps, grab the skull close to the olfactory bulbs and tear the skull carefully, taking care not to damage the underlying brain (Figure 1G-I).
- Remove cerebellum and olfactive bulb with a razor blade and cut the brain into two pieces (Figure 1J).
- Place the brain pieces in a Petri dish containing 30-40 mL of HBSS-/- (Figure 1K).
3. Brain dissociation and magnetic microglial isolation
NOTE: All cell manipulations and resuspensions must be performed with a 1,000 µL pipette with great caution. Applying a high mechanical action may activate or kill microglia cells.
- Transfer 12 brain pieces (~1.2 g) per dissociation tube containing dissociation mixture according to Table 1. For 24 pups, four C-Tubes were needed (Figure 2A-B).
- Place C-Tubes on the dissociator (with the heating mode). Start the optimized NTDK program in the equipment according to the manufacturer's instruction (Figure 2D).
- Centrifuge at 300 x g for 20 s (at 4 °C) to collect all the cells. Complete the mechanical dissociation by pipetting three times up and down with a 1,000 µL pipette (Figure 2E).
- Transfer the cells to four 15 mL tubes + strainers. Rinse the strainers with 10 mL of HBSS+/+ (Figure 2F).
- Centrifuge at 300 x g for 10 min (at 4 °C) and remove the supernatant. Carefully resuspend the pellet with 10 mL of HBSS+/+ (Figure 2G).
- Centrifuge at 300 x g for 10 min (at 4 °C) and remove the supernatant. Carefully resuspend the pellet with 6 mL of sorting buffer (step 1.4) (Figure 2H).
- Centrifuge at 300 x g for 10 min (at 4 °C) and remove the supernatant. Add 200 µL of CD11b-microbead solution (step 1.5) per tube and resuspend carefully (Figure 2I).
- Incubate the tubes for 15-20 min at 4 °C. Carefully resuspend the pellet with 6 mL of sorting buffer (Figure 2I-J).
- Centrifuge at 300 x g for 10 min (at 4 °C) and remove the supernatant. Carefully resuspend the pellet with 8 mL of sorting buffer (Figure 2K).
- Follow the POSSEL program on the separator (see Table of Materials) to prepare eight columns. Transfer cells 1 mL by 1 mL on the column. Wait for all the cells to pass through before adding another mL. Elute CD11b+ cells on sterile elution plate with 1 mL of sorting buffer (Figure 2L).
- Pool CD11b+ cells in a new 50 mL tube (Figure 2M).
- Centrifuge at 300 x g for 10 min (at 4 °C) and remove the supernatant. Carefully resuspend the pellet with 10 mL of cold microglia medium (step 1.6) (Figure 2N).
- Count the CD11b+ cells. At P8, one should obtain ~650,000 cells per brain.
NOTE: In the present protocol, the cells were counted using an automated cell counter (see Table of Materials). - Resuspend the cells in cold microglia medium to a final concentration of 650,000-700,000 cells/mL and dispense in cell culture plates.
NOTE: The 6-well plates are for Western Blot (2 mL per well); the 12-well plates are for RT-qPCR analysis (1 mL per well), and the 96-well plates are for phagocytic assay (250 µL per well); all three were used for this work. However, in this manuscript, the results from the Western Blot analysis are not shown, but it is possible to perform with this protocol. - Place the plates at 37 °C with 5% CO2 overnight. Change the medium carefully with a 1,000 µL pipette with pre-heated microglia medium.
- Place the plates at 37 °C with 5% CO2 overnight before stimulation.
NOTE: Isolating microglia from more than 36 pups and after P9 are not recommended. This will increase the risk of contamination and accumulation of cell debris to activate microglia.
4. Cell stimulations
- Prepare stimulation reagents according to Table 3 using the commercially available reagents (see Table of Materials).
- Add the appropriate volume in each stimulated well.
NOTE: The appropriate volume depends on the concentration of the stimulus and the size of the well. - Place the plates at 37 °C with 5% CO2until the end of stimulation at 6 h, 24 h, or 48 h.
- For the Western Blot analysis, aspirate the supernatant and add 50 µL of protein lysis buffer (see Table of Materials) with a pipette. Using tips, scratch the bottom of the plate to detach the lysed cells and transfer them to 1.5 mL tubes. Store at -80 °C.
NOTE: The culture plates can be stored for years at -80 °C if the supernatant is aspirated correctly. - For RT-qPCR analysis6,31, aspirate the supernatant and store the culture plates directly at -80 °C until mRNA extraction (step 5).
- For the phagocytic assay, please see step 6.
5. mRNA extraction and RT-qPCR analysis
- Perform RNA extraction, RT-PCR, and RT-qPCR following the manufacturer's protocol (see Table of Materials). Refer to Table 4 for the primer sequences5,6,31.
- Perform RT-qPCR analysis by following the previously published report5.
6. Phagocytic assay
- Stimulate the cells and perform phagocytic assay during the last 3 h of the stimulation. For example, for stimulation of 6 h, start the phagocytic assay after 3 h of stimulation; and for stimulation of 12 h, start the phagocytic assay 9 h after the beginning of the stimulation.
- Calculate the number of beads needed to prepare considering the ratio (1 cell: 50 beads). For one well of the 96-well plate, 8.1 x 106 beads are required. Prepare the beads mixture according to Table 5.
NOTE: Carefully vortex the beads stock vial before adding to the PBS/FBS mixture. - Incubate for 1 h in a water bath at 37 °C. Vortex every 10 min.
- Calculate the solution volume to add to each well. Add the solution and incubate for 3 h.
- Rinse the wells three times with 1x PBS. Read the fluorescence emission at 550 nm (Cy3 emission wavelength).
7. Purity quality control
- Evaluate the purity of CD11b magnetic isolation by flow cytometry (FACS) (before and after cell sorting), and then perform the RT-qPCR.
- Count the cells and resuspend in FACS buffer (PBS + 2 mM of EDTA + 0.5% of Bovine Serum Albumin) in order to obtain a dilution of 10 x 106 cells/mL after step 3.5 and step 3.14.
- After 15 min of the conventional Fc blocking32,33, incubate the cells for 15 min with viability probe (FVS780) and fluorophore-conjugated antibodies against mouse CD45, CD11b, CX3CR1, ACSA-2, O4 or their corresponding control isotypes32,33 at the concentration recommended by the manufacturers (see Table of Materials).
- Wash the cells with FACS buffer, fix, and permeabilize with a commercially available permeabilization kit (see Table of Materials).
- Perform Fc blocking again on the permeabilized cells, and then incubate with fluorophore-conjugated antibody against NeuN (see Table of Materials) or its control isotype for 15 min.
- Perform FACS analysis after washing with FACS buffer.
- After excluding the doublets and the dead cells based on morphological parameters and FVS780 staining, respectively, select the gating strategy for the surface expression of CX3CR1 (microglia), ACSA-2 (astrocytes), O4 (oligodendrocytes), and intracellular presence of NeuN (neurons) for the analysis of the percentage of positive cells.
- Following step 5, extract mRNA and perform RT-qPCR to quantify Itgam (CD11b), Cx3cr1, Olig2, Synaptophysin, and Gfap mRNA. Normalize Cq using Rpl13a mRNA as a reporter, and perform a relative expression to Itgam mRNA.
Representative Results
Microglia is the CNS resident macrophage that gets activated when exposed to environmental challenges (trauma, toxic molecules, inflammation )4,5,6,34 (Figure 3A). In vitro studies on microglia are commonly used to evaluate cell-autonomous mechanisms related to those environmental challenges and characterize activation state after pharmacological or genetic manipulation. Here, an approach is presented to isolate primary microglia at the juvenile stage using magnetic coupled beads.
An easy readout for microglial activation in vitro is to quantify mRNA expression of several microglial reactivity markers associated with a pro-inflammatory phenotype (Cd86, Nos2, Ptgs2, Tnf) or an anti-inflammatory phenotype (Cd206, Igf1, Arg1, Lgasl3)5,6,31. These phenotypes were described depending on mRNA expression after pro-inflammatory (IL-1β, LPS) or anti-inflammatory (IL-4 or IL-10) stimulus. Some markers are classified as immuno-regulatory markers because they are upregulated through pro- and anti-inflammatory stimulation (Figure 3A). This classification was described previously by our team5,6,31. After 48 h, isolated microglial were stimulated with pro- and anti-inflammatory stimulus for 4 h or 24 h. mRNA were extracted, and RT-qPCR quantified gene expression. At 4 h and 24 h, pro-inflammatory and immuno-regulatory markers are induced by IL-1β, IL-1β + IFN-γ, and LPS5,6,31. At 4 h, IL-4 stimulation also induces the immuno-regulatory marker Il-1rn6. They are induced strongly at 4 h after IL-4 stimulation regarding anti-inflammatory markers. Interestingly, Cd206 is also significantly upregulated 24 h after IL-1β stimulation6 (Figure 3B).
To evaluate the phagocytic activity of microglia in vitro, fluorescent Cy3 PVC beads were used. They were pre-treated with fetal bovine serum (FBS) to facilitate their phagocytosis by microglia. Microglia were polarized toward pro-inflammatory phenotype by stimulation with IL-1β + IFN-γ or LPS for 3 h or 21 h. Three hours before the end of stimulation, Cy3-beads were incubated with microglial cells. After rinsing with 1x PBS, the fluorescence intensity in each well was quantified. Quantification relative to wells with no beads was expressed, and representative images were taken (Figure 4). After 6 h stimulation, microglia start to phagocyte Cy3-beads only under IL-1β + IFN-γ. After a 24 h stimulation, there is an increase of Cy3 fluorescence for both kinds of stimulation. That increase of Cy3 fluorescence highlights increased phagocytic activity (Figure 4C).
Flow cytometry (FACS) and RT-qPCR were performed to evaluate the microglial culture's purity. Different brain cellular populations can be distinguished by flow cytometry: CX3CR135 for microglia, O4 for oligodendrocytes36, NeuN for neurons37, and ACSA-2 for astrocytes38. After dissociation, all brain cells are present; however, after cell sorting using CD11b antibody, only microglia and small amounts of O4 cells are present (Figure 5A). 48 h after primary cell culture, only microglia markers are found as evaluated by RT-qPCR (Figure 5B).
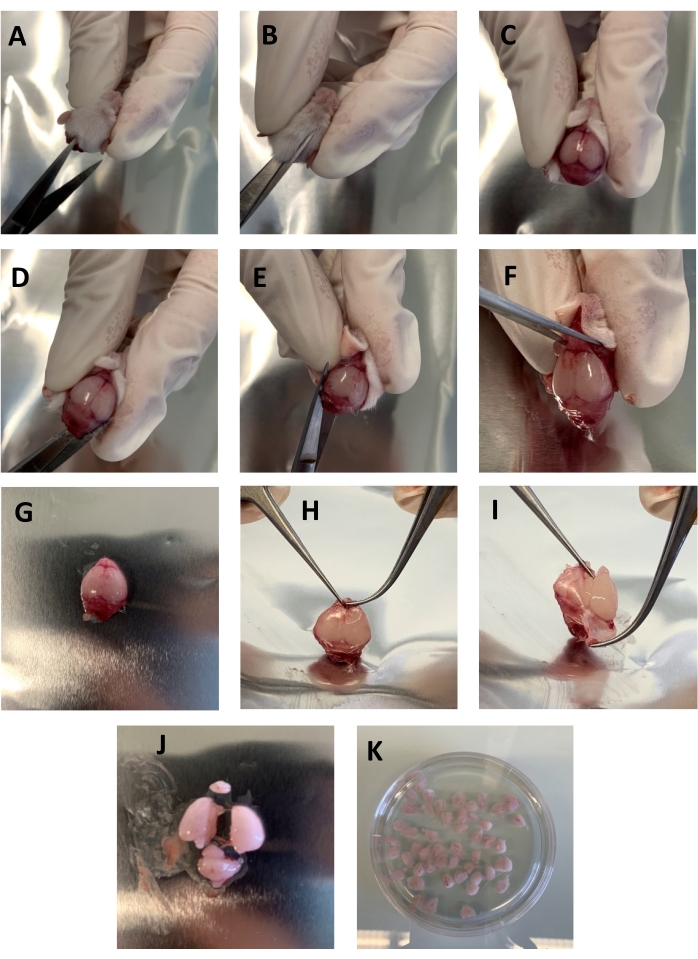
Figure 1: Step-by-step representation showing the removal of the brain from the skull. (A–C) Small scissors were used to cut the skin from the neck to the nose following the sagittal suture. (D–E) The small scissor's tips were inserted within the Foramen magnum parallel to the skull, and from each side to the eyes were carefully cut. (F) The skull and brain were detached from the head by cutting between the eyes with small scissors. (G–I) The skull was grabbed close to the olfactive bulbs with two forceps and torn carefully not to damage the underlying brain. (J) The cerebellum and the olfactive bulb were removed with a razor blade, and the brain was cut into two pieces. (K) The brain pieces were placed in a Petri dish containing 30-40 mL of HBSS-/-. Please click here to view a larger version of this figure.
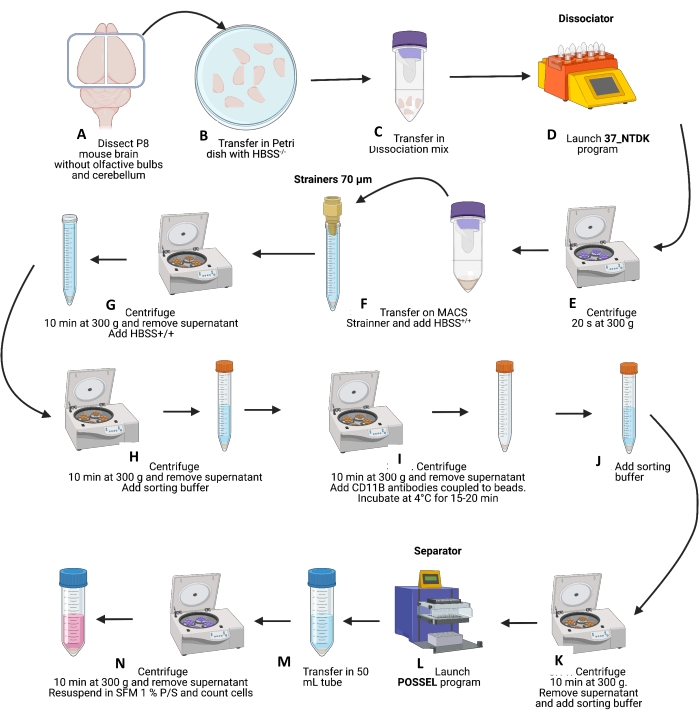
Figure 2: Schematic representation of brain dissociations and microglial cells isolation. (A–C) After P8 mouse brain dissection and removing olfactive bulbs and cerebellum, brains were first transferred to a Petri dish with HBSS+/+, and then to dissociation tubes containing the dissociation mixture. The C-tubes were placed on the dissociator (with the heating mode), and the NTDK program was started (D). (E) At the end of the program, tubes were centrifuged at 300 x g for 20 s at 4 °C; dissociation was then completed by pipetting three times up and down with a 1,000 µL pipette. (F) Cells were then transferred to 15 mL tubes + strainers of 70 µm and rinsed with 10 mL of HBSS+/+. (G) Samples were then centrifuged at 300 x g for 10 min at 4 °C, the supernatant was removed, and the pellet was resuspended with 10 mL of HBSS+/+. (H) Tubes were again centrifuged at 300 x g for 10 min at 4 °C; the supernatant was removed, and then the pellet was resuspended in 6 mL of sorting buffer. (I) Tubes were centrifuged at 300 x g for 10 min at 4 °C, the supernatant was removed, and the CD11b-microbead (200 µL) solution was added. Tubes were incubated for 15-20 min at 4 °C, and then resuspended in 6 mL of sorting buffer (J) and centrifuged at 300 x g for 10 min at 4 °C. (K) The supernatant was removed, and the pellet was carefully resuspended in 8 mL of sorting buffer. (L) Then, launch the POSSEL program on the separator to prepare columns. Cells were transferred 1 mL by 1 mL on the column, and CD11b cells were eluted on a sterile elution plate with 1 mL of sorting buffer. (M) CD11b cells were pooled in a new 50 mL tube and centrifuged at 300 x g for 10 min at 4 °C, and the supernatant was removed. (N) The pellet was carefully resuspended in 10 mL of cold microglia medium in the last step. The cells were counted and diluted in the microglial medium to a final concentration of 650,000-700,000 cells/mL dispensed in cell culture plates. Please click here to view a larger version of this figure.
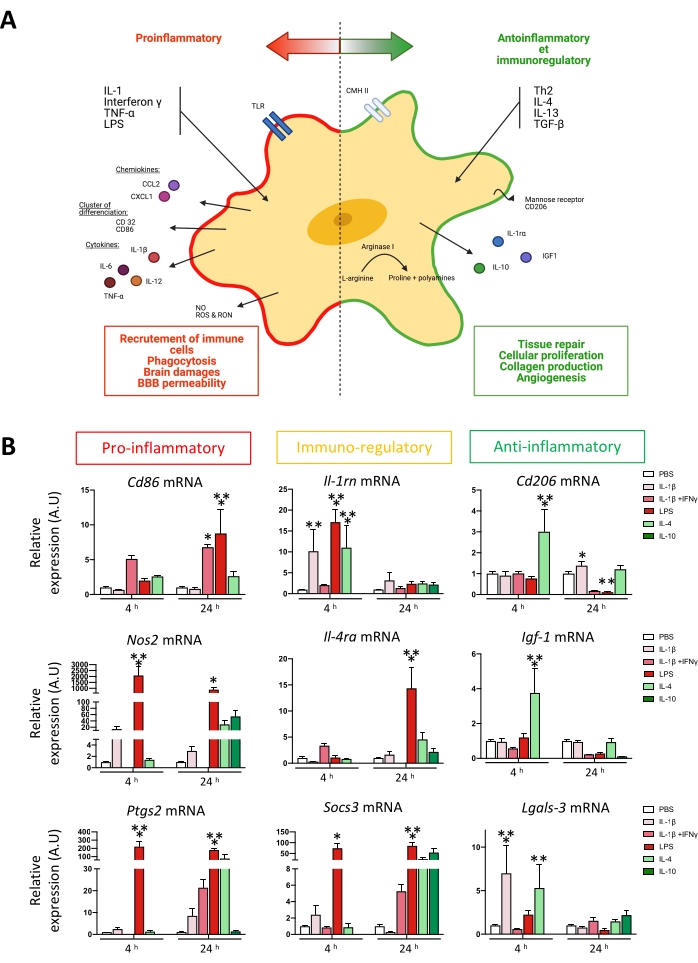
Figure 3: Microglial activation following exposition to environmental challenges. (A) Simplified schematic representation of microglial activation spectrum. (B) Relative quantification of microglial activation markers after 4 h or 24 h stimulation. Two-way ANOVA followed by Dunnett's multiple comparisons test39 (n = 5-15); error bars represent SEM; *p < 0.05, **p < 0.01, ***p < 0.001. Please click here to view a larger version of this figure.
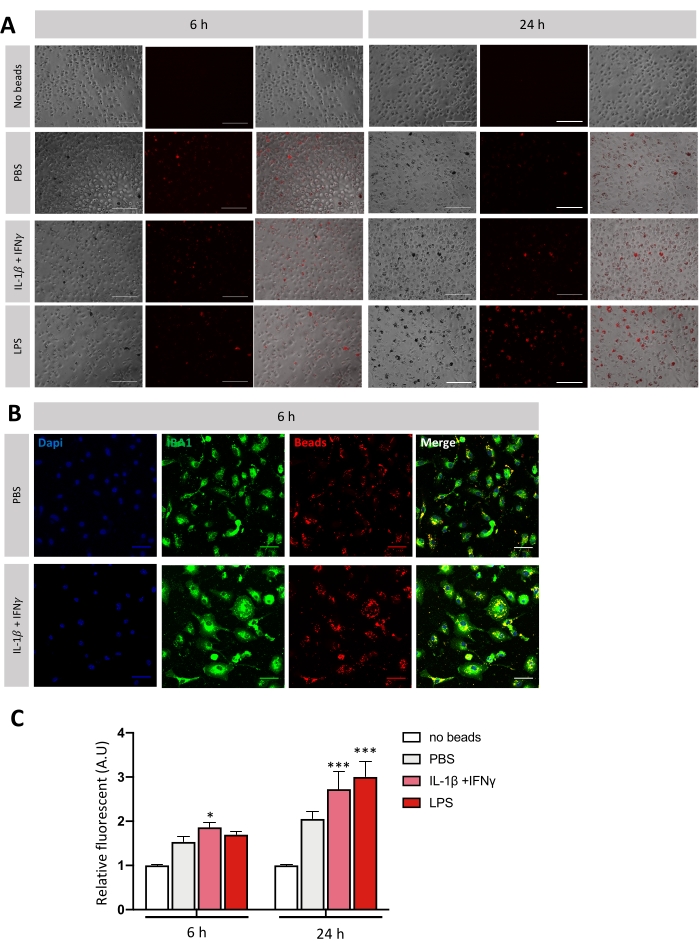
Figure 4: Evaluation of the phagocytic activity of microglia in vitro. (A) Representative images of microglia Cy3-beads (in red) phagocytosis after stimulation of 6 h or 24 h. The images were acquired using 20x objective of the fluorescence microscope. Scale bar = 100 μm. (B) Relative quantification of fluorescence emits per well. Statistics was performed using two-way ANOVA followed by Dunnett's multiple comparisons test (n = 4-7); error bars represent SEM; *p < 0.05, ** p < 0.01, ***p < 0.001. (C) Representative picture of microglia (IBA-1 + cells in green) Cy3-beads (in red) phagocytosis after 6 h stimulation. The images are acquired using a 40x objective of the confocal microscope. Scale bar = 300 μm. Please click here to view a larger version of this figure.
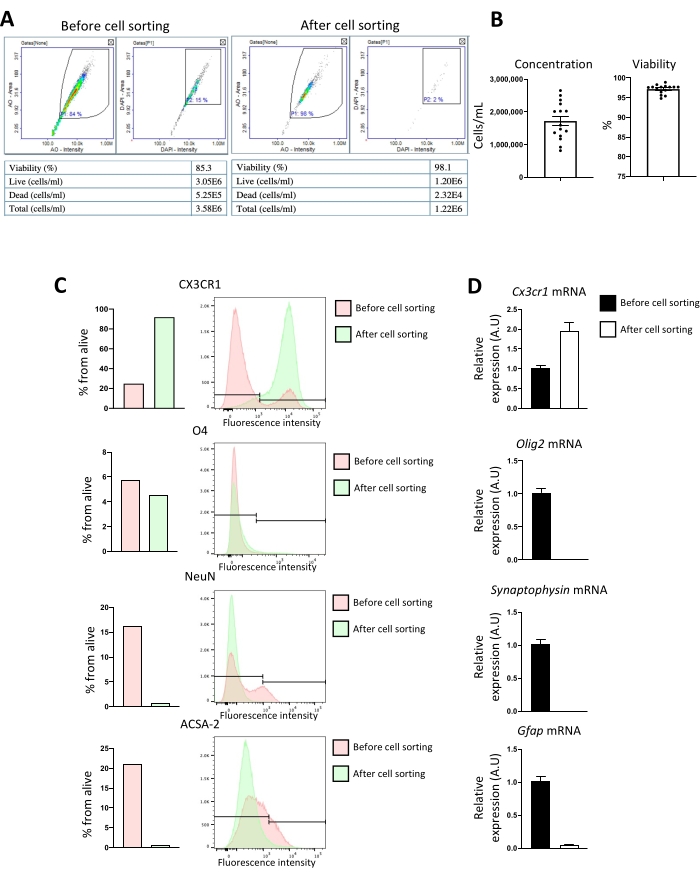
Figure 5: Evaluation of CD11b magnetic isolation purity by flow cytometry before and after cell sorting. (A) Reports examples from the cell counter before and after cell sorting, highlighting cell concentration and viability. (B) The x-axis represent cell concentration (cell/mL) and viability (%) after CD11b cell sorting. Column bar graph; error bars represent SEM (n = 16). Phenotypic and gene expression analysis of cell population markers before and after CD11b+ cell sorting. After dissociation, CD11b+ cells from P8 mice brains were magnetically sorted. Expression of microglia (CX3CR1), oligodendrocytes (O4 or Olig2 mRNA), neurons (NeuN or Synaptophysin mRNA)), and astrocytes (ACSA-2 or Gfap mRNA) markers was analyzed before and after sorting. (C) FACS analysis of CX3CR1, O4, NeuN, and ACSA-2 expression. The x-axis represents the percentage of alive cells and cell numbers. (D) Relative quantification of CD11b+ cells expression of Cx3cr1, Olig2, Synaptophysin, and Gfap mRNA (x-axis represent relative target mRNA expression to Itgam mRNA). Column bar graph; error bars represent SEM (n = 7). Please click here to view a larger version of this figure.
| Solution | For one C-Tube (µL) | For four C-tubes (µL) |
| Buffer X | 2850 | 11400 |
| Enzyme P (Papain) | 75 | 300 |
| Enzyme A (DNAse) | 15 | 60 |
| Buffer Y | 30 | 120 |
| Total | 2970 | 11880 |
Table 1: Preparation of the dissociation mixture.
| Solution | For one tube (µL) | For four tubes (µL) |
| CD11b Microbeads | 20 | 80 |
| Sorting Buffer | 180 | 720 |
| Total | 200 | 800 |
Table 2: Preparation of the CD11b microbead solution.
| Stimulation | Concentration (ng/mL) |
| IL-1β | 50 |
| IFN-γ | 20 |
| LPS | 10 |
| IL-4 | 30 |
| IL-10 | 20 |
Table 3: The stimulation reagents.
| Genes | Protein | Forward | Reverse |
| Arg1 | Arginase 1 | GTG AAG AAC CCA CGG TCT GT | GCC AGA GAT GCT TCC AAC TG |
| Cd206 | Cluster of differentiation 206 | CTT CGG GCC TTT GGA ATA AT | TAG AAG AGC CCT TGG GTT GA |
| Cd32 | Cluster of differentiation 32 | CTG GAA GAA GCT GCC AAA AC | CCA ATG CCA AGG GAG ACT AA |
| Cd86 | Cluster of differentiation 86 | GAG CGG GAT AGT AAC GCT GA | GGC TCT CAC TGC CTT CAC TC |
| Gfap | Glial fibrillary acidic protein | AAGCCAAGCACGAAGCTAAC | CTCCTGGTAACTGGCCGACT |
| Igf-1 | Insulin like growth factor 1 | TGG ATG CTC TTC AGT TCG TG | GCA ACA CTC ATC CAC AAT GC |
| Il1-rn | Interleukine 1 receptor antagonist | TTG TGC CAA GTC TGG AGA TG | TTC TCA GAG CGG ATG AAG GT |
| Il4-ra | Interleukine 4 receptor antagonist | GGA TAA GCA GAC CCG AAG C | ACT CTG GAG AGA CTT GGT TGG |
| Itgam | Integrin alpha M | CTGGTGCTCTTGGCTCTCAT | GGCAGCTTCATTCATCATGT |
| Lgals3 | Lectin Glactoside-Binding soluble 3 | GAT CAC AAT CAT GGG CAC AG | ATT GAA GCG GGG GTT AAA GT |
| Nos2 | Nitric oxide synthase 2 | CCC TTC AAT GGT TGG TAC ATG G | ACA TTG ATC TCC GTG ACA GCC |
| Olig2 | Oligodendrocyte transcription factor 2 | CAGCGAGCACCTCAAATCTA | GATGGGCGACTAGACACCAG |
| Ptgs2 | Prostaglandin endoperoxide synthase 2 | TCA TTC ACC AGA CAG ATT GCT | AAG CGT TTG CGG TAC TCA TT |
| Rpl13 | Ribosomal protein L13a | ACA GCC ACT CTG GAG GAG AA | GAG TCC GTT GGT CTT GAG GA |
| Socs3 | Suppressor of cytokine 3 | CGT TGA CAG TCT TCC GAC AA | TAT TCT GGG GGC GAG AAG AT |
| Sphk1 | Sphingosine kinase 1 | TCC AGA AAC CCC TGT GTA GC | CAG CAG TGT GCA GTT GAT GA |
| Syp | Synaptophysin | ATCTCAGTGTCCCGATCCCA | GCTGTCTTCCTGGTGGGTAC |
| Tnf-α | Tumor necrosis factor α | GCC TCT TCT CAT TCC TGC TT | AGG GTC TGG GCC ATA GAA CT |
Table 4: RT-qPCR primer sequences.
| Stimulation | For 1-well | For n-wells |
| Cy3 Beads (1 cell : 50 beads) | 8, 100, 000 | X = (8, 100, 000) x n |
| 1x PBS | Y =(100+X)/n | 50 |
| FBS | 50 |
Table 5: Preparation of the beads mixture for phagocytic assay.
Discussion
The current work presents a primary microglial cell culture using magnetically sorted CD11b+ cells. In addition to the microglial functional evaluation (RT-qPCR and phagocytic assays), microglial culture purity was also determined.
Classical microglia cell cultures are commonly generated from P1 or P2 rodent neonate brain and co-culture with astrocytes for at least 10 days. Microglia are then separated mechanically using an orbital shaker. The method to isolate and culture microglia in vitro was described for the first time at the end of the 1990s from the brains of neonate rats40,41. Since then, it has been widely used to analyze microglial phenotypes such as activated/resting microglia or pro-inflammatory/anti-inflammatory microglia. Furthermore, this experiment has been fundamental to defining microglia as neurotoxic cells co-cultured with neurons. However, in 2014, Biber and collaborators described three significant disadvantages of studying microglia in vitro using classical methods42. (1) The experiment uses rat/mouse neonate brains (P1/P2), and these cells did not pass through all the maturation processes that can occur in vivo. (2) In the co-culture medium, 10% FBS is used; however, in vivo, microglia never encounter such components in a "normal" environment. (3) Recent studies demonstrate that in vivo microglia are restrained by several inhibitory components not present in the culture43,44. Moreover, many studies (electrophysiological45 and transcriptomic46,47) described non-stimulated primary microglia as "activated".
The present method proposes an alternative technique to generate microglia cultures using magnetically cell sorted technology. With this protocol, microglia are harvested only a few hours after the animal dies without any co-culture step. Moreover, microglia are only cultured 2 days in vitro (DIV) before stimulation compared to 10-14 DIV, including co-culture in other protocols. This allows getting closer to the microglial physiological conditions. This procedure of culturing microglia in vitro was previously published25,26,27,28,29. The current work presented an optimized protocol for neonate mice, standardized for reducing the number of animals used and without myelin removal step.
To reduce the number of mice used for primary microglia culture, we decided to work at P8 in the OF1 strain. At this stage of brain development in OF1 strain, up to 750,000 microglial cells per brain was obtained, in opposition to 500,000 microglial cells per C57BL6/J strain mouse brain at the same age. C57BL6/J strain has been published at a different developmental stage for microglia culture27,28. The present protocol has also been used with Fmr1KO mice to evaluate phagocytic properties of microglia linked to this mutation and LysMCre: Dicerfl/+ mice to assess microglial activation phenotypes by RT-qPCR. Thus, working with C57BL/6 J mouse strain is a little more challenging than working on OF1 strain due to (1) fewer microglial cells per brain at the same developmental stage and (2) fragility of microglia that are more prone to die due to mechanical activation (see the troubleshooting section).
Depending on the developmental stage chosen for primary microglia culture, myelination must be considered since it starts around P5. In a classical microglial culture (at P0-2), myelination is not an issue; however, in other published methods to isolate microglia, myelin is removed using Percoll gradient or anti-myelin antibodies25,26,28,29. In the present protocol, animals are euthanized at P8 when myelination has already begun; large volumes are used to rinse pellets, and the newly formed myelin can be removed without any additional procedure. This can be one troubleshooting method (see the Debris section).
The manufacturer and previously published microglial magnetic isolation used DMEM F-12 media supplemented with 10% FBS – 1% P/S25,26,27,28,29. Biber et al.42 described that microglia rarely contact serum in vivo. Indeed, the central nervous system's extracellular fluid rarely contains protein and bioactive factors29. That is why serum-free macrophage (SFM) medium + 1% P/S is used in the present protocol.
Magnetic-activated cell sorting (MACS) protocol can also isolate and culture postnatal rat microglia with some modifications25,29. Rat microglia are indeed isolated using Microbead-coated anti-rat CD11b/c. However, due to the brain size being bigger than P8 mice, only one brain per dissociation tube per column should be used. Microglia were isolated from P10-14 rat brains in previously published protocols, including the myelination removal step29. Despite the previous comments on culture medium, for rat microglial culture, F-12 media + 10% FBS and 550,000-600,000 cells per 12-well plate are recommended for downstream applications.
Troubleshooting
Dissociation: The dissociation mixture as described in Table 1 is calculated for the maximal amount of brain that can be dissociated and proceed at P8 in OF1 mice. If more brain is added, dissociation cannot be completed at the end of step 3.2, and non-dissociated brain tissue will be found. In this case, it is recommended to run the NTDK program on the tube with non-dissociated tissue and keep the other tubes at 4 °C.
Clogged strainer and/or column: As described before, the dissociation mixture is calculated for the maximal amount of brain tissue that can pass through the strainer during step 3.5. If more brain is added, the filtration may take longer, but eventually, it will pass through. It is recommended to carefully resuspend cell suspension on the top of the strainer until all cell suspension is filtered.
During step 3.12, the experimenter must pass labeled cell suspension through a column inside the magnetic field. The column can be clogged at this stage of the procedure (1) if too much brain tissue is dissociated per tube, (2) if resuspension volume is not correct, or (3) if there is some mechanically induced cell death (some DNA can be present); in that case, it is recommended to remove the visible DNA with a 200 µL tip carefully.
Mechanically induced cell death and/or activation: The protocol described here presents one main challenge: avoiding mechanical activation. To do so, it is critical to pipette gently and keep the centrifugation speed low. If not, immature microglia can die due to the mechanical stress or get activated, leading to high variability in RT-qPCR results.
Debris: In this procedure, animals are euthanized at P8 when myelination is just beginning, and therefore there is no procedure to precisely remove myelin. At this stage of development, myelin can easily be removed by centrifugation. Resuspension volumes described in this procedure are calculated to remove the maximum myelin. If not respected, myelin could appear as cell debris in culture wells. The experimenter cannot completely remove it when observed in culture wells by changing the medium on day 2. Microglial cells tend to phagocyte this debris, which may affect the microglial activation state before stimulation and thus affect experimental reproducibility.
All previously described troubleshooting (Dissociation, Clogged strainer and/or column, Mechanically induced cell death and/or activation, and debris) can lead to variability in the number of CD11b+ cells obtained when cell counting (Figure 5B). We recommend paying great attention to this to optimize the final cell yield.
Contamination: In this protocol, microglial cells are cultured in SFM medium + 1% P/S. SFM medium can easily be contaminated. Therefore, it is recommended to aliquot the medium in 50 mL tubes after adding P/S to avoid medium contamination. Moreover, all experimentation needs to be performed as much as possible under sterile conditions.
mRNA quantity/quality: Our team works on miRNA or siRNA transfections. The present protocol is highly compatible with such applications using magnetic transfection with a slight modification of the protocol; the experimenter needs to harvest 700,000 cells per 12-well plate to get high-quality mRNA for RT-qPCR. Although RNAseq is not performed in the present study, previously, our team performed mRNA extraction for RNAseq using 500,000 cells per 12-well plate48.
In conclusion, this protocol can be reproduced, and it is expected to be a new standard for isolating microglia at a juvenile developmental stage.
Divulgations
The authors have nothing to disclose.
Acknowledgements
Figures were created using BioRender. Research is funded by Inserm, Université de Paris, Horizon 2020 (PREMSTEM-874721), Fondation de France, Fondation ARSEP, Fondation pour la Recherche sur le Cerveau, Fondation Grace de Monaco, and an additional grant from Investissement d'Avenir -ANR-11-INBS-0011-NeurATRIS and Investissement d'Avenir -ANR-17-EURE-001-EUR G.E.N.E.
Materials
| Anti mouse ACSA-2 PE Vio 615 | Miltenyi Biotec | 130-116-246 | |
| Anti mouse CD11b BV421 | Sony Biotechnology | 1106255 | |
| Anti mouse CD45 BV510 | Sony Biotechnology | 1115690 | |
| Anti mouse CX3CR1 PE Cy7 | Sony Biotechnology | 1345075 | |
| Anti mouse NeuN PE | Milli-Mark | FCMAB317PE | |
| anti mouse O4 Vio Bright B515 | Miltenyi Biotec | 130-120-016 | |
| BD Cytofix/Cytoperm permeabilization kit | BD Biosciences | 554655 | |
| Bovine Serum Albumin | Miltenyi Biotec | 130-091-376 | |
| CD11b (Microglia) MicroBeads, h, m | Miltenyi Biotec | 130-093-634 | |
| Confocal microscope | Leica TCS SP8 | ||
| D-PBS (10x) | Thermo Scientific | 14200067 | |
| EDTA | Sigma-Aldrich | E1644 | |
| Falcon Cell culture 12-well plate, flat bottom + lid | Dutscher | 353043 | |
| Falcon Cell culture 96-well plate, flat bottom + lid | Dutscher | 353072 | |
| Falcon tubes 50 mL | Dutscher | 352098 | |
| Fc blocking reagent (Mouse CD16/32) | BD Biosciences | 553142 | |
| Fluorescence microscope | Nikon ECLIPSE TE300 | ||
| gentleMACS C Tubes (4 x 25 tubes) | Miltenyi Biotec | 130-096-334 | |
| gentleMACS Octo Dissociator with Heaters | Miltenyi Biotec | 130-096-427 | |
| Hanks' Balanced Salt Solution (HBSS) +CaCl2 +MgCl2 10x | Thermo Scientific | 14065049 | |
| Hanks' Balanced Salt Solution (HBSS) -CaCl2 -MgCl2 10x | Thermo Scientific | 14185045 | |
| iQ SYBR Green Supermix | Bio-rad | 1725006CUST | |
| Iscript c-DNA synthesis | Bio-rad | 1708890 | |
| Latex beads, amine-modified polystyrene, fluorescent red | Sigma-Aldrich | L2776-1mL | |
| Lipopolysaccharides (LPS) from Escherichia coli O55:B5 | Sigma-Aldrich | L2880 | |
| Macrophage-SFM serum-free medium | Thermo Scientific | 12065074 | |
| MACS BSA Stock Solution | Miltenyi Biotec | 130-091-376 | |
| MACS SmartStrainers (70 μm), 4 x 25 pcs | Miltenyi Biotec | 130-110-916 | |
| Mouse IgG1 PE | Millipore | MABC002H | |
| Mouse IgG2a PE Cy7 | Sony Biotechnology | 2601265 | |
| Mouse IL1 beta | Miltenyi Biotec | 130-101-684 | |
| Multi-24 Column Blocks | Miltenyi Biotec | 130-095-691 | |
| MultiMACS Cell24 Separator | Miltenyi Biotec | ||
| Neural Tissue Dissociation Kit – Papain | Miltenyi Biotec | 130-092-628 | |
| Nucleocounter NC-200 | Chemometec | ||
| Nucleospin RNA Plus XS | Macherey Nagel | 740990.5 | |
| Nun EZFlip Top Conical Centrifuge Tubes | Thermo Scientific | 362694 | |
| OPTILUX Petri dish – 100 x 20 mm | Dutscher | 353003 | |
| Pénicilline-streptomycine (10 000 U/mL) | Thermo Scientific | 15140122 | |
| Rat IgG2b, k BV421 | BD Biosciences | 562603 | |
| Rat IgG2b, k BV510 | Sony Biotechnology | 2603230 | |
| REA control (S) PE vio 615 | Miltenyi Biotec | 130-104-616 | |
| REA control (S) Vio Bright B515 | Miltenyi Biotec | 130-113-445 | |
| Recombinant Mouse IFN-gamma Protein | R&D System | 485-MI | |
| Recombinant Mouse IL-10 Protein | R&D System | 417-ML | |
| Recombinant Mouse IL-4 Protein | R&D System | 404-ML | |
| RIPA Buffer | Sigma-Aldrich | R0278 | |
| Viability probe (FVS780) | BD Biosciences | 565388 |
References
- Kierdorf, K., et al. Microglia emerge from erythromyeloid precursors via Pu.1- and Irf8-dependent pathways. Nature Neuroscience. 16 (3), 273-280 (2013).
- Wright-Jin, E. C., Gutmann, D. H. Microglia as dynamic cellular mediators of brain function. Trends in Molecular Medicine. 25 (11), 967-979 (2019).
- Hellstrom Erkenstam, N., et al. Temporal characterization of microglia/macrophage phenotypes in a mouse model of neonatal hypoxic-ischemic brain injury. Frontiers in Cellular Neuroscience. 10, 286 (2016).
- Chhor, V., et al. Role of microglia in a mouse model of paediatric traumatic brain injury. Brain, Behavior, and Immunity. 63, 197-209 (2017).
- Van Steenwinckel, J., et al. Decreased microglial Wnt/beta-catenin signalling drives microglial pro-inflammatory activation in the developing brain. Brain. 142 (12), 3806-3833 (2019).
- Chhor, V., et al. Characterization of phenotype markers and neuronotoxic potential of polarised primary microglia in vitro. Brain, Behavior, and Immunity. 32, 70-85 (2013).
- Di Pietro, P., et al. Bisphenol A induces DNA damage in cells exerting immune surveillance functions at peripheral and central level. Chemosphere. 254, 126819 (2020).
- Roque, P. J., Dao, K., Costa, L. G. Microglia mediate diesel exhaust particle-induced cerebellar neuronal toxicity through neuroinflammatory mechanisms. Neurotoxicology. 56, 204-214 (2016).
- Yun, H. S., Oh, J., Lim, J. S., Kim, H. J., Kim, J. S. Anti-inflammatory effect of wasp venom in BV-2 microglial cells in comparison with bee venom. Insects. 12 (4), 297 (2021).
- Nair, S., et al. Lipopolysaccharide-induced alteration of mitochondrial morphology induces a metabolic shift in microglia modulating the inflammatory response in vitro and in vivo. Glia. 67 (6), 1047-1061 (2019).
- Fleiss, B., et al. The anti-inflammatory effects of the small molecule pifithrin-micro on BV2 microglia. Developmental Neuroscience. 37 (4-5), 363-375 (2015).
- Dean, J. M., et al. Microglial MyD88 signaling regulates acute neuronal toxicity of LPS-stimulated microglia in vitro. Brain, Behavior, and Immunity. 24 (5), 776-783 (2010).
- Tang, Y., Wolk, B., Nolan, R., Scott, C. E., Kendall, D. A. Characterization of subtype selective cannabinoid CB2 receptor agonists as potential anti-inflammatory agents. Pharmaceuticals (Basel). 14 (4), 378 (2021).
- Liu, C. P., et al. miR146a reduces depressive behavior by inhibiting microglial activation. Molecular Medicine Reports. 23 (6), 463 (2021).
- Aquino, G. V., Dabi, A., Odom, G. J., Zhang, F., Bruce, E. D. Evaluating the endothelial-microglial interaction and comprehensive inflammatory marker profiles under acute exposure to ultrafine diesel exhaust particles in vitro. Toxicology. 454, 152748 (2021).
- You, J. E., Jung, S. H., Kim, P. H. The effect of Annexin A1 as a potential new therapeutic target on neuronal damage by activated microglia. Molecules and Cells. 44 (4), 195-206 (2021).
- Xie, Z., et al. By regulating the NLRP3 inflammasome can reduce the release of inflammatory factors in the co-culture model of tuberculosis H37Ra strain and rat microglia. Frontiers in Cellular and Infection Microbiology. 11, 637769 (2021).
- Ogunrinade, F. A., et al. Zanthoxylum zanthoxyloides inhibits lipopolysaccharide- and synthetic hemozoin-induced neuroinflammation in BV-2 microglia: roles of NF-kappaB transcription factor and NLRP3 inflammasome activation. Journal of Pharmacy and Pharmacology. 73 (1), 118-134 (2021).
- Fernandez-Arjona, M. D. M., Leon-Rodriguez, A., Lopez-Avalos, M. D., Grondona, J. M. Microglia activated by microbial neuraminidase contributes to ependymal cell death. Fluids Barriers CNS. 18 (1), 15 (2021).
- Du, S., et al. Primary microglia isolation from postnatal mouse brains. Journal of Visualized Experiments: JoVE. (168), e62237 (2021).
- Boccazzi, M., et al. The immune-inflammatory response of oligodendrocytes in a murine model of preterm white matter injury: the role of TLR3 activation. Cell Death & Disease. 12 (2), 166 (2021).
- Timmerman, R., Burm, S. M., Bajramovic, J. J. An overview of in vitro methods to study microglia. Frontiers in Cellular Neuroscience. 12, 242 (2018).
- Nikodemova, M., Watters, J. J. Efficient isolation of live microglia with preserved phenotypes from adult mouse brain. Journal of Neuroinflammation. 9, 147 (2012).
- Bennett, M. L., et al. New tools for studying microglia in the mouse and human CNS. Proceedings of the National Academy of Sciences of the United States of America. 113 (12), 1738-1746 (2016).
- Bohlen, C. J., Bennett, F. C., Bennett, M. L. Isolation and culture of microglia. Current Protocols in Immunology. 125 (1), 70 (2019).
- Schroeter, C. B., et al. One brain-all cells: A comprehensive protocol to isolate all principal CNS-resident cell types from brain and spinal cord of adult healthy and EAE mice. Cells. 10 (3), 651 (2021).
- Harms, A. S., Tansey, M. G. Isolation of murine postnatal brain microglia for phenotypic characterization using magnetic cell separation technology. Methods in Molecular Biology. 1041, 33-39 (2013).
- Pan, J., Wan, J. Methodological comparison of FACS and MACS isolation of enriched microglia and astrocytes from mouse brain. Journal of Immunological Methods. 486, 112834 (2020).
- Montilla, A., Zabala, A., Matute, C., Domercq, M. Functional and metabolic characterization of microglia culture in a defined medium. Frontiers in Cellular Neuroscience. 14, 22 (2020).
- Krishnan, M. L., et al. Integrative genomics of microglia implicates DLG4 (PSD95) in the white matter development of preterm infants. Nature Communications. 8 (1), 428 (2017).
- Bokobza, C., et al. miR-146b protects the perinatal brain against microglia-induced hypomyelination. Annals of Neurology. 91 (1), 48-65 (2021).
- Villapol, S., et al. Early sex differences in the immune-inflammatory responses to neonatal ischemic stroke. International Journal of Molecular Sciences. 20 (15), 3809 (2019).
- Rosiewicz, K. S., et al. Comparison of RNA isolation procedures for analysis of adult murine brain and spinal cord astrocytes. Journal of Neuroscience Methods. 333, 108545 (2020).
- Fleiss, B., et al. Microglia-mediated neurodegeneration in perinatal brain injuries. Biomolecules. 11 (1), 99 (2021).
- Pawelec, P., Ziemka-Nalecz, M., Sypecka, J., Zalewska, T. The Impact of the CX3CL1/CX3CR1 axis in neurological disorders. Cells. 9 (10), 2277 (2020).
- Reynolds, R., Cenci di Bello, I., Dawson, M., Levine, J. The response of adult oligodendrocyte progenitors to demyelination in EAE. Progress in Brain Research. 132, 165-174 (2001).
- Duan, W., et al. Novel insights into NeuN: From neuronal marker to splicing regulator. Molecular Neurobiology. 53 (3), 1637-1647 (2016).
- Kantzer, C. G., et al. Anti-ACSA-2 defines a novel monoclonal antibody for prospective isolation of living neonatal and adult astrocytes. Glia. 65 (6), 990-1004 (2017).
- Lee, S., Lee, D. K. What is the proper way to apply the multiple comparison test. Korean Journal of Anesthesiology. 71 (5), 353-360 (2018).
- Chao, C. C., Hu, S., Molitor, T. W., Shaskan, E. G., Peterson, P. K. Activated microglia mediate neuronal cell injury via a nitric oxide mechanism. Journal of Immunology. 149 (8), 2736-2741 (1992).
- Boje, K. M., Arora, P. K. Microglial-produced nitric oxide and reactive nitrogen oxides mediate neuronal cell death. Brain Research. 587 (2), 250-256 (1992).
- Biber, K., Owens, T., Boddeke, E. What is microglia neurotoxicity (Not). Glia. 62 (6), 841-854 (2014).
- Biber, K., Neumann, H., Inoue, K., Boddeke, H. W. Neuronal ‘On’ and ‘Off’ signals control microglia. Trends in Neurosciences. 30 (11), 596-602 (2007).
- Ransohoff, R. M., Cardona, A. E. The myeloid cells of the central nervous system parenchyma. Nature. 468 (7321), 253-262 (2010).
- Boucsein, C., Kettenmann, H., Nolte, C. Electrophysiological properties of microglial cells in normal and pathologic rat brain slices. European Journal of Neuroscience. 12 (6), 2049-2058 (2000).
- Beutner, C., et al. Unique transcriptome signature of mouse microglia. Glia. 61 (9), 1429-1442 (2013).
- Schmid, C. D., et al. Differential gene expression in LPS/IFNgamma activated microglia and macrophages: in vitro versus in vivo. Journal of Neurochemistry. 109, 117-125 (2009).
- Srivastava, P. K., et al. A systems-level framework for drug discovery identifies Csf1R as an anti-epileptic drug target. Nature Communications. 9 (1), 3561 (2018).

