Comparison of Methods for Isolating Entomopathogenic Fungi from Soil Samples
Summary
Entomopathogenic fungal colonies are isolated from tropical soil samples using Tenebrio bait, Galleria bait, as well as selective artificial medium, i.e., potato dextrose agar enriched with yeast extract supplemented with chloramphenicol, thiabendazole, and cycloheximide (CTC medium).
Abstract
The goal of the present study is to compare the effectiveness of using insect baits versus artificial selective medium for isolating entomopathogenic fungi (EPF) from soil samples. The soil is a rich habitat for microorganisms, including EPF particularly belonging to the genera Metarhizium and Beauveria, which can regulate arthropod pests. Biological products based on fungi are available in the market mainly for agricultural arthropod pest control. Nevertheless, despite the high endemic biodiversity, only a few strains are used in commercial bioproducts worldwide. In the present study, 524 soil samples were cultured on potato dextrose agar enriched with yeast extract supplemented with chloramphenicol, thiabendazole, and cycloheximide (CTC medium). The growth of fungal colonies was observed for 3 weeks. All Metarhizium and Beauveria EPF were morphologically identified at the genus level. Additionally, some isolates were molecularly identified at the species level. Twenty-four out of these 524 soil samples were also surveyed for EPF occurrence using the insect bait method (Galleria mellonella and Tenebrio molitor). A total of 51 EPF strains were isolated (41 Metarhizium spp. and 10 Beauveria spp.) from the 524 soil samples. All fungal strains were isolated either from croplands or grasslands. Of the 24 samples selected for comparison, 91.7% were positive for EPF using Galleria bait, 62.5% using Tenebrio bait, and 41.7% using CTC. Our results suggested that using insect baits to isolate the EPF from the soil is more efficient than using the CTC medium. The comparison of isolation methods in addition to the identification and conservation of EPF has a positive impact on the knowledge about biodiversity. The improvement of EPF collection supports scientific development and technological innovation.
Introduction
Soil is the source of several microorganisms, including entomopathogenic fungi (EPF). This particular group of fungi is recognized by their ability to colonize and often kill arthropod hosts, especially insects1. After isolation, characterization, selection of virulent strains, and registration, EPF are mass-produced for arthropod-pest control, which supports their economical relevance2. Accordingly, the isolation of EPF is considered the first step to the development of a biopesticide. Beauveria spp. (Hypocreales: Cordycipitaceae) and Metarhizium spp. (Hypocreales: Clavicipitaceae) are the most common fungi used for arthropod-pest control3. EPF have been successfully isolated from soil, arthropods with visible mycosis, colonized plants, and plant rhizosphere4,5.
Isolation of EPF can also be useful to study the diversity, distribution, and ecology of this particular group. Recent literature reported that the use of EPF is underestimated, citing several unconventional applications of EPF such as their capacity to improve plant growth4, to remove toxic contaminants from the soil, and to be used in medicine6. The present study aims to compare the efficiency of isolating EPF from soil using insect baits versus artificial culture medium7,8,9. The use of Galleria mellonella L. (Lepidoptera: Phyralidae) as an insect bait in the context of EPF isolation has been well accepted. These larvae are used worldwide by the scientific community as an experimental model to study host-pathogen interactions10,11. Tenebrio molitor L. (Coleoptera: Tenebrionidae) larva is considered another insect model for studies involving virulence and for isolation of EPF since this insect is easy to rare in the laboratory at a low cost7,12.
Culture-independent methods such as using a variety of PCR techniques can be applied to detect and quantify EPF on their substrates, including soil13,14. Nevertheless, to properly isolate these fungal colonies, their substrate should be cultured onto a selective artificial medium9, or the fungi present in the samples can be baited using sensitive insects15. On one hand, CTC is a dodine-free artificial medium that consists of potato dextrose agar enriched with yeast extract supplemented with chloramphenicol, thiabendazole, and cycloheximide. This medium was developed by Fernandes et al.9 to maximize the recovery of naturally occurring Beauveria spp. and Metarhizium spp. from the soil. On the other hand, G. mellonella and T. molitor larvae can also to be successfully used as baits to obtain EPF isolates from the soil. Nevertheless, according to Sharma et al.15, fewer studies reported the concomitant use and comparison of these two bait insects. Portuguese vineyards soils exhibited significant recoveries of Metarhizium robertsii (Metscn.) Sorokin using T. molitor larvae in comparison to G. mellonella larvae; in contrast, Beauveria bassiana (Bals. -Criv.) Vuill isolation was linked to the use of G. mellonella baits15. Therefore, the decision on which EPF isolation method to use (i.e., G. mellonella-bait, T. molitor-bait or CTC medium) should be considered according to the study's goal and the laboratory infrastructure. The goal of the present study is to compare the effectiveness of using insect baits versus artificial selective medium for isolating EPF from soil samples.
Protocol
As the present study accessed Brazilian genetic heritage, the research was registered at the National System for the Management of Genetic Heritage and Associated Traditional Knowledge (Sisgen) under the code AA47CB6.
1. Soil sampling
- Collect 800 g of soil (with or without incident secondary plant roots) to a depth of 10 cm using a small shovel. Store them in polypropylene bags at room temperature until the start of the experiment.
NOTE: Small roots can also be collected as EPF are reported to have rhizosphere competence. The faster the processing of the samples, the better because the fungal spores may be less viable over time. In the present study, samples were analyzed no more than 7 days after the collection. - Use a GPS to identify the location of the collected samples in latitude and longitude and classify the collected area according to the type of soil (for example, grasslands, native rainforest, lakeshores, or croplands).
2. Isolation methods for entomopathogenic fungi
- Isolation using CTC selective artificial medium.
- To prepare the CTC medium [potato dextrose agar plus yeast extract (PDAY) supplemented with 0.5 g/L of chloramphenicol, 0.001 g/L of thiabendazole, and 0.25 g/L of cycloheximide9], weigh all the reagents individually, mix them in distilled water, and sterilize the medium in autoclave. In a biosafety cabinet, plate 23 mL of the medium into 60 mm x 15 mm Petri plates.
CAUTION: While weighing CTC reagents, use a lab coat, mask, gloves, and goggles because cycloheximide and chloramphenicol are toxic. - Weigh 0.35 ± 0.05 g of each soil sample (with or without roots) and place it in a 1.5 mL microtube.
- In a biosafety cabinet, add 1 mL of sterile 0.01% (vol/vol) polyoxyethylene sorbitan monooleate aqueous suspension to the microtube containing soil and vortex for 30 s.
- Remove 50 µL of the supernatant and pipette it onto the center of Petri plates with CTC medium. Homogenously disperse the suspensions onto the surface of the medium using a sterile Drigalski spatula (6 mm in diameter).
NOTE: At least three replicates for each soil sample should be prepared. - Incubate the plates in climate chambers (25 ± 1 °C, relative humidity ≥80%) in the dark and observe the growth of fungal colonies after 7, 14, and 21 days of incubation.
- Observe the macromorphology and micromorphology of the fungal colonies seeking EPF. Transfer the EPF cultures to potato dextrose agar medium plus 0.05% chloramphenicol (PDAC) until pure cultures are obtained.
NOTE: Use the description keys presented below in step 3 for the identification of EPF colonies.
- To prepare the CTC medium [potato dextrose agar plus yeast extract (PDAY) supplemented with 0.5 g/L of chloramphenicol, 0.001 g/L of thiabendazole, and 0.25 g/L of cycloheximide9], weigh all the reagents individually, mix them in distilled water, and sterilize the medium in autoclave. In a biosafety cabinet, plate 23 mL of the medium into 60 mm x 15 mm Petri plates.
- Isolation using insect baits
- Use surface-disinfected G. mellonella and T. molitor late-stage larvae. Immerse the larvae into 0.5% sodium hypochlorite for 1 min for sterilization. Wash the larvae twice using sterile water.
NOTE: G. mellonella larvae from the fourth stage were used in the present study. T. molitor larval stages were not standardized. - Use plastic pots to assemble the baits. Add 250 g of collected soil to each plastic pot (98 mm width x 47 mm height x 142 mm length). Separate 15 larvae of each species (T. molitor and G. mellonella) and deposit five larvae per plastic pot. Store the pots at 25 ± 1 °C and relative humidity ≥ 80% in the dark.
NOTE: Drill 10 small holes (2 mm in diameter) in the pot lids to allow ventilation. A sharp heated iron device can be used to drill the holes. - Homogenize the soil every other day to allow maximum contact of larvae with soil.
NOTE: Moisture is important to support the fungal infection of larvae. To maintain moisture in the soil, spray sterile distilled water on the soil surface whenever necessary. Do not soak the soil sample in water. - Analyze the pots daily seeking dead insects.
NOTE: Observe the remaining larvae in the colony daily for invertebrate pathological signs to make sure the insects are not infected. As an alternative, control pots with sterile soil can be included in the study to check the health status of the insect larvae. - Remove dead insects and superficially sterilize them with 0.5% sodium hypochlorite for 1 min. Place the sterile insects in a humid chamber (relative humidity ≥ 80%) at 25 ± 1 °C for 7 days to favor the exteriorization of entomopathogenic fungi (mycosis).
- Upon mycosis, harvest the conidia from the insect surface. Use a microbiological loop to place the conidia on PDAC medium under a stereoscopic microscope. As an alternative, place the whole infected larvae on the PDAC medium. Incubate the culture plates in a climate chamber at 25 ± 1 °C and relative humidity ≥ 80%.
- Observe the macromorphology and micromorphology of the fungal colonies on the plates to confirm the identity of EPF. Repeat culturing on PDAC until pure fungal colonies are obtained.
NOTE: Use the description keys presented below in step 3 for the identification of EPF colonies.
- Use surface-disinfected G. mellonella and T. molitor late-stage larvae. Immerse the larvae into 0.5% sodium hypochlorite for 1 min for sterilization. Wash the larvae twice using sterile water.
3. Identification of EPF (Metarhizium spp. and Beauveria spp.)
- Analyze the macromorphological characteristics of the fungal cultures on the plates (i.e., surface and reverse of colonies, their shape, edge, growth rate, color, texture, diffusible pigment, exudates, and aerial conidia) after 14 days at 25 ± 1 °C and relative humidity ≥ 80%.
- Transfer the aerial conidia to slide cultures (microculture technique)16 for 3 days at 25 ± 1 °C and relative humidity ≥ 80% and stain with lactophenol blue to observe the microscopic features (i.e., arrangement of conidia, conidiophores, shape, and size of conidia)17,18,19,20.
- Observe the microscopic fungal structures at 400x using an optical microscope to confirm the EPF identification.
NOTE: Morphological keys for EPF are described in the reports by Bischoff et al., Rehner et al., Seifert et al., and Humber17,18,19,20. The macro and micromorphology of fungal colonies are the most frequent criteria used to identify filamentous fungi at the genus level. Depending on the genus of the EPF, these morphological characteristics will change. Humber20 presents an identification key to major genera of fungal entomopathogens. Metarhizium spp. colonies, for example, are usually circular, powdery, exhibiting varying shades of green, and can present exudate. Microscopically, these colonies have conidiogenous cells apical on broadly branched, densely intertwined conidiophores forming a compact hymenium, and cylindrical to ellipsoid conidia in parallel chains forming columns or plate-like masses. Beauveria spp. colonies are usually white, powdery, or cotton-like. They exhibit conidiogenous cells with a dilated basal portion extending apically in a zigzag direction. Beauveria conidiophores form dense clusters of globe-shaped conidia. Molecular analyses are needed for the identification of EPF at the species level. - Perform molecular analyses on the isolates for taxonomic identification at the species level. For the EPF strains isolated in this study, namely, Metarhizium spp. and Beauveria spp., perform molecular analyses based on the reports of Bischoff et al.17 and Rehner et al.18.
- After confirming the isolates to be EPF, deposit the isolates in a collection of fungal cultures. In the present study, the isolates were deposited in the entomopathogenic fungal cultures collection from the Laboratory of Microbial Control (LCM) at the Federal Rural University of Rio de Janeiro.
Representative Results
A total of 524 soil samples were collected from grassland: livestock pasture (165 samples), native tropical forest (90 samples), lakeside (42 samples), and cultivated/cropland (227 samples) between 2015 and 2018 in the Rio de Janeiro State, Brazil. Details of geographic coordinates of samples positive for EPF are given in Supplementary Table 1.
Of the 524 soil samples, 500 samples were analyzed only using CTC medium, and 24 samples were concomitantly analyzed using three forms of isolation (Galleria-bait, Tenebrio-bait, and the selective CTC culture medium), so the relative efficiency of these methods could be evaluated. A total of 51 EPF strains were isolated from 524 samples (41 Metarhizium spp. and 10 Beauveria spp.) (Figure 1). Micromorphological characteristics of some isolates are shown in Figure 2. All fungal strains were isolated from grassland or cropland (Supplementary Table 1). The results revealed that Metarhizium spp. is more prevalent than Beauveria spp. (Supplementary Table 1). Nine of the Metarhizium isolates (LCM S01 to LCM S09) were molecularly identified using the ef1-a (eukaryotic translation elongation factor 1-alpha) gene21. Of these, seven isolates (LCM S01-LCM S06 and LCM S08) were identified as Metarhizium anisopliae sensu stricto while two isolates (LCM S07 and LCM S09) were identified as Metarhizium pingshaense21.
The occurrence of EPF (% of positive EPF samples) in the 24 soil samples studied using the three different methods of isolation is shown in Table 1. Recovery rates of EPF were analyzed by chi-square test. As shown in Table 1, Galleria bait proved to be more efficient in the isolation of EPF (91.7% (22/24) of positive samples) followed by T. molitor bait (62.5% (15/24) of EPF positive samples) and CTC medium (41.7% (14/24) of EPF positive samples). These 24 soil samples showed no recovery of Beauveria spp., but only Metarhizium.

Figure 1: Entomopathogenic fungal colonies of strains isolated from soil samples. Colonies were cultivated on CTC artificial medium. (1) Petri plate exhibiting fungal colonies from soil samples 14 days after incubation on CTC selective medium before pure cultures are obtained; (2-42) Pure Metarhizium spp. colonies; (43-52) Pure Beauveria spp. colonies. Please click here to view a larger version of this figure.
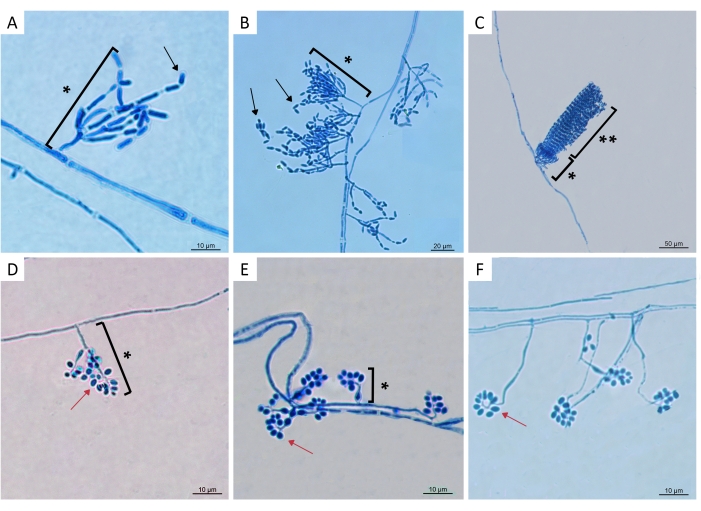
Figure 2: Micromorphological characteristics of entomopathogenic fungi isolated from soil samples. Colonies were incubated for 3 days on potato dextrose agar at 25 ± 1 °C and relative humidity ≥ 80%. The microscope slide was stained with lactophenol blue solution. Images show conidiophores and conidia of (A) Metarhizium anisopliae sensu stricto (s.s) isolate LCM S01; (B) Metarhizium anisopliae s.s. isolate LCM S03; (C) Metarhizium sp. isolate LCM S27; (D–F) Beauveria spp. isolates LCM S23, LCM S24, and LCM S20, respectively. All strains represented here were isolated using the CTC medium. LCM S27 was also recovered from soil using insect baits. * Conidiophores and conidia. ** Conidial chains show the characteristic side-by-side placement of Metarhizium spores in adjacent chains. Black arrows indicate Metarhizium cylindrical to ellipsoid conidia. Red arrows indicate Beauveria globe-shaped conidia. Please click here to view a larger version of this figure.
| Method of isolation | Entomopathogenic fungi* | χ2** | |
| Positive | Negative | ||
| Galleria-bait | 91.7% (22/24) | 8.3% (2/24) | 13.4 |
| Tenebrio-bait | 62.5% (15/24) | 37.2% (9/24) | |
| CTC selective medium | 41.7% (10/24) | 58.3% (14/24) | |
| * Only Metarhizium spp. were isolated | |||
| ** Chi-square analysis, DF2. P = 0.0013 | |||
Table 1: Occurrence of entomopathogenic fungi (% of positive samples) in 24 soil samples using different isolation methods.
Supplementary Table 1: Geographical coordinates, isolation method, code, year of collection, and land-use types of samples positive for entomopathogenic fungi. Please click here to download this Table.
Discussion
Natural and agricultural soil habitats are typical environments for EPF22 and an excellent natural reservoir. In the present study, two methods of EPF isolation using insect baits versus selective medium were addressed. The first step for isolation is the collection of the soil samples. Proper storage and identification of soil samples are crucial. Information on the latitude, longitude, soil type, and biome is essential for studies involving epidemiological, modeling, and geospatial subjects23,24. After collection, it is recommended that the samples are processed as soon as possible (preferably within 7 days) because the viability of conidia in these soil samples can eventually decrease. Critical steps in the EPF isolation using CTC include: a) investigation of CTC plates 1 and 2 weeks after incubation (the first weeks are critical because, at later stages, other fungal colonies can narrow EPF development), and b) accurately identifying EPF colonies based on their macromorphology and micromorphology. For isolation using insect baits, it is essential to keep the soil sample humid but not soak it in water.
The results reported by several studies have led to an interpretation that M. anisopliae is more common in cultivated soils than natural ecosystems8,25,26. Differences in the distribution and occurrence of these fungi can occur. In the present study, all strains were isolated either from cultivated soil (crops) or grasslands, and there was a predominance of Metarhizium spp. over Beauveria spp. It is suggested that cultivation practices and the high content of organic matter favor the presence of saprophytic fungi in the soil27. Accordingly, effective isolation techniques seeking EPF should consider reducing fungal contaminants.
Selective artificial media are commonly used for isolation because they are easy to use and have proven efficient in isolating entomopathogenic fungi, mainly Metarhizium spp. and Beauveria spp.28. These selective media use specific chemicals to reduce the growth of contaminants. In the 1980s and 1990s, the fungicide dodine became a widely used selective medium to isolate Metarhizium spp. and Beauveria spp.29,30. Although these artificial media are effective, some EPF species such as Metarhizium acridum can be susceptible to dodine31. That is why the dodine-free CTC medium was chosen in the present study. According to Fernandes et al.9, CTC was developed to maximize the isolation of naturally occurring entomopathogenic fungi, including M. acridum. Using a selective medium rather than insect baits in the isolation of EPF is convenient because the former requires less space in the sample processing. The main disadvantage in CTC use relies on the fact that some of its components (i.e., cycloheximide and chloramphenicol) are toxic, so the use of personal protection equipment is mandatory.
As observed in the present study, a higher percentage of positive samples has been reported with insect baits as compared to artificial selective media for isolation of EPF15,32,33,34,35. The use of insect baits is considered a low-cost and high-efficiency alternative in the search for new EPF. Despite this, there are disadvantages associated with the use of insect baits over selective media. As the amount of soil to analyze using insects is higher, it is also necessary to have more physical space to store the samples and incubate the pots. The acquisition of insects can also be a limitation. In Brazil, for example, G. mellonella is not commercially available, so it is necessary to establish a colony in the lab to use this insect as bait. It is essential to keep the salubrity of the insects' colonies, avoiding natural infection by EPF. An EPF infection in the colony can make the isolation results unreliable. Therefore, one has to observe the remaining larvae in the colony seeking invertebrate pathological signs. As an alternative, control pots with sterile soil can be included in the study to check the health status of the insect larvae.
Seeking new fungal isolates with outstanding biocontrol traits is crucial to increase the effectiveness of fungi in arthropod-pest control. Fungi isolated from soil can be well adapted to growing in this environment22, and they are likely to have high field persistence, which is an essential characteristic of successful EPF in pest control21. Accordingly, locally isolated EPF can improve the biological control of local pests because of their geographic and temporal congruence, increasing the chances of success and reducing the environmental impacts otherwise caused by the application of synthetic insecticides.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This study was financed in part by the Coordenacão de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) from Brazil, finance code 001, Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) (project number E-26/010.001993/2015), and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) from Brazil.
Materials
| Autoclave | Phoenix Luferco | 9451 | |
| Biosafety cabinet | Airstream ESCO | AC2-4E3 | |
| Chloramphenicol | Sigma-Aldrich | C0378 | |
| Climate chambers | Eletrolab | EL212/3 | |
| Coverslip | RBR | 3871 | |
| Cycloheximide | Sigma-Aldrich | C7698 | |
| Drigalski spatula | Marienfeld | 1800024 | |
| GPS app | Geolocation app | 2.1.2005 | |
| Lactophenol blue solution | Sigma-Aldrich | 61335 | |
| Microscope | Zeiss Axio star plus | 1169 149 | |
| Microscope camera | Zeiss Axiocam 105 color | 426555-0000-000 | |
| Microscope softwere | Zen lite Zeiss 3.0 | ||
| Microscope slide | Olen | k5-7105-1 | |
| Microtube | BRAND | Z336769-1PAK | |
| Petri plates | Kasvi | K30-6015 | |
| Pipette tip | Vatten | VT-230-200C/VT-230-1000C | |
| Pippette | HTL – Labmatepro | LMP 200 / LMP 1000 | |
| Plastic pots | Prafesta descartáveis | 8314 | |
| Polypropylene bags | Extrusa | 38034273/5561 | |
| Potato dextrose agar | Kasvi | K25-1022 | |
| Prism software 9.1.2 | Graph Pad | ||
| Shovel | Tramontina | 77907009 | |
| Tenebrio mollitor | Safari | QP98DLZ36 | |
| Thiabendazole | Sigma-Aldrich | T8904 | |
| Tween 80 | Vetec | 60REAVET003662 | |
| Vortex | Biomixer | QL-901 | |
| Yeast extract | Kasvi | K25-1702 |
References
- Roberts, D. W., St. Leger, R. J. Metarhizium spp., cosmopolitan insect-pathogenic fungi: Mycological aspects. Advances in Applied Microbiology. 54, 1-70 (2004).
- do Nascimento Silva, J., et al. New cost-effective bioconversion process of palm kernel cake into bioinsecticides based on Beauveria bassiana and Isaria javanica. Applied Microbiology and Biotechnology. 102 (6), 2595-2606 (2018).
- Faria, M. R., Wraight, S. P. Mycoinsecticides and Mycoacaricides: A comprehensive list with worldwide coverage and international classification of formulation types. Biological Control. 43 (3), 237-256 (2007).
- Vega, F. V. The use of fungal entomopathogens as endophytes in biological control: a review. Applied Mycology. 110 (1), 4-30 (2018).
- Sharma, L., et al. Advances in entomopathogen isolation: A case of bacteria and fungi. Microorganisms. 9 (1), 1-28 (2021).
- Litwin, A., Nowak, M., Różalska, S. Entomopathogenic fungi: unconventional applications. Reviews in Environmental Science and Bio/Technology. 19, 23-42 (2020).
- Kim, J. C., et al. Tenebrio molitor-mediated entomopathogenic fungal library construction for pest management. Journal of Asia-Pacific Entomology. 21 (1), 196-204 (2018).
- Meyling, N., Eilenberg, J. Ocurrence and distribution of soil borne entomopathogenic fungi within a single organic agroecosystem. Agriculture, Ecosystems and Environment. 113 (1), 336-341 (2006).
- Fernandes, E. K. K., Keyser, C. A., Rangel, D. E. N., Foster, R. N., Roberts, D. W. CTC medium: A novel dodine-free selective medium for isolating entomopathogenic fungi, especially Metarhizium acridum, from soil. Biological Control. 54 (3), 197-205 (2010).
- Ortiz-Urquiza, A., Keyhani, N. O. Molecular genetics of Beuveria bassiana infection of insects. Advantages in Genetics. 94, 165-249 (2016).
- Pereira, M. F., Rossi, C. C., Silva, G. C., Rosa, J. N., Bazzolli, M. S. Galleria mellonella as infection model: an in depth look at why it works and practical considerations for successful application. Pathogens and Disease. 78 (8), (2020).
- Souza, P. C., et al. Tenebrio molitor (Coleoptera: Tenebrionidae) as an alternative host to study fungal infections. Journal of Microbiological Methods. 118, 182-186 (2015).
- Canfora, L., et al. Development of a method for detection and quantification of B. brongniartii and B. bassiana in soil. Scientific Reports. 6, 22933 (2016).
- Garrido-Jurado, I., et al. Transient endophytic colonization of melon plants by entomopathogenic fungi after foliar application for the control of Bemisia tabaci Gennadius (Hemiptera: Aleyrodidae). Journal of Pest Science. 90, 319-330 (2016).
- Sharma, L., Oliveira, I., Torres, L., Marques, G. Entomopathogenic fungi in Portuguese vineyards soils: suggesting a ‘Galleria-Tenebrio-bait method’ as bait-insects Galleria and Tenebrio significantly underestimate the respective recoveries of Metarhizium (robertsii) and Beauveria (bassiana). MycoKeys. 38, 1-23 (2018).
- Riddell, R. W. Permanent stained mycological preparations obtained by slide culture. Mycologia. 42 (2), 265-270 (1950).
- Bischoff, J., Rehner, S. A., Humber, R. A. A multilocus phylogeny of the Metarhizium anisopliae lineage. Mycologia. 101 (4), 512-530 (2009).
- Rehner, S. A., et al. Phylogeny and systematics of the anamorphic, entomopathogenic genus Beauveria. Mycologia. 103 (5), 1055-1073 (2011).
- Seifert, K. A., Gams, W., Seifert, K. A., Morgan-Jones, G., Gams, W., Kendrick, B. Anamorphs of Clavicipitaceae, Cordycipitaceae and Ophiocordycipitaceae. The Genera of Hyphomycetes. CBS Biodiversity Series. CBS-KNAW Fungal Biodiversity Centre. 9, 903-906 (2011).
- Humber, R. A., Lacey, L. A. Identification of entomopathogenic fungi. Manual of Techniques in Invertebrate Pathology., 2nd ed. , 151-187 (2012).
- Mesquita, E., et al. Efficacy of a native isolate of the entomopathogenic fungus Metarhizium anisopliae against larval tick outbreaks under semifield conditions. BioControl. 65 (3), 353-362 (2020).
- St Leger, R. J. Studies on adaptations of Metarhizium anisopliae to life in the soil. Journal of Invertebrate Pathology. 98 (3), 271-276 (2008).
- Mar, T. T., Suwannarach, N., Lumyong, S. Isolation of entomopathogenic fungi from Nortern Thailand and their production in cereal grains. World Journal of Microbiology and Biotechnology. 28 (12), 3281-3291 (2012).
- Rocha, L. F. N., Inglis, P. W., Humber, R. A., Kipnis, A., Luz, C. Occurrence of Metarhizium spp. in central Brazilian soils. Journal of Basic Microbiology. 53 (3), 251-259 (2013).
- Quesada-Moraga, E., Navas-Cortés, J. A., Maranhao, E. A. A., Ortiz-Urquiza, A., Santiago-Álvarez, C. Factors affecting the occurrence and distribution of entomopathogenic fungi in natural and cultivated soils. Mycological Research. 111 (8), 947-966 (2007).
- Mora, M. A. E., Rouws, J. R. C., Fraga, M. E. Occurrence of entomopathogenic fungi in atlantic forest soils. Microbiology Discovery. 4 (1), 1-7 (2016).
- Goble, T. A., Dames, J. F., Hill, M. P., Moore, S. D.The effects of farming system, habitat type and bait type on the isolation of entomopathogenic fungi from citrus soils in the Eastern Cape Province, South Africa. BioControl. 55 (3), 399-412 (2010).
- Medo, J., Cagáň, L. Factors affecting the occurrence of entomopathogenic fungi in soils of Slovakia as revealed using two methods. Biological Control. 59 (2), 200-208 (2011).
- Chase, A. R., Osborne, L. S., Ferguson, V. M. Selective isolation of the entomopathogenic fungi Beauveria bassiana and Metarhizium anisopliae from an artificial potting medium. Florida Entomologist. 69, 285-292 (1986).
- Liu, Z. Y., Milner, R. J., McRae, C. F., Lutton, G. G. The use of dodine in selective media for the isolation of Metarhizium spp. from soil. Journal of Invertebrate Pathology. 62, 248-251 (1993).
- Rangel, D. E. N., Dettenmaier, S. J., Fernandes, E. K. K., Roberts, D. W. Susceptibility of Metarhizium spp. and other entomopathogenic fungi to dodine-based selective media. Biocontrol Science and Technology. 20 (4), 375-389 (2010).
- Keller, S., Kessler, P., Schweizer, C. Distribution of insect pathogenic soil fungi in Switzerland with special reference to Beauveria brongniartii and Metharhizium anisopliae. BioControl. 48 (3), 307-319 (2003).
- Enkerli, J., Widmer, F., Keller, S. Long-term field persistence of Beauveria brongniartii strains applied as biocontrol agents against European cockchafer larvae in Switzerland. Biological Control. 29 (1), 115-123 (2004).
- Imoulan, A., Alaoui, A., El Meziane, A. Natural occurrence of soil-borne entomopathogenic fungi in the Moroccan endemic forest of Argania spinosa and their pathogenicity to Ceratitis capitata. World Journal of Microbiology and Biotechnology. 27 (11), 2619-2628 (2011).
- Keyser, C. A., De Fine Licht, H. H., Steinwender, B. M., Meyling, N. V. Diversity within the entomopathogenic fungal species Metarhizium flavoviride associated with agricultural crops in Denmark. BMC Microbiology. 15 (1), 1-11 (2015).

