Protocol for Developing a Femur Osteotomy Model in Wistar Albino Rats
Summary
Here, we present a protocol to iatrogenically fracture the shaft of the femur of Wistar albino rats and follow up on the development of the callus. This femur osteotomy model can help researchers evaluate the process of fracture healing and to study how a drug could influence fracture healing.
Abstract
Fracture healing is a physiological process resulting in the regeneration of bone defects by the coordinated action of osteoblasts and osteoclasts. Osteoanabolic drugs have the potential to augment the repair of fractures but have constraints like high costs or undesirable side effects. The bone healing potential of a drug can initially be determined by in vitro studies, but in vivo studies are needed for the final proof of concept. Our objective was to develop a femur osteotomy rodent model that could help researchers understand the development of callus formation following fracture of the shaft of the femur and that could help establish whether a potential drug has bone healing properties. Adult male Wistar albino rats were used after Institutional Animal Ethics Committee clearance. The rodents were anesthetized, and under aseptic conditions, complete transverse fractures at the middle one-third of the shafts of the femurs were created using open osteotomy. The fractures were reduced and internally fixed using intramedullary K-wires, and secondary fracture healing was allowed to take place. After surgery, intraperitoneal analgesics and antibiotics were given for 5 days. Sequential weekly x-rays assessed callus formation. The rats were sacrificed based on radiologically pre-determined time points, and the development of the fracture callus was analyzed radiologically and using immunohistochemistry.
Introduction
Bone is a dense connective tissue consisting of bone-forming cells, the osteoblasts, and bone-resorbing cells, the osteoclasts. Fracture healing is a physiological process resulting in the regeneration of bone defects by the coordinated action of osteoblasts and osteoclasts1. When there is a fracture, osteoblastic and osteoclastic activity at the fracture site are some of the important factors that determine bone healing2. When fracture healing deviates from its normal course, it results in a delayed union, malunion, or nonunion. A fracture is said to be in nonunion when there is a failure of union of the fracture for 9 months, with no progression of repair in the last 3 months3. Approximately 10%-15% of all fractures experience a delay in repair that may progress to nonunion4. The nonunion rate for all fractures is 5%-10% and varies depending on the bone involved and the site of fracture5.
The current regimen for the treatment of fracture nonunion comprises surgical and/or medical modalities. Currently, delayed or nonunion of fractures can be overcome by surgical strategies like bone grafting. However, bone grafting has its limitations and complications like availability of graft tissue, donor site pain, morbidity, and infection6. Medical treatment comprises osteoanabolic drugs like bone morphogenetic protein (BMP) and teriparatide (parathormone analog). Currently used osteoanabolic agents have the potential to augment the repair of fractures but have constraints like exorbitant costs or undesirable side effects7. Hence, there is scope for identifying cost-effective, nonsurgical alternatives for bone healing. The bone healing potential of a drug can initially be determined by in vitro studies, but in vivo studies are needed for the final proof of concept. A drug that is known to enhance bone healing should be evaluated in vitro and, if found promising, can be used for in vivo animal model studies. If the drug proves to promote bone formation and remodeling in the in vivo model, it could proceed to the next stage (i.e., clinical trials).
Assessing fracture healing in animals is a logical step forward to evaluate a novel agent introduced for bone healing before it undergoes human trials. For in vivo animal model studies of fracture healing, rodents have become an increasingly popular model8. The rodent models have generated increasing interest due to the low operational costs, limited need for space, and less time needed for bone healing9. In addition, rodents have a broad spectrum of antibodies and gene targets, which allow studies on the molecular mechanisms of bone healing and regeneration10. A consensus meeting comprehensively highlighted various small animal bone healing models and focused on the different parameters influencing bone healing, as well as emphasizing several small animal fracture models and implants11.
Basic fracture models can be broadly divided into open or closed models. Closed fracture models use a three- or four-point bending force on the bone and do not require a conventional surgical approach. They lead to oblique or spiral fractures, resembling long bone fractures in humans, but the lack of standardization of fracture location and dimensions may act as a confounding factor in them12. Open fracture models require surgical access for osteotomy of the bone, help to achieve a more consistent fracture pattern at the fracture site, but are associated with delayed healing compared to the closed models13. The choice of bone used to study fracture healing mainly remains the tibia and femur due to their dimensions and accessibility. The choice of the site of fracture is usually the diaphysis or metaphysis. The metaphyseal region is specially chosen in cases where fracture healing is studied in osteoporotic subjects, as the metaphysis is more affected by osteoporosis14. Several implants like intramedullary pins and external fixators can be used to stabilize the fracture11,15.
The objective of this study was to develop a simple and easy-to-follow rodent model that could help researchers not only understand the development of the callus following fracture of the femur but could help to determine whether a potential drug has bone healing properties by understanding the mechanism by which it acts.
Protocol
Animal experiments were done after taking ethical approval from the Institutional Animal Ethics Committee (IAEC), AIIMS, New Delhi, India (286/IAEC-1/2021).
1. Preoperative procedure
- House male Wistar albino rats 6-8 weeks of age, weighing between 150-200 g each, at a Central Animal Facility (CAF) in separate individual cages. This ensures no surgical/fracture site injury when multiple rats share cages.
- Keep the rats at a temperature of 23 °C ± 2 °C in a humidity-controlled environment with a relative humidity of 50% ± 5%, expose them to a 12 h dark/light cycle, and give ad libitum access to food (standard semi-synthetic diet): pellet diet (dry) and water. The composition of the standard semi-synthetic diet is as follows: roasted Bengal gram flour (60%), wheat flour (22%), casein (4%), skim milk powder (5%), refined oil (4%), salt mixture with starch (4.8%), and vitamin choline mixture with starch (0.2%).
- Acclimatize the rats for a period of at least 48 h before surgery.
- Weigh each rat on a digital weighing scale and note the weight.
- Administer intraperitoneal (IP) injections of cefuroxime (100 mg/kg body weight), tramadol (25 mg/kg body weight), and a combination of ketamine (75 mg/kg body weight) with xylazine (10 mg/kg body weight) to the rats 15 min before starting the surgical procedure. Apply ophthalmic ointment to both eyes to prevent dry eye.
- Remove the hair from the right lower limb, from the flank region up to the knee joint, with topical application of a hair removal cream.
NOTE: Blood (0.5 mL) can be collected from the tail vein of each rat for the baseline analysis of different parameters. Blood can be collected again every 2 weeks after the surgery.
2. Surgical procedure for creating complete transverse fracture through open osteotomy
NOTE: Use a designated operation room with an operating table and optimal ambient temperature (26 °C) for performing the procedure.
- Place the wax block (aluminum tray 30 cm x 30 cm x 4 cm containing wax up to a depth of 2.5 cm) on the operating table and cover it with sterile drapes. The wax block prevents any change in the animal's position during surgery.
- Confirm the onset of anesthesia (by checking loss of toe pinch). Place the anesthetized rat on a sterile drape in the left lateral position. Ask an assistant to hold the right lower limb (knee and hip) in extension. Keep a sterile hard support (marble block) under the right leg to support the femur. Clean the surgical site with alcohol and betadine.
- Inject local anesthesia (0.25 mL of 1% lignocaine) at the site of incision (lateral aspect of the right thigh), cut a hole in another sterile drape, and expose only the right leg of the rat through it for surgery.
- Give a 1 cm vertical skin incision on the lateral side of the right thigh, and extend it as per need with a no. 15 surgical blade.
- Expose the vastus lateralis muscle by separating the deep fascia using Metzenbaum scissors. Split the vastus lateralis in line with the muscle fibers using artery forceps until the shaft of the femur is reached.
- Free the bone from the muscles attached to it using the periosteal elevator.
- Inject local anesthesia (0.2 mL of 1% lignocaine) in and around the periosteum to prevent vasovagal reflex.
- Create an indentation in the middle third of the shaft of the femur using the no.15 surgical blade, and fracture the bone in the middle-third of the shaft (complete fracture) by placing a chisel on the indentation made (so that the chisel does not slip) and gently tapping the chisel with a hammer. Use the sterile hard support (marble block) to support the bone while fracturing it to ensure a clean break.
NOTE: The sterile hard support usually does not cause a significant injury to the muscles underneath. - Internally fix the fracture using a sterile K-wire (1.0 mm) held with the help of a battery-operated power drill. Pass the K-wire into the medullary canal of the distal fragment through the fracture site. Then, drill out the K-wire through the distal end of the femur using the battery-operated power drill.
NOTE: Disinfect the surface of the power drill with alcohol before use. Change gloves after the K-wire is fixed. - After reducing the fracture, advance the K-wire from the distal end into the canal of the proximal fragment until it obtains purchase in the trochanteric region. Cut off the distal part of the K-wire protruding through the skin using a wire cutter.
- Bend the tip of the K-wire to around 90° using pliers and use a gauze bandage soaked in betadine for pin-site dressing. The K-wire acts as an intramedullary splint for keeping the fracture in a reduced position.
- Ensure complete hemostasis before closing the skin using a 3-0 nylon suture. Apply pressure on the bleeding area using sterile gauze or artery forceps to stop any bleeding.
- Clean the wound with betadine, and cover it with sterile gauze and micropore adhesive tape.
3. Postoperative care
- Return the rats to their cages, allow normal ambulation, and continue giving a standard semi-synthetic diet until sacrificing them, as well as antibiotics (injection cefuroxime 100 mg/kg) and analgesics (injection tramadol 25 mg/kg/day in two divided doses) intraperitoneally for 5 days after the procedure.
NOTE: The rats can be divided into treatment and control groups to test a particular drug. If the drug is water soluble, it can be given orally through gavage. The weight of the individual animals may be noted to calculate the dose of the drug to be used. Inclusion and exclusion criteria can be followed to ensure homogeneity of the animal groups. - House the animals in individual cages under similar conditions to the preoperative period. Inspect the surgical site every day to look for any signs of postoperative pain, wound infection, slipping of sutures, or any abdominal swelling or discomfort.
- Assess bone healing by X-ray of the fractured site once weekly.
4. Radiological procedure
- Before the X-ray, anesthetize the rats with an intraperitoneal injection of ketamine (50 mg/kg body weight) and xylazine (5 mg/kg body weight).
- Keep the hip joint of the rat in a flexed and abducted position while the knee joint is kept semi-flexed to take the X-ray of the fractured limb with the following exposure settings: Ref. kVp ≈ 62; Ref. mAS = 6.4; and automatic exposure settings (Ref. mA=160).
NOTE: X-rays were taken at baseline (1 day after surgery) and then once weekly until sacrifice or 5 weeks.
5. Animal euthanasia and callus retrieval
- Sacrifice the rats by an overdose of carbon dioxide (administer 100% CO2 at a flow rate of 7-8 L/min for 1 min, followed by a waiting period of 4-5 min), at two previously determined time points, based on the radiological appearance of soft and hard bridging calluses, respectively.
- Incise the skin parallel to the femur and separate the overlying muscles carefully to avoid damage to the callus tissue.
- Fracture the bone between the hip joint and callus tissue using a hammer and chisel. Similarly, fracture the bone between the callus and the knee joint. Remove the K-wire and clean the bone piece in saline to remove blood clots and soft tissue.
- Transfer the callus immediately to a labeled container with 10% neutral buffered formalin (20 mL per sample) and keep it for 3 days at room temperature (RT).
6. Decalcification of bone and callus tissue
- Take the callus tissue from formalin and keep it at RT in 20% ETDA solution, pH 7, for decalcification of the bone tissue.
- Change fresh EDTA solution every 2 days for approximately 3 weeks, and check for bone decalcification by poking the bone with a needle without disturbing the callus tissue. Optimal decalcification is denoted by the loss of the normal gritty sensation of the bone tissue.
- After complete decalcification, cut the sagittal section of the callus and prepare paraffin blocks of the callus tissue. Cut 4 µm thick sections of the callus tissue for histopathological16 and any other comparative analysis17.
Representative Results
This study was undertaken to develop a femur osteotomy model in Wistar albino rats. This model can be used to evaluate bone healing, as well as the osteogenic effect of a promising osteoanabolic drug in bone healing. Standard surgical precautions and protocols were followed. Sterile gowns, drapes, and surgical equipment were used for the procedure (Figure 1). The equipment (Table 1) was sterilized 48 h before surgery. Anesthetic, analgesic, and antibiotics were used as per the protocol to ensure that animals were kept pain- and infection-free at all times. Blood (0.5 mL) can be collected from the tail vein of each rat for baseline analysis and sequential chronological comparative follow-up of different parameters as fracture healing occurs. Hair was removed from the flank region to the knee region using hair removal cream (Figure 2). The osteotomy procedure took approximately 10 min to complete (from the first incision to the suture). Infection and mortality were negligible on following aseptic precautions. An incision was made after local anesthesia (lignocaine), and the shaft of the femur was exposed after retracting the fibers of the vastus lateralis (Figure 3). An indentation (groove) was created in the bone using a surgical blade to ensure that the chisel did not slip. Sterile hard support (marble block) was used to support the bone while fracturing it to ensure a clean break (Figure 1). A complete transverse fracture was induced in the middle third of the shaft of the femur using a chisel and hammer (Figure 4).
The fracture was internally fixed using a sterile K-wire (1.0 mm). The K-wire was passed into the medullary canal of the distal fragment through the fracture site. The K-wire was then drilled out through the distal end of the femur. The fracture was reduced, and then the K-wire was advanced from the distal end into the canal of the proximal fragment until it obtained purchase in the trochanteric region. The distal part of the K-wire that protruded through the skin was cut off. The K-wire acted as an intramedullary splint to keep the fracture in a reduced position (Figure 5).
An X-ray of the fractured area was taken 1 day post surgery and weekly thereafter to evaluate the appearance of the callus (beginning of fracture healing) and appearance of the bridging callus (the first time point when the fracture gap was healed), as evaluated by the radiologist (Figure 6). Two radiological time points for comparative evaluation of fracture healing were the appearance (visualization) of the (soft) callus and the appearance of the bridging (hard) callus.
After sacrifice, the femur was carefully preserved in formalin, followed by the protocol for bone decalcification (Figure 7). The K-wire was removed during the sacrifice, taking care not to disturb the callus. After complete decalcification, the bone was cut into sagittal sections and preserved in paraffin blocks for sectioning (4 µm thick sections) as and when required. A hematoxylin-and-eosin-stained section of the fracture site and callus confirmed cartilage and new bone formation at the end of 5 weeks (Figure 8).
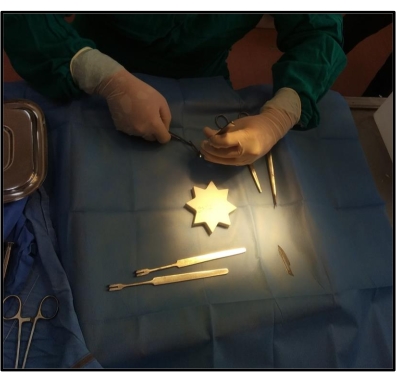
Figure 1: Sterile surgical instruments kept on the surgical drape on the operating table. The surgeon is ready to start the surgical procedure in a sterile environment with sterile instruments. Please click here to view a larger version of this figure.
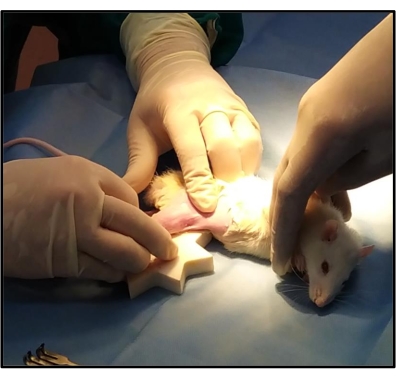
Figure 2: Anesthetized rat kept on the operating table. After giving anesthesia to the rat and removing the hair around the incision site, it is kept on the operating table in a left lateral position, exposing the right leg for osteotomy. Another surgical drape is used to pass the right leg through a hole in that drape to ensure that only the leg is exposed, thereby minimizing wound infections. Please click here to view a larger version of this figure.
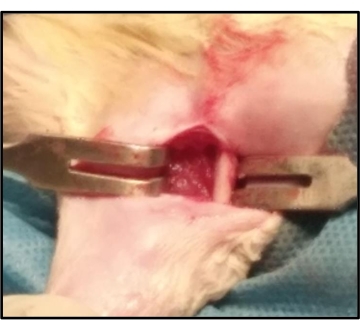
Figure 3: Surgical procedure: Exposure of the shaft of the rat femur. During osteotomy, after the vastus lateralis is exposed, it is split in line with the muscle fibers to expose the shaft of the femur. The bone is freed from the attached muscles using the periosteal elevator. Please click here to view a larger version of this figure.
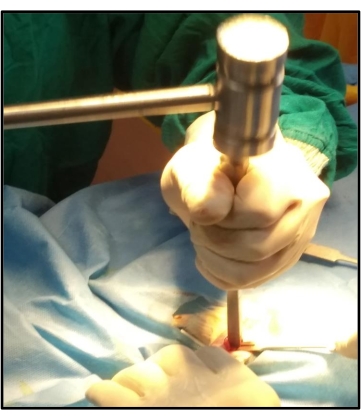
Figure 4: Creating a complete transverse osteotomy in the middle third of the shaft of the femur using a chisel and hammer. A complete transverse fracture is created in the middle one-third of the shaft of the femur by gently tapping the chisel with the hammer. Please click here to view a larger version of this figure.
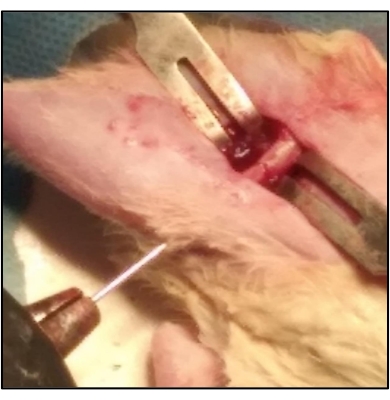
Figure 5: K-wire acts as an intramedullary splint for keeping the fracture in a reduced position. The K-wire is passed into the medullary canal of the distal fragment through the fracture site. The K-wire is then drilled out through the distal end of the femur. The fracture is reduced and then the K-wire is advanced from the distal end into the canal of the proximal fragment until it obtained purchase in the trochanteric region. This is done using a battery-operated power drill. Please click here to view a larger version of this figure.
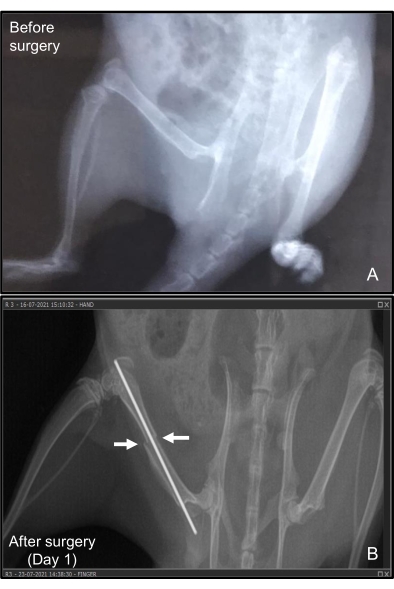
Figure 6: X-ray of the femur of the rat with the K-wire in situ. (A) Before inducing the fracture and (B) 1 day after surgery. The fracture healing is monitored radiologically by taking sequential weekly X-rays of the operated site to radiologically assess callus formation. The fracture remains reduced and immobilized with the intramedullary K-wire. The before and after representative data are not from the same animal. Please click here to view a larger version of this figure.
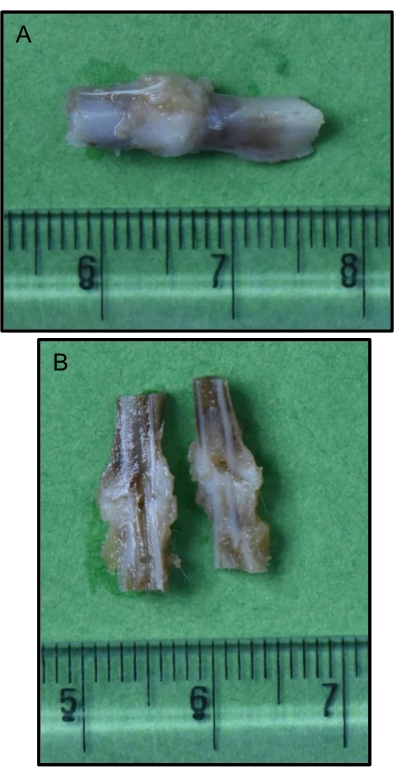
Figure 7: Bone with callus (after optimal decalcification) obtained after sacrificing the animal at a pre-determined time point. (A) Intact callus; (B) Sagittal section of the callus. After sacrificing the animal, the area of the fracture site is obtained, preserved, and decalcified using the described methodology. The callus is intermittently assessed to ensure optimal decalcification before evaluating it by any other techniques (reference scale in centimeters). Please click here to view a larger version of this figure.
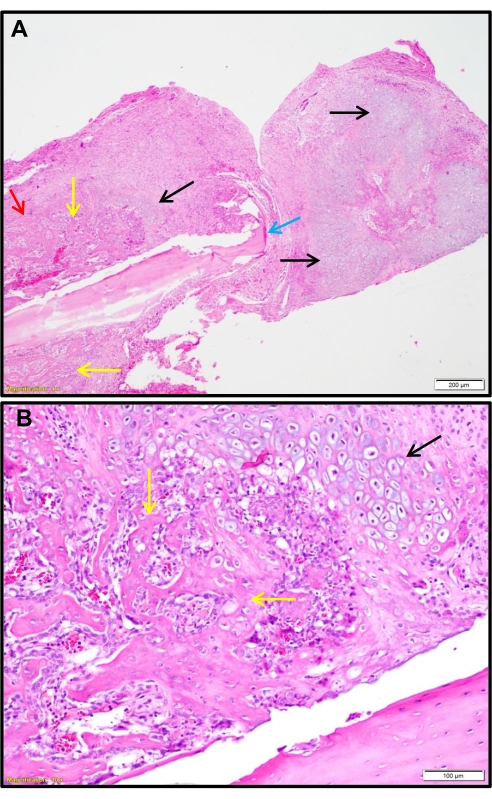
Figure 8: Hematoxylin-and-eosin-stained section of the fracture site showing a hard callus with the formation of cartilage and new bone. (A) Low magnification; (B) High magnification. The hematoxylin-and-eosin-stained sections of the fracture site show a hard callus with the formation of cartilage (black arrows) and new bone (yellow arrows) (A: 40x; B: 100x). The blue arrow shows the fracture end of the bone, and the red arrow shows the second cortical region. Please click here to view a larger version of this figure.
Discussion
This method lucidly describes the details needed to develop a fracture osteotomy model in Wistar albino rats. This model can be used to evaluate the osteogenic effect of a promising osteoanabolic drug in fracture healing, as well as understand the intricacies of bone healing. The salient feature of this method is that it is simple and does not need too much time or sophisticated equipment. In this method, adult male Wistar albino rats were selected as the rodent model for the experiments. Uniform gender was selected to remove any confounding factor on bone healing related to the sex hormones.
This study followed the procedure of open osteotomy, which is similar to that followed by other groups, as well as other small animal models11,18,19. The advantage of the open osteotomy followed in this method over other bone healing models is that the injury-induced (complete cortical bone fracture) resembles a regular long bone fracture, and fracture healing in this method resembles that of a regular fracture, where there is secondary bone healing (enchondral ossification) by callus formation, compared to drill hole bone injury, where there is primary bone healing (intramembranous ossification)20. The method of open osteotomy is also better than the closed osteotomy or three-point pressure-inducing method, where there is a possibility of bone shattering and wide disparity in the fracture line, thus leading to the difference in fracture healing21. Open osteotomy increases the chance of wound infection compared to closed osteotomy, but, similar to other studies, we observed that with proper precautions, wound infection was negligible22. It was also observed in this methodology that creating a groove (indentation) on the bone with a blade before fracturing it with a chisel and hammer served to create a uniform fracture line and avoided slipping of the chisel on the bone. Another modification we introduced in this method was keeping a hard, sterile block below the bone to be fractured. This not only provided a counterforce while creating the fracture but also helped to avoid shattering, crushing, or an irregular fracture line. This usually does not cause any significant injury to the muscles underneath.
Our study used X-rays to determine the time points of sacrifice based on the radiological appearance of the callus as the first time point and the appearance of the bridging (hard) callus as the second time point in the animals before starting the complete experiment. The entire comparative group of animals should be sacrificed when any treatment or control group reaches a particular time point to compare their calluses using immunohistochemical analysis of osteoblastic and osteoclastic markers. This will ensure unbiased comparison among the different treatment and control groups. X-ray of the fractured site of the rats should be done at weekly intervals, and blood samples (tail vein) should be taken at 2 weekly intervals until they reach the respective time points of sacrifice. Weekly x-rays were done (under anesthesia) to evaluate callus formation by the radiologist (who was blinded to the control and treatment groups). The X-rays also helped substantiate and corroborate the biological parameters of bone healing.
This method involves the application of a K-wire as an intramedullary splint to keep the fracture immobilized in a reduced position. However, intramedullary pin fixation does not provide absolute stability to the fracture, as do plating and external fixators, and may at times be associated with complications like wound infection, pin migration, perforation of the femoral shaft cortex, etc. Our study also suggests that it is better to inject lignocaine in and around the periosteum, which is extremely pain-sensitive. This prevents severe pain and the possibility of neurogenic shock during the osteotomy. It was also observed that keeping the volume of intraperitoneal injections low helped minimize subsequent respiratory distress in the rats. Analgesics and antibiotics were continued for 5 days after surgery to prevent any pain or infection. For this study, the femur was chosen for inducing fracture as it was easy to access, easy to break cleanly, and because of its straight contour, which is easier for K-wire insertion. Caution must be exercised when the K-wire is advanced into the proximal fragment of the femur, as there is a risk of bleeding by injuring the femoral artery. It was observed that rats tend to pull out the K-wire if too much residual wire is left protruding out of the skin.
The parameters of the outcome of bone healing are osteoblastic and osteoclastic markers in the blood and callus of the animals (of various groups and different time points). For osteoblastic markers, osteocalcin, Col1A1, RANKL, P1NP, and bone-specific alkaline phosphatase could be selected, while CTX and RANK could be assessed to evaluate osteoclastic activity. Some of these parameters can be evaluated in serum, while others can be evaluated by immunohistochemistry in callus tissue. These parameters give a holistic view of bone remodeling by simultaneously evaluating osteoblastic and osteoclastic activity.
The limitation of this study is that it does not evaluate the tensile strength of the callus. Ideally, biomechanical studies add value to data. Caution needs to be exercised while processing the callus and adjoining bone tissue for decalcification, as incomplete decalcification will not give optimal results in immunohistochemistry.
This protocol to assess fracture healing using the rodent model will be useful for all groups that are trying to evaluate promising drugs with osteoanabolic activity. It is a simple model to accurately assess bone and fracture healing in the rodent model while evaluating osteoblastic and osteoclastic activity and bone remodeling, which give useful mechanistic insights. If resources and logistics revolving around the number of animals are permissible, biological indicators can also be reinforced by the radiological assessment of fracture healing, as well as comparing tensile strength, which assesses the mechanical stature of the healed bone. Studies that elucidate the mechanism of action are preferred to purely observational studies.
Divulgations
The authors have nothing to disclose.
Acknowledgements
The authors would like to thank Central Council for Research in Homoeopathy (CCRH), Ministry of AYUSH, Govt. of India, for research funding. The authors are grateful for the help and support of Central Animal Facility, AIIMS, New Delhi, for their help and support with the animal experiments and CMET, AIIMS, New Delhi, for their help and support in photography and videography.
Materials
| Alcohol | Raman & Weil Pvt. Ltd, Mumbai, Maharashtra, India | MFG/MD/2019/000189 | Sterillium hand disinfectant |
| Artery forceps | Nebula surgical, Gujarat, India | G.105.05S | 5", straight |
| Bard-Parker handle | Nebula surgical, Gujarat, India | G.103.03 | Size number 3 |
| Betadine solution | Win-medicare New Delhi, India | UP14250000001 | 10% w/v Povidone iodine solution |
| Cat's-paw skin retractor | Nebula surgical, Gujarat, India | 908.S | Small |
| EDTA | Sisco research laboratories Pvt. Ltd, Maharashtra, India | 43272 | Disodium salt |
| Eosin | Sigma Aldrich, Merck Life Sciences Pvt Ltd, Mumbai, Maharashtra, India | 115935 | For preparing the staining solution |
| Forceps (plain) | Nebula surgical, Gujarat, India | 115.06 | 6", plain |
| Forceps (toothed) | Nebula surgical, Gujarat, India | 117.06 | 6", toothed |
| Formaldehyde | Sisco research laboratories Pvt. Ltd, Maharashtra, India | 84439 | For preparing the neutral buffered formalin |
| Haematoxylin | Sigma Aldrich, Merck Life Sciences Pvt Ltd, Mumbai, Maharashtra, India | 104302 | For preparing the staining solution |
| Hammer | Nebula surgical, Gujarat, India | 401.M | |
| Injection Cefuroxime | Akumentis Healthcare Ltd, Thane, Maharashtra, India | 48/UA/SC/P-2013 | Cefuroxime sodium IP, 1.5 g/vial |
| Injection Ketamine | Baxter Pharmaceuticals India Private Limited, Gujarat, India | G/28-B/6 | Ketamine hydrochloride IP, 50 mg/mL |
| Injection Xylazine | Indian Immunologicals Limited, Hyderabad, Telangana, India | 28/RR/AP/2009/F/G | Xylazine hydrochloride USP, 20 mg/mL |
| Injection Lignocaine | Jackson laboratories Pvt Limited, Punjab, India | 1308-B | 2% Lignocaine Hydrochloride IP, 21.3 mg/mL |
| Injection Tramadol | Intas Pharmaceuticals Limited, Ahmedabad, Gujarat, India | MB/07/500 | Tramadol hydrochloride IP, 50 mg/mL |
| K-wire | Nebula surgical, Gujarat, India | 166 (1mm) | 12", double ended |
| Mechanical drill for inserting K-wire | Bosch, Germany | 06019F70K4 | GSR 120-LI Professional |
| Metzenbaum cutting scissors | Nebula surgical, Gujarat, India | G.121.06S | 6", straight |
| Needle holder | Nebula surgical, Gujarat, India | G.108.06 | 6", straight |
| Ophthalmic ointment | GlaxoSmithKline Pharmaceutical Limited, Bengaluru, Karnataka, India | KTK/28a/467/2001 | Neomycin, Polymixin B sulfate and Bacitracin zinc ophthalmic ointment USP |
| Osteotome (chisel) | Nebula surgical, Gujarat, India | 1001.S.10 | 10 mm, straight |
| Periosteal elevator | Nebula surgical, Gujarat, India | 918.10.S | 10 mm, straight |
| Pliers cum wire cutter | Nebula surgical, Gujarat, India | 604.65 | |
| Reynold’s scissors | Nebula surgical, Gujarat, India | G.110.06S | 6", straight |
| Standard semi-synthetic diet | Ashirvad Industries, Chandigarh, India | No catalog number available | Detailed composition provided in materials used |
| Steel cup for keeping betadine for application | Local purchase | No catalog number available | |
| Steel tray with lid for autoclaving instruments | Local purchase | No catalog number available | |
| Sterile gauze | Ideal Healthcare Industries, Delhi, India | E(0047)/14/MNB/7951 | Sterile, 5cmx5cm, 12 ply |
| Sterile marble block for support | Local purchase | No catalog number available | Locally fabricated; autoclavable |
| Syringe and needle (1 mL) | Becton Dickinson India Pvt. Ltd., Haryana, India | REF 303060 | 1 mL sterile Syringe with 26 G x 1/2 (0.45 mm x 13 mm) needle |
| Syringe and needle (2 mL) | Becton Dickinson India Pvt. Ltd., Haryana, India | REF 307749 | 2 mL sterile syringe with 24 G x 1'' (0.55 mm x 25 mm) needle |
| Syringe and needle (10 mL) | Hindustan Syringes & Medical Devices Ltd. Faridabad, India | 334-B(H) | 10 mL sterile syringe with 21 G x1.5" (0.80 mm x 38 mm) needle |
| Surgical blades (size no.15) | Paramount Surgimed Ltd, New Delhi, India for Medline Industries Inc, IL, USA | REF MDS15115E | Sterile, Single use |
| Surgical blades (size no.24) | Paramount Surgimed Ltd, New Delhi, India for Medline Industries Inc, IL, USA | REF MDS15124E | Sterile, Single use |
| Sutures | Healthium Medtech Pvt Ltd, Bangalore, Karnataka, India | SN 3318 | 4-0, 16 mm, 3/8 circle cutting needle, monofilament polyamide suture |
| Wax block in aluminium tray | Locally fabricated | No catalog number available | 30 cm x 30 cm x 4 cm aluminium tray containing wax (to prevent animal from slipping) |
| X-ray machine | Philips India Ltd, Gurugram, Haryana | SN19861013 | Model: Philips Digital Diagnost R 4.2 |
References
- Wang, T., Zhang, X., Bikle, D. D. Osteogenic differentiation of periosteal cells during fracture healing. Journal of Cellular Physiology. 232 (5), 913-921 (2017).
- Fakhry, M., Hamade, E., Badran, B., Buchet, R., Magne, D. Molecular mechanisms of mesenchymal stem cell differentiation towards osteoblasts. World Journal of Stem Cells. 5 (4), 136-148 (2013).
- Bishop, J. A., Palanca, A. A., Bellino, M. J., Lowenberg, D. W. Assessment of compromised fracture healing. JAAOS – Journal of the American Academy of Orthopaedic Surgeons. 20 (5), 273-282 (2012).
- Fong, K., et al. Predictors of nonunion and reoperation in patients with fractures of the tibia: an observational study. BMC Musculoskeletal Disorders. 14 (1), 103 (2013).
- Ramoutar, D. N., Rodrigues, J., Quah, C., Boulton, C., Moran, C. G. Judet decortication and compression plate fixation of long bone nonunion: Is bone graft necessary. Injury. 42 (12), 1430-1434 (2011).
- Goulet, J. A., Senunas, L. E., DeSilva, G. L., Greenfield, M. L. V. H. Autogenous iliac crest bone graft: Complications and functional assessment. Clinical Orthopaedics and Related Research. 339, 76-81 (1997).
- Stevenson, M., et al. A systematic review and economic evaluation of alendronate, etidronate, risedronate, raloxifene and teriparatide for the prevention and treatment of postmenopausal osteoporosis. Health Technology Assessment. 9 (22), 1 (2005).
- Haffner-Luntzer, M., Kovtun, A., Rapp, A. E., Ignatius, A. Mouse models in bone fracture healing research. Current Molecular Biology Reports. 2 (2), 101-111 (2016).
- Mills, L. A., Simpson, A. H. R. W. In vivo models of bone repair. The Journal of Bone and Joint Surgery. British Volume. 94 (7), 865-874 (2012).
- Houdebine, L. -. M., Sioud, M. Transgenic Animal Models in Biomedical Research. Target Discovery and Validation Reviews and Protocols: Volume 1, Emerging Strategies for Targets and Biomarker Discovery. , (2007).
- Histing, T., et al. Small animal bone healing models: Standards, tips and pitfalls results of a consensus meeting. Bone. 49 (4), 591-599 (2011).
- Bonnarens, F., Einhorn, T. A. Production of a standard closed fracture in laboratory animal bone. Journal of Orthopaedic Research. 2 (1), 97-101 (1984).
- Klein, M., et al. Comparison of healing process in open osteotomy model and open fracture model: delayed healing of osteotomies after intramedullary screw fixation. Journal of Orthopaedic Research. 33 (7), 971-978 (2015).
- Kolios, L., et al. Do estrogen and alendronate improve metaphyseal fracture healing when applied as osteoporosis prophylaxis. Calcified Tissue International. 86 (1), 23-32 (2010).
- Holstein, J. H., et al. Advances in the establishment of defined mouse models for the study of fracture healing and bone regeneration. Journal of Orthopaedic Trauma. 23, 31-38 (2009).
- Umiatin, U., Dilogo, I. H., Sari, P., Wijaya, S. K. Histological analysis of bone callus in delayed union model fracture healing stimulated with pulsed electromagnetic fields (PEMF). Scientifica. 2021, 4791172 (2021).
- Han, W., et al. The osteogenic potential of human bone callus. Scientific Reports. 6, 36330 (2016).
- Haffner-Luntzer, M., et al. A novel mouse model to study fracture healing of the proximal femur. Journal of Orthopaedic Research. 38 (10), 2131-2138 (2020).
- Aurégan, J. C., et al. The rat model of femur fracture for bone and mineral research: An improved description of expected comminution, quantity of soft callus and incidence of complications. Bone & Joint Research. 2 (8), 149-154 (2013).
- Li, Z., Helms, J. A. Drill hole models to investigate bone repair. Methods in Molecular Biology. 2221, 193-204 (2021).
- Handool, K. O., et al. Optimization of a closed rat tibial fracture model. Journal of Experimental Orthopaedics. 5 (1), 13 (2018).
- Kobata, S. I., et al. Prevention of bone infection after open fracture using a chitosan with ciprofloxacin implant in animal model. Acta Cirurgica Brasileira. 35 (8), 202000803 (2020).

