Quantifying Myeloperoxidase-DNA and Neutrophil Elastase-DNA Complexes from Neutrophil Extracellular Traps by Using a Modified Sandwich ELISA
Summary
We present a protocol for a modified sandwich enzyme-linked immunosorbent assay technique to quantitatively measure two components of neutrophil extracellular trap remnants, myeloperoxidase conjugated-DNA and neutrophil elastase conjugated-DNA complexes,derived from activated neutrophils.
Abstract
Certain stimuli, such as microorganisms, cause neutrophils to release neutrophil extracellular traps (NETs), which are basically web-like structures composed of DNA with granule proteins, such as myeloperoxidase (MPO) and neutrophil elastase (NE), and cytoplasmic and cytoskeletal proteins. Although interest in NETs has increased recently, no sensitive, reliable assay method is available for measuring NETs in clinical settings. This article describes a modified sandwich enzyme-linked immunosorbent assay to quantitatively measure two components of circulating NETs, MPO-DNA and NE-DNA complexes, which are specific components of NETs and are released into the extracellular space as breakdown products of NETs. The assay uses specific monoclonal antibodies for MPO or NE as the capture antibodies and a DNA-specific detection antibody. MPO or NE binds to one site of the capture antibody during the initial incubation of samples containing MPO-DNA or NE-DNA complexes. This assay shows good linearity and high inter-assay and intra-assay precision. We used it in 16 patients with COVID-19 with accompanying acute respiratory distress syndrome and found that the plasma concentrations of MPO-DNA and NE-DNA were significantly higher than in the plasma obtained from healthy controls. This detection assay is a reliable, highly sensitive, and useful method for investigating the characteristics of NETs in human plasma and culture supernatants.
Introduction
This article outlines a method to quantify neutrophil extracellular trap (NET) formation in biological fluids by using sandwich enzyme-linked immunosorbent assay (ELISA) to detect complexes of myeloperoxidase (MPO) and neutrophil elastase (NE) with DNA1,2. NETs are composed of a DNA backbone decorated with antimicrobial proteases originating from neutrophil granules3,4. Both MPO-DNA and NE-DNA complexes are important and specific components of NETs and are released into the extracellular space as breakdown products of NETs3,4.
Besides their important physiological role in antimicrobial defense3, NETs also have various pathological effects4,5, including the promotion of thrombogenesis6 and the worsening of sepsis7. Accordingly, NETs have been gaining attention recently. Nevertheless, the in vivo quantification of NETs has proven challenging because of the lack of a sensitive, reliable quantitative assay method.
A few methods are available, including the direct measurement of NETs by fluorescence microscopy8,9 and flow cytometry10 and the indirect measurement of circulating cell-free DNA, nucleosomes, and citrullinated histone H3, but each method has its own advantages and limitations11. Although the immunofluorescence microscopic method is specific to NETs and clearly shows the localization and degree of NET formation, samples are limited to biopsy tissue and secreted materials. Moreover, this method needs to be performed by skilled researchers and requires a long time for results to be obtained. Measuring circulating levels of NET-related components by flow cytometry is easy and provides results quickly; however, the method is not specific to NETs12.
We13 and others1,2 have developed a highly sensitive and reliable assay to measure the circulating NET components, MPO-conjugated or NE-conjugated DNA, in human plasma with a modified ELISA technique that uses specific antibodies for MPO or NE as the capture antibodies and a DNA-specific detection antibody. This assay can also be used ex vivo to identify NET components in cell culture supernatants released by activated neutrophils in response to phorbol 12-myristate 13-acetate (PMA) stimulation.
Protocol
This study was conducted in conformity with the Declaration of Helsinki and was approved by the institutional review boards of Aichi Medical University (2017-H341, 2019-H137). Written informed consent was obtained from each participant.
1. Reagent preparation
NOTE: To perform the sandwich ELISA assay, the reagents are prepared as described below.
- Coating buffer:
- To make a 0.1 mol/L carbonate-bicarbonate buffer, weigh 10.6 g of anhydrous sodium carbonate (molecular weight, 106 g/mol) and 8.4 g of sodium bicarbonate (molecular weight, 84 g/mol).
- Add it to approximately 900 mL of distilled water in a beaker.
- Adjust the pH of the buffer to 9.6 by adding the required quantity of diluted hydrochloric acid or sodium hydroxide.
- Check the pH with a pH meter.
- Then, add the required volume of distilled water to achieve a total volume of 1,000 mL.
- Store this 0.1 mol/L carbonate-bicarbonate buffer in the refrigerator at 4 °C.
- Blocking buffer:
- Add approximately 250 µL of 20% sodium azide and 1 g of bovine serum albumin (BSA) to 70 mL of phosphate-buffered saline (PBS) composed of 137 mmol/L NaCl, 8.1 mmol/L Na2HPO4, 2.68 mmol/L KCl, and 1.47 mol/L KH2PO4 (pH 7.4), and mix gently.
- Add distilled water to achieve a total volume of 100 mL. The final concentrations of bovine serum albumin and sodium azide are 1% and 0.05%, respectively. Wait for 10 min, and then the solution is ready to use.
- Washing solution: Prepare 0.5% t-octylphenoxypolyethoxyethanol, polyethylene glycol tert-octylphenyl ether (Triton X-100) in distilled water.
- Dilute 100% Triton X-100 to 10% with distilled water, and allow the 10% solution to stand for 1 day before use.
- Then, re-dilute the 10% Triton X-100 solution 20-fold with distilled water to achieve a concentration of 0.5%.
- Antibody dilution:
- Dilute the capture antibody (anti-MPO antibody [1 mg/mL] or anti-NE antibody [1 mg/mL]) to 1:2,000 in the coating buffer (pH 9.6) before use on day 1 of the assay (see below) and the detection antibody (peroxidase-conjugated anti-DNA antibody) to 1:40 with PBS before use on day 3 of the assay (see below).
NOTE: For the preliminary experiment, dilute the iso-type antibodies (IgG rabbit [5 mg/mL] and IgG1 mouse clone Ci4 [0.5 mg/mL]) to 1:10,000 and 1:1,000 in the coating buffer (pH 9.6).
- Dilute the capture antibody (anti-MPO antibody [1 mg/mL] or anti-NE antibody [1 mg/mL]) to 1:2,000 in the coating buffer (pH 9.6) before use on day 1 of the assay (see below) and the detection antibody (peroxidase-conjugated anti-DNA antibody) to 1:40 with PBS before use on day 3 of the assay (see below).
- ABTS substrate solution: Depending on the number of samples, dissolve one, two, or three 2,2'-azino-bis(3 ethylbenzothiazoline-6-sulfonic acid (ABTS) tablets in 5 mL, 10 mL, or 15 mL of ABTS buffer (containing sodium perborate, citric acid, and disodium hydrogen phosphate); 100 µL is needed per sample. Store the prepared solution at 2-8 °C protected from light. The solution is stable for up to 3 months.
2. Sample collection and storage
- Plasma isolation:
- Collect 4 mL of peripheral blood samples into a lithium heparin blood collection tube by venipuncture with a 22 G syringe, and centrifuge at 1,600 x g for 10 min at 4 °C.
- Transfer the supernatant into a 2 mL safe-lock sample tube.
- Re-centrifuge the supernatant at 16,000 x g for 10 min at 4 °C to remove any residual cells.
- Collect the supernatant into a new safe-lock sample tube without disturbing the pellet at the bottom of the tube.
- Store the obtained plasma at −80 °C until further use.
NOTE: To obtain high-throughput results with this assay, good-quality samples are required. If plasma samples are used, perform a second centrifugation to remove any residual cells. Avoid repeated thawing of the samples. Use heparin as the anticoagulant because EDTA affects the DNase activity.
- Isolation of polymorphonuclear neutrophils
- Collect the peripheral blood samples into a lithium heparin blood collection tube by venipuncture with a 22 G syringe.
- Layer 6 mL of blood onto 6 mL of one-step polymorphs, and centrifuge the tubes at 1,000 x g for 45 min at room temperature (RT).
- Harvest the third buffy coat layer containing neutrophils, and wash it three times in phenol red-free RPMI-1640 at 400 x g for 10 min at RT.
- Resuspend the neutrophils in phenol red-free RPMI-1640 containing 2 mM L-glutamine supplemented with 6% heat-inactivated fetal bovine serum, and count the cells in the microscope field with a hemocytometer.
NOTE: This method yields 96% to 98% purity with greater than 95% viability, as assessed by trypan blue dye exclusion.
- Activation of polymorphonuclear neutrophils and NETosis in vitro
- Dilute freshly isolated polymorphonuclear neutrophils to 1 × 105 cells/mL in phenol red-free RPMI-1640 containing 2 mM L-glutamine supplemented with 6% heat-inactivated fetal bovine serum, and seed them in 35 mm culture dishes.
- To induce NETs, stimulate polymorphonuclear neutrophils with 25 nM PMA for 4 h at 37 °C in 5% CO2/95% air.
- After 4 h of incubation, partially digest the NETs by adding 0.6 µg/mL DNase I directly to the culture dishes for 15 min at RT, and stop the DNase I activity by adding 5 mM EDTA.
- Collect the medium containing the synthesized NETs, and centrifuge at 400 x g for 10 min at 4 °C to remove the cell debris.
- Obtain the supernatants from four healthy controls, mix, and store at −80 °C until use.
NOTE: DNase-digested PMA-stimulated neutrophils obtained from four healthy controls are used as the NET-standard1,14,15. Generate a calibration curve from a serially diluted NET-standard with PBS, and assign the optical density (OD) values obtained from an undiluted NET-standard as 100% NET.
3. Assay method
NOTE: The steps for performing the assay are described in detail below.
- Day 1
- Apply a total of 100 µL of diluted anti-MPO or anti-NE antibody containing 0.05 µg of antibody to each well of the plate, including the blank well.
NOTE: The coating buffer must not contain any kind of detergent because, otherwise, the antibodies may not bind equally and smoothly to the walls of each well. To prevent the hook effect, the coating protein concentration must not be more than 20 µg/mL, because concentrations above that level will saturate most of the available sites on the microtiter plate. The typical concentration range of protein coating solutions is 2-10 µg/mL. For the preliminary experiment, use diluted iso-type control antibody IgG rabbit for coating instead of specific monoclonal antibodies against MPO. Use diluted iso-type control antibody IgG1 mouse for coating instead of specific monoclonal antibodies against NE. - Cover the plate with an adhesive plastic cover to prevent the evaporation of the sample, and incubate overnight at 4 °C to allow the binding of the capture antibodies.
- Apply a total of 100 µL of diluted anti-MPO or anti-NE antibody containing 0.05 µg of antibody to each well of the plate, including the blank well.
- Day 2
- Discard the diluted antibody solution from the wells, and then pipette 300 µL of wash solution per well, and repeat this washing procedure three to four times.
- Remove excess PBS by tapping the plate dry on a paper towel.
- Block each well of the ELISA plate with 200 µL of the blocking buffer.
- Cover the plate closely with an adhesive plastic cover, and incubate it for 1.5-2 h at RT to obstruct the wells.
- Discard the blocking solution completely from the wells, and then pipette 300 µL of wash solution per well, and repeat this washing procedure three to four times.
- Remove excess PBS by tapping the plate dry on a paper towel.
- Apply a total of 25 µL of plasma or medium to each well except the blank well, and add 75 µL of PBS as a diluent to make the final volume 100 µL; add 100 µL of PBS to the blank well.
- Then, mix the samples at RT for 10 s by placing the plate on a shaker at 250 rpm.
- Apply 2 µL of 100-fold diluted DNase I (0.03 mg/mL) to each well, including the blank well (final concentration of DNase I in the reaction mixture: 0.6 µg/mL).
- Seal the plate with an adhesive plastic cover, and thoroughly mix the samples at RT for 10 s by placing the plate on a shaker at 250 rpm.
NOTE: To assess the optimum conditions for DNA digestion, when developing the assay, we incubated samples containing MPO-associated DNA from patients with COVID-19 with 0-0.9 µg/mL DNase I for 15 min at RT and used plates coated with specific capture antibody for MPO or a species-matched iso-type control antibody. - Incubate the samples for 15 min at RT.
- Remove the adhesive plastic cover, and apply 1 µL of 0.5 M EDTA to each well to stop the DNase reaction.
- Reseal the plate with an adhesive plastic cover, and shake it at RT for 15 s by placing it on a shaker at 250 rpm.
- Then, incubate the plate overnight at 4 °C to allow the protein components of the NETs to attach to the capture antibodies.
- Day 3
- Remove the adhesive plastic cover, and discard the solution from the wells.
- Discard the solution completely from the wells, and then pipette 300 µL of the wash solution per well, and repeat this washing procedure three to four times.
- Apply a total of 100 µL of the diluted peroxidase-conjugated anti-DNA detection antibody to each well.
- Seal the plate closely with an adhesive plastic cover, and incubate it for 1.5 h at RT.
- Discard the solution completely from the wells, and then pipette 300 µL of wash solution per well, and repeat this washing procedure three to four times.
- Carefully remove the residual solution.
- Apply a total of 100 µL of ABTS substrate solution to each well, and cover the plate with an adhesive plastic cover.
- Incubate the plate in the dark at RT for 20-30 min on a shaker at 250 rpm.
NOTE: Monitor the plate at 5 min intervals until the desired OD readings are obtained. - Add a total of 50 µL of 2 M sulfuric acid to stop the reaction.
- Mix the liquid contents of the wells by carefully tapping the side of the plate.
- Switch on the microplate reader, and connect it to the computer.
- Open the software application on the computer.
- From the status bar, create a new experiment and name it.
- Set all the following parameters for plate reading: Read type, Absorbance; Read mode, Endpoint; Wavelengths, 2; Lm-1, 405 nm; Lm-2, 490 nm; Auto mix & Blanking before, Off; Pre-read plate, Off; Auto calibration, On; Strips, Read entire plate; Column wavelength priority, Column priority; Carriage speed, Normal; and Auto read, Off.
- Then, put the 96-well plate containing the samples into the drawer and close it.
- Last, select the Read button so that the plate is read immediately.
- Read the absorbance of each well at a wavelength of 405 nm by using a microplate reader (at this stage, use a wavelength of 490 nm as a reference).
- Perform automatic subtraction of the absorbance value of the assay medium alone from all the unknown samples, and save the data.
NOTE: Use a standard curve obtained from serially diluted NET-standard to calculate the relative concentration of NET in the sample1,14,15. Here, the final results are presented as "MPO- or NE-DNA complexes (% of NET-standard)." The limit of detection (LOD) was calculated from the standard deviation (SD) and the slope of the calibration curve (S) as follows: LOD = 3.3(SD/S).
4. Statistics
- Perform all the statistical analyses using appropriate statistical analysis software. Here SigmaPlot v14.5 was used. Percentages and continuous variables are shown as the median with the interquartile range due to the skewed distribution of most of the parameters.
- Compare the plasma MPO-DNA and NE-DNA levels between COVID-19 patients and healthy controls using the Wilcoxon rank sum test. Use correlation analysis to explore the relationship between optical density and the percentage of NET-standard.
Representative Results
This method used a sandwich ELISA with anti-MPO, anti-NE, and anti-DNA monoclonal antibodies to measure MPO-associated and NE-associated DNA (Figure 1). In this method, the wells of a microtiter plate were coated with an MPO-specific or NE-specific monoclonal antibody to capture DNA-associated MPO and DNA-associated NE, as well as non-DNA-associated MPO and NE. To calculate the intra-assay coefficient of variability (CV), duplicate measurements were performed within the same plate for 30 samples collected from patients with COVID-19 and healthy controls, and the %CV was calculated as the mean of the duplicate measurements; to calculate the inter-assay CV (i.e., the plate-to-plate consistency), two types of samples from patients with COVID-19 and healthy controls were measured in quadruplicate on 10 different plates; to demonstrate the specificity of this assay, various concentrations of MPO-DNA and NE-DNA complexes were assayed by using plates coated with iso-type control antibodies instead of specific monoclonal antibodies against MPO and NE; and to calculate the sensitivity and linearity of the assay, a serially diluted NET-standard made by stimulating isolated human neutrophils with PMA and mild DNase digestion was assayed, and the correlation coefficient and limit of detection (LOD) were calculated.
The calibration curves for MPO-DNA and NE-DNA drawn from a serially diluted NET-standard are shown in Figure 2. Reliable standard curves for MPO-DNA and NE-DNA (R2 = 0.958 and 0.963, respectively) are obtained when the absorbance values do not exceed concentrations of 0.93 and 0.90, respectively. The LOD values calculated from the SD and the slope of the calibration curve were 0.132% and 0.126% for MPO-DNA and NE-DNA (%NET standard), respectively.
The highest OD was obtained when 0.6 µg/mL DNase I was applied (Figure 3). Therefore, the DNA digestion was limited to adding a 0.6 µg/mL reaction mixture of DNase I for 15 min at room temperature. When the plates were coated with the iso-type control antibody instead of the specific monoclonal antibody against MPO, very low OD values (<0.09 absorbance units [AU]) were detected at various concentrations of DNase I.
To calculate the intra-assay coefficient of variability (CV) for MPO-DNA and NE-DNA, duplicate measurements were performed in 30 samples from patients with COVID-19 and healthy controls in the same plate; the %CV was calculated by using the duplicate mean. The intra-assay CVs for MPO-DNA and NE-DNA were 1.871 and 0.987, respectively, in healthy controls and 2.532 and 2.010, respectively, in COVID-19 patients (Table 1). The mean intra-assay CVs of MPO-DNA and NE-DNA were 2.202 ± 0.467 and 1.497 ± 0.723, respectively (mean ± SD) (Table 1).
To calculate the inter-assay CV for MPO-DNA and NE-DNA, two types of samples collected from patients with COVID-19 and healthy controls were measured in quadruplicate on 10 different plates to monitor the plate-to-plate consistency. The mean inter-assay CVs of MPO-DNA and NE-DNA were 6.524 ± 2.672 and 4.389 ± 0.923, respectively (mean ± SD) (Table 2).
To evaluate the specificity of the capture antibodies to MPO-DNA and NE-DNA complexes, we assayed various concentrations of MPO-DNA and NE-DNA complexes by using plates coated with iso-type control antibodies instead of specific monoclonal antibodies against MPO and NE. Table 3 shows that the iso-type control antibodies reacted little with the MPO-DNA and NE-DNA complexes at the various concentrations (0.035 AU to 0.078 AU and −0.007 AU to 0.096 AU, respectively).
The levels of MPO-DNA and NE-DNA in the supernatant of DNase-digested PMA-stimulated neutrophils were defined as 100% NET-standard, and the plasma sample data were expressed as %NET-standard. The levels of MPO-DNA (%NET-standard) were significantly higher in the plasma from patients with COVID-19 (n = 16; 29.1% [IQR, 25.8, 41.5]) than in the plasma from healthy controls (n = 10; 13.4% [IQR, 12.4, 14.8]), as were the levels of NE-DNA (%NET-standard) (46.4% [IQR, 32.7, 53.7] vs. 12.1% [IQR, 9.9, 14.7], respectively, P < 0.01; Figure 4).
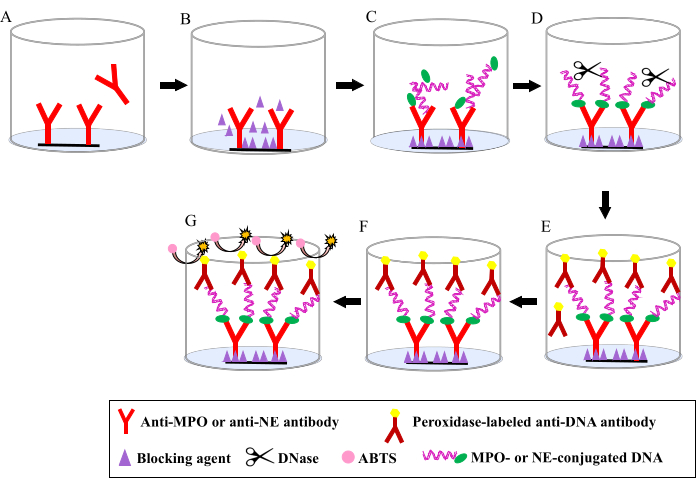
Figure 1: Basic stages of a sandwich enzyme-linked immunosorbent assay method for measuring myeloperoxidase-DNA or neutrophil elastase-DNA in samples. (A) The wells are coated with an anti-myeloperoxidase (MPO) or anti-neutrophil elastase (NE) capture antibody. (B) The remaining protein binding sites are blocked by blocking agents. (C) Samples containing NE-conjugated and MPO-conjugated DNA are added. (D) Limited DNase digestion is performed, and the sample is incubated in the wells to bind with the capture antibody. (E) A secondary peroxidase-labeled anti-DNA antibody is added. (F) Unbound secondary peroxidase-labeled anti-DNA antibody is removed. (G) The substrate 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) is added, and the color development is monitored. Abbreviations: ABTS = 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid); MPO = myeloperoxidase; NE = neutrophil elastase Please click here to view a larger version of this figure.
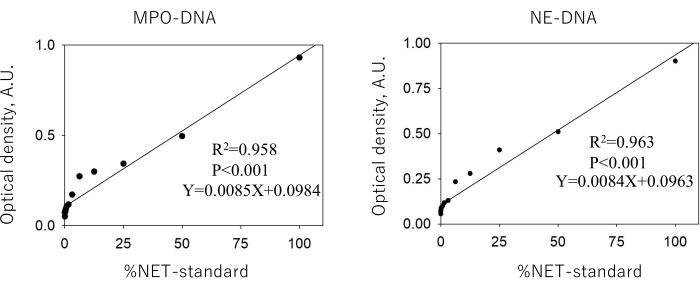
Figure 2: Linearity of the relationships between the intensity values and the various dilutions of the neutrophil extracellular trap standard. The supernatant of DNase-digested phorbol 12-myristate 13-acetate-stimulated neutrophils was serially diluted, and the levels of myeloperoxidase-DNA and neutrophil elastase-DNA were assayed to generate a calibration curve. The absorbance units (AU) obtained from the undiluted neutrophil extracellular trap standard (NET-standard) were assigned as 100%NET, and plotting the intensity value at 405 nm against the %NET-standard revealed a linear relationship. Abbreviations: MPO = myeloperoxidase; NE = neutrophil elastase; NET = neutrophil extracellular trap Please click here to view a larger version of this figure.
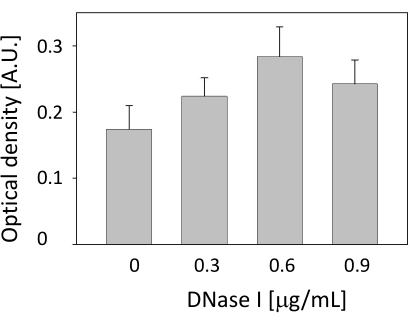
Figure 3: Dose-response of DNase I in neutrophil extracellular trap-DNA digestion. The sample was inserted into myeloperoxidase-coated wells. Then, DNase I (0 µg/mL, 0.3 µg/mL, 0.6 µg/mL, or 0.9 µg/mL) was added. After 15 min of digestion, the enzyme activity was stopped, and the remaining steps of the sandwich enzyme-linked immunosorbent assay were performed accordingly. The data are shown as the mean ± SD. N = 12. Please click here to view a larger version of this figure.
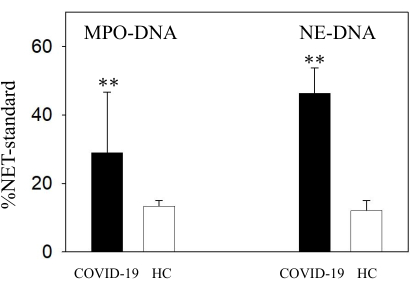
Figure 4: Plasma levels of myeloperoxidase-DNA and neutrophil elastase-DNA complexes in patients with COVID-19. The data are expressed as percentages of neutrophil extracellular trap standard (NET-standard) content and presented as medians and interquartile ranges. Patients with COVID-19, n = 16; healthy controls, n = 10. ** P < 0.01 versus healthy controls. Abbreviations: HC = healthy controls; NET = neutrophil extracellular trap. Please click here to view a larger version of this figure.
Table 1: Intra-assay coefficient of variability. Thirty samples were measured in duplicate to monitor individual coefficients of variability and, thus, determine the intra-assay coefficient of variability. Abbreviations: AU = absorbance units; CV = coefficients of variability Please click here to download this Table.
Table 2: Inter-assay coefficient of variability. The samples were measured in quadruplicate on 10 different plates to monitor plate-to-plate variation and, thus, determine the inter-assay coefficient of variability. Abbreviations: AU = absorbance units; CV = coefficient of variability. Please click here to download this Table.
Table 3: Specificity test for capture antibodies. The plates were coated with iso-type control antibodies for anti-myeloperoxidase (MPO) and anti-neutrophil elastase (NE) to evaluate the specificity of the capture antibodies for the MPO-DNA and NE-DNA complexes. Abbreviations: AU = absorbance units; MPO = myeloperoxidase; NE = neutrophil elastase Please click here to download this Table.
Discussion
We have described a sandwich ELISA method in which MPO or NE binds to one site of the capture antibody during the initial incubation of samples containing MPO-DNA or NE-DNA complexes. After washing, the "sandwich" is completed by incubating the samples with a peroxidase-associated anti-DNA monoclonal antibody. After the removal of unbound secondary antibody, the bound peroxidase conjugate is detected by the addition of a chromogenic ABTS peroxidase substrate, which yields a soluble end-product that can be read spectrophotometrically at 405 nm. The good linearity and high inter-assay and intra-assay precision indicate that the ELISA assay described in this paper and others1,2 is reliable and feasible for clinical application. Furthermore, when the supernatant of DNase-treated PMA-stimulated neutrophils is assigned as a maximal signal for the NET-standard, this ELISA assay can be used as a semi-quantitative method1,14,15.
The long chromatin threads of NETs are decorated with MPO and NE proteins. To increase the binding between the capture antibody and the MPO-DNA or NE-DNA complexes, the threads are cut into shorter pieces by limited DNA digestion with the enzyme DNase; the addition of a DNase concentration that is too high could lead to excessive digestion of the DNA and, thus, a decrease in the absorbance. The results of a preliminary experiment (see Figure 3) showed that an appropriate amount of DNase addition is necessary to loosen the remnants derived from NETs. The experiments that used iso-type control antibodies instead of specific capture antibodies showed that the capture antibodies used in this assay were specific for MPO-DNA and NE-DNA complexes.
The early stage of NET formation is characterized by decondensed chromatin and the preservation of the intensity of the plasma membrane9,16,17. Neutrophils with a condensed nucleus have been identified as neutrophils that are undergoing NET formation and can be quantified by flow cytometry10. Although flow cytometry can analyze a large number of images from cells in a short time, it cannot be used to evaluate the later stages of NET formation after cell membrane rupture and the extrusion of chromatin. Citrullinated histones H3, which are known to be NET-specific markers, can be detected by western blotting18 and ELISA19; however, these methods are only specific to PAD4-related NET formation and cannot be used to assess PAD4-independent NETs20.
The finding that the levels of serum NET remnants were higher in patients with COVID-19 than in healthy controls agrees with previous reports21,22. This finding suggests that neutrophil activation, including NET formation, may play an important role in the pathogenesis of COVID-19.
This assay has a limitation. Recent reports have shown that immune-related genes, including MPO and ELANE, which encode MPO and NE, respectively, are highly expressed under uncontrolled inflammatory conditions23 and are positively correlated with disease severity and mortality24. As MPO and NE are involved in the formation of NETs independent of those enzymatic activities15, increased protein levels of NE and MPO in neutrophils may affect the results of this assay.
We conclude that this sandwich ELISA method directly measures extracellular DNA with granule proteins, including NE and MPO, which are specific for NET formation. This detection assay is a reliable, highly sensitive, and useful method for investigating the characteristics of NETs in human samples and culture supernatants.
Divulgations
The authors have nothing to disclose.
Acknowledgements
The authors thank Dr. Huq Muhammad Aminul for providing assistance in reviewing the manuscript.
Materials
| 1-Step Polymorphs | Accurate Chemical and Scientific Corporation | AN221725 | Isolation of PMN's from human blood. |
| 96-well microtiter plate | Thermo Fisher Scientific | 467466 | flat bottom |
| ABTS buffer solution | Sigma-Aldrich Merck | 11 204 530 001 | Contains sodium perborate, citric acid, and disodium hydrogen phosphate. |
| ABTS tablets | Sigma-Aldrich Merck | 11 204 521 001 | Each tablet contains 5 mg ABTS substrate and 60 mg vehicle substances. |
| Adhesive plastic cover, Axygen | Thermo Fisher Scientific | 14222348 | |
| Anti-MPO antibody | Sigma-Aldrich Merck | 07-496-I | Store at 2-8 °C. stable for 1 year. Host species is rabbit. |
| Anti-NE antibody, clone AHN-10 | Sigma-Aldrich Merck | MABS461 | Store at 2-8 °C. stable for 1 year. Host species is mouse. |
| Bovine serum albumin | Biomedical Science | BR-220700081 | Albumin from bovine fraction V. Store at 2–8 °C. stable for 2 year. |
| DNase I | New England BioLabs | M0303M | Store at -20 °C |
| IgG, rabbit, Isotype Control | GENETEX, Inc. | GTX35035 | Store as concentrated solution at 2–8 °C. |
| IgG1, mouse Isotype Control, clone Ci4 | Merck | MABC002 | Store as concentrated solution at 2–8 °C. |
| Lithium heparin blood collection tube | Becton Dickinson and Company | ||
| Microplate mixer | As one corporation | NS-P | |
| Microplate Reader | Molecular Devices | SpectraMax 190 | Any microplate plate reader capable of reading wavelengths from 405–490 nm can use. |
| Microplate reader application | Molecular Devices | SoftMax pro | |
| Peroxidase-conjugated anti-DNA antibody, Cell death Detection ELISA | Roche Diagnostics | 1154467500 | bottle 2. Store at 2–8 °C. stable for 1 year. |
| Phorbol 12-myristate 13-acetate | Sigma-Aldrich Merck | P8139 | Activation of PMN's from human blood. |
| Phosphate buffered solution | Takara Bio | T9181 | Store at room temperature. Stable for 6 months. |
| SigmaPlot v14.5 | Systat Software Inc. San Jose, CA, USA | ||
| Sodium azide | Fujifilm Wako Chemicals | 190-14901 | Store at room temperature. |
| t-Octylphenoxypolyethoxyethanol, Polyethylene glycol tert-octylphenyl ether | Fujifilm Wako Chemicals | 9002-93-1 | Store at room temperature. |
References
- Sil, P., Yoo, D. G., Floyd, M., Gingerich, A., Rada, B. High throughput measurement of extracellular DNA release and quantitative NET formation in human neutrophils in vitro. Journal of Visualized Experiments. (112), e52779 (2016).
- Yoo, D. G., Floyd, M., Winn, M., Moskowitz, S. M., Rada, B. NET formation induced by Pseudomonas aeruginosa cystic fibrosis isolates measured as release of myeloperoxidase-DNA and neutrophil elastase-DNA complexes. Immunology Letters. 160 (2), 186-194 (2014).
- Brinkmann, V., et al. Neutrophil extracellular traps kill bacteria. Science. 303 (5663), 1532-1535 (2004).
- Papayannopoulos, V. Neutrophil extracellular traps in immunity and disease. Nature Reviews Immunology. 18, 134-147 (2018).
- Chamardani, T. M., Amiritavassoli, S. Inhibition of NETosis for treatment purposes: Friend or foe. Molecular and Cellular Biochemistry. 477 (3), 673-688 (2022).
- Rao, A. N., Kazzaz, N. M., Knight, J. S. Do neutrophil extracellular traps contribute to the heightened risk of thrombosis in inflammatory diseases. World Journal of Cardiology. 7 (12), 829-842 (2015).
- Sørensen, O. E., Borregaard, N. Neutrophil extracellular traps – The dark side of neutrophils. Journal of Clinical Investigation. 126 (5), 1612-1620 (2016).
- Abrams, S. T., et al. A novel assay for neutrophil extracellular traps (NETs) formation independently predicts disseminated intravascular coagulation and mortality in critically ill patients. American Journal of Respiratory and Critical Care Medicine. 200 (7), 869-880 (2019).
- Brinkmann, V., Goosmann, C., Kühn, L. I., Zychlinsky, A. Automatic quantification of in vitro NET formation. Frontiers in Immunology. 3, 413 (2012).
- Zhao, W., Fogg, D. K., Kaplan, M. J. A novel image-based quantitative method for the characterization of NETosis. Journal of Immunological Methods. 423, 104-110 (2015).
- Masuda, S., et al. NETosis markers: Quest for specific, objective, and quantitative markers. Clinica Chimica Acta. 459, 89-93 (2016).
- Rada, B. Neutrophil extracellular traps. Methods in Molecular Biology. 1982, 517-528 (2019).
- Kano, H., Huq, M. A., Tsuda, M., Noguchi, H., Takeyama, N. Sandwich ELISA for circulating myeloperoxidase- and neutrophil elastase-DNA complexes released from neutrophil extracellular traps. Advanced Techniques in Biology & Medicine. 5 (1), 1000196 (2016).
- Prevel, R., et al. Plasma markers of neutrophil extracellular trap are linked to survival but not to pulmonary embolism in COVID-19-related ARDS patients. Frontiers in Immunology. 13, 851497 (2022).
- Schechter, M. C., et al. et al. extracellular trap (NET) levels in human plasma are associated with active TB. PLoS One. 12, e0182587 (2017).
- Papayannopoulos, V., Metzler, K. D., Hakkim, A., Zychlinsky, A. Neutrophil elastase and myeloperoxidase regulate the formation of neutrophil extracellular traps. Journal of Cell Biology. 191 (3), 677-691 (2010).
- Gupta, S., Chan, W., Zaal, K. J., Kaplan, M. J. A high-throughput real-time imaging technique to quantify NETosis and distinguish mechanisms of cell death in human neutrophils. Journal of Immunology. 200 (2), 869-879 (2018).
- Li, M., Lin, C., Leso, A., Nefedova, Y. Quantification of citrullinated histone H3 bound DNA for detection of neutrophil extracellular traps. Cancers. 12 (11), 3424 (2020).
- Thålin, C., et al. Quantification of citrullinated histones: Development of an improved assay to reliably quantify nucleosomal H3Cit in human plasma. Journal of Thrombosis and Haemostasis. 18 (10), 2732-2743 (2020).
- Li, P., et al. PAD4 is essential for antibacterial innate immunity mediated by neutrophil extracellular traps. Journal of Experimental Medicine. 207 (9), 1853-1862 (2010).
- Zuo, Y., et al. Neutrophil extracellular traps in COVID-19. JCI Insight. 5 (11), e138999 (2020).
- Masso-Silva, J. A., et al. Increased peripheral blood neutrophil activation phenotypes and neutrophil extracellular trap formation in critically ill Coronavirus disease 2019 (COVID-19) patients: A case series and review of the literature. Clinical Infectious Diseases. 74 (3), 479-489 (2022).
- Gong, F. C., et al. Identification of potential biomarkers and immune features of sepsis using bioinformatics analysis. Mediators of Inflammation. 2020, 3432587 (2020).
- Almansa, R., et al. Transcriptomic correlates of organ failure extent in sepsis. Journal of Infection. 70 (5), 445-456 (2015).

