Analysis of Extracellular Vesicle-Mediated Vascular Calcification Using In Vitro and In Vivo Models
Summary
This paper presents the methodology for obtaining and assessing vascular calcification by isolating murine aortas followed by extracting calcified extracellular vesicles to observe the mineralization potential.
Abstract
Cardiovascular disease is the leading cause of death in the world, and vascular calcification is the most significant predictor of cardiovascular events; however, there are currently no treatment or therapeutic options for vascular calcification. Calcification begins within specialized extracellular vesicles (EVs), which serve as nucleating foci by aggregating calcium and phosphate ions. This protocol describes methods for obtaining and assessing calcification in murine aortas and analyzing the associated extracted EVs. First, gross dissection of the mouse is performed to collect any relevant organs, such as the kidneys, liver, and lungs. Then, the murine aorta is isolated and excised from the aortic root to the femoral artery. Two to three aortas are then pooled and incubated in a digestive solution before undergoing ultracentrifugation to isolate the EVs of interest. Next, the mineralization potential of the EVs is determined through incubation in a high-phosphate solution and measuring the light absorbance at a wavelength of 340 nm. Finally, collagen hydrogels are used to observe the calcified mineral formation and maturation produced by the EVs in vitro.
Introduction
Calcification is the most significant predictor of cardiovascular disease mortality and morbidity1. Calcification alters the arterial wall mechanics due to the buildup of calcium and phosphate minerals2. In atherosclerosis, calcification can exacerbate local stress and result in plaque rupture, which is the leading cause of heart attacks. Medial calcification-often resulting from chronic kidney disease-is more widespread and leads to significant arterial stiffening, dysfunction, and cardiac overload2,3. Currently, there are no therapeutic options for the treatment or prevention of vascular calcification.
Vascular smooth muscle cells (VSMCs) adopt an osteoblast-like phenotype and release calcifying extracellular vesicles (EVs) that nucleate nascent minerals, thus driving calcification4,5,6. This process resembles the physiological mineralization of osteoblasts in bone7. Although the endpoint of mineralization is similar in the vascular wall and bone matrix, the mechanisms by which the calcifying EVs originate differ in the two tissues8. There are many types of models that are used to study vascular calcification. In vitro, cell culture models mimic the osteogenic transition of VSMCs and subsequent mineral formation with specialized media.
When studying in vivo calcification, the model used depends upon the type of calcification being studied. Hyperlipidemic mouse models are often used to study atherosclerotic calcification, which appears more focal within lipid-rich plaques9. In contrast, medial calcification is more widespread throughout the vasculature and is often studied using chronic kidney disease models that employ an adenine-rich diet regimen to induce renal failure or surgical techniques to remove significant portions of the kidneys10,11. Aggressive models of vascular calcification have used a combination of both hyperlipidemic and chronic kidney disease models12. This protocol provides a method for assessing vascular calcification in murine aortas for both medial and atherosclerotic calcification, extracting EVs from the aortic wall, and observing the mineralization potential in the EVs obtained from in vitro cell culture models. Future studies can use these procedures in mechanistic analyses of vascular calcification and to assess potential therapeutic interventions.
Protocol
The in vivo work was approved and overseen by the Institutional Animal Care and Use Committee (IACUC) at Florida International University and conformed to current National Institutes of Health (NIH) guidelines. For this protocol, the procedure does not differ depending on the strain, weight, age, and sex of the mouse. The type of calcification being studied, diets, and treatments may alter the length of the study and the weight of the mouse used and may be dependent on a specific strain and sex of mouse used in the study. For this protocol, both male and female C57BL/6J mice were used, and they were fed a calcification diet. The mice were sacrificed between 20 weeks and 24 weeks of age.
1. Isolation and excision of the aorta
- Fluorescent markers
- Forty-eight hours before euthanasia, inject the mice with 20 µM OsteoSense 680, a fluorescent imaging agent, at the recommended dose of 100 µL per 25 g via an intravenous injection.
- Under the fume hood, place a Styrofoam board with four t-pins, a bucket lined with a paper towel, a 50 mL tube stuffed with a paper towel, a 1.5 mL microcentrifuge tube, dissection tools (see the Table of Materials), and ethanol in a spray bottle. Place beakers with PBS on ice.
- Blood draw
- Add 2 mL of isoflurane into a 50 mL conical tube stuffed with the paper towel. Place the tube in the bucket, and close it with its lid.
CAUTION: Isoflurane should be handled in a well-ventilated space with no recirculation of exhaust air due to the toxicity of short-term exposure. - Transfer one mouse into the bucket at a time. Once sedated, place the mouse on the paper towel in the dorsal position. Holding the mouse with the palm of the non-dominant hand, place the ears back using the thumb and pointer finger.
- Using the tweezers, excise one of the eyes. Hold the mouse above the 1.5 mL tube to begin blood collection, during which 1 mL of blood should be collected. Once the blood stops draining, place the mouse back in the bucket for further sedation.
- Add 2 mL of isoflurane into a 50 mL conical tube stuffed with the paper towel. Place the tube in the bucket, and close it with its lid.
- Euthanasia
- Place a paper towel on the dissection mat. Once the mouse is sedated, place the mouse on the mat in a supine position. Secure the mouse in the supine position using a t-pin to hold down each limb.
- Spray down the abdomen of the mouse with alcohol. Using the tweezers, lift the skin at the chest region. With the scissors, cut down the medial line into the thoracic cavity. Once the sternum has been cut down the center, cut the diaphragm on either side of the ribcage.
- If necessary, cut away the frontal portion of the ribcage to expose the heart and lungs.
- Organ removal
- Using tweezers, lift the skin and make an incision down the midline. Remove any excess skin or fat. Remove the reproductive organs and bladder, as well as any fat outside of the midline.
- Move the gastrointestinal (GI) organs to the right-hand side, and follow the intestines in the midline. Once found, lift the midline, and excise the GI tract up to the stomach.
- Excise the kidneys by lifting them away from the midline. Cut as close to the kidney as possible.
- Next, begin the removal of the liver by lifting the lobes and cutting away from the midline.
- Perfuse the heart and aorta. Using a 10 mL syringe, inject cold PBS into the right ventricle. Wait for the lungs to inflate and turn white. Remove the lungs, as well as any excess diaphragm or ribcage.
- Bone removal
- Open a pair of scissors wide, and place them at the lower region of the mouse above the tail. Cut downward, and begin lifting the spine from the skin. Remove the fat from the sides of the midline. Once the skin is detached, excise the spine and intact organs by cutting right above the heart. If transporting to a stereoscope, place the spine and intact organs in a beaker of PBS in the ice bucket.
- Move the spine and organs into the dissecting dish. Fill with ice-cold PBS. Pin down the spine and ribcage using needles.
- Under the dissection microscope, use tweezers to lift the heart carefully, and begin cutting above the spine and underneath the aorta. Continue this until the bottom of the spine is reached. Remove the spine from the dissecting dish, and pin down the heart and any extraneous fat or muscle.
- Microdissection
- Starting from the area right below the heart, identify the three tubular structures: the esophagus, vena cava, and aorta. Remove the esophagus and vena cava to have a clear visual field of the aorta.
- Using the microdissection forceps and scissors, begin lifting the adipose tissue and cutting as close to the aorta as possible. Be sure not to cut the aorta. Continue this down the medial line until all the aorta as well as the femoral arteries are exposed.
- At the heart, remove all the adipose tissue, exposing the three branches at the aortic arch. Remove the aortic root from the left ventricle by inserting the microdissection scissors into the left ventricle and cutting the muscle surrounding the aorta.
- Once the aorta is isolated and cleaned, image the aorta using a near-infrared scanner to visualize vascular calcification.
- Use the custom MATLAB script (Supplemental File 1) to quantify the total signal of the calcium tracer, which is normalized to the total scanned aorta area. The MATLAB script functions using the Bioinformatics toolbox add-in. MATLAB is first directed to the location of the TIFF files from the near-infrared scan of the aortas, the individual files are then opened, and the pixel intensity values are extracted from the TIFF files.
- Once the image is presented to the user with a color map, select the aorta with the greatest calcification as the scale maximum.
- When MATLAB asks "How many specimens are in the image?" in the command window, input the number of aortas in the current image, and select each aorta one by one. Once an aorta is selected, MATLAB will create a binary image with a mask of the total area of the aorta and another binary image with a mask of the calcified area of the aorta. The values from these masked images are then used to determine the total calcified area, the total area of the aorta, the percentage area positive for calcification, and the mean intensity of the calcified area.
- To select an aorta, make a box around the aorta of interest on the most recent figure on the screen by selecting four corners with one left click and creating a box around the image. To close the box, double-click on the first corner.
NOTE: The threshold may be different depending on the model of calcification used. It is up to the user to determine what value determines a positive signal for vascular calcification. Once the threshold is set for an experiment, it must remain the same throughout the duration of the experiment.
- Once the quantitative data are extracted, perform a one-way ANOVA with Tukey's post-hoc test to show significance between the groups.
2. Isolating and extracting EVs from aortas
NOTE: Once the aortas have been isolated and removed, EVs can be extracted from the tissue. Using a digestive solution and multiple centrifuge cycles, EVs can be collected and used for many different techniques, including calcification assays, gel electrophoresis, and immunoblotting13. The protocol for isolating and extracting EVs is as follows:
- Immediately after scanning the aortas, pool two to three aortas to yield a sufficient protein concentration.
- Prepare a digestive solution containing 0.25 M sucrose, 0.12 M sodium chloride (NaCl), 0.01 M potassium chloride (KCl), 0.02 M Tris hydrochloride, and 600 U/mL collagenase13.
- Incubate two to three pooled aortas in 1.5 mL of digestive solution for 2 h at 37 °C. Collect the solution. Centrifuge the solution at 1,000 × g for 15 min to remove cell debris.
- Centrifuge the supernatant for an additional 30 min at 33,000 × g to remove microvesicles. Collect and assess the supernatant for calcification potential (see section 3).
NOTE: Follow the subsequent steps if gel electrophoresis and protein immunoblotting of the EVs are to be performed. - Ultracentrifuge the supernatant at 100,000 × g for 1 h to isolate the EVs of interest.
- While the supernatant is undergoing ultracentrifugation, prepare RIPA lysis and extraction buffer (see the Table of Materials) by adding the protease inhibitor and vortexing until dissolved.
- Once the ultracentrifugation is complete, aspirate the supernatant, leaving the pellet, which contains the EVs. Suspend the pellet in the prepared RIPA lysis and protease inhibitor mixture. The samples are ready for gel electrophoresis and western blotting.
3. Assessing the calcification potential of extracellular vesicles with light scattering absorbance
NOTE: To measure the real-time mineral formation of EVs, we used an assay originally developed to study mineral formation from growth plate cartilage EVs from cell culture14. Since calcium phosphate deposition is the hallmark of calcification, an increase in light scattering absorbance as the result of calcium phosphate compound formation is indicative of the calcification potential14. In vitro VSMC models are convenient for measuring the calcification potential of EVs. In this technique, a plate reader with a short wavelength filter can quantify the in vitro calcification of EVs. The absorbency reading is recorded at 340 nm, and a higher absorbance is indicative of more calcium phosphate mineral formation. The protocol for the light scattering absorbance assay is as follows:
- Centrifuge the conditioned collected medium of VSMCs at 1,000 × g for 5 min to remove cell debris14.
- Centrifuge the supernatant at 33,000 × g for 30 min, collect the supernatant, and transfer it into a new tube.
NOTE: The presence of EVs in the collected supernatant can be confirmed through dynamic light scattering (DLS) or nanoparticle tracking analysis (NTA) - Prepare a 300 mM sodium phosphate monobasic (NaH2PO4) sterile solution. Filter the solution to ensure sterile conditions if necessary.
- Add 1% (v/v) of 300 mM NaH2PO4 to the EV samples, and gently pipette the solution to mix well. Transfer 200 µL of the mixture solution to a 96-well plate. Incubate the 96-well plate in the microplate reader at 37 °C.
- Set the plate reader to record the absorbance at a wavelength of 340 nm every 1 min.
4. Assessing the calcification mineral formation of extracellular vesicles with collagen hydrogels
NOTE: The aggregation of EVs and the formation of microcalcifications are observed through collagen hydrogels. These hydrogels act as a scaffold that mimics the collagen density observed in vivo15. This demonstrates the effect of collagen on calcification growth. The protocol for assessing the mineral formation of EVs in hydrogels is as follows:
- Day 1
- Prepare a 2.5 mg/mL collagen solution in DMEM. Slowly add 5 M sodium hydroxide to the collagen/DMEM mixture until the color changes to red. Check that the pH of the solution is approximately 7.
NOTE: If the solution turns purple and becomes too basic, restart the process. - Pipette 300 µL of the 2.5 mg/mL collagen solution with a pH of 7 into each well of an 8-well chamber glass. Incubate at 37 °C and 5% CO2 controlled humidity for 72 h.
- Prepare a 2.5 mg/mL collagen solution in DMEM. Slowly add 5 M sodium hydroxide to the collagen/DMEM mixture until the color changes to red. Check that the pH of the solution is approximately 7.
- Day 4
- Add 300 µL of DMEM as a control into the wells.
- Add 300 µL of the EV medium samples collected previously to the remaining wells.
- Incubate at 37 °C and 5% CO2 controlled humidity for 6 days.
- Day 10
- Prepare 2.5 µL of Osteosense 680EX with 22.5 µL of DMEM.
- Pipette 2.5 µL of the Osteosense and DMEM mixture into each well. Incubate at 37 °C and 5% CO2 controlled humidity for 24 h.
- Day 11
- Image the hydrogels with filters appropriate for Osteosense fluorescence. Image them through the bottom of a coverglass chamber using an inverted microscope or from the top with a long working distance objective.
- Assess the number of calcifications and the calcification size in the collected images using the Particle Analysis options available in ImageJ.
- Use the Image | Adjust | Threshold command to binarize the images such that only the Osteosense signal appears white. Use the Analyze | Analyze particles command to obtain information for each calcification in the image. Be sure to use the same threshold parameters for each image analyzed for consistency.
Representative Results
Once the aortas are extracted, imaging using a near-infrared optical scanner shows a visual representation of the aorta as well as the vascular calcification (Figure 1). The pixel intensity values within the scanned fluorescent image represent the distribution of calcification and are shown here using a colored heatmap. Quantification methods include identifying a positive threshold and reporting the percentage area of the aorta with values greater than this threshold value and/or reporting the mean fluorescence intensity of the pixels within the aorta. As shown in Figure 2 and Figure 3, commercially available primary human coronary artery smooth muscle cells were used to demonstrate the techniques. Conditioned media from VSMCs can be used to measure the calcification potential of EVs. Mineral formation over time is measured by the light absorbance at 480 nm using a microplate reader (Figure 2A,B). A linear regression during the rapid phase of mineralization revealed that the absorbance increase occurred 1.5-fold faster in the pro-calcific sample compared to a control sample. Within collagen hydrogels, calcium deposits can be visualized by imaging Osteosense fluorescence (Figure 3A,B). Individual calcifications appear as Osteosense-positive regions within the hydrogel.
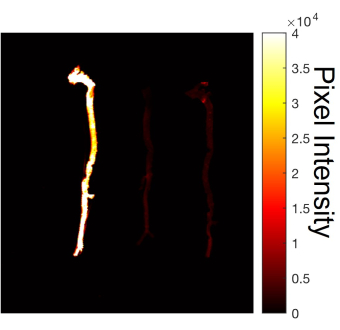
Figure 1: Imaged aortas following dissection. After scanning, calcification can be visualized within the aortas of C57BL/6J mice. A higher Osteosense signal is observed throughout the aorta of a mouse with chronic kidney disease (far left) compared to the two aortas from control mice (right). Please click here to view a larger version of this figure.
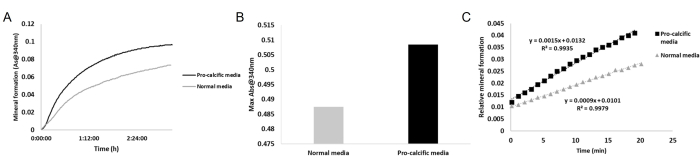
Figure 2: Mineral absorbance to measure the EV calcification potential. (A,B) Light scattering at 340 nm shows a higher absorbance in the conditioned medium obtained from VSMCs cultured in pro-calcific medium (dark line) compared to the normal control medium (light gray line). (C) A linear regression of the first 20 min of the assay shows faster mineral formation in the pro-calcific sample. Abbreviations: EV = extracellular vesicle; VSMCs = vascular smooth muscle cells. Please click here to view a larger version of this figure.

Figure 3: Mineral formation from conditioned media in collagen hydrogels. Conditioned media from (A) control cells and (B) cells cultured for 21 days in a pro-calcific medium were placed in collagen hydrogels. The imaging of the far-red Osteosense fluorescence shows (A) a minimal signal in the control sample and (B) the presence of minerals that formed from the conditioned medium of cells cultured in pro-calcific conditions. Scale = 5 µm. Please click here to view a larger version of this figure.
Supplemental File 1: A custom MATLAB script to quantify the total signal of the calcium tracer. Please click here to download this File.
Discussion
When performing the protocol, it is important to note the critical steps for obtaining successful results. During the isolation of the murine aorta, it is vital that the perfusion is performed properly. When injecting the PBS, care must be taken not to puncture the right ventricle. This would cause the liquid to leak directly out of the ventricle and fail to circulate through the lungs, leaving blood within the aorta. Once the perfusion has been conducted properly and microdissection has begun, all the adipose and fatty tissue must be removed from the aorta before completion and scanning. Remaining fat will cause the aorta to appear larger when normalizing the data to the total tissue size.
Due to the limited size of the mouse aorta during dissection, it is common to pierce or cut the aorta in half. If this occurs, it is recommended to continue the dissection as normal. The aorta pieces can be placed close together for imaging. The aorta can also be placed into the digestive solution in pieces for EV isolation. When beginning the isolation of the EVs, multiple aortas are needed to yield sufficient protein. This limitation means more mice must be dissected to collect the appropriate amount of tissue to produce reliable results.
In the demonstrated method, we show how to measure the mineralization potential directly from EVs in conditioned media from VSMCs in culture. The light scattering protocol is a simple method used to measure the mineral formation of EVs in real time14. This provides insight into the period in which calcification occurs within EVs. The collagen hydrogels provide a platform to direct the aggregation and calcification of EVs for the three-dimensional analysis of mineralization15. Thus far, we have only performed these analyses with EVs from in vitro studies; however, in future studies, we will seek to adapt these methods to analyze the mineralization potential of EVs isolated from mouse aortas.
Cardiovascular disease is the leading cause of death in the world, with calcification being the most significant predictor of morbidity and mortality. By identifying the mechanisms through which EVs lead to calcification, future studies can be conducted focusing on controlling mineral growth as a therapeutic strategy by inhibiting the pathways that lead to the release of calcified EVs, as well as by directly interacting with the mineralization process.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was supported by grants from the National Heart, Lung, and Blood Institute of the National Institutes of Health (NIH) (1R01HL160740 and 5 T32GM132054-04) and the Florida Heart Research Foundation. We would like to thank Kassandra Gomez for her help synthesizing and imaging the hydrogels.
Materials
| 8-well chambered coverglass | Thermo Scientific | 155409PK | |
| 10 mL Syringe | BD | 302995 | |
| 20 G 1 inch Needle | BD | 305175 | |
| Collagen, High Concentraion, Rat Tail | Corning | 354249 | |
| Collagenase | Worthington Biochemical | LS004174 | |
| Curved Forceps | Roboz Surgical Instrument | RS-8254 | |
| Dissection Dish | Living Systems Instrumentation | DD-90-S | |
| Dissection Pan and Wax | United Scientific Supplies | DSPA01-W | |
| DMEM | Cytiva | SH30022.FS | |
| Isoflurane | Sigma-Aldrich | 26675-46-7 | |
| LI-COR Odyssey | LI-COR | DLx | |
| Micro Dissecting Curved Scissors (24 mm Blade) | Roboz Surgical Instrument | RS-5913 | |
| Micro Dissecting Spring Scissors (13 mm Blade) | Roboz Surgical Instrument | RS-5677 | |
| Micro Dissecting Spring Scissors (5 mm Blade) | Roboz Surgical Instrument | RS-5600 | |
| Micro Dissecting Tweezers (0.10 x 0.06 mm Tip) | Roboz Surgical Instrument | RS-4976 | |
| Optima MAX-TL Ultracentrifuge | Beckman Coulter | B11229 | |
| OsteoSense 680EX | Perkin Elmer | NEV10020EX | |
| Pierce Protease Inhibitor | Thermo Scientific | A32963 | |
| Potassium Chloride | Fischer Chemical | P217 | |
| RIPA Lysis and Extraction Buffer | G Biosciences | 786-489 | |
| Sodium Chloride | Fischer Chemical | BP358 | |
| Sodium Hydroxide | Thermo Scientific | A4782602 | |
| Sodium phosphate monobasic | Sigma-Aldrich | S0751 | |
| Sucrose | Sigma | S7903 | |
| Synergy HTX Multimode Reader | Agilent | ||
| Tissue culture plate, 96-well | Thermo Fisher | 167008 | |
| T-Pins | United Scientific Supplies | TPIN02-PK/100 | |
| Tris Hydrochloride | Fischer Chemical | BP153 |
References
- Bakhshian Nik, A., Hutcheson, J. D., Aikawa, E. Extracellular vesicles as mediators of cardiovascular calcification. Frontiers in Cardiovascular Medicine. 4, 78 (2017).
- Ho, C. Y., Shanahan, C. M. Medial arterial calcification: an overlooked player in peripheral arterial disease. Arteriosclerosis, Thrombosis, and Vascular Biology. 36 (8), 1475-1482 (2016).
- Marinelli, A., et al. Diagnosis of arterial media calcification in chronic kidney disease. Cardiorenal Medicine. 3 (2), 89-95 (2013).
- Moe, S. M., Chen, N. X. Mechanisms of vascular calcification in chronic kidney disease. Journal of the American Society of Nephrology. 19 (2), 213-216 (2008).
- Mir, B., Goettsch, C. Extracellular vesicles as delivery vehicles of specific cellular cargo. Cells. 9 (7), 1601 (2020).
- Ruiz, J. L., Hutcheson, J. D., Aikawa, E. Cardiovascular calcification: Current controversies and novel concepts. Cardiovascular Pathology. 24 (4), 207-212 (2015).
- New, S. E., Aikawa, E. Role of extracellular vesicles in de novo mineralization: An additional novel mechanism of cardiovascular calcification. Arteriosclerosis, Thrombosis, and Vascular Biology. 33 (8), 1753-1758 (2013).
- Aikawa, E., Hutcheson, J. D. . Cardiovascular Calcification and Bone Mineralization. , (2020).
- Johnson, T. P. The P-407-induced murine model of dose-controlled hyperlipidemia and atherosclerosis: A review of findings to date. Journal of Cardiovascular Pharmacology. 43 (4), 595-606 (2004).
- Shobeiri, N., et al. Vascular calcification in animal models of CKD: A review. American Journal of Nephrology. 31 (6), 471-481 (2010).
- El-Abbadi, M. M., et al. Phosphate feeding induces arterial medial calcification in uremic mice: role of serum phosphorus, fibroblast growth factor-23, and osteopontin. Kidney International. 75 (12), 1297-1307 (2009).
- Veseli, B. E., et al. Animal models of atherosclerosis. European Journal of Pharmacology. 816, 3-13 (2017).
- Chen, N. X., et al. Transglutaminase 2 accelerates vascular calcification in chronic kidney disease. American Journal of Nephrology. 37 (3), 191-198 (2013).
- Wu, L. N., et al. Physicochemical characterization of the nucleational core of matrix vesicles. Journal of Biological Chemistry. 272 (7), 4404-4411 (1997).
- Hutcheson, J. D., et al. Genesis and growth of extracellular-vesicle-derived microcalcification in atherosclerotic plaques. Nature Materials. 15 (3), 335-343 (2016).

