Murine Intrapulmonary Tracheal Transplantation: A Model for Investigating Obliterative Airway Disease After Lung Transplantation
Summary
The murine intrapulmonary tracheal transplantation (IPTT) model is valuable for studying obliterative airway disease (OAD) after lung transplantation. It offers insights into lung-specific immunological and angiogenic behavior in airway obliteration after allotransplantation with high reproducibility. Here, we describe the IPTT procedure and its expected results.
Abstract
Murine intrapulmonary tracheal transplantation (IPTT) is used as a model of obliterative airway disease (OAD) following lung transplantation. Initially reported by our team, this model has gained use in the study of OAD due to its high technical reproducibility and suitability for investigating immunological behaviors and therapeutic interventions.
In the IPTT model, a rodent tracheal graft is directly inserted into the recipient’s lung through the pleura. This model is distinct from the heterotopic tracheal transplantation (HTT) model, wherein grafts are transplanted into subcutaneous or omental sites, and from the orthotopic tracheal transplantation (OTT) model in which the donor trachea replaces the recipient’s trachea.
Successful implementation of the IPTT model requires advanced anesthetic and surgical skills. Anesthetic skills include endotracheal intubation of the recipient, setting appropriate ventilatory parameters, and appropriately timed extubation after recovery from anesthesia. Surgical skills are essential for precise graft placement within the lung and for ensuring effective sealing of the visceral pleura to prevent air leakage and bleeding. In general, the learning process takes approximately 2 months.
In contrast to the HTT and OTT models, in the IPTT model, the allograft airway develops airway obliteration in the relevant lung microenvironment. This allows investigators to study lung-specific immunological and angiogenic processes involved in airway obliteration after lung transplantation. Furthermore, this model is also unique in that it exhibits tertiary lymphoid organs (TLOs), which are also seen in human lung allografts. TLOs are comprised of T and B cell populations and characterized by the presence of high endothelial venules that direct immune cell recruitment; therefore, they are likely to play a crucial role in graft acceptance and rejection. We conclude that the IPTT model is a useful tool for studying intrapulmonary immune and profibrotic pathways involved in the development of airway obliteration in the lung transplant allograft.
Introduction
Lung transplantation has been established as an effective treatment for patients with end-stage respiratory diseases. However, the median survival rate for human lung transplant recipients is only approximately 6 years, with the development of obliterative bronchiolitis (OB), a type of obstructive airway disease (OAD), being a major cause of death after the first year post transplantation1.
Several animal models have been utilized to investigate the mechanism underlying OAD. One such model is the heterotopic tracheal transplantation (HTT) model2. In this model, tracheal grafts are implanted into the recipient's subcutaneous tissue or omentum. Ischemia-induced loss of tracheal graft epithelial cells occurs, followed by alloreactive lymphocyte infiltration and apoptosis of donor epithelial cells. Fibroblasts and myofibroblasts migrate around the trachea, producing an extracellular matrix. Finally, complete fibrous obliteration of the airway lumen occurs. The HTT model is technically simple, provides an in vivo environment, and offers high reproducibility.
Another model for studying OAD is the rat orthotopic tracheal transplantation (OTT) model, where tracheal grafts are interposed into the recipient's trachea to maintain physiological ventilation3. In this model, ischemia-induced depletion of donor epithelial cells results in their replacement by recipient epithelial cells within the trachea, forming an unobstructed airway accompanied by moderate fibrosis. Although these models have contributed to the understanding of airway obliteration after lung transplantation, they have limitations in terms of recapitulation of the lung parenchymal microenvironment.
Our research group introduced the rat intrapulmonary tracheal transplantation (IPTT) model, where tracheal grafts are implanted into the recipient lung4 (Figure 1). The IPTT model exhibits fibrous obliteration of the airway lumen occurring within the lung microenvironment. Furthermore, it has been successfully applied to mice that are technically more challenging than rat IPTT5,6,7,8,9,10. This adaptation of the murine IPTT model enabled us to delve deeper into the intricate details of the lung immunological environment of OAD after lung transplantation using transgenic mice.
The IPTT model possesses some unique features. One is neoangiogenesis, which is facilitated by pulmonary circulation and plays a crucial role in airway obliteration4,10. Additionally, the IPTT model exhibits lymphoid aggregates, some of which have high endothelial venules expressing peripheral node addressin, indicating that they are tertiary lymphoid organs (TLOs)7,8. TLOs resemble lymph nodes and consist of T cells, B cells, and frequently, a germinal center accompanied by follicular dendritic cells11,12. TLOs have been reported in various chronic inflammatory diseases, including airway obliteration, making the IPTT model suitable for investigating the role of TLOs in airway obliteration7,8,11,12,13. This paper presents the methodology of the murine IPTT model, with the goal of familiarizing researchers with this model and facilitate further investigations into airway obliteration following lung transplantation.
Protocol
All animals were treated in accordance with the guidelines set forth by the Canadian Council on Animal Care in the Guide to the Care and Use of Experimental Animals. The experimental protocol was approved by the Animal Care Committee of the Toronto General Hospital Research Institute, University Health Network.
1. Donor surgery
NOTE: BALB/c mice are used as an example of donors for the experiment. All procedures must be performed utilizing a sterile technique.
- Prior to the procedure, record the weight of each mouse.
- Euthanize the mouse using a CO2 chamber.
- Once death is confirmed, position the mouse in a supine position and secure the limbs with tape.
- Prep the surgical area by sterilizing it with 70% isopropyl alcohol.
NOTE: If needed and as recommended by the local animal ethics committee, clip the fur from the incision site. - Make a midline incision on the skin, starting from the mid-abdomen and extending to the anterior cervical region.
- Access the trachea by carefully retracting the fat pads, laterally moving the strap muscles, and separating the trachea from surrounding connective tissue. Use forceps to create space between the trachea and the esophagus.
- Lift the xiphoid and cut the diaphragm.
- Raise the sternum, ensuring a clear path from the sternum to the neck region by inserting a hemostat. Clamp the rib cage on both sides and cut through the sternum, extending up through the neck muscles.
- Remove the thymus and any fat or muscle obstructing the trachea to expose the tracheal bifurcation.
- Cut both the main bronchi and carefully separate the airway from the esophagus.
- Cut the larynx and remove it.
- Spray the dissected trachea with sterile saline solution or preservation solution with sterile gauze soaked in sterile saline or preservation solution and place it on ice to preserve its viability.
2. Recipient surgery
NOTE: C57BL/6 mice are used as an example of recipients for the experiment.
- Administer sustained-release buprenorphine subcutaneously at a dose of 1 mg/kg on the morning of the surgery day.
- Induce anesthesia in an induction chamber using 5% isoflurane.
- Once the mouse is lightly anesthetized, intraperitoneally inject a cocktail consisting of (0.1 mg/g) xylazine and (0.01 mg/g) ketamine.
- Return the mouse to the induction chamber with 2-3% isoflurane maintained.
- Shave the fur at the surgical site. Also, administer bupivacaine as a line block subcutaneously along the planned insicion site at a dose of 7 mg/kg.
- Confirm the absence of reflex response to a toe pinch before orotracheal intubation. Intubate the mouse orotracheally using a 20 G intravenous catheter and connect it to a ventilator with a tidal volume of 500 µL, a respiratory rate of 120 bpm, 100% oxygen and 2% isoflurane. Use a stand with a clamp applied to the tongue, holding the animal in a vertical position with the neck extended, to facilitate this procedure.
- Activate a heating pad and position the mouse in a right lateral position on top of the pad, with the head away from the surgeon and the tail facing the surgeon (Figure 2). Secure the limbs with tape. Put veterinary ointment on the eyes to prevent dryness while under anesthesia.
- Scrub the surgical area with 7.5% povidone iodine, sterilize with 70% isopropyl alcohol, and re-scrub with 10% povidone iodine. Apply sterile surgical drapes to cover the surgical area.
- Load the donor trachea into a 16 G intravenous catheter during this time (Figure 3C,D).
- Use scalpel to make an incision in the recipient's skin and cauterize the muscle and connective tissue.
- Open the fifth or sixth intercostal space and hold the rib cage open using two retractors.
- Dissect the inferior pulmonary ligament using cotton swabs and scissors.
- Simulate the creation of the pathway for the donor trachea (Figure 3G,H).
- Secure the ventilator outflow tube by partially occluding it with a three-way stopcock to facilitate inflation of the left lung.
- Create a pathway by puncturing the left lung with a 20 G needle. Ensuring that the puncture depth is roughly equivalent to the length of the tracheal allograft. Select the puncture site at the lung's edge (as indicated in Figure 3I), ensuring the pathway runs parallel to the tabletop (as marked by a blue circle in Figure 3J).
NOTE: An upward insertion angle will result in penetration of the pleural layer, while a deeper angle may lead to bleeding from major vessels (as marked by red crosses in Figure 3J). - Insert the 16 G intravenous catheter into the left lung and extrude the donor trachea into the left lung. After insertion of the tracheal allograft, release the three-way stopcock to allow unobstructed expiratory flow through the outflow tube.
- Close the pleural injection site with a clip (Figure 3K,L). Position the clip precisely onto the puncture site, with its edge aligned to match the contour of the lung's edge (indicated by the blue circle in Figure 3L).
NOTE: An incorrect location for the clipping site can result in ineffective sealing and air leakage, while insufficient clip depth may lead to the clip detaching post-surgery (as shown by the red crosses in Figure 3L). - Fill the thoracic cavity with saline solution and absorb the saline with a gauze.
- Reinflate the left lung and close the ribs using a running suture technique.
- Close the muscle and skin with interrupted sutures.
- Administer meloxicam analgesic subcutaneously at a dose of 5 mg/kg at the end of the surgery.
- Observe the recipient mouse until it is awake. Then, remove the tracheal tube and put the recipient mouse in a cage.
NOTE: Recipient mice should be housed individually. - Administer meloxicam (5 mg/kg) once daily via subcutaneous injection, starting 24 h after the surgery and continue for 3 days postoperatively.
3. Collection of samples from recipient mice
- Induce anesthesia in an induction chamber using 5% isoflurane.
- Confirm the absence of reflex response to the toe pinch before orotracheal intubation. The intubation method and respirator setting are the same as in recipient surgery.
- Position the mouse in a supine position and secure the limbs.
- Prep the surgical area by sterilizing it with 70% isopropyl alcohol.
- Make a midline incision on the skin, starting from the mid-abdomen and extending to the anterior cervical region.
- Exsanguinate the mouse via the inferior vena cava using a 1 ml syringe connected to a 25 G needle, resulting in euthanasia.
- Open the chest and access the trachea in the same way as a donor mouse. Tie the trachea around the intubating tube with 7-0 silk.
- Remove the thymus, fat, and muscles to expose the heart.
- Cut the left atrium, right atrium, and inferior vena cava. Perfuse the lungs with 3 mL of sterile saline via the right ventricle.
- For histologic analysis, inflate the lungs with 10% formalin via an intubating tube.
- Extubate the ventilation tube and tie off the trachea with 7-0 silk.
- Divide the larynx and esophagus. Pull them in an inferior direction, and then extract the heart and lung block, placing it into 10% formalin.
Representative Results
Based on our experience, proficiency in this model typically requires approximately 2 months of training. Once proficiency is achieved, the donor procedures typically require 15 min, while the recipient procedures require approximately 30 min. The expected mortality rate for a trained operator is 0%.
In Figure 4A, a tracheal allograft exhibits complete obstruction with fibroblastic tissue, and the epithelial cells are visibly destroyed. Conversely, in Figure 4B, a tracheal isograft remains patent, and the epithelial cells are preserved.
Figure 5 illustrates a lung in which the tracheal allograft has been transplanted, showing the presence of lymphoid aggregates.
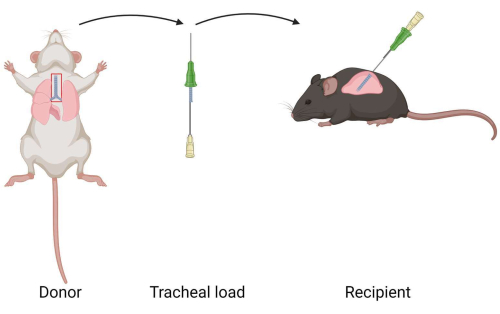
Figure 1: Diagram of murine intrapulmonary tracheal transplantation model. A tracheal allograft is extracted from a donor mouse. The tracheal allograft is loaded into a catheter. The tracheal allograft is transplanted to the lung of a recipient. Please click here to view a larger version of this figure.

Figure 2: Position of the recipient during surgery. The mouse is placed in a right lateral decubitus position. The recipient mouse's head is oriented away from the surgeon and the tail faces the surgeon. Please click here to view a larger version of this figure.
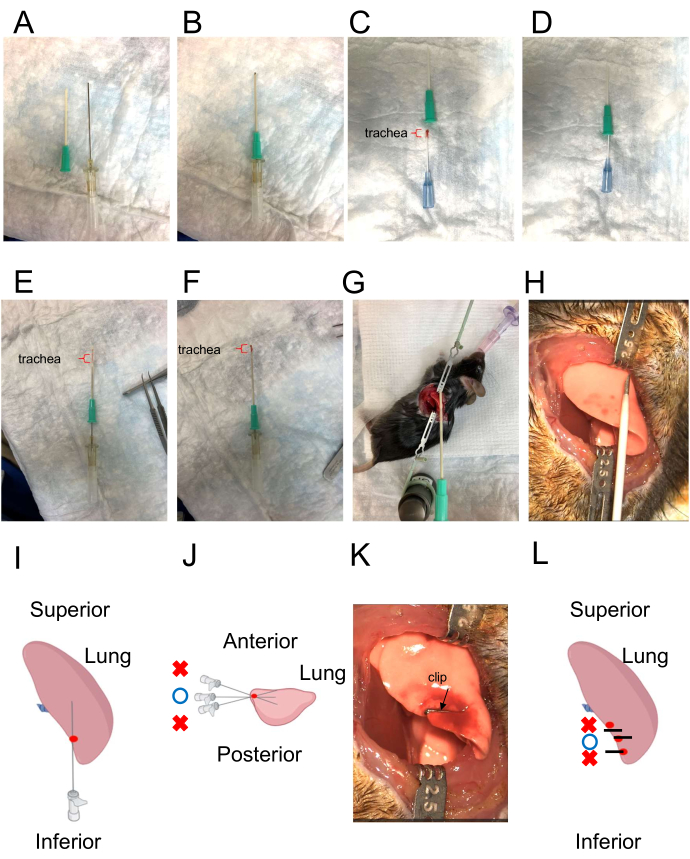
Figure 3: Catheter for tracheal insertion and illustration of the direction of the insertion of the tracheal graft. (A) The catheter for tracheal insertion. The outer catheter has a sharp tip (left), while the inner catheter has a blunt tip (right). (B) Combination of outer and inner catheters. The inner catheter protrudes slightly from the outer catheter. (C,D) Loading the donor trachea into the catheter. The red bracket indicates the tracheal graft. (E) Tracheal graft inside the catheter. The red bracket indicates the tracheal graft. (F) Extruding the inner tracheal graft using the inner catheter as a "pusher." The red bracket indicates the tracheal graft. (G, H) Simulation of the direction for placement of the tracheal graft. (I,J) Creating the pathway using a 22 G needle. The depth should closely match the length of a tracheal allograft. The needle's direction should be opposite from the surgeon and parallel to the tabletop. The correct puncture site is denoted by the red point. The appropriate insertion angle is indicated by the blue circle. Incorrect angles are indicated by red crosses. (K,L) Closing the pleural injection site with a clip. Black lines represent clips. Red point represents the puncture site. The blue circle indicates the correct clipping point. Red crosses indicate incorrect clipping points. Please click here to view a larger version of this figure.
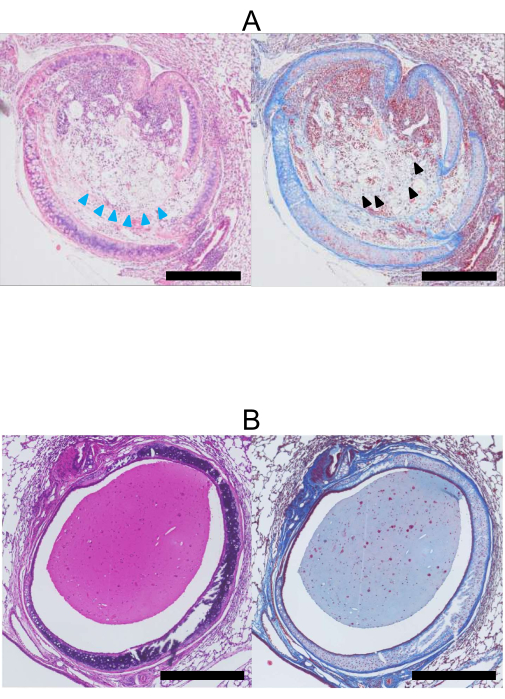
Figure 4: Histopathology at 28 days post lung transplantation. (A) H&E (left) and Masson's Trichrome (right) stained images of a tracheal allograft (Donor: BALB/c, Recipient: C57BL/6). The lumen of the allograft is occluded with collagen and fibrous tissue stained blue by Masson's Trichrome (black arrowhead). Additionally, the epithelial cells have been lost (blue arrowheads). (B) H&E (left) and Masson's Trichrome (right) stained images of a tracheal isograft. In contrast to the allograft, the lumen of the isograft (Donor, Recipient: C57BL/6) remains open, and the epithelial cells are preserved. Stained tissue is mucus. Scale bars = 500 µm. Abbreviation: H&E = hematoxylin and eosin. Please click here to view a larger version of this figure.
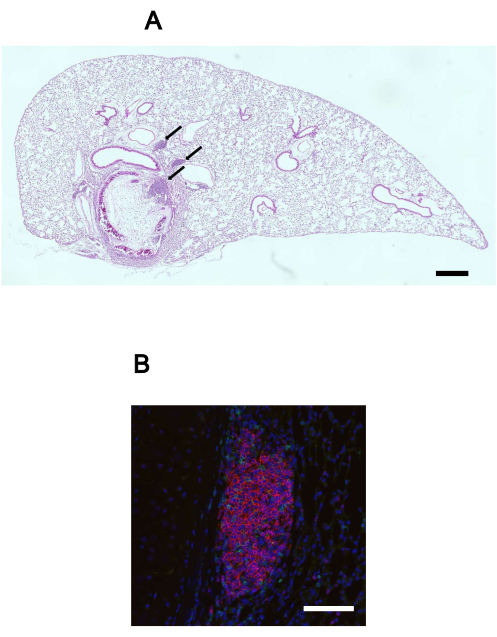
Figure 5: Lymphoid aggregates in the lung with the transplanted tracheal allograft. (A) H&E stained image of a lung with a transplanted tracheal allograft in situ. Lymphoid aggregates (black arrows) are observed within the lung tissue. Scale bar = 500 µm. (B) Immunofluorescence image of a lymphoid aggregate, highlighting the presence of B cell (B220, red), T cell (CD3, green), and nuclei (DAPI, blue). Scale bar = 100 µm. Abbreviations: H&E = hematoxylin and eosin; DAPI = 4',6-diamidino-2-phenylindole. Please click here to view a larger version of this figure.
Discussion
The murine IPTT procedure includes critical steps. Regarding anesthesia, the first crucial step is endotracheal intubation. It is essential to hold the mouse at an appropriate height with its legs on the table to visualize the vocal cords and facilitate immediate intubation. Additionally, careful respiratory volume and positive end-expiratory pressure (PEEP) adjustment is necessary. Typically, a respiratory volume of 500 µL and a PEEP of 2 cmH2O are sufficient for mice weighing 25-30 g. However, larger recipient mice may experience "hiccup" episodes, indicating oxygen deficiency. In such cases, it may be necessary to increase the respiratory volume, PEEP, and temporarily clamp the expiratory tube to recruit the lung. If hiccups persist, the depth of the tracheal tube should be adjusted. Lastly, the timing of tracheal tube extubation is critical, and confirmation of the recipient's respiratory rate is essential to ensure sufficient recovery. Failure to do so may result in postextubation mortality.
Regarding the surgical procedure, proper placement of the transplanted trachea is crucial. Simulating the appropriate needle direction is necessary to accurately position the tracheal graft (Figure 3G). If the path is too shallow, the tracheal graft may penetrate the pleura, while inserting it too deep may lead to lung vessel perforation and severe bleeding. Another critical step is ensuring accurate sealing of the insertion point by securely fixing the pleural incision into which the tracheal allograft was inserted with a stainless steel clip (Figure 3H). Insufficient sealing can cause air leakage, potentially resulting in recipient mortality after chest closure.
While the IPTT model offers some advantages, such as relative simplicity, high reproducibility, and an immunological environment similar to that seen in lung transplantation, it does have some limitations. Tracheal allografts in the IPTT model differ from the clinical situation, where OB occurs in the small airways. Moreover, in this major histocompatibility complex (MHC) mismatch IPTT model, the tracheal allograft is completely occluded with fibroblastic tissue, which is in contrast with clinical OAD where fibroblastic tissue occlusion is usually partial. To address this issue, another strain combination (minor or different major mismatch) might be used for IPTT (depending on the intended study), as it could result in a milder alloimmune response and milder fibroblastic tissue occlusion compared to the BALB/c-to-C57BL/6 combination. We are unaware of any published studies examining different strain combinations, so this possibility would need to be tested empirically.
Finally, it is a nonvascularized graft, similar to the HTT and OTT models. To address this limitation, orthotopic lung transplantation (OLT) can be utilized14. However, the development of OAD-like pathology has been variable in the mouse OLT model15,16,17. Given the IPTT model's relative simplicity and reproducibility compared to OLT, it remains a reasonable model to study the mechanisms of transplant-related OAD. In conclusion, the IPTT model serves as a useful research model for studying intrapulmonary immune and profibrotic pathways involved in the development of airway obliteration after lung transplantation.
Divulgations
The authors have nothing to disclose.
Acknowledgements
The authors would like to thank Jerome Valero for editing this manuscript. Figure 1 and Figure 3I,J,L were created with BioRender.com.
Materials
| BALB/cJ | The Jackson Laboratory | 8-10 weeks 25-30 g | Male, Donor |
| BD 1 mL Syringe | Becton Dickinson | 309659 | |
| BD PrecisionGlide Needle Aiguile BD PrecisionGlide |
Becton Dickinson | 305122 | |
| Bovie Change-A-Tip Deluxe High-Temperture | Bovie | DEL1 | |
| C57BL/6J | The Jackson Laboratory | 8-10 weeks 25-30 g | Male, Recipient |
| Dumont #5/45 Forceps | F·S·T | 11251-35 | |
| Ethicon Ligaclip Multiple -Clip Appliers- | Ethicon | LX107 | |
| Extra Fine Graefe Forceps | F·S·T | 11150-10 | |
| Glover Bulldog Clamp | Integra | 320-127 | |
| Halsted-Mosquito Hemostats | F·S·T | 13009-12 | |
| Horizon Titanium Ligating Clips | Teleflex | 001201 | |
| Leica M651 Manual surgical microscope for microsurgical procedures | Leica | ||
| Magnetix Fixator with spring lock | CD+ LABS | ACD-001 | |
| Microsurgical Scissor | Jarit | 277-051 | |
| Mouse and Perinatal Rat Ventilator Model 687 | Harvard | 55-0001 | |
| Perfadex Plus | XVIVO | 19850 | |
| Retractor Tip Blunt – 2.5 mm | CD+ LABS | ACD-011 | |
| small animal table | CD+ LABS | ACD-003 | |
| Surgipro Blue 24" CV-1 Taper, Double Armed | Covidien | VP702X | |
| Systane ointment | Alconn | 1444062 | |
| System Elastomer | CD+ LABS | ACD-007 | |
| Terumo Surflo IV Catheter, 20 G x 1 in | Terumo Medical Corporation | SR-OX2025CA | |
| VMT table Top | benson | 91803300 |
References
- Chambers, D. C., et al. The International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation: Thirty-fifth adult lung and heart-lung transplant report-2018; Focus theme: Multiorgan Transplantation. J Heart Lung Transplant. 37 (10), 1169-1183 (2018).
- Hertz, M. I., Jessurun, J., King, M. B., Savik, S. K., Murray, J. J. Reproduction of the obliterative bronchiolitis lesion after heterotopic transplantation of mouse airways. American J Pathol. 142 (6), 1945-1951 (1993).
- Ikonen, T. S., Brazelton, T. R., Berry, G. J., Shorthouse, R. S., Morris, R. E. Epithelial re-growth is associated with inhibition of obliterative airway disease in orthotopic tracheal allografts in non-immunosuppressed rats. Transplantation. 70 (6), 857 (2000).
- Dutly, A. E., et al. A novel model for post-transplant obliterative airway disease reveals angiogenesis from the pulmonary circulation. Am J Transplant. 5 (2), 248-254 (2005).
- Wagnetz, D., et al. Rejection of tracheal allograft by intrapulmonary lymphoid neogenesis in the absence of secondary lymphoid organs. Transplantation. 93 (12), 1212-1220 (2012).
- Hirayama, S., et al. Local long-term expression of lentivirally delivered IL-10 in the lung attenuates obliteration of intrapulmonary allograft airways. Hum Gene Ther. 22 (11), 1453-1460 (2011).
- Watanabe, T., et al. Recipient bone marrow-derived IL-17 receptor A-positive cells drive allograft fibrosis in a mouse intrapulmonary tracheal transplantation model. Transpl Immunol. 69, 101467 (2021).
- Matsuda, Y., et al. Spleen tyrosine kinase modulates fibrous airway obliteration and associated lymphoid neogenesis after transplantation. Am J Transplant. 16 (1), 342-352 (2016).
- Suzuki, Y., et al. Effect of CTLA4-Ig on Obliterative bronchiolitis in a mouse intrapulmonary tracheal transplantation model. Ann Thorac Cardiovasc Surg. 27 (6), 355-365 (2021).
- Watanabe, T., et al. A potent anti-angiogenic factor, vasohibin-1, ameliorates experimental bronchiolitis obliterans. Transplant Proc. 44 (4), 1155-1157 (2012).
- Aloisi, F., Pujol-Borrell, R. Lymphoid neogenesis in chronic inflammatory diseases. Nat Rev Immunol. 6 (3), 205-217 (2006).
- Cupedo, T., Jansen, W., Kraal, G., Mebius, R. E. Induction of secondary and tertiary lymphoid structures in the skin. Immunity. 21 (5), 655-667 (2004).
- Sato, M., et al. The role of intrapulmonary de novo lymphoid tissue in obliterative bronchiolitis after lung transplantation. J Immunol. 182 (11), 7307-7316 (2009).
- Okazaki, M., et al. Maintenance of airway epithelium in acutely rejection orthotopic vascularized mouse lung transplants. A J Resp Cell Mol Biol. 37 (6), 625-630 (2007).
- Yamada, Y., et al. Chronic airway fibrosis in orthotopic mouse lung transplantation models-an experimental reappraisal. Transplantation. 102 (2), e49-e58 (2018).
- Watanabe, T., et al. A B cell-dependent pathway drives chronic lung allograft rejection after ischemia-reperfusion injury in mice. Am J Transplant. 19 (12), 3377-3389 (2019).
- Guo, Y., et al. Vendor-specific microbiome controls both acute and chronic murine lung allograft rejection by altering CD4+Foxp3+ regulatory T cell levels. Am J Transplant. 19 (10), 2705-2718 (2019).

