The Establishment of a Murine Maxillary Orthodontic Model
Summary
Here we demonstrate step-by-step a manageable, orthodontic tooth movement protocol operated on a murine maxillary model. With the explicit explanation of each step and visual demonstration, researchers can master this model and apply it to their experimental needs with a few modifications.
Abstract
Due to the lack of reproducible protocols for establishing a murine maxillary orthodontic model, we present a reliable and reproducible protocol to provide researchers with a feasible tool to analyze mechanical loading-associated bone remodeling. This study presents a detailed flowchart in addition to different types of schematic diagrams, operation photos, and videos. We performed this protocol on 11 adult wide-type C57/B6J mice and harvested samples on postoperative days 3, 8, and 14. The micro-CT and histopathological data have proven the success of tooth movements coupled with bone remodeling using this protocol. Furthermore, according to the micro-CT results on days 3, 8, and 14, we have divided bone modeling into three stages: preparation stage, bone resorption stage, and bone formation stage. These stages are expected to help researchers concerned with different stages to set sample collection time reasonably. This protocol can equip researchers with a tool to carry out regenerative analysis of bone remodeling.
Introduction
Bone is a highly active reconstructed tissue that adapts its size, shape, and properties through the lifetime of the individual1,2. In addition to hormones, aging, nutrition, and other biological or biochemical factors3, the idea that mechanical load is the most determining factor has garnered general acceptance4,5. Under some circumstances with abnormal mechanical load, the imbalance between bone resorption and bone formation may lead to abnormal bone remodeling and bone disorders. Bone diseases such as disuse osteoporosis and bone loss during long-term bed rest or in the presence of microgravity at spaceflight have a close relationship with abnormal mechanical load6,7,8.
Mechanical load has also been used to treat bone-related diseases such as distraction treatment and orthodontic treatment. Distraction treatment has been used in developmental diseases such as craniosynostosis and mandibular hypoplasia9,10, while orthodontic treatment has been widely used to rectify abnormal teeth position and any malocclusion11. The core of orthodontic treatment is also the management of mechanical load. When the bone tissue is subjected to mechanical load, a highly coordinated bone remodeling process is induced by coupling of bone resorption followed by bone formation, which can move teeth to achieve the orthodontic purpose12,13.
Although orthodontic treatment has been widely applied for clinical practice, as our knowledge of the biological effects of mechanical load is limited, the results of orthodontic treatment are uncontrollable. To overcome these limitations, several animal models such as mouse, rat, rabbit, cat, dog, monkey, and pig have been established to investigate the underlying mechanism of mechanical load-induced bone remodeling (Table 1)14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32. Large animals such as dogs, monkeys, and pigs have some advantages over small animals in orthodontic operation-they have more human-like teeth and dentition so that the surgical procedure is easy to replicate in humans. Additionally, a wide view can reduce the operation difficulty and make it possible to apply a variety of orthodontic schemes33,34. However, large animals are difficult to obtain, leading to challenges related to sample size, and they are subject to ethical restrictions35. Furthermore, routine extraction procedures and complex instruments make the experiments difficult to perform due to which large animals are rarely used.
Under such circumstances, rodents are mainly used to establish orthodontic models. Among these models, rats and rabbits have lower operating difficulty and more tooth movement schemes compared with mice. However, the murine model has the unique advantage that there are a large number of genetically modified mice available, which is especially crucial for investigating the underlying mechanisms36. However, the murine model is the most difficult model to manipulate because of its small size. Reviewing the current methods, moving the first molar in the mesial direction is the only practical method for an orthodontic model. Two devices are mainly used to move the tooth-coil spring and elastic band. Using an elastic band is easier, but the orthodontic force varies greatly, which makes it difficult to obtain stable results.
Xu et al.15 have established a murine model with a coil spring on the mandible. However, due to the mobility of the mandible and the obstructive nature of the tongue, operation on the maxilla is always the first choice for both intraoperative and postoperative considerations. Taddei et al.16 described a more detailed protocol on the murine maxilla 10 years ago and more visual and pellucid details should be added. In summary, this protocol has systematically described a detailed orthodontic tooth movement protocol in a murine maxillary model to help researchers master the modeling method in a standardized way and enable the comparative evaluation between different studies.
Protocol
The animal procedures in this study were reviewed and approved by the Ethical Committee of the West China School of Stomatology, Sichuan University (WCHSIRB-D-2017-041). Adult C57BL/6 mice were used in this study (see the Table of Materials). This protocol adds mechanical load to the right maxillary first molar (M1) for mesial movement where a highly coordinated bone remodeling process is induced by coupling of bone resorption and bone formation (Figure 1).
1. Preoperative preparation
- Surgical items
- Prepare the following surgical items for the operation: surgical platform (Figure 2A), fastener (Figure 2B), surgical instruments (Figure 2C and Supplementary Figure S1), orthodontic supplies (Figure 2C), and dental restoration supplies (Figure 2D).
NOTE: The customized coil spring is custom-made and provides a force of 10cN when stretched to 10 mm.
- Prepare the following surgical items for the operation: surgical platform (Figure 2A), fastener (Figure 2B), surgical instruments (Figure 2C and Supplementary Figure S1), orthodontic supplies (Figure 2C), and dental restoration supplies (Figure 2D).
- Sterilization
- Sterilize the surgical instruments by autoclaving and all the surgical items with ultraviolet irradiation for at least 30 min.
- Anesthesia
- Anesthetize the mouse by administering ketamine (100 mg/kg) and diazepam (5 mg/kg) by intraperitoneal injection.
- Apply vet ointment to the eyes of the murine with a cotton stick to avoid eye dryness.
- Proceed with the surgery only when the mouse does not respond when its toes are pinched with forceps.
2. Surgical process
- Spread and tape the limbs of the anesthetized mouse in a supine position to the surgical platform using adhesive tape.
- Pin a 27 G needle on either side above the head and another 27 G needle on either side below the axilla.
- Wind a rubber band around the above two needles and the upper incisors and another one around another two needles and the lower incisors. Change the needle positions to control the degree of opening and the orientation of the mouth (Figure 3A).
NOTE: For the orthodontic tooth movement operation, keep the mouth open to the maximum extent before the buccinator becomes completely tight. The tongue should be pulled towards the non-operative side to expose the surgical field and prevent ischemia. - Bend the 1.5 mm end of a 3 cm 304 stainless steel wire and push the bent end through the interproximate space between the M1 and the maxillary second molar (M2) from the buccal side with curved ophthalmic tweezers (Figure 3B). When the palatal end of the ligature wire is seen from the palatal side, pull it out up to about half of its length and pass it through one end of the customized coil spring.
- Tie a square knot with the two ends of the ligature wire in the mesial direction of the maxillary M1 until the spring is firmly fixed to the tooth (Figure 3C). Subtract the excess wire.
- Similarly, pierce a second 3 cm 304 stainless steel wire through the other end of the coil spring.
- Clean and dry the incisors' surfaces with cotton balls. Apply adhesives on all those surfaces with cotton sticks and light-cure them.
- Push the second stainless steel wire through the interproximate space between the maxillary incisors and tie a slip knot in the labial direction (Figure 3D). Subtract the excess wire and make the rest of the wire lie close to the tooth surface.
- Inject light-cured resin to cover the knot and incisors; light-cure the resin (Figure 3E).
3. Postoperative management
- After surgery, inject the mice with 0.05 mg/kg buprenorphine intraperitoneally for postoperative analgesia.
- Place the anesthetized mouse on a 37 °C thermostatic electric blanket. When the murine regains consciousness with ambulation, return it to a separate housing cage.
- Due to the limited functioning of the incisors after the surgery, replace regular hard fodder with only a soft diet.
- Check the orthodontic appliances every day. If any condition are observed during the inspection that affects the conduction of orthodontic force, such as spring deformation, spring loosening, and device falling off, the mouse should be excluded from the experiment.
- In order to maintain the comparability of experiments, assess the weight of the mice daily post-surgery. Any mice exhibiting a weight loss exceeding 30% of their preoperative weight must be excluded from the experiment.
Representative Results
We have performed the OTM surgery on 11 adult male mice (C57/BL6, 3 months old). They were euthanized for results on days 3, 8, and 14 post surgery. In these experiments, the right maxillary side is the operation side, while the left maxillary side is the control side. The micro-CT showed that there was a temporal consecutive increase in the distance between M1 and M2: 30 µm, 70 µm, and 110 µm at days 3, 8, and 14 post surgery, respectively (Figure 4). The low-density periodontal ligament showed broadening at the distal side and narrowing at the mesial side of roots as a result of the mechanical load (Figure 5). Further, the periodontal ligament was continuous and there was no absorption occurring in any root. These results prove that it is feasible and safe to move the M1 physically with this protocol.
Furthermore, we have analyzed the bone area enclosed within the roots of M1 with parameters shown in Figure 6. The percentage of bone volume and bone mineral density of the operation side on day 8 showed a significant decrease compared with the control side (Figure 6A,B). In contrast, the percent bone volume of the operation side on days 3 and 14 showed a significant increase compared with the operation side on day 8 (Figure 6A). These results suggest that bone remodeling is inactive before day 3 post surgery. After day 3 post surgery, bone absorption starts to dominate the bone remodeling process. After day 8 post surgery, bone formation gains an edge in bone remodeling and the alveolar bone almost returns to the physiological level, which also implies that tooth movement almost comes to a stop. By day 14 of this protocol, the bone remodeling enclosed within the roots of M1 goes through three stages, which can roughly be divided into the preparation, bone resorption, and bone formation stages. Researchers can thus study different stages of bone remodeling with this model.
Figure 7 shows the results of hematoxylin-eosin staining and Masson-trichrome staining. We chose the alveolar bone between the mesial buccal root (MB) and the distal buccal root (DB) of M1 as the region of interest. The periodontal ligaments at the distal end of the MB and the mesial end of the DB are the fronts of force transmission of the bone region of interest. The control side of each group showed a similar manifestation: These periodontal ligaments shared a similar width with wave-like fiber and spindle-shaped cells in alignment, and the surface of the alveolar bone was intact linear. This suggests that the periodontal tissues enclosed within the roots of M1 were not subjected to unbalanced and excessive mechanical load under physiological conditions.
On day 3 post surgery, the periodontal ligament fiber was stretched tightly at the tension side, while the periodontal ligament fiber was compressed with morphological ambiguity. Hyalinization was noted in the area of greatest pressure. The surface of the alveolar bone had still retained its integrity on both sides. Consistent with the micro-CT results, in the initial 3 days post surgery, M1 moved within the alveolar socket by compressing the periodontal ligament on the pressure side, while bone resorption or formation was not yet observed.
On day 8 post surgery, the periodontal ligaments on both sides showed the same features as those on day 3 although the surface of the alveolar bone had begun to look rough. Moreover, the marrow cavity was enlarged and the number of trabecular bones seemed to reduce as seen in the CT data. Therefore, on day 8 post surgery, the histopathological phenotype of bone remodeling shows an increase in bone resorption. The alveolar bone also indicates that M1 moves at a high speed.
On day 14 post surgery, the widths of the periodontal ligaments on both sides seemed to be almost equal. The surface of the alveolar bone had become much rougher compared with that on day 8 post surgery. However, the bone was restored to the physiological level on the control side, which was also indicated by the CT data. This stage shows that bone formation dominated the bone modeling process. As the mechanical load was applied only once at the time of the operation, the load decreased as the moving distance increased. As the alveolar bone returned to normal, the movement of M1 also stopped.
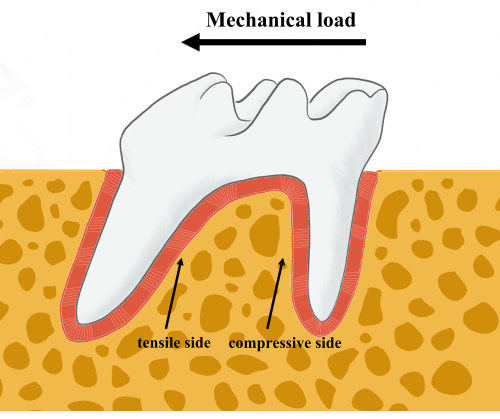
Figure 1: Schematic representation of tooth movement. When a mechanical load is applied to the molar, the tensile and compressive sides of the alveolar bone remodeling can be defined. The thick arrow indicates the direction of the mechanical load. Thin arrows indicate the tensile and the compressive sides of the bone remodeling front. Please click here to view a larger version of this figure.
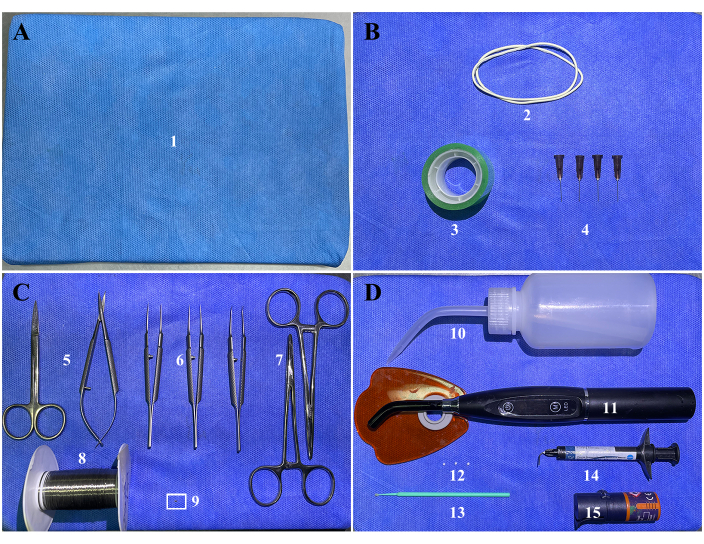
Figure 2: Surgical items. (A) (1) Surgical platform: a foam board or corkboard wrapped in medical non-woven fabric. (B) Fasteners: (2) two rubber bands, (3) tape, and (4) four 27 G needles. (C) Surgical instruments and orthodontic supplies: (5) surgical scissors, (6) ophthalmic tweezers, (7) needle holders, (8) 304 stainless steel wire, and (9) a customized coil spring. The white rectangle refers to the customized coil spring. Enlarged versions of the spring with and without force are shown in Supplementary Figure S1. (D) Dental restoration supplies: (10) air pump bottle, (11) light curer, (12) cotton balls, (13) cotton sticks, (14) light-cured fluid resin, and (15) adhesives. Please click here to view a larger version of this figure.
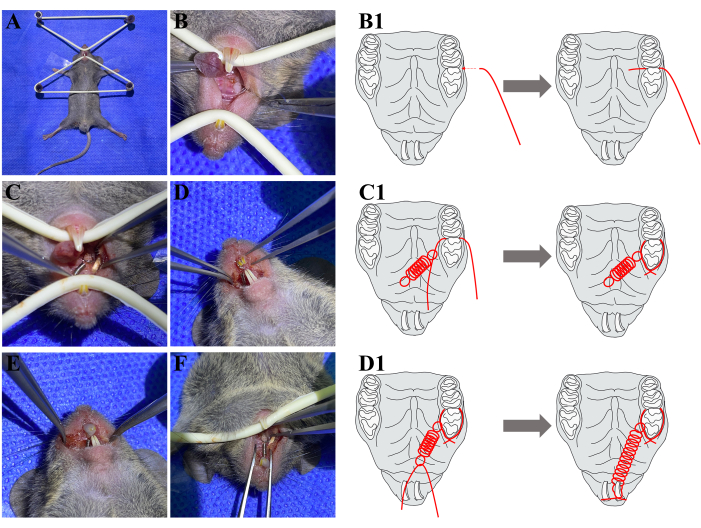
Figure 3: Surgical process. (A) Fasten the mouse to the surgical platform. (B) Push the 304 stainless steel wire through the interproximate space between M1 and M2 from the buccal side. (B1) A schematic diagram has been added to help understanding. (C) A coil spring is fastened to M1 and no occlusal interference occurs at M1. (C1) A schematic diagram has been added to help understanding. (D) The other end of the coil spring is fastened to the ipsilateral upper incisor. (D1) A schematic diagram has been added to help understanding. (E) Apply fluid resin to wrap the incisors and stainless steel together. (F) The final view of all the orthodontic appliances. Abbreviations: M1 = the maxillary first molar; M2 = the maxillary second molar. Please click here to view a larger version of this figure.
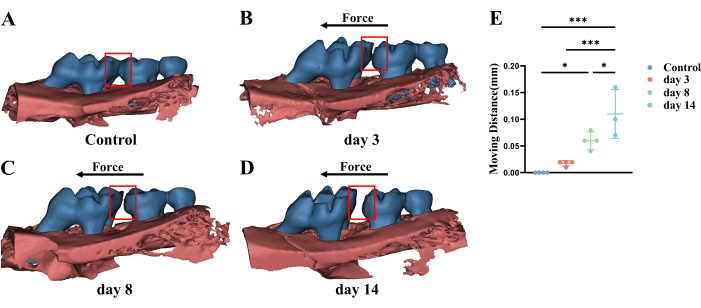
Figure 4: Representative micro-CT three-dimensional images and statistical analysis of different stages of M1 movement. (A) Under physiological circumstances, there is no space between M1 and M2. (B–D) M1 starts to move and the moving distance increases according to the mutual positional relationship between M1 and M2 over time. The red box refers to the distance between M1 and M2. The black arrow refers to the direction of the mechanical load. (E) The statistical analysis of M1 moving distance. Abbreviations: M1 = the maxillary first molar; M2 = the maxillary second molar; OTM = orthodontic tooth movement. Please click here to view a larger version of this figure.
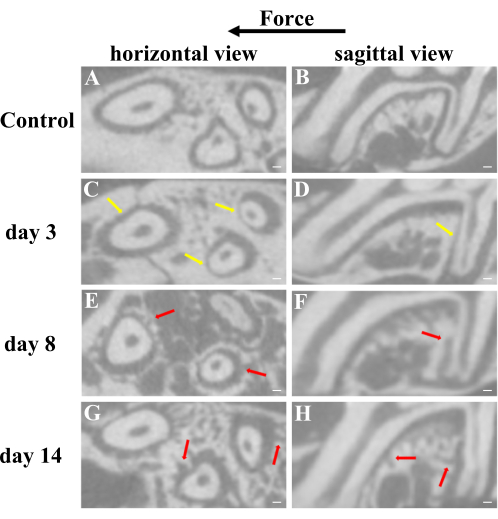
Figure 5: Representative micro-CT two-dimensional images from horizontal and sagittal views of different stages of M1 movement. (A,B) Under physiological circumstances, the low-density periodontal ligament is aequilatus and continuously occupies some space instead of being compressed and the surface of the alveolar bone is intact linear. (C,D) The periodontal ligament is broadening at the distal side and narrowing at the mesial side of roots, which can be observed on day 3 after surgery. (E–H) The lopsided periodontal ligament starts to revert and the surface of the alveolar bone becomes rough as a result of absorption and deposition of bone at days 8 and 14 after surgery. Yellow arrows refer to the compressed periodontal ligament. Red arrows refer to the rough surface of alveolar bone for the absorption and deposition of bone. * P < 0.05; *** P < 0.005. One-way ANOVA. Data are mean ± SD, n ≥ 3. Scale bar = 100 µm. Abbreviations: M1 = the maxillary first molar; M2 = the maxillary second molar; OTM = orthodontic tooth movement. Please click here to view a larger version of this figure.
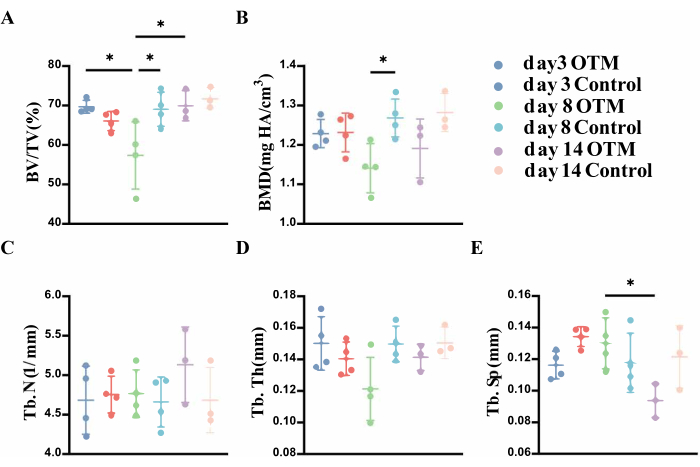
Figure 6: Statistical analysis of alveolar bone enclosed within the roots of M1 at different stages of M1 movement from micro-CT. (A) The significant decrease in the percent bone volume on day 8 indicates the active bone resorption between day 3 and day 8. The significant increase in the percent bone volume on day 14 indicates active bone formation between day 8 and day 14. (B) The significant difference on day 8 in the bone mineral density compared with the control side. also supports the above conclusion. (C–E) Three supplementary indicators were used for evaluation. Few significant differences were found, but the trend still supports the above conclusions. *P < 0.05. One-way ANOVA. Data are mean± SD, n ≥ 3. Abbreviations: M1 = the maxillary first molar; OTM = orthodontic tooth movement; BV/TV = percent bone volume; BMD = bone mineral density; Tb. N = trabecular number; Tb. Th = trabecular thickness; Tb. Sp = trabecular separation. Please click here to view a larger version of this figure.
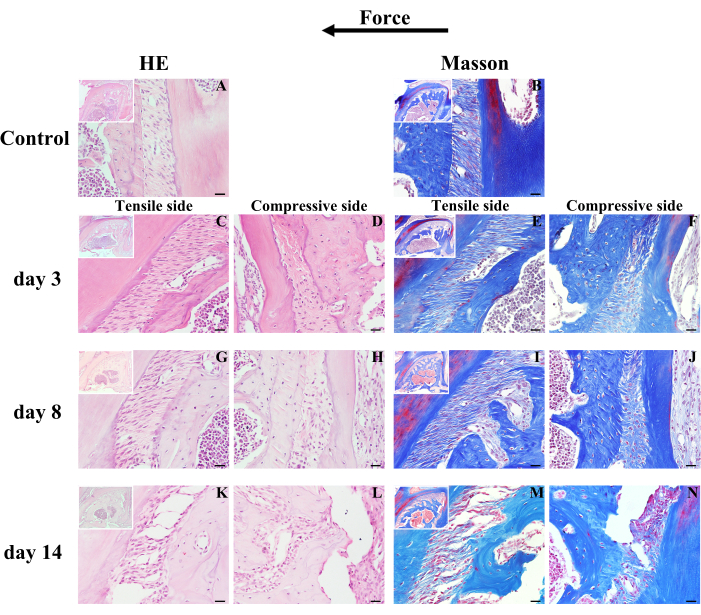
Figure 7: Representative results of hematoxylin-eosin staining and Masson-trichrome staining of the different stages of M1 movement. (A,B) Under physiological conditions, the periodontal ligament fibers are subjected to certain forces with a distinct wave-like shape like "~", and the surface of the alveolar bone is intact linear. When M1 is subjected to mechanical load, (C,E,G,I,K,M) fiber has been stretched tightly at the tension side, while (D,F,H,J,L,N) periodontal ligament fiber has been compressed with morphological ambiguity. (C–N) The surface of the alveolar bone becomes more and more uneven as bone modeling proceeds. Scale bar = 20 µm. Abbreviations: M1 = the maxillary first molar; OTM = orthodontic tooth movement. Please click here to view a larger version of this figure.
| Species | Moving tooth | Anchorage | Device | Moving direction | Reference |
| Murine | first molar | incisors | coil spring | mesial | 14,15 |
| first molar | second molar | elastic band | mesial | 16 | |
| Rat | first molar | mini implant | coil spring | mesial | 17 |
| first molar | incisors | coil spring | mesial | 18 | |
| second and third molar | contralateral homonymous teeth | spring expansion appliance | buccal | 19 | |
| first molar | second molar | orthodontic wire | mesial | 20 | |
| Rabbit | first premolar | incisors | coil spring | mesial | 21 |
| first premolar | mini implant | coil spring | mesial | 22 | |
| incisor | contralateral homonymous teeth | coil spring | distal | 23 | |
| incisor | contralateral homonymous teeth | Omega loop | distal | 24 | |
| Dog | second premolar and first molar | mini implant | coil spring | mesial | 25 |
| second premolar | canine | coil spring | mesial | 26 | |
| first premolar | mini implant | elastic band | distal | 27 | |
| lateral incisor | canine | elastic band | distal | 28 | |
| Pig | first molar | deciduous third molar and mini implant | coil spring | mesial | 29 |
| first molar | second molar | orthodontic wire | buccal | 30 | |
| Monkey | central incisor | first molar, premolar, canine and lateral incisor | coil spring and orthodontic wire | labial | 31 |
| Cat | canine | mini implant | coil spring | mesial | 32 |
Table 1: Summary of the existing animal orthodontic models. The table lists the commonly used models of conventional laboratory animals that focus on simple orthodontic tooth movement. They always consist of three elements: the target moving tooth, the anchorage, and the connecting device to add mechanical load. Various orthodontic programs have been derived by changing the three elements. Complex orthodontic tooth movements with multiple teeth have been excluded.
Supplementary Figure S1: Enlarged versions of the spring. (A) Without and (B) with mechanical load. Scale bar = 5 mm. Please click here to download this File.
Supplementary Figure S2: The method of clamping the ligature wire with forceps. During protocol step 2.4., the safest and most convenient way to clamp the bend of the ligature wire before piercing is shown here. Please click here to download this File.
Supplementary Figure S3: The scope of resin coating. During protocol step 2.9, the incisor end of the spring (A) without and (B) with a covering with resin is shown here. The resin must not be added to the elastic part. Please click here to download this File.
Discussion
In this paper, we tried to describe the simplest orthodontic tooth movement protocol on murine maxillary model step by step to study the latent mechanisms of mechanical load-induced bone remodeling. Apart from research on bone remodeling, there are some other mainstream applications of this method: 1) methodological research on the acceleration of orthodontic tooth movement; 2) research on orthodontic root resorption; 3) biological mechanisms of orthodontic tooth movement and pain; 4) research on the transgenic model.
Compared with other mechanical loading-associated treatments such as mandibular distraction osteogenesis37, orthodontic tooth movement is the simplest and mildest method without wound and bleeding. Moreover, the murine model has the advantages of being easy to operate on with less time and less cost38. The maxillary model can provide a wide visual field and stable fixation during operation and the least interference on the appliance from the tongue after operation14.
Based on the model established here, we have further described three representative time points. Tooth movement could be macroscopically measured from the third postoperative day and the moving distance increases with time. On day 3 after the operation, mechanical load was added to the bone through the periodontal ligament fiber without any obvious changes in the bone. On day 8 after the operation, bone remodeling had already started and bone resorption was in the dominant position, while bone formation was dominant on day 14 after the operation. This model can show the characteristics of different stages of bone remodeling during orthodontic tooth treatment.
There are some critical operation steps to be considered. Before protocol step 2.7, the head of the mouse should be towards the operator for a better surgical field of view. After protocol step 2.4, the area of operation is near the incisors and the tail of the mouse must be toward the operator. When the stainless steel wire must be pushed through the interproximate space between M1 and M2 from the buccal side, prebending is necessary to locate the target area safely and reduce the space occupied by the instruments in the mouth. The bending angle should be >45° to make sure that the stainless steel wire cannot pierce the gingiva when passing through the interproximate space. Piercing in a parallel manner is the way of least resistance. The occlusal wire piercing in a small angle can also be led to the palatal side by the smooth and tough tooth surface. The cusp of the curved ophthalmic tweezer should clamp the bend to reduce the occupied space in the mouth and make it convenient for exertion (Supplementary Figure S2).
As the stainless steel wire may be unable to pass through the interproximate space between the maxillary incisors, toothed ophthalmic tweezers are helpful for incisor separation. Additionally, a square knot is not necessary as resin bonding is the major method of retention here. A slip knot can be made almost close to the tooth surface where a square knot will increase the volume of the coating resin.
However, this model has its shortcomings as well. The orthodontic appliances may be destroyed by the mice because of the sensation of the presence of foreign material in the mouth. The part of the molar side stays beneath the occlusal plane, which is difficult to destroy. However, the lower incisors bite exactly on the fixing part of the incisor side, including the end of the coil spring. Therefore, we suggest that all surfaces of both upper incisors should be wrapped in resin to Increase the retention force. The incisor end of the spring-the weakest part-may be covered in resin (Supplementary Figure S3). In conclusion, this protocol has demonstrated the details of orthodontic tooth movement operated on the murine maxillary model step by step. With the explicit explanation of each step and visual demonstration, researchers can master this model and apply it to their experimental needs with a few modifications.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This work was supported by the National Natural Science Foundation of China grant 82100982 to F.L.
Materials
| Experimental Models: Mouse Lines | |||
| C57/B6J | Gempharmatech Experimental Animals Company | C57/B6J | |
| Critical Commercial Assays | |||
| Hematoxylin and Eosin Stain Kit | Biosharp | BL700B | |
| Masson’s Trichrome Stain Kit | Solarbio | G1340 | |
| Instruments | |||
| 27 G needle | Chengdu Xinjin Shifeng Medical Apparatus & Instruments Co. LTD. | SB1-074(IV) | |
| Adhesives | Minnesota Mining and Manufacturing Co., Ltd. | 41282 | |
| Corkboard | DELI Group Co., Ltd. | 8705 | |
| Cotton balls | Haishi Hainuo Group Co., Ltd. | 20120047 | |
| Cotton sticks | Lakong Medical Devices Co., Ltd. | M6500R | |
| Customized coil spring | Chengdu Mingxing Spring Co., Ltd. | 1109-02 | |
| Forceps | Chengdu Shifeng Co., Ltd. | none | |
| Light-cured fluid resin | Shofu Dental Trading (SHANGHAI) Co., Ltd. | 518785 | |
| Light curer | Liang Ya Dental Equipment Co., Ltd. | LY-A180 | |
| Medical adhesive tapes | Haishi Hainuo Group Co., Ltd. | 0008-2014 | |
| Medical non-woven fabric | Henan Yadu Industrial Co., Ltd. | 01011500018 | |
| Needle holders | Chengdu Shifeng Co., Ltd. | none | |
| Rubber bands | Haishi Hainuo Group Co., Ltd. | 32X1 | |
| Surgical scissors | Chengdu Shifeng Co., Ltd. | none | |
| Tweezers | Chengdu Shifeng Co., Ltd. | none |
References
- Kenkre, J. S., Bassett, J. The bone remodeling cycle. Annals of Clinical Biochemistry. 55 (3), 308-327 (2018).
- Feng, X., McDonald, J. M. Disorders of bone remodeling. Annual Review of Pathology. 6 (1), 121-145 (2011).
- Alliston, T. Biological regulation of bone quality. Current Osteoporosis Reports. 12 (3), 366-375 (2014).
- Duncan, R. L., Turner, C. H. Mechanotransduction and the functional response of bone to mechanical strain. Calcified Tissue International. 57 (5), 344-358 (1995).
- García-Aznar, J. M., Nasello, G., Hervas-Raluy, S., Pérez, M. &. #. 1. 9. 3. ;., Gómez-Benito, M. J. Multiscale modeling of bone tissue mechanobiology. Bone. 151 (10), 1-12 (2021).
- Rolvien, T., Amling, M. Disuse osteoporosis: clinical and mechanistic insights. Calcified Tissue International. 110 (5), 592-604 (2022).
- Vico, L., Hargens, A. Skeletal changes during and after spaceflight. Nature Reviews Rheumatology. 14 (4), 229-245 (2018).
- Iwaniec, U. T., Turner, R. T. Influence of body weight on bone mass, architecture and turnover. Journal of Endocrinology. 230 (3), R115-R130 (2016).
- Governale, L. S. Craniosynostosis. Pediatric Neurology. 53 (5), 394-401 (2015).
- Sahoo, N. K., Issar, Y., Thakral, A. Mandibular Distraction osteogenesis. Journal of Craniofacial Surgery. 30 (8), e743-e746 (2019).
- Roberts-Harry, D., Sandy, J. Orthodontics. Part 1: Who needs orthodontics. British Dental Journal. 195 (8), 433-437 (2003).
- Li, Y., Jacox, L. A., Little, S. H., Ko, C. C. Orthodontic tooth movement: The biology and clinical implications. Kaohsiung Journal of Medical Sciences. 34 (4), 207-214 (2018).
- Will, L. A. Orthodontic tooth movement: a historic prospective. Frontiers of Oral Biology. 18, 46-55 (2016).
- Xu, H., Lee, A., Sun, L., Naveh, G. R. S. 3D Imaging of PDL collagen fibers during orthodontic tooth movement in mandibular murine model. Journal of Visualized Experiments. (170), e62149 (2021).
- Taddei, S. R., et al. Experimental model of tooth movement in mice: a standardized protocol for studying bone remodeling under compression and tensile strains. Journal of Biomechanics. 45 (16), 2729-2735 (2012).
- Deguchi, T., Takeshita, N., Balam, T. A., Fujiyoshi, Y., Takano-Yamamoto, T. Galanin-immunoreactive nerve fibers in the periodontal ligament during experimental tooth movement. Journal of Dental Research. 82 (9), 677-681 (2003).
- Gudhimella, S., et al. A rodent model using skeletal anchorage and low forces for orthodontic tooth movement. American Journal of Orthodontics and Dentofacial Orthopedics. 155 (2), 254-263 (2019).
- Lira Dos Santos, E. J., et al. Orthodontic tooth movement alters cementocyte ultrastructure and cellular cementum proteome signature. Bone. 153 (12), 116-139 (2021).
- Danz, J. C., Bibby, B. M., Katsaros, C., Stavropoulos, A. Effects of facial tooth movement on the periodontium in rats: a comparison between conventional and low force. Journal of Clinical Periodontology. 43 (3), 229-237 (2016).
- Kohno, T., Matsumoto, Y., Kanno, Z., Warita, H., Soma, K. Experimental tooth movement under light orthodontic forces: rates of tooth movement and changes of the periodontium. Journal of Orthodontics. 29 (2), 129-135 (2002).
- Gad, A. M., Soliman, S. O. Evaluation of systemic Omega-3 PUFAs effect on orthodontic tooth movement in a rabbit model: RCT. Angle Orthodontist. 93 (4), 476-481 (2023).
- Huang, C. Y., et al. Comparison of tooth movement and biological response resulting from different force magnitudes combined with osteoperforation in rabbits. Journal of Applied Oral Science. 29 (2), 20200734 (2021).
- Alhasyimi, A. A., Pudyani, P. P., Asmara, W., Ana, I. D. Enhancement of post-orthodontic tooth stability by carbonated hydroxyapatite-incorporated advanced platelet-rich fibrin in rabbits. Orthodontics & Craniofacial Research. 21 (2), 112-118 (2018).
- Elkattan, A. E., et al. Effects of Different Parameters of Diode Laser on Acceleration of Orthodontic Tooth Movement and Its Effect on Relapse: An Experimental Animal Study. Open Access Macedonian Journal of Medical Sciences. 7 (3), 412-420 (2019).
- von Böhl, M., Maltha, J. C., Von Den Hoff, J. W., Kuijpers-Jagtman, A. M. Focal hyalinization during experimental tooth movement in beagle dogs. American Journal of Orthodontics and Dentofacial Orthopedics. 125 (5), 615-623 (2004).
- Machibya, F. M., et al. Effects of bone regeneration materials and tooth movement timing on canine experimental orthodontic treatment. Angle Orthodontist. 88 (2), 171-178 (2018).
- Deguchi, T., et al. Histomorphometric evaluation of alveolar bone turnover between the maxilla and the mandible during experimental tooth movement in dogs. American Journal of Orthodontics and Dentofacial Orthopedics. 133 (6), 889-897 (2008).
- Tanimoto, K., et al. Experimental tooth movement into new bone area regenerated by use of bone marrow-derived mesenchymal stem cells. Cleft Palate-craniofacial Journal. 52 (4), 386-394 (2015).
- Oltramari, P. V., et al. Orthodontic movement in bone defects filled with xenogenic graft: an experimental study in minipigs. American Journal of Orthodontics and Dentofacial Orthopedics. 131 (3), e10-e17 (2007).
- Verna, C., Dalstra, M., Lee, T. C., Melsen, B. Microdamage in porcine alveolar bone due to functional and orthodontic loading. European Journal of Morphology. 42 (1-2), 3-11 (2005).
- Steiner, G. G., Pearson, J. K., Ainamo, J. Changes of the marginal periodontium as a result of labial tooth movement in monkeys. Journal of Periodontology. 52 (6), 314-320 (1981).
- Celebi, A. A., Demirer, S., Catalbas, B., Arikan, S. Effect of ovarian activity on orthodontic tooth movement and gingival crevicular fluid levels of interleukin-1β and prostaglandin E(2) in cats. Angle Orthodontist. 83 (2), 70-75 (2013).
- Holmes, H. D., Tennant, M., Goonewardene, M. S. Augmentation of faciolingual gingival dimensions with free connective tissue grafts before labial orthodontic tooth movement: an experimental study with a canine model. American Journal of Orthodontics and Dentofacial Orthopedics. 127 (5), 562-572 (2005).
- Wennström, J. L., Lindhe, J., Sinclair, F., Thilander, B. Some periodontal tissue reactions to orthodontic tooth movement in monkeys. Journal of Clinical Periodontology. 14 (3), 121-129 (1987).
- Ibrahim, A. Y., Gudhimella, S., Pandruvada, S. N., Huja, S. S. Resolving differences between animal models for expedited orthodontic tooth movement. Orthodontics & Craniofacial Research. 20, 72-76 (2017).
- Kirschneck, C., Bauer, M., Gubernator, J., Proof, P., Schröder, A. Comparative assessment of mouse models for experimental orthodontic tooth movement. Scientific Reports. 10 (1), 1-12 (2020).
- Ransom, R. C., et al. Mechanoresponsive stem cells acquire neural crest fate in jaw regeneration. Nature. 563 (7732), 514-521 (2018).
- Mardas, N., et al. Experimental model for bone regeneration in oral and cranio-maxillo-facial surgery. Journal of Investigative Surgery. 27 (1), 32-49 (2014).

