Techniques for Rapidly Sampling Six Crucial Organs in Adult Xenopus
Summary
This article presents a guide for sampling six significant and diverse organs in adult Xenopus that can be rapidly and easily accessed: the heart ventricle, liver lobe, pancreas, fat bodies, paired kidneys, and skin.
Abstract
Xenopus has been a powerful model organism for understanding vertebrate development and disease for over a hundred years. While experimental analysis and dissection techniques of the embryo have been well documented, descriptions of adult Xenopus structures and organs, together with techniques for working with adults, have not been updated to take into consideration the requirements of such modern approaches as quantitative proteomics and single-cell transcriptomics. The cell-type and gene-centric perspectives require contrasting observations in embryonic stages to those in adult tissues. The organs of the larva undergo significant changes in their overall structure, morphology, and anatomical location all along the larval to adult transition, most notably during massive metamorphosis remodeling. Establishing robust standards for organ identification and dissection is crucial to ensure datasets resulting from studies performed at different laboratories can be consistent. The present protocol identifies six of the organs in the adult Xenopus, demonstrating methods for dissection and sampling of the heart ventricle, liver, fat body, pancreas, paired kidney, and skin of the adult Xenopus. Depending on the preservation methods, the dissected organs can be used for quantitative proteomics, single cell/nuclei transcriptomics, in situ hybridization, immunohistochemistry, histology, etc. This protocol aims to standardize tissue sampling and facilitate multi-lab investigations of the adult organ systems.
Introduction
Though the "digital dissection" of adult Xenopus” is available1, replicable organ and tissue sampling of adult Xenopus remains challenging without the detailed instruction available for other adult models (e.g. mice2,3,4). This article aims to provide clear guidance for accurate and replicable organ sampling of adult Xenopus similar to what is currently available for their larvae5. An emphasis is placed on ease of completion to maintain maximum freshness and make the protocol accessible to all users.
Though there is a thorough dissection guide for Rana sp.6, as well as numerous classroom dissection guides for other anurans7, no Xenopus dissection and sampling guide is currently available. For those not familiar with sampling practices or amphibian anatomy, the small differences between Xenopus and other anurans render these resources suboptimal for replicable tissue sampling.
Many valuable tissues are not included and are even discarded in the present guide; this is to ensure tissue freshness. Six samples are limited enough to ensure that these tissues can be collected in under an hour after the heart starts beating, regardless of the experience or skill level of the user. More advanced and detailed guides for collecting many other tissues are under preparation as separate companion papers.
For less experienced users, it is always recommended that this protocol be first attempted on animals being euthanized for reasons other than experimentation before sampling any animals that are challenging to replace (i.e., transgenics, animals of advanced age, etc). Ideally, all animals sampled will be healthy and, if female, will not have been ovulated in the past two weeks.
Protocol
All experiments were performed in accordance with the rules and regulations of the Harvard Medical School IACUC (Institutional Animal Care and Use Committee) (IS 00001365_3). The representative results are shown for both a perfused and unperfused mature albino male Xenopus laevis.
1. Experimental preparation
NOTE: If perfusion protocol8 is being followed before sampling, skip to step 2.2.
- Ensure that the research institution has approved the euthanasia technique described in this protocol.
- Prepare a solution of 5 g/L MS-222 (tricaine methanesulfonate) and 5 g/L sodium bicarbonate (see Table of Materials). The volume must be greater than the volume required to cover the animals being euthanized completely. Check the pH to ensure that it is ≥7.
- Perform primary euthanasia by placing the Xenopus in the euthanasia solution; the animal will remain submerged for a total of 1 h.
- Set up the dissection station so that immediately after sampling, all tissues can be rinsed in chilled PBS or 0.7x PBS9 (depending on experimental needs), checked, and trimmed under a 5x (or greater) magnification light. This station must also enable the user to either replace all forceps and scissors or wipe them clean between uses.
- Once the frog has been in the solution for 1 h, primary euthanasia has been completed. Remove the frog and check the loss of pain response by performing a foot pinch.
- Record the appropriate details for the animal, such as species, strain, sex, age, and health status as well as if it was perfused. Weigh the Xenopus and take any additional measurements, such as snout-vent length.
- Place the frog on its back and pin down the limbs proximal to the body (Figure 1).
- Using dissection scissors, cut through the skin, up the midline, and then laterally, making two flaps.
- Referring to Figure 2, identify the linea alba and use forceps to grasp it and pull it away from the coelomic cavity. Cut up through the musculature carefully using scissors. Make two flaps out of the cavity wall. Cut or pin all flaps out of the way.
- Identify the heart which will still be beating. Use dissecting scissors to reduce the coracoid bones (Figure 2) to gain better access to the heart.
NOTE: If the heart has stopped beating before sampling, it should be noted that the sample freshness has been compromised.
2. Sampling
NOTE: If the animal has been perfused, skip to step 2.2.
- Identify the thin pericardium and pull it taut with tissue forceps (Figure 3).
- Using the tip of the iridectomy scissors, gently perforate the pericardium, being careful not to cut the underlying tissues. Peel the pericardium up away from the 3 chambers of the heart.
- Use forceps to grasp the ventricle by the apex, identify where it attaches to the auricles and arterial trunk (Figure 4), and cut it below these attachments (Figure 5). If required, trim the ventricle so that no tissues from the auricles or arterial trunk are visible, and light-colored valve tissue will still be visible inside the ventricle.
NOTE: In unperfused animals, the removal of the ventricle may qualify as secondary euthanasia. - The 3 lobes of the liver will be visible (Figure 6 and Figure 7). Grasp the lip of the left lobe (on the viewer's right) and gently lift it so that the hepatic and cystic ducts are visible (Figure 8). Sample the bottom 1/3 of the lobe below these attachments (Figure 9).
- To gain better access to the tissues of a female frog, it is helpful to remove the ovary. Identify the ovary which is enveloped in a layer of visceral peritoneum called the germinal epithelium. Gently shift the lobes until they are on their respective sides to make the area of attachment visible (Figure 10). These attachments are directly ventral to the paired kidney.
- Using scissors, remove the ovaries as close to the kidneys as possible without damaging them (Figure 11).
- Inspect the medial lobe (also called the anterior lobe) of the liver and note how it connects to the stomach and duodenum through the mesentery and hepatopancreatic duct (also called the common bile duct) (Figure 6, Figure 7, and Figure 8).
- Sever the mesentery, hepatoduodenal ligament using iridectomy scissors as well as the hepatopancreatic duct where it meets the duodenum. Sever the connection of the pancreas and hepatopancreatic duct to the medial lobe of the liver so that no dark liver tissue is attached (Figure 12).
- Grasp the stomach with toothed forceps and the superior end of the pancreas with tissue forceps. Under 5x magnification, gently tease the pancreas off of the stomach (Figure 13).
NOTE: If it does not come away cleanly, the remaining pancreatic tissue will be visible and can be picked off in fragments. Alternatively, the pancreas can be methodically detached using iridectomy scissors and tissue forceps. - Referring to Figure 14A, identify the urinary bladder and remove it, cutting as close to the cloaca as possible. Discard this tissue.
- Referring to Figure 14B, identify the large intestine and pull it taut to sever the large intestine as close to the cloaca as possible. Remove and discard the entire alimentary canal, severing the peritoneum where it attaches to the spleen. The fat bodies will now be fully accessible.
- Tease apart the fat bodies so that they are on their respective sides. The area over the kidney, where the fat body connects to the peritoneum, will be visible. Grasp the base of the left fat body (on the viewer's right) and use scissors to cut it away from the peritoneum, leaving a small margin so that the kidney is not damaged (Figure 15).
- Remove and discard the remaining fat body. The paired kidneys will now be fully visible.
- In female frogs or males with distinct vestigial oviducts, grasp an oviduct and pull it away from the kidney and cloaca (Figure 16). Cut the oviduct where it meets the cloaca and continue to pull it away from the kidney, cutting any clear peritoneal attachments as they become apparent. Discard this tissue.
- Repeat this process with the remaining oviduct.
- The kidneys are still covered with clear peritoneum (retroperitoneal)10. Use forceps to grasp the kidneys and sever the peritoneum at their inferior end.
- Lift the kidneys out of the coelomic cavity, using scissors to sever the peritoneum as close to the kidneys as possible without damaging them (Figure 17).
- Under 5x magnification, cut away excess peritoneum and any other remaining tissue (fat bodies, spleen). If the frog is female, ensure any remaining ovary tissue is removed (Figure 18). If the frog is male, carefully remove the testis and check for a vestigial oviduct, which may not be visible without magnification (Figure 19).
- Remove the pins from the animal, flip it onto its ventrum, and re-pin the animal's limbs.
- Select either hindlimb to sample from and pin the foot of that limb.
- Remove an almond-shaped flap of skin from over the gastrocnemius/tibiofibula (Figure 20).
Representative Results
By utilizing Figure 1 à Figure 20 and following all steps of this protocol, the heart ventricle, the left lobe of the liver, the pancreas, the left fat bodies, paired kidneys, and a flap of skin were cleanly excised within an hour of euthanasia. Within this time, the samples are rinsed and trimmed so that they will appear, as shown in Figure 21.
Figure 1: Pinned Xenopus. A mature female X. tropicalis pinned through each limb. Please click here to view a larger version of this figure.
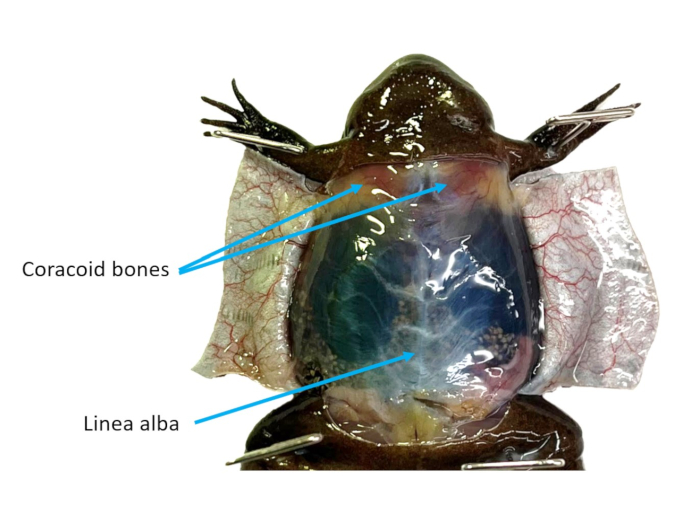
Figure 2: Abdominal wall. The ventral skin of an X. tropicalis female is cut into flaps, making the linea alba and coracoid bones visible. Please click here to view a larger version of this figure.
Figure 3: Pericardium enclosed heart. The apex of the heart ventricle is grasped through the pericardium. Please click here to view a larger version of this figure.
Figure 4: Heart ventricle and arterial trunk. The ventricle of a perfused X laevis, being grasped, showing its attachment to the arterial trunk. Please click here to view a larger version of this figure.
Figure 5: Heart diagram. A diagram of the relevant structures of the heart with a dashed line indicating where to sample the ventricle. Please click here to view a larger version of this figure.
Figure 6: Hepatopancreatic diagram. A diagram of the 3 lobes of the liver, pancreas, and associated organs. Please click here to view a larger version of this figure.
Figure 7: Hepatopancreatic organs. A perfused, albino X. laevis male with 3 lobes of liver, pancreas, and associated organs labeled. Please click here to view a larger version of this figure.
Figure 8: Cystic and hepatic ducts. The left lobe of the liver is being lifted to show the cystic and hepatic ducts in perfused X. laevis. Please click here to view a larger version of this figure.
Figure 9: Liver sampling. The left liver lobe of an unperfused X. tropicalis is severed under the attachments of the hepatic ducts. Please click here to view a larger version of this figure.
Figure 10: Ovary attachment. With the ovary lobes on their respective sides, the continuity of the germinal epithelium to the peritoneal wall (over the kidneys) is visible. Two white dashed lines indicate where to sever these attachments. Please click here to view a larger version of this figure.
Figure 11: Ovary removal. The ovary of an unperfused X. laevis, is pulled away from the paired kidneys. Please click here to view a larger version of this figure.
Figure 12: Mesentery incisions. The coelomic cavity of an unperfused X. laevis, following the sampling of the heart ventricle and left lobe of the liver as well as the removal of the ovary. A white dashed line indicates where to sever the hepatopancreatic ligament and duct, while a green dashed line indicates where to sever the pancreas from the medial lobe of the liver. Please click here to view a larger version of this figure.
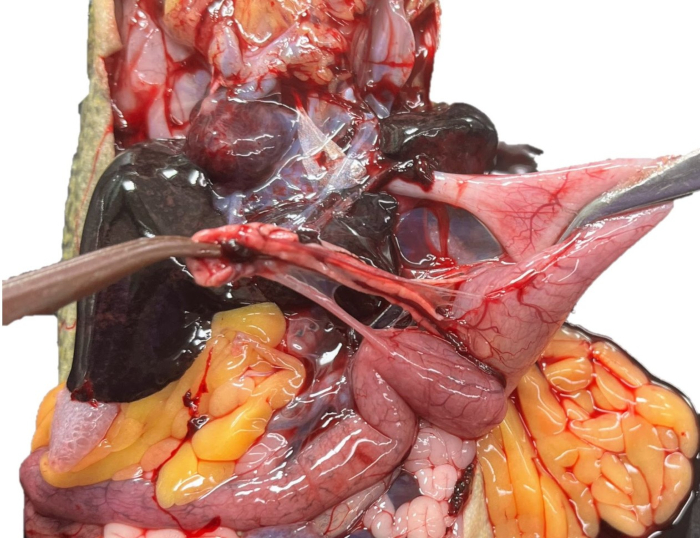
Figure 13: Pancreas sampling. The pancreas of an unperfused X. laevis is being teased off of the stomach. Please click here to view a larger version of this figure.
Figure 14: Organ removal. (A) The urinary bladder of an unperfused X. laevis is pulled away from the cloaca with a dashed line indicating where to cut it. (B) The large intestine of an unperfused X. laevis, is being pulled away from the cloaca with a dashed line indicating where to sever it. Please click here to view a larger version of this figure.
Figure 15: Fat body sampling. The fat bodies, attached to the peritoneum at the superior end of the paired kidneys, are pulled out of the coelomic cavity with a dashed line showing where to cut them. Note that adjacent to this attachment, this male X. tropicalis has 1 testis as well as a pair of distinct vestigial oviducts. Please click here to view a larger version of this figure.
Figure 16: Oviduct removal. The oviduct of a perfused X. laevis is tugged away from the paired kidney, making the clear peritoneum visible. A dashed line indicates where to incise the peritoneum. Please click here to view a larger version of this figure.
Figure 17: Kidney sampling. The paired kidneys of an unperfused X. laevis are being lifted out of the coelomic cavity. Please click here to view a larger version of this figure.
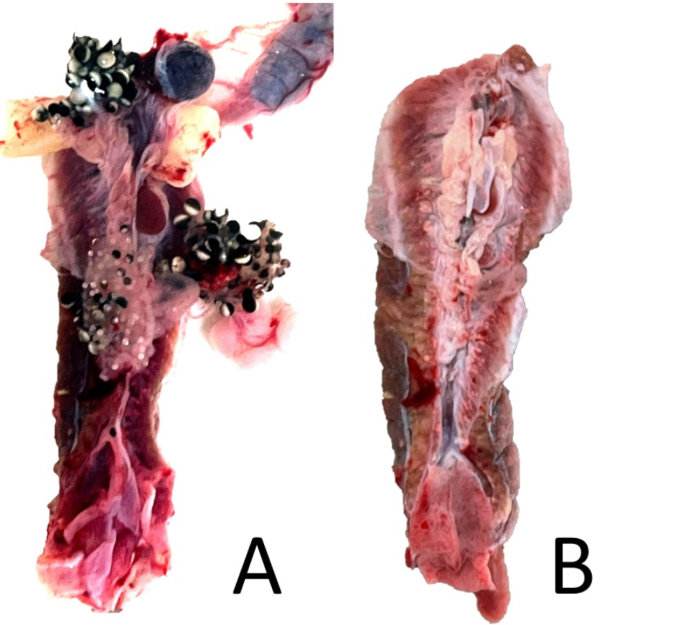
Figure 18: Kidney trimming. (A) A ventral view of an unperfused female X. laevis’s paired kidney with associated peritoneal organs attached. (B) The same kidney with associated organs removed but with some peritoneal tissue remaining. Please click here to view a larger version of this figure.
Figure 19: Testis removal. The paired kidneys of unperfused X. tropicalis with one testis removed. Please click here to view a larger version of this figure.
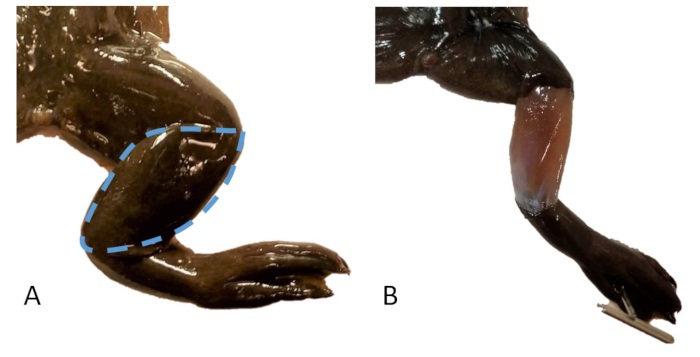
Figure 20: Skin sampling. (A) The right leg of an X. tropicalis with a dashed line indicating the area of skin to be sampled. (B) The right leg of an X. tropicalis with a skin sample removed over the tibiofibula. Please click here to view a larger version of this figure.
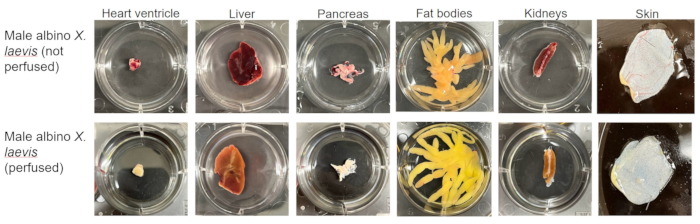
Figure 21: Representative results of organ sampling. Samples of heart ventricle, liver, pancreas, fat body, paired kidney, and skin taken from a perfused and unperfused albino X. laevis. Please click here to view a larger version of this figure.
Discussion
As this protocol aims to maximize freshness, some samples may include undesired tissues. For example, the hepatopancreatic duct and some mesentery are sampled with the pancreas, and some peritoneal tissue, adrenal glands, and ureters will always be sampled with the paired kidneys. If freshness is not a concern, then more precise sampling can be achieved using modified techniques.
The appearance and location of organs are comparable between sexes and species of Xenopus. However, the color of tissues varies significantly based on whether or not the animals have been perfused. It is for this reason that images of both perfused and unperfused animals are included.
A constraint of this protocol is that speed and reproducibility are prioritized over collecting samples that best represent the entirety of the desired tissue. For example, the section of the left lobe of the liver sampled here cannot adequately represent all three lobes of liver tissue. If there are errors in sampling, options for troubleshooting are affected by the potential for variation between different sections of tissue. For example, it is not known if the right lobe of the liver, the right fat body, or a different portion of skin would be functional alternatives to the desired tissues. In these cases, discretion should be used, based on the needs of the research, before substituting sections of tissue.
Another limitation of this protocol is that if the animals being sampled have drastic anatomical defects or clinically significant health issues, the organs in the coelomic cavity may not appear as described here. Granulomas have been found in the tissues of frogs infected with Mycobacterium spp.11,12, and previous cases of ovarian hyperstimulation syndrome appear to lead to an abnormal presentation of organs13.
Though this method has been developed for laboratory Xenopus, there are significant similarities in the appearance of these organs within many non-caecilian amphibians and limbed reptiles14. The sampling portion of this protocol could easily be modified for other models, such as axolotls or the green anole.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This work was supported by NIH's OD grant R24OD031956. We thank Samantha Jalbert, Jill Ralston, and Cora Anderson for their assistance and support as well as our editor and anonymous peer reviewers for their helpful feedback
Materials
| 5x Magnifying Glass with LED Light and Stand | amazon.com | B08QJ6J8P1 | light must not produce heat |
| Disposable Transfer Pipets | VWR | 414004-036 | |
| Dissecting Fine-Pointed Forceps | Fisher Scinetific | 08-875 | |
| Dissecting scissors sharp piont, straight 6.5" | VWR | 76457-374 | |
| Dissection Tray | Fisher Scinetific | 14-370-284 | styrofoam sheets are an acceptable alternative |
| Euthanasia container | US Plastic | Item 2860 | alternative opaque containers acceptable |
| Euthanasia container lid | US Plastic | Item 3047 | |
| Iridectomy Scissors 6" | vwr | 470018-938 | iris scissors are an acceptable alternative |
| MS-222: Syncaine (formerly tricaine) | Pentair AES | TRS1 | |
| PBS 1x | Corning | 21-040-CV | |
| Sodium Bicarbonate, Powder, USP | Fisher Scientific | 18-606-333 | |
| Specimen Forceps, Serrated | VWR | 82027-442 | |
| T-Pins for Dissecting | Fisher Scinetific | S99385 |
References
- Porro, L. B., Richards, C. T. Digital dissection of the model organism Xenopus laevis using contrast-enhanced computed tomography. J Anat. 231 (2), 169-191 (2017).
- Ruehl-Fehlert, C., et al. Revised guides for organ sampling and trimming in rats and mice–part 1. Exp Toxicol Pathol. 55 (23), 91-106 (2003).
- Kittel, B., et al. Revised guides for organ sampling and trimming in rats and mice–Part 2. A joint publication of the RITA and NACAD groups. Exp Toxicol Pathol. 55, 413-431 (2004).
- Morawietz, G., et al. Revised guides for organ sampling and trimming in rats and mice – Part 3 – A joint publication of the RITA and NACAD groups. Exp Toxicol Pathol. 55, 433-449 (2004).
- Patmann, M. D., Shewade, L. H., Schneider, K. A., Buchholz, D. R. Xenopus tadpole tissue harvest. Cold Spring Harb Protoc. 2017 (11), 097675 (2017).
- Lőw, P., Molnár, K., Kriska, G. Dissection of a Frog (Rana sp.). Atlas of Animal Anatomy and Histology. , 213-263 (2016).
- O’Rourke, D. P. Amphibians used in research and teaching. ILAR J. 48 (3), 183-187 (2007).
- Jonas-Closs, R. A., Peshkin, L. Effective rapid blood perfusion in Xenopus. JoVE. (issue), e65287 (2023).
- Balls, M., Worley, R. S. Amphibian cells in vitro. II. Effects of variations in medium osmolarity on a permanent cells line isolated from Xenopus. Exp Cell Res. 76 (2), 333-336 (1973).
- Holz, P. H., Raidal, S. R. Comparative renal anatomy of exotic species. Vet North Am Exot Anim Pract. 9 (1), 1-11 (2006).
- Trott, K. A., et al. Characterization of a Mycobacterium ulcerans-like infection in a colony of African tropical clawed frogs (Xenopus tropicalis). Comp Med. 54 (3), 309-317 (2004).
- Fremont-Rahl, J. J., et al. Mycobacterium liflandii outbreak in a research colony of Xenopus (Silurana) tropicalis frogs. Vet Pathol. 48 (4), 856-867 (2011).
- Green, S. L., Parker, J., Davis, C., Bouley, D. M. Ovarian hyperstimulation syndrome in gonadotropin-treated laboratory South African clawed frogs (Xenopus laevis). J Am Assoc Lab Anim Sci. 46 (3), 64-67 (2007).
- Vitt, L. J., Caldwell, J. P. Anatomy of amphibians and reptiles. Herpetol. Herpetol. , 35-81 (2009).

