Methodology to Metabolically Inactivate Bacteria for Caenorhabditis elegans Research
Summary
The food source for Caenorhabditis elegans in the lab is live Escherichia coli. Since bacteria are metabolically active, they present a confounding variable in metabolic and drug studies in C. elegans. A detailed protocol to metabolically inactivate bacteria using paraformaldehyde is described here.
Abstract
Caenorhabditis elegans is a common model organism for research in genetics, development, aging, metabolism, and behavior. Because C. elegans consume a diet of live bacteria, the metabolic activity of their food source can confound experiments looking for the direct effects of various interventions on the worm. To avoid the confounding effects of bacterial metabolism, C. elegans researchers have used multiple methods to metabolically inactivate bacteria, including ultraviolet (UV)-irradiation, heat-killing, and antibiotics. UV treatment is relatively low-throughput and cannot be used in liquid culture because each plate must be examined for successful bacterial killing. A second treatment method, heat-killing, negatively affects the texture and nutritional quality of the bacteria, leading to the developmental arrest of C. elegans. Finally, antibiotic treatment can directly alter C. elegans physiology in addition to preventing bacterial growth. This manuscript describes an alternative method to metabolically inactivate bacteria using paraformaldehyde (PFA). PFA treatment cross-links proteins within bacterial cells to prevent metabolic activity while preserving cellular structure and nutritional content. This method is high-throughput and can be used in liquid culture or solid plates, as testing one plate of PFA-treated bacteria for growth validates the whole batch. Metabolic inactivation through PFA treatment can be used to eliminate the confounding effects of bacterial metabolism on studies of drug or metabolite supplementation, stress resistance, metabolomics, and behavior in C. elegans.
Introduction
Caenorhabditis elegans was originally proposed as a model organism in 19651 and has since been widely adopted in studies of genetics, development, behavior, aging, and metabolism2. Due to their large brood size and transparent cuticle, C. elegans is particularly well-suited for high-throughput screening with fluorescent reporters3. Their short life cycle, hermaphroditic reproduction, and genetic homology with humans also make C. elegans a valuable model system for studies on development4 and aging biology5. Moreover, C. elegans are relatively easy to maintain. Worms can be grown in liquid culture or on solid agar plates and consume a diet of live Escherichia coli OP50 bacteria4.
However, the live food source of C. elegans can confound studies of metabolism, drug supplementation, and behavior. Because live bacteria have their own metabolism, experimental conditions that affect the bacteria also alter the nutrients and metabolites available to the worms. For example, differences in bacterial iron, amino acid, and folate concentrations have diverse effects on C. elegans' development, physiology, and lifespan6. Many common lab practices can elicit such changes in the nutrient composition and metabolites produced by OP50. Specifically, exposure to 5-fluoro-2'-deoxyuridine (FUdR), a compound commonly used to prevent reproduction in C. elegans, elicits broad changes in OP50 gene expression, including amino acid biosynthesis pathways7. Live bacteria can also confound studies in which C. elegans are supplemented with small molecules because bacteria can partially or completely metabolize the active compounds. Moreover, the effects of these small molecules on the bacteria can, in turn, alter C. elegans physiology, as was reported with the lifespan-extending drug metformin8. Finally, live bacteria can change the worm's environment in ways that alter behavior, such as secreting attractive odorants9, producing exogenous neuromodulators10, and creating oxygen gradients in a dense bacteria lawn11.
To mitigate the confounding effects of bacterial metabolism on C. elegans research, multiple methods for killing bacteria have been developed (Table 1). Three common strategies for killing OP50 are UV-irradiation, heat-killing, and antibiotic treatment. While straightforward and relatively low-cost, each of these methods can have undesirable effects on both bacteria and C. elegans. UV-killing via a UV crosslinker12 is low-throughput and the rate is limited by the number of plates that can fit in the UV crosslinker. Moreover, the efficacy of UV-killing can vary from plate to plate within a batch, and testing for growth on all plates can become difficult in large experiments. Heat-killing OP50 by exposing culture to temperatures of >60 °C comes with a separate set of challenges. High heat can damage nutrients essential for the worm and destroy the cellular structure of bacteria, creating a softer texture that decreases the amount of time worms spend on the food13. This method also cannot be used throughout the life cycle of C. elegans because worms fed heat-killed bacteria can arrest early in development13. Antibiotic treatment is a third common method for suppressing bacterial metabolism14, but antibiotics can also alter worm growth and metabolism15.
One solution to eliminate the metabolic effects of live bacteria while preserving bacterial structure and essential nutrients is to kill OP50 with paraformaldehyde (PFA)16. PFA is a polymer of formaldehyde that can crosslink proteins within cells17 to prevent bacterial replication without destroying internal cell structures like the inner plasma membrane18. Due to this preservation of internal cellular structure, PFA-treated bacteria exhibit no growth or metabolic activity but remain an edible and nutrient-rich food source for C. elegans16. Here, a detailed protocol is provided which shows how to metabolically inactivate bacteria using paraformaldehyde.
| Method | Required Materials | Scalable? | Nutritional? | Effects on Worm? | ||||
| UV | UV-crosslinker | Limited by: | Yes | Variable effects on lifespan on NGM12, 23, 24 | ||||
| Number of plates that fit in UV-crosslinker | Variable effects on lifespan on FUdR24, 26, 27 | |||||||
| Irradiation time per plate | Decreased food preference16 | |||||||
| Ability to check every plate for growth8 | ||||||||
| Heat | >60 °C incubator | Yes | No: destroys cell wall, decreased nutritional value | Developmental arrest 13 | ||||
| Decreased food preference13 | ||||||||
| Extends lifespan on NGM31 | ||||||||
| Antibiotics | Antibiotics (kanamycin, carbenicillin, etc.) | Yes | Yes | Delays growth and development15 | ||||
| Extends lifespan in liquid media19 | ||||||||
| Extends lifespan on NGM15 | ||||||||
| PFA | 0.5% Paraformaldehyde | Yes | Yes | Small brood size decrease16 | ||||
| Small development time increase16 | ||||||||
| Decreased food preference16 | ||||||||
Table 1. Comparisons of methods to kill OP50. UV-killing, heat-killing, antibiotic-treatment, and PFA-treatment have varied effects on the nutritional status of the bacteria and the health of worms fed treated bacteria. These methods for replicatively inactivating E. coli also differ in their required materials and scalability.
Protocol
1. Bacteria inoculation
- Prepare Luria broth (LB) by dissolving 10 g of tryptone, 5 g of yeast extract, and 10 g of sodium chloride (NaCl) in 950 mL of distilled water.
- Adjust the pH of the LB to 7.0 by adding 5M sodium hydroxide (NaOH). This should only require about 0.2 mL of NaOH.
- Autoclave the pH-adjusted LB media on a liquid cycle for 45 min at 15 psi. Allow the solution to cool and store at room temperature.
- Inoculate a single colony of bacteria in 100 mL of LB in a 500 mL Erlenmeyer flask. Culture the bacteria overnight in a 37 °C shaker incubator.
- Depending on the health of the bacterial colony, the size of the flask and the speed that the shaker is set to, the time that it takes for the bacteria to grow may vary. After ~14 h, check the optical density (OD) of the bacteria at 600 nm (OD600).
- Remove the bacteria from the shaker when the OD600 is 3.0 (1 x 109 colony forming units (CFU)/mL). If the OD600 is less than 3.0, return the flask to the shaker incubator until the desired OD is reached.
- Aliquot the bacteria in 50 mL conical tubes and store at 4 °C or proceed to the next step.
2. Working with paraformaldehyde
NOTE: The concentration of paraformaldehyde (PFA) used, and the duration of exposure may vary somewhat depending on climate, location, and type of bacteria being treated. A good starting point for OP50 is exposure to 0.5% PFA for 1 h, whereas 0.25% PFA for 1 h may be sufficient for HT115.
- Prepare 32% PFA stock or use commercially purchased 32% PFA solution. Use proper personal protective equipment (PPE) when working with PFA. Wear gloves and eye protection.
- Add PFA inside a chemical fume hood with proper ventilation. Dispose of PFA containing washes and tips in proper chemical hazard containers in the fume hood.
3. Bacterial treatment with paraformaldehyde
- Once the bacteria reach an OD600 of 3.0, use a serological pipette to transfer 50 mL into a new 250 mL Erlenmeyer flask. Save the rest for a live control, a mock-treated control (see step 4), or to treat as well with PFA as needed.
- Avoid pouring from one flask to another and be cautious not to splash the sides of the new flask with bacteria. Colonies on the side of the flask may receive a lower dose of PFA.
- In the chemical hood, add 781 µL of 32% PFA to 50 mL bacteria to bring the final concentration to 0.5%. Dispose of the tip used in a solid waste chemical hazard container.
- Cover the flask with foil and return to the 37 °C shaker incubator for 1 h. After 1 h, remove the flask from the incubator and proceed to step 5.
4. Mock-treated control
- Once the bacteria reach an OD600 of 3.0, use a serological pipette to transfer 50 mL of the bacteria to a 50 mL conical tube.
- Proceed to step 5.3 to complete the washing steps similar to the PFA-treated group.
5. Washing the bacteria to remove residual PFA
- In the chemical hood, use a serological pipette to transfer the treated bacteria from the Erlenmeyer flask to a 50 mL conical tube. Using a serological pipette instead of pouring the bacteria will prevent contamination from any bacterial colonies on the edge of the flask that may have avoided direct treatment with PFA.
- Centrifuge treated bacteria at approximately 3000 x g for 20 min. Remove the supernatant by disposing of it in a liquid waste chemical hazard container in the chemical hood.
- Add 25 mL of LB and vortex to resuspend the bacterial pellet (Filling the tube fully makes it harder to resuspend the pellet). Repeat centrifugation and pellet resuspension 4x.
- Resuspend the pellet in volumes optimal for different assays. For lifespan assays, seed 60 mm plates with 200 µL of bacteria resuspended in 10 mL of LB. Resuspending the bacteria in 10 mL of LB results in a 5x concentration from the original 50 mL culture.
- Store the bacteria at 4 °C.
6. Quality check of bacterial growth
- After the final wash and resuspension, streak an LB plate (using a sterile pipette tip) with the prepared bacteria. It is good practice to streak the LB used for washing and resuspending on a separate plate as well to make sure that the LB used was not contaminated.
- Place the plates in a 37 °C incubator overnight. Check for any growth. The bacteria are considered replicatively dead when colonies do not grow on the LB plate.
7. Quality check for bacterial metabolism using a respirometer
- After the final wash and resuspension from step 5.4, confirm that the bacteria are metabolically dead using available tools such as respirometers19,20 and measuring the basal oxygen consumption rate (OCR).
- Prepare M9 solution: Dissolve 3 g of potassium phosphate monobasic (KH2PO4), 6 g of sodium phosphate dibasic (Na2HPO4), and 5 g of sodium chloride (NaCl) in 950 mL of distilled water. Autoclave on a liquid cycle for 45 min at 15 psi, then allow the solution to cool to room temperature. Add 1 mL of 1 M magnesium sulfate (MgSO4) and store at room temperature.
- Hydrate the respirometer cartridge: Add 200 µL of calibrant to all wells of a 96-well plate. Place the cartridge in the 96-well plate and incubate overnight in a 37 °C incubator.
- Assay calibration: On the following day, place the hydrated cartridge in the machine and begin calibration.
- Assay test plate setup: Using a new 96-well plate, add 160 µL of M9 and 40 µL of the prepped bacteria (1 x 109 CFU/mL) to test wells. Add 200 µL of M9 to the 4 corner wells to use as blank wells. Add 160 µL of M9 and 40 µL of LB used for washing and resuspending to use as negative controls. Add 200 µL of M9 to the rest of the wells that will not be used.
- Run the assay: Once the cartridge calibration is complete, insert the assay plate from step 7.5 into the machine for analysis. The settings include steps to mix, wait, measure and loop. The results will be shown as oxygen consumption rate (OCR). The bacteria are metabolically dead and ready to use when the OCR is zero.
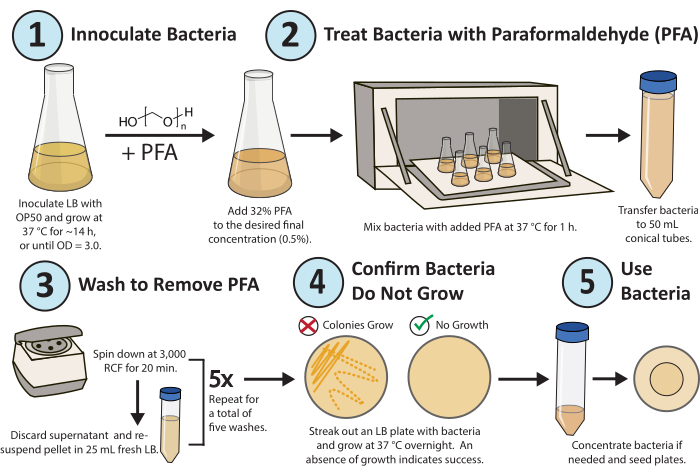
Figure 1. Workflow for paraformaldehyde treatment. A single colony of E. coli OP50 bacteria is grown overnight. PFA is added to a final concentration of 0.5%, and the PFA-treated culture is shaken for 1 h at 37 °C. Finally, the PFA is removed by washing the culture with fresh LB 5x. To confirm the treated bacteria are replicateively inactive, streak out an LB plate of the treated bacteria and grow overnight. Please click here to view a larger version of this figure.
Representative Results
A detailed workflow of the protocol is shown in Figure 1. A high-throughput method was developed and optimized to consistently inactivate bacterial replication (Figure 2A) and metabolism (Figure 2B) for metabolic and drug studies in C. elegans research using paraformaldehyde16. The goal was to determine the lowest concentration of PFA needed and the shortest amount of time required to consistently kill the bacteria without impacting various measures of health in the worms. These values may vary from one location to another depending on lab environment, and from one bacterial strain to another. For example, exposure of HB101 bacteria to 0.25% for 1 h is sufficient to render it metabolically inactive (Figure 2C), while Enterococcus faecalis (EF) requires a 1.0% PFA concentration (Figure 2D) for a consistent effect. An approach for individual lab optimization was previously established16 and is detailed in this current work.
Food attraction and consumption
As bacteria grow and replicate, they release various metabolites and pheromones that are attractive to the worms. To determine whether the worms remain attracted to the PFA-treated OP50, plates were seeded with PFA-treated or mock-treated OP50 and the percentage of worms present on the food lawn as compared to off the lawn was tabulated. Data show that worms are attracted to the PFA-treated OP50, since most of the worms remain on the bacterial lawn after 1 h (Figure 3A), similar to what is observed with the mock-treated control (Figure 3B). However, as expected, a sensitive pairwise assay21 shows that C. elegans have a stronger preference for the mock-treated live control (Figure 3C) when seeded with the PFA-treated on the same plate. Having established that worms are attracted to PFA-killed bacteria, albeit less than live bacteria, it was imperative to establish that they eat the PFA-killed food. To determine whether the worms consume the PFA-treated OP50, the pumping rate of the worms on different food types was measured. As shown in Figure 3D, worms on PFA-treated OP50 have a higher pumping rate (+25%) than worms on the mock-treated control. This indicates that worms are not purposefully slowing their rate of eating on PFA-killed food and could either indicate a higher rate of eating or a compensatory response to a change in the ease of pumping the treated food.
Fecundity, development, and lifespan
Changes in food conditions can have physiological effects on worms13,22. To test for broad effects, changes in the rate of development, fecundity, and lifespan were measured. PFA-treated OP50 slightly delays development time (~4 h) of wildtype N2 worms as shown in Figure 4A. Monitoring the egg-laying capacity of worms grown on different bacterial conditions show a slight but significant decrease in fecundity (-21%) in the worms fed PFA-treated OP50 (Figure 4B). For labs interested in C. elegans aging, the effect of PFA-treated OP50 on longevity was then determined. Interestingly, the lifespan of wildtype worms fed PFA-treated OP50 was not significantly different from the worms fed mock-treated OP50 on standard nematode growth media (NGM) plates (Figure 4C), however, PFA-treated OP50 increases wildtype lifespan (+23%) when FUdR is present (Figure 4D). Feeding PFA-treated OP50 to a long-lived fmo-2 overexpressing (OE) strain20 does not alter its longevity compared to wildtype worms (Figure 4E). UV-, heat-, and antibiotic-killed OP50 have also been shown to alter C. elegans lifespan. Specifically, UV-treated bacteria have been reported to extend lifespan on NGM12,23 and on FUdR24. However, there is conflicting data on the interaction of FUdR and UV-killed bacteria25,26 that may be due to varied methods of UV-killing and validation of metabolic inactivity in the bacteria. Additionally, heat-killed bacteria extend lifespan on NGM27, and antibiotics extend lifespan on NGM15 and in liquid culture28.
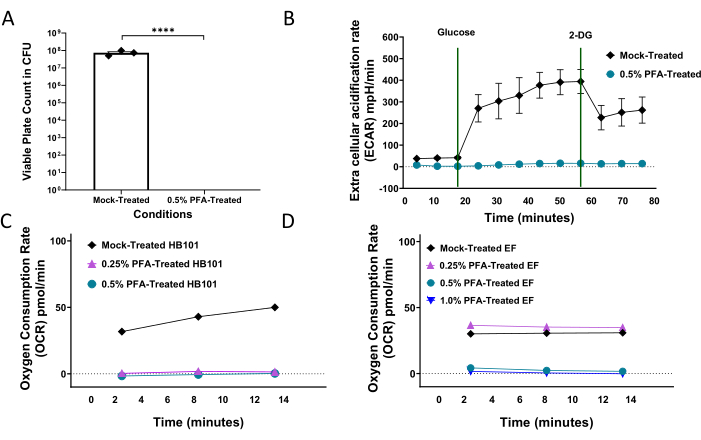
Figure 2. Treatment of bacteria with PFA inhibits proliferation and metabolism. (A) Bacteria growth in colony forming units (CFU) of mock-treated and 0.5% PFA-treated OP50. (B) Extracellular acidification rate (ECAR) in mpH/min of mock-treated and 0.5% PFA-treated OP50. (C) Basal oxygen consumption rate (OCR) in pmol/min of mock-treated, 0.25% PFA-treated, and 0.5% PFA-treated HB101. (D) Basal oxygen consumption rate (OCR) in pmol/min of mock-treated, 0.25% PFA-treated, 0.5% PFA-treated, and 1.0% PFA-treated enterococcus faecalis (EF). All error bars shown in figures represent the standard error of the mean (SEM); a two-tailed t-test was used to derive p-values. **** denotes p-value < 0.0001. This figure has been modified from 16. Please click here to view a larger version of this figure.
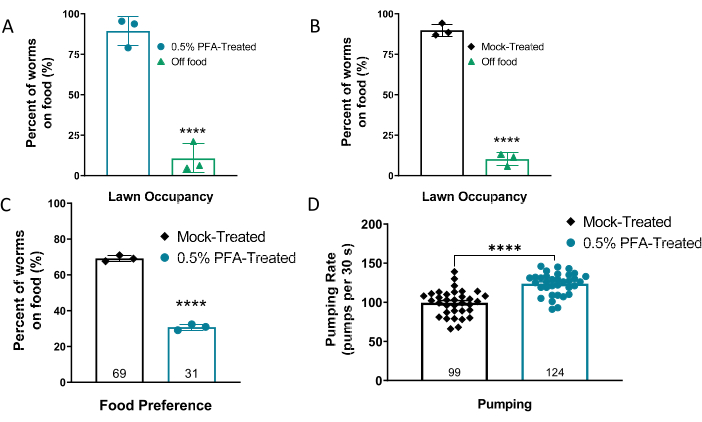
Figure 3. Worms are attracted to PFA-treated OP50. Percent of worms attracted to (A) PFA-treated or (B) mock-treated OP50 bacterial lawn. (C) Pairwise sensitized assay testing worm preference for mock-treated and PFA-treated OP50. (D) Pumping rate (pumps per 30 s) of worms on PFA-treated or mock-treated OP50. A two-tailed t-test analysis was used to derive p-values for all comparisons. All error bars shown in figures represent the standard error of the mean (SEM); **** denotes p-value < 0.0001. Panels A-C have been modified from 16. Please click here to view a larger version of this figure.
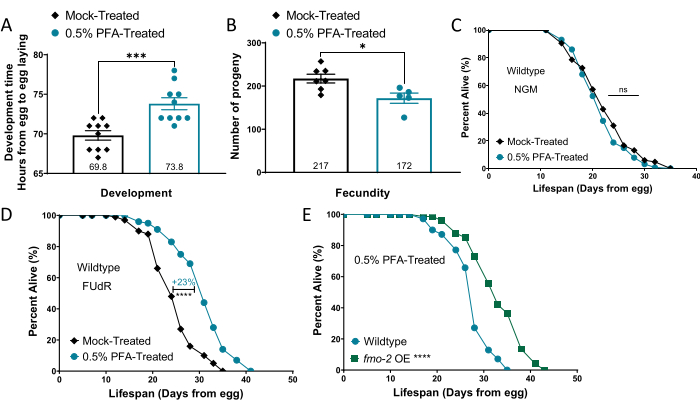
Figure 4. Worms grow and develop on PFA-treated OP50. (A) Development time (h) of worms from egg-to-egg-laying adults on mock-treated and PFA-treated OP50. (B) Average progeny (number of worms) of worms fed mock-treated or PFA-treated OP50. (C, D) Percent alive of worms fed mock-treated or PFA-treated OP50 on (C) NGM and (D) FUdR lifespan plates. (E) Percent alive of wildtype and fmo-2 OE worms fed PFA-treated OP50. A two-tailed t-test analysis was used to derive p-values for development and fecundity comparisons. The log-rank test was used to derive p-values for lifespan comparisons. All error bars shown in figures represent the standard error of the mean (SEM); * denotes p-value < 0.05, *** denotes p-value <0.001, and **** denotes p-value < 0.0001. Panels A, B, and D have been modified from16. Please click here to view a larger version of this figure.
Discussion
Benefits of PFA-killing relative to other bacterial-killing methods
PFA-treatment is a high-throughput method to prevent bacterial metabolism while maintaining a nutritious food source for C. elegans. Killing bacteria via PFA-treatment has multiple advantages over other methods. Unlike UV-treatment, where every plate must be tested for successful killing, a single plate from a batch of PFA-treated bacteria can be tested to validate the batch16. PFA-treatment is also very effective at eliminating bacterial metabolism (Figure 2B), and PFA-treated bacteria exhibit decreased metabolic activity relative to antibiotic-treated bacteria19. Another benefit of PFA-treatment is that the bacteria maintain their cellular structure and nutritional profile. Consequently, worms can develop on PFA-treated bacteria, whereas feeding heat-killed bacteria from egg results in developmental arrest13,16.
Critical steps and troubleshooting
While PFA-treating bacteria is a relatively straightforward protocol, it is important to validate that each batch of bacteria is replicatively and metabolically dead (steps 6 and 7) and to ensure the washing steps after PFA-treatment are done thoroughly (step 5). If colonies do grow in step 6, indicating the bacteria is not completely dead, it is possible to troubleshoot the concentration of PFA and duration of PFA-treatment in step 3. When optimizing the PFA-treatment protocol, a 0.5% concentration of PFA was ideal for successfully killing OP50 with minimal side effects. 0.25% PFA was not sufficient to metabolically kill all strains of bacteria (Figure 2C-D) and increasing the concentration to 0.5% PFA did not alter developmental time, lifespan, or food preference of worms on OP50 relative to 0.25% PFA. Also, 1 h of inoculation with PFA was the shortest time sufficient to prevent all bacterial growth. If bacteria are not completely killed by a 1 h inoculation with 0.5% PFA, the inoculation time and/or PFA concentration can be increased to troubleshoot killing efficiency. A second key step in the protocol is washing the bacteria after PFA-treatment (step 5). It is very important to complete all five washes and to completely remove the supernatant with each wash to ensure no formaldehyde remains in the bacteria. If worms are not growing well or not pumping on the PFA-treated bacteria, additional washes can be performed.
Limitations of the method
While PFA-killed bacteria are ideal for eliminating the confounding effects of bacterial metabolism (Figure 2B) from C. elegans studies, there are limitations to this method. Specifically, C. elegans have slightly slower development on PFA-treated bacteria relative to live bacteria (Figure 4A)16. PFA-treatment also extends worm lifespan if FUdR is present (Figure 4D), but not on NGM (Figure 4C) plates16. However, other methods also present these challenges; UV-killed bacteria also alter C. elegans lifespan. The majority of reports suggest UV-treated bacteria extend worm lifespan on NGM12,23 and on FUdR24, with other studies indicating UV-treatment does not affect lifespan24 and that there is an interaction between UV-treatment and FUdR on C. elegans lifespan25,26. In addition to the lifespan effects of PFA-treated bacteria, worms prefer to spend time on live bacteria relative to PFA-treated bacteria (Figure 3C)16 but have a higher pumping rate on the PFA-treated food (Figure 3D). This suggests that they may consume a similar amount of food or that PFA-treated bacteria are easier or harder to eat. Many of these effects are also observed with other methods of killing bacteria12,13,14, suggesting that metabolically active bacteria decrease development time, shorten lifespan, and are more attractive to C. elegans.
Potential applications of PFA-treated bacteria
Overall, the ability to quickly and reliably kill large batches of bacteria has great potential for eliminating the confounding effects of bacterial metabolism in C. elegans studies. This ability is particularly useful for drug screens, supplementation experiments, toxicity assays, metabolomics studies, and examining the effects of microbial metabolism on host behavior. More information on the use of PFA-treated bacteria in each of these applications is below.
Drug, supplementation, and toxicity experiments: Because only one plate per batch of PFA-treated bacteria needs to be tested for killing success, PFA-killed bacteria can be added to liquid culture for high-throughput drug screening. PFA-treated bacteria can also be seeded onto solid plates with small molecules or nutrients incorporated into the agar media. Solid plates seeded with PFA-treated bacteria could also be useful for toxicity or stress-response assays. Unpublished data suggest that PFA-treated bacteria can help distinguish between the role of bacterial stress-response pathways and worm stress-resistance pathways in paraquat and tunicamycin resistance. Using PFA-killed bacteria in these drug supplementation and toxicity studies prevent bacteria from metabolizing the molecule of interest and will ensure that any phenotype observed results from a compound acting directly on C. elegans. Using metabolically dead bacteria for drug and supplementation experiments also minimizes variability in dosing, as live bacteria can metabolize different amounts of the compound of interest from experiment to experiment.
Metabolomics studies: C. elegans metabolomics studies can also be confounded by bacterial metabolism: observing changes in worm metabolism requires the absence of bacterial metabolic activity29. Even small changes to the bacterial food source can alter the worm metabolome. For example, the metabolome of worms fed live washed bacteria differs from the metabolome of worms fed live unwashed bacteria16. Consuming PFA-treated bacteria also changes the worm metabolome relative to consuming live food16 but allows for comparisons between the effects of experimental conditions on C. elegans metabolism in isolation from bacterial metabolism.
Host-microbe behavioral studies: Finally, PFA-treated bacteria could be used to identify interactions between the bacterial metabolism and host behavior. Metabolites secreted by both pathogenic and non-pathogenic bacteria can be attractive or repellent to worms to affect their foraging and feeding behavior10,11,30. Moreover, bacteria can produce neuromodulators that drive changes in locomotion30. Additionally, growing C. elegans on different strains of bacteria results in altered brood size, development time, and physiology31,32. Treating these bacteria with PFA can help determine whether the effects of a given bacteria on C. elegans require active bacterial metabolism.
Disclosures
The authors have nothing to disclose.
Acknowledgements
This work was funded by NIH R21AG059117 and the Paul F. Glenn Laboratories for Biology of Aging Research at the University of Michigan. SB was funded by T32AG000114. ESK was funded by NSF DGE 1841052.
Materials
| Aluminum Foil | Staples | 2549291 | |
| Bunsen burner | VWR | 470121-700 | |
| Cell Density Meter | Denville | 80-3000-45 | |
| Centrifuge | Eppendorg | 5430 | |
| Chemical fume hood | Labcono | 975050411384RG | |
| Conincal tubes (50 mL) | Fisher | 339652 | |
| Cuvettes | Fisher | 14-955-127 | |
| E. coli OP50 | CGC | OP50 | |
| Erlenmyer flasks | Fisher | 250 mL: FB501250 500 mL: FB501500 1000 mL: FB5011000 |
|
| Inoculation loop | Fisher | 22-363-605 | |
| LB Agar | Fisher | BP1425500 | |
| Liquid waste collection bottle | Thomas Scientific | 1230G50 | |
| Magnesium Sulfate (MgSO4) | Sigma | M7506 | |
| Paraformaldehyde (32%) | Electron Microscopy Sciences | 15714-S | Paraformaldehyde – methanol free solution |
| Pipettor | Eppendorf | Eppendorf Easypet 3 | |
| Plastic dishes (100 mm) | Fisher | FB0875712 | |
| Potassium Phosphate Monobasic (KH2PO4) | Fisher | P2853 | |
| Seahorse XF Calibrant | Agilent | 100840-000 | |
| Seahorse XFe96 Extracellular Flux Assay Kit and Cell Culture Microplate | Agilent | 101085-004 | |
| Serological pipettes (50 mL) | Genesee Scientific | 12-107 | |
| Shaker incubator | Thermo | 11 676 083 | |
| Sodium Chloride (NaCl) | Fisher | S640-3 | |
| Sodium Hydroxide (NaOH) | Fisher | S318500 | |
| Sodium Phosphate Dibasic Anhydrous (Na2HPO4) | Sigma | S374-500 | |
| Solid waste collection bucket | M&M Industries | 5.0 Gallon M1 Traditional Pail | |
| Tryptone | Genesee Scientific | 20-251 | |
| Vortex | Thermo | 11676331 | |
| Weighing balance | C Goldenwall | HZ10K6B | |
| Yeast Extract | Genesee Scientific | 20-255 |
References
- Riddle, D. L., Blumenthal, T., Meyer, B. J., Priess, J. R. C. . Elegans II. 33, (1997).
- Corsi, A. K., Wightman, B., Chalfie, M. A transparent window into biology: A primer on caenorhabditis elegans. WormBook. , 1-31 (2015).
- Kaletta, T., Hengartner, M. O. Finding function in novel targets: C. elegans as a model organism. Nature reviews. Drug discovery. 5 (5), 387-398 (2006).
- Meneely, P. M., Dahlberg, C. L., Rose, J. K. Working with worms: Caenorhabditis elegans as a model organism. Current Protocols Essential Laboratory Techniques. 19 (1), (2019).
- Zhang, S., Li, F., Zhou, T., Wang, G., Li, Z. Caenorhabditis elegans as a useful model for studying aging mutations. Frontiers in Endocrinology. 11, 554994 (2020).
- Feng, M., Gao, B., Garcia, L. R., Sun, Q. Microbiota-derived metabolites in regulating the development and physiology of Caenorhabditis elegans. Frontiers in Microbiology. 14, 1035582 (2023).
- McIntyre, G., Wright, J., Wong, H. T., Lamendella, R., Chan, J. Effects of FUdR on gene expression in the C. elegans bacterial diet OP50. BMC Research Notes. 14 (1), 207 (2021).
- Cabreiro, F., et al. Metformin retards aging in c. Elegans by altering microbial folate and methionine metabolism. Cell. 153 (1), 228-239 (2013).
- Worthy, S. E., et al. Identification of attractive odorants released by preferred bacterial food found in the natural habitats of c. Elegans. PLoS One. 13 (7), e0201158 (2018).
- Chen, Y. C., Seyedsayamdost, M. R., Ringstad, N. A microbial metabolite synergizes with endogenous serotonin to trigger C. elegans reproductive behavior. Proceedings of the National Academy of Sciences of the United States of America. 117 (48), 30589-30598 (2020).
- Kim, D. H., Flavell, S. W. Host-microbe interactions and the behavior of Caenorhabditis elegans. Journal of Neurogenetics. 34 (3-4), 500-509 (2020).
- Gems, D., Riddle, D. L. Genetic, behavioral, and environmental determinants of male longevity in Caenorhabditis elegans. Genetics. 154 (4), 1597-1610 (2000).
- Qi, B., Kniazeva, M., Han, M. A vitamin-b2-sensing mechanism that regulates gut protease activity to impact animal’s food behavior and growth. eLife. 6, e26243 (2017).
- Garigan, D., et al. Genetic analysis of tissue aging in caenorhabditis elegans: A role for heat-shock factor and bacterial proliferation. Genetics. 161 (3), 1101-1112 (2002).
- Virk, B., et al. Folate acts in E. coli to accelerate C. elegans aging independently of bacterial biosynthesis. Cell Reports. 14 (7), 1611-1620 (2016).
- Beydoun, S., et al. An alternative food source for metabolism and longevity studies in Caenorhabditis elegans. Communications Biology. 4 (1), 258 (2021).
- Thavarajah, R., Mudimbaimannar, V. K., Elizabeth, J., Rao, U. K., Ranganathan, K. Chemical and physical basics of routine formaldehyde fixation. Journal of Oral and Maxillofacial Pathology. 16 (3), 400-405 (2012).
- Felix, H. Permeabilized and immobilized cells. Methods in Enzymology. 137, 637-641 (1988).
- Lobritz, M. A., et al. Antibiotic efficacy is linked to bacterial cellular respiration. Proceedings of the National Academy of Sciences of the United States of America. 112 (27), 8173-8180 (2015).
- Nadanaciva, S., et al. Assessment of drug-induced mitochondrial dysfunction via altered cellular respiration and acidification measured in a 96-well platform. Journal of Bioenergetics and Biomembranes. 44 (4), 421-437 (2012).
- Shtonda, B. B., Avery, L. Dietary choice behavior in Caenorhabditis elegans. The Journal of Experimental biology. 209 (Pt 1), 89-102 (2006).
- MacNeil, L. T., Watson, E., Arda, H. E., Zhu, L. J., Walhout, A. J. Diet-induced developmental acceleration independent of tor and insulin in C. elegans. Cell. 153 (1), 240-252 (2013).
- Kumar, S., et al. Lifespan extension in C. elegans caused by bacterial colonization of the intestine and subsequent activation of an innate immune response. Developmental Cell. 49 (1), 100-117 (2019).
- Nakagawa, H., et al. Effects and mechanisms of prolongevity induced by Lactobacillus gasseri sbt2055 in Caenorhabditis elegans. Aging Cell. 15 (2), 227-236 (2016).
- Kaeberlein, T. L., et al. Lifespan extension in Caenorhabditis elegans by complete removal of food. Aging Cell. 5 (6), 487-494 (2006).
- Beaudoin-Chabot, C., et al. The unfolded protein response reverses the effects of glucose on lifespan in chemically-sterilized C. elegans. Nature Communication. 13 (1), 5889 (2022).
- Komura, T., Takemoto, A., Kosaka, H., Suzuki, T., Nishikawa, Y. Prolonged lifespan, improved perception, and enhanced host defense of Caenorhabditis elegans by Lactococcus cremoris subsp. cremoris.Microbiology Spectrum. 10 (3), e0045421 (2022).
- Ye, X., Linton, J. M., Schork, N. J., Buck, L. B., Petrascheck, M. A pharmacological network for lifespan extension in Caenorhabditis elegans. Aging Cell. 13 (2), 206-215 (2014).
- Hastings, J., et al. Wormjam: A consensus C. elegans metabolic reconstruction and metabolomics community and workshop series. Worm. 6 (2), e1373939 (2017).
- O’Donnell, M. P., Fox, B. W., Chao, P. H., Schroeder, F. C., Sengupta, P. A neurotransmitter produced by gut bacteria modulates host sensory behaviour. Nature. 583 (7816), 415-420 (2020).
- Stuhr, N. L., Curran, S. P. Bacterial diets differentially alter lifespan and healthspan trajectories in C. elegans. Communications Biology. 3 (1), 653 (2020).
- Dirksen, P., et al. Cembio – the Caenorhabditis elegans microbiome resource. G3 (Bethesda). 10 (9), 3025-3039 (2020).

