Embryo Microinjection: A Technique to Deliver a Compound into the Zebrafish Yolk
Published: April 30, 2023
Abstract
Source: Sorlien, E. L., et al. Efficient Production and Identification of CRISPR/Cas9-generated Gene Knockouts in the Model System Danio rerio. J. Vis. Exp. (2018).
This video describes a technique of Embryo microinjection to deliver compounds into Zebrafish yolk sac.
Protocol
1. Microinjection of CRISPR-components into Zebrafish Embryos
- Set up breeding tanks the night prior to injecting by placing the number of desired males and females (typically 2 females and 1 or 2 males) in a breeding tank with a divider in place.
- Prepare a microinjection plate with 1.5% agarose in 1x E3 media (see Table of Materials) with 0.01% methylene blue (a fungicide) by pouring 35 mL of the melted agarose into a 10 cm Petri dish and gently lay a plastic mold to create wedge-shaped troughs into the solution, tapping the mold to eliminate air bubbles.
- Allow the agarose to set, and store the dish with a small amount of media and wrapped in paraffin film to prevent the plate from drying out at 4 °C.
NOTE: Injection plates are reusable for several weeks, until the wells become deformed or dry, or the plate begins to grow mold. - On the morning of injecting, thaw purified sgRNA and Cas9 protein on ice. Remember to handle all materials with gloves to prevent RNase contamination and to use RNase-free tips and tubes.
- Generate a 5 µL injection solution by combining Cas9 protein and the sgRNA in a 2:1 ratio of Cas9:sgRNA to obtain final concentrations of 400 pg/nL Cas9 protein and 200 pg/nL sgRNA. Incubate the Cas9/sgRNA solution at room temperature for 5 min to allow the Cas9 and sgRNA to form a ribonucleoprotein complex. Add 0.5 µL of 2.5% wt/vol phenol red solution (see Table of Materials), and RNase-free water to a final volume of 5 µL.
NOTE: The ionic strength of the solution has been shown to affect the solubility of the Cas9/sgRNA complex, therefore the addition of KCl may increase the cutting efficiency of sgRNAs that exhibit low indel formation. - Make an injection needle by pulling a 1.0 mm glass capillary using a micropipette puller. Cut the tip of the freshly-made needle using a new razor blade or forceps to obtain an angled opening that will easily pierce the chorion and yolk sac.
- Place the needle in a micromanipulator attached to a microinjector with the air source turned on. Under a light microscope using the magnification suitable for the calibration determined for the particular apparatus, adjust the injection pressure until the needle consistently ejects a 1 nL solution into a Petri dish filled with mineral oil.
NOTE: The quality of the needle is critical. Practice producing a needle and injecting into the yolk sac of embryos until this is skill is mastered before attempting further experiments. - Remove the divider and allow the fish to breed for approximately 15 min.
NOTE: Longer breeding times will produce more embryos, however the injection should be completed while the embryos are at the 1-cell stage to maximize the chance that Cas9 cutting will occur early and therefore decrease genetic mosaicism. Embryos can be injected at later stages (2–4 cell stages), but this may possibly decrease the germline transmission rate of the modified allele. - Collect the eggs using a strainer and rinse them into a 10 cm Petri dish using 1x E3 media with 0.0001% methylene blue. Examine the health of the eggs under the light microscope, removing any unfertilized eggs and debris.
- Set aside 10–15 embryos as an uninjected control in a separate, labeled Petri dish.
- Using a transfer pipette, gently line up the eggs on the injection plate warmed to room temperature.
- Under a dissection microscope at 2.5X magnification, inject 1 nL of the solution into the yolk sac of each embryo to inject a total of 400 pg of Cas9 protein and 200 pg of sgRNA.
NOTE: To increase cutting if desired or necessary, increase the final concentration of Cas9 protein to 800 pg/nL and of sgRNA to 400 pg/nL in the injection solution; however, this may also increase off-target cutting and/or decrease embryo health. Cutting efficiency may also be increased by injecting directly into the cell. However, injection into the yolk sack is technically less demanding and gives sufficient cutting to produce fish with high germline transmission (>70% of offspring containing a modified allele). - Return the injected embryos to a properly labeled Petri dish, cover them with 1x E3 media with methylene blue, and put them in an embryo incubator set to 28 °C.
- At 24 h post fertilization (hpf), inspect the health of the injected embryos, removing dead or abnormally developing individuals and change the media (See Figure 1). Check the rate of survival against the uninjected control.
NOTE: When targeting a nonessential gene, less than 10% lethality is expected relative to the uninjected control. If elevated levels of lethality are observed in the guide-injected populations compared to the uninjected control, it may indicate that the targeted gene is essential for development, or off-target effects are leading to failed development. Reducing the amount of injected CRISPR-reagents may be necessary or generation of a new sgRNA with reduced off-target effects may be required. - Return the embryos to the incubator and continue growing the embryos to 72 hpf, changing the media daily to maintain embryo health.
Representative Results
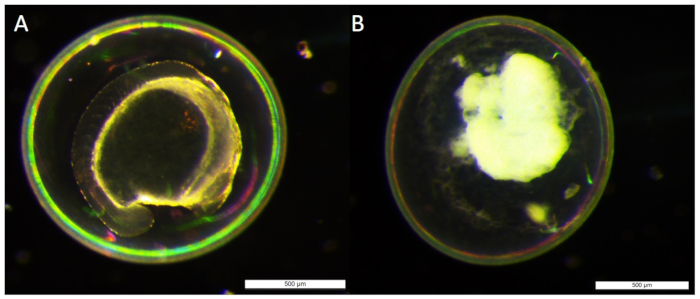
Figure 1: Comparison of the health of 24 hpf injected embryos. A living embryo (A) developed to 24 hpf, is easily distinguished from an embryo that has aborted development (B). Embryos that resemble (B) or have drastically altered features to (A), such as spinal curvature or altered head and eye development should be removed from dish
Materials
| RNase-free water | Any brand | Synthesis of guide RNAs. Thermo Fisher and Qiagen both carry suitable water. |
|
| E3 embryo media | Made in house | Media for raising embryos and making injeciton plate. Recipe: http://cshprotocols.cshlp.org/ content/2011/10/pdb.rec66449 |
|
| Methylene Blue | Sigma-Aldrich | M9140 | Added to 1x E3 media to prevent fungal growth on embryos. |
| Petri dish | Thermo Fisher | FB0875711Z | Store embryos, cast injection plate |
| Agarose | Denville | CA3510-8 | Casting injection plate, agarose gels. |
| Microinection mold | Adaptive Science Tools | TU-1 | To create wells to hold embryos during injection |
| Phenol Red | Sigma-Aldrich | P0290 | Dye for visualization of injection |
| Mineral Oil | Sigma-Aldrich | M5904-5ML | To calibrate needle injection volume. |
| Transfer pipettes | Any brand | Moving embryos | |
| Razor blade | Thermo Fisher | 11295-10 | Cutting injection needle, tail clipping adult fish. |
| Incubator | Maintaining embryos at 28.5 C. | ||
| Verticle pipette puller | David Kopf Instruments | 700C | Geneate needles for injection. Additional needle pulling instructions: http:// cshprotocols.cshlp.org/ content/2006/7/pdb.prot4651.long |
| Capillary tubes | Sutter Instruments | BF100-58-10 | Geneate needles for injection. |
| Microloader tips | Eppendorf | 930001007 | Load solution into injection needles |
| Microinjector | World Precision Instruments | PV 820 | Injecting embryos. |
| Disecting microscope | Leica | Injecting embryos. | |
| Microwave | Any brand | Casting injection plate, agarose gels. | |
| Scale | Any brand | Casting injection plate, agarose gels. | |
| Gloves | Any brand | For all aspects of the protocol. | |
| N2 | Any brand | To expell liquid from the capillary for embryo injection | |
| PCR strip tubes | Any brand | For all aspects of the protocol. | |
| 200 µL tips | Any brand | For all aspects of the protocol. | |
| 10 µL tips | Any brand | For all aspects of the protocol. | |
| Micropipettes | Any brand | For all aspects of the protocol. |
Citer Cet Article
Embryo Microinjection: A Technique to Deliver a Compound into the Zebrafish Yolk. J. Vis. Exp. (Pending Publication), e20196, doi: (2023).

