The Hemagglutination Inhibition Assay to Detect Serum Antibodies Against a Target Antigen
Abstract
Source: Kaufmann, L. et al., An Optimized Hemagglutination Inhibition (HI) Assay to Quantify Influenza-specific Antibody Titers. J. Vis. Exp. (2017)
The video demonstrates a hemagglutination inhibition assay for detecting serum antibodies against specific antigens. Confirmation of hemagglutination inhibition in the test well and hemagglutination in the control well is achieved through distinct visual patterns. This assay plays a crucial role in assessing immune response, vaccine effectiveness, and studying viral infections in virology and immunology.
Protocol
All procedures involving human participants have been performed in compliance with the institutional, national, and international guidelines for human welfare and have been reviewed by the local institutional review board.
1. Serum Collection
- Collect serum samples from humans at time points of interest. For this study, we collected sera at days 0 (time of influenza vaccination), +7, +30, +60, and +180 after vaccination.
- To obtain the serum, centrifuge the sample tubes at 1,200 x g for 10 min at room temperature (20 – 25 °C).
NOTE: Non-centrifuged blood samples should be stored at 4 °C and for no longer than 24 h. - Aliquot the serum into different tubes (cryo-vials) and freeze at -80 °C until use.
- Perform the subsequent assays batch-wise, including all the time points of one person to reduce variability within a patient.
2. Preparation of Antigens
CAUTION: Five different antigens are used (see Table of Materials). Prepare antigens in a Biosafety Level 2 (BSL-2) laboratory.
- According to the manufacturer's instructions, reconstitute the total contents of one lyophilized influenza antigen ampoule with 1.0 mL of distilled water and allow the dissolved antigen to stand for a minimum of 5 min at room temperature before proceeding.
- Aliquot the antigen solution to 1.5 mL tubes and freeze at -80 °C until further use.
3. Preparation of Cholera Filtrate
NOTE: Cholera filtrate is used as a receptor-destroying enzyme (RDE) according to the World Health Organization (WHO) protocol. This removes innate inhibitors from the serum which would interfere with the assay.
- Reconstitute the lyophilized RDE according to the manufacturer's instructions.
- Store the RDE solution in a 15 mL tube at 4 °C until further use.
4. HA Assay
NOTE: To ensure that the hemagglutination inhibition (HI) assays are comparable between several plates, the same amount of virus particles must be used for each plate. The hemagglutination (HA) assay (also called HA titration) is performed to quantify the virus particles necessary for hemagglutination and is recorded in HA units. A "unit" of hemagglutination is an operational unit dependent on the method used for HA titration and is not a measurement of an absolute amount of virus. Thus, an HA unit is defined as the amount of virus needed to agglutinate an equal volume of a standardized RBC suspension. According to the WHO, the standard amount used for the HI assay is 4 HA units per 25 µL. For an illustration of the principle of the HA assay see Figure 1.
NOTE: The red blood cells (RBCs) used are dependent on the type of influenza virus in the assay (Table 1). Further, for various types of 96-well microtiter plates, the incubation time as well as the appearance of the non-agglutinated cells differ (Table 2).
- Preparation of the RBC Suspension
- Dilute the RBC stock suspension (10%, v/v; except human type O) (see Table of Materials) with phosphate-buffered saline (PBS) to make the proper concentrations for avian and mammalian RBCs of 0.75% and 1%, respectively.
- Preparation of the 96-well Microtiter Plate
NOTE: See Figure 2 for an overview of the plate design.- Add 25 µL of PBS to wells 1 to 12 of each used row of a 96-well microtiter plate by using a multichannel pipette (Figure 2). Use the V-shaped microtiter plate when working with avian RBCs, like chicken and turkey. Use the U-shaped microtiter plate when working with mammalian RBCs, like guinea pigs and human type O (Table 2).
- Add 25 µL of influenza antigen to the first well of the antigen rows, which are arranged in duplicates. No antigen is added to the control rows. The control rows should not show a hemagglutination effect and serve as negative controls (Figure 2).
- Perform a serial 2-fold dilution by transferring 25 µL from the first well of the antigen rows to successive wells by using a multichannel pipette. Mix each dilution step by pipetting up and down gently 10 times.
- Discard the final 25 µL of the last wells.
- Add 25 µL of PBS to wells 1 to 12 of each used row by using a multichannel pipette, to reach a total volume of 50 µL per well.
- Add 50 µL of the RBC suspension to each used well by using a multichannel pipette.
- Tap the plate carefully 10 times on all four sides to mix.
- Cover the plate with a lid and incubate at room temperature for the appropriate amount of time depending on the RBC species used (see Table 2). Do not move the plate while incubating.
- Reading the plate
NOTE: The readout is slightly different when using avian RBCs compared to mammalian RBCs, because of the different shaped microtiter wells (Figure 3).- Readout of avian RBCs
- Tilt the plate 90° for 25 s.
NOTE: The HA titration endpoint is the last well where complete hemagglutination occurs. This well contains 1 HA unit of virus. Because of the 2-fold dilutions of the antigen, two wells ahead of the HA titration endpoint is the well that contains 4 HA units of virus (Figure 4). - Mark the results immediately, while the plate is still in a tilted position, on a printed scheme of the 96-well plate. The agglutination patterns of the avian RBCs are shown in Figure 3.
- Tilt the plate 90° for 25 s.
- Readout of mammalian RBCs
- Mark the results on a printed scheme of the 96-well plate, without tilting the plate (horizontal position on the bench).
NOTE: When hemagglutination occurs, the agglutinated cells do not settle to the bottom, whereas non-agglutinated cells appear as a halo at the bottom of the well. The halo of the partially agglutinated cells is less intense and has a larger diameter (Figure 3).
- Mark the results on a printed scheme of the 96-well plate, without tilting the plate (horizontal position on the bench).
- Determination of 4 HA units.
NOTE: Tilting the plate is crucial for the differentiation of avian patterns because all three different types of agglutination patterns (completely agglutinated, partially agglutinated, and non-agglutinated) appear as a button when not tilted.
- Readout of avian RBCs
5. HI Assay
NOTE: The workflow of the protocol has been optimized to allow more efficient handling of multiple samples at the same time, by using polymerase chain reaction (PCR) tube stripes and a thermocycler (see below).
- Preparation of the Serum Samples
NOTE: Prepare serum samples in a biosafety level-2 (BSL-2) laboratory.- Thaw the frozen serum samples of each time point of every person (see step 1.2) at room temperature.
- Add an aliquot of 10 µL of each thawed serum sample to a tube of PCR tube strip (10 tubes in one strip).
NOTE: The big advantage of using PCR tube strips is that a multichannel pipette can be used for the following steps in the HI assay; this saves a lot of time when testing a large number of serum samples and when performing repeated measures of the same samples for antibody titers against different virus strains. - Store the aliquoted serum samples in the PCR tube strips at -80 °C until use.
- One day before the HI assay is performed, thaw the serum sample aliquots of interest at room temperature.
- Add 10 µL of the appropriate anti-serum to an empty PCR tube.
NOTE: To serve as a positive control, the anti-serum against a specific virus must match the used virus. The positive control allows for standardization of plate performance over multiple plates. - Add 30 µL of cholera filtrate solution to each serum aliquot and to the anti-serum (3 volumes of cholera filtrate to 1 volume of serum) by using a multichannel pipette.
- Keep the PCR tubes in a PCR 96-well rack or an empty tip-box and vortex for 5 s.
- Incubate the samples overnight at 37 °C using a thermocycler.
- Incubate the samples at 56 °C for 30 min to inactivate the cholera filtrate using a thermocycler.
NOTE: Depending on the thermocycler, this step can be programmed to further automate the process. - Keep the PCR tubes in a PCR 96-well rack or an empty tip-box and vortex for 5 s.
- Store the samples at 4 °C in the fridge until use for the HI assay.
- HI assay
NOTE: For an illustration of the principle of the HI assay see Figure 1. Depending on the virus, different species of RBCs are used for the assay (Table 1). The different species of RBCs are used in various types of 96-well plates, and the incubation time as well as the appearance of the non-agglutinated cells differs (Table 2). For the HI assay, 4 HA units of virus or antigen are added to the 2-fold dilution series of the samples.- Preparation of the antigen solution
- Calculate the volume of antigen solution needed according to the number of 96-well plates used (25 µL antigen per well × 96 = 2,400 µL antigen per 96-well plate; add 100 µL per plate extra due to the usage of a reservoir for the multichannel pipette; a total 2.5 mL of antigen per plate).
NOTE: For example, if measuring 100 serum samples then 10 plates are needed (10 samples per plate): 2.5 mL x 10 = 25 mL of antigen solution needed in total. - Prepare the proper dilution of 4 HA units for the calculated volume using PBS.
NOTE: 4 HA units are determined for the HA assay. For the appropriate amount of antigen, divide the calculated volume by the titer corresponding to 4 HA units. For example, 4 HA units correspond to a dilution of 1/64, and we needed 15,000 µL of antigen solution are needed: 15,000/64 = 234.4 µL of the dissolved lyophilized influenza antigen are added.
- Calculate the volume of antigen solution needed according to the number of 96-well plates used (25 µL antigen per well × 96 = 2,400 µL antigen per 96-well plate; add 100 µL per plate extra due to the usage of a reservoir for the multichannel pipette; a total 2.5 mL of antigen per plate).
- Preparation of the RBC suspension
- Calculate the volume of RBC suspension needed according to the number of 96-well microtiter plates used (50 µL RBC suspension per well × 96 = 4,800 µL RBC suspension per 96-well plate; add 200 µL per plate extra due to the usage of a reservoir for the multichannel pipette).
- Dilute the RBC stock suspension (normally 10%, v/v; except human type O) with PBS to make the proper concentrations for avian and mammalian RBCs of 0.75% and 1%, respectively.
- Preparation of the 96-well microtiter plate
- Label the 96-well microtiter plates (sample ID, positive control, and back titration). Please check the plate orientation in Figure 5 carefully.
- Add 25 µL of PBS to every well except to the first well of the "back titration" row (Figure 5, 12th row) using the multichannel pipette.
NOTE: A back titration was performed to check if the used antigen dilution equals 4 HA units. An antigen titer of 4 HA units is indicated if hemagglutination occurs in the first three wells of the "back titration" row, but not in the fourth well. - Add 50 µL of the prepared antigen solution (described in 5.2.1) to the first well of the "back titration" row (12th row).
- Add 25 µL of the RDE-treated serum samples to the first wells of rows 1 to 10 on each plate, using the multichannel pipette.
- Add 25 µL of the appropriate anti-serum to the first well of the 11th row as a positive control.
- Perform serial 2-fold dilutions by transferring 25 µL from the first well of each row (1 – 12) to successive wells by using a multichannel pipette. Mix by pipetting up and down 10 – 15 times for each dilution step. The same tips can be used for each dilution step per sample.
- Discard the final 25 µL of the last wells.
- Add 25 µL of the antigen solution by using a multichannel pipette to each well of rows 1 to 11 (serum samples and anti-serum). The same tips can be used if they do not touch the wells.
- Add 25 µL of PBS instead of antigen to each well of the "back titration" row (12th row).
- Tap the plate carefully 10 times on all four sides to mix.
- Cover the plate with a lid and incubate at room temperature for 30 min. Do not move the plate while incubating.
- Add 50 µL of the RBC suspension to every well.
- Tap the plate carefully 10 times on all 4 sides to mix.
- Cover the plate with a lid and incubate at room temperature for the appropriate amount of time depending on the RBC species used (see Table 2). Do not move the plate while incubating.
- Reading the plate
NOTE: The HI titer is the reciprocal of the last dilution of (anti-) serum that completely inhibits hemagglutination. It is important to consider that the RDE-treated sera were already diluted 1:4 and after the serial dilution step, the sera in the first wells are diluted 1:8, which corresponds to a HI titer of 8.- Readout of avian RBCs
- Tilt the plate 90° for 25 s.
NOTE: Tilting the plate is crucial for the differentiation of avian patterns because all three different types of agglutination patterns (completely agglutinated, partially agglutinated, and non-agglutinated) appear as a button when not tilted. - Mark the results immediately, while the plate is still in a tilted position, on a printed scheme of the 96-well plate. The agglutination patterns of avian RBCs are shown in Figure 3.
- Tilt the plate 90° for 25 s.
- Readout of avian RBCs
- Preparation of the antigen solution
Table 1: Influenza antigens and corresponding species of RBCs. According to the manufacturer's instructions (NIBSC).
| Influenza antigen | A/California/7/09 (H1N1) | A/Switzerland/9715293/2013 (H3N2) | A/Texas/50/2012 (H3N2) | B/Brisbane/60/08 | B/Massachusetts/02/2012 |
| RBC species | Chicken | Guinea Pig | Guinea Pig | Turkey | Turkey |
Table 2: Assay conditions with different species of RBCs. According to the WHO protocol. (* flows when tilted).
| RBC species | Chicken | Turkey | Guinea pig | Human type O |
| Concentration of RBCs (v/v) | 0.75% | 0.75% | 1% | 1% |
| Type of microtiter plate | V bottom | V bottom | U bottom | U bottom |
| Incubation time, RT | 30 min | 30 min | 1 hour | 1 hour |
| Appearance of non-agglutinated cells | Button* | Button* | halo | halo |
Representative Results
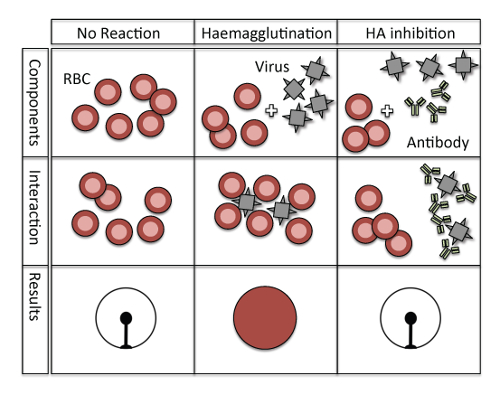
Figure 1: Principle of hemagglutination and hemagglutination inhibition. No hemagglutination occurs in a negative control situation without viruses and antibodies (left column), and erythrocytes hemagglutinate only in the presence of influenza virus (middle column). However, when the hemagglutinin of the influenza virus is blocked by virus-specific antibodies then no hemagglutination can occur (right column).
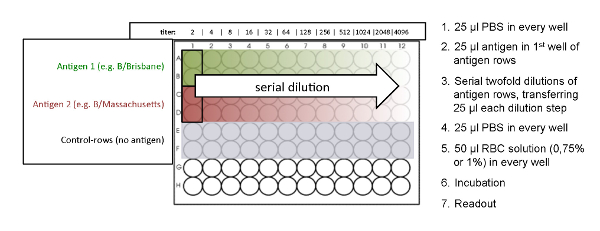
Figure 2: Plate design of the HA assay. The HA titration is performed in duplicates. No antigen was added to the control rows. Also, see Figure 4 for the determination of the best antigen concentration.
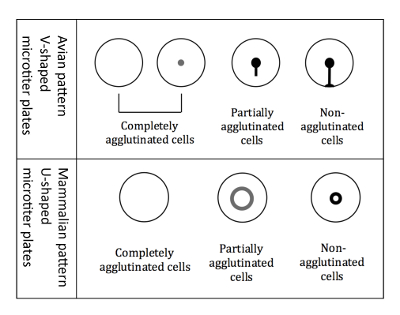
Figure 3: Agglutination patterns of avian and mammalian RBCs. V-shaped microtiter plates are used when working with avian RBCs. The readout is performed in a tilted plate position, and non-agglutinated RBCs start to run down forming a tear-like shape. U-shaped microtiter plates are used when working with mammalian RBCs. The readout is then performed in a non-tilted position, and non-agglutinated RBCs form a small halo.
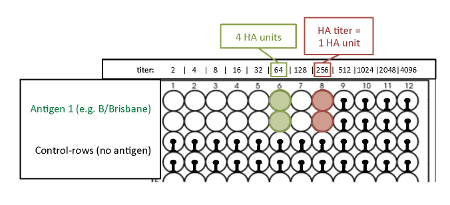
Figure 4: Readout of the HA titration with avian RBCs to determine the titer of 4 HA units. The optimal antigen amount required for hemagglutination is measured by the hemagglutination assay (antigen titration assay). The last well where complete hemagglutination occurs is the HA titration endpoint which contains 1 HA unit. Because of the 2-fold dilutions of the antigen, two wells ahead of the HA titration endpoint, the titer corresponds to 4 HA units.

Figure 5: Plate design and workflow of the HI assay. Five timepoints of two people can be measured on one plate. The HI titer ranges from 8 to 1,024. An anti-serum of the used antigen served as a positive control and a back titration was performed to check if the antigen dilution equals 4 HA units. The serial dilution of the serum sample is shown for 2 individual vaccine recipients.
Divulgations
The authors have nothing to disclose.
Materials
| 25 ml Disposable Multichannel Pipette Reservoirs | Integra | 4312 | |
| 8-well PCR tubes | Brand GMBH | 781332 | For serum aliquots |
| 96-well microtiter plate, U-shaped | TPP | 92097 | For HI assay when using mammalian RBCs |
| 96-well microtiter plate, V-shaped | Corning Costar | 3897 | For HI assay when using avian RBCs |
| Aqua ad iniect. Steril | Bichsel AG | 1000004 | For preparing influenza antigen and cholera filtrate solutions |
| Chicken RBC (10%) | Cedarlane | CLC8800 | 10% suspension of chicken red blood cells in Alsever's solution |
| Cholera filtrate | Sigma-Aldrich | C8772 | Used as receptor destroying enzyme (RDE) |
| Dulbecco's PBS | Sigma-Aldrich | D8537 | For diluting the serum samples, RBCs and antigens |
| Eppendorf Multichannel pipette, 12-channel, 10-100 µl | Sigma-Aldrich | Z683949 | |
| Eppendorf Multichannel pipette, 8-channel, 10-100 µl | Sigma-Aldrich | Z683930 | |
| Guinea Pig RBC (10%) | Cedarlane | CLC1800 | 10% suspension of guinea pig red blood cells in Alsever's solution |
| Influenza Anti-A/California/7/09 HA serum | NIBSC | 14/134 | Used as positive control at the HI assay |
| Influenza Anti-A/Switzerland/9715293/2013-like HA serum | NIBSC | 14/272 | Used as positive control at the HI assay |
| Influenza Anti-A/Texas/50/2012-Like HA Serum | NIBSC | 13/178 | Used as positive control at the HI assay |
| Influenza Anti-B/Brisbane/60/2008-HA serum | NIBSC | 13/254 | Used as positive control at the HI assay |
| Influenza Anti-B/Massachusetts/02/2012 HA serum | NIBSC | 13/182 | Used as positive control at the HI assay |
| Influenza antigen A/California/7/09 (H1N1)(NYMC-X181) | NIBSC | 12/168 | Inactivated, partially purified A/California/7/09 (H1N1)(NYMC-X181) virus (ca. 46µgHA/ml) |
| Influenza antigen A/Switzerland/9715293/2013 (NIB88) | NIBSC | 14/254 | Inactivated, partially purified A/Switzerland/9715293/2013 (NIB88) virus (ca. 55µgHA/ml) |
| Influenza antigen A/Texas/50/2012 (H3N2)(NYMCX-223) | NIBSC | 13/112 | Inactivated, partially purified A/Texas/50/2012 (H3N2)(NYMCX-223) virus (ca. 74µgHA/ml) |
| Influenza antigen B/Brisbane/60/2008 | NIBSC | 13/234 | Inactivated, partially purified B/Brisbane/60/2008 virus (ca. 42µgHA/ml) |
| Influenza antigen B/Massachusetts/02/2012 | NIBSC | 13/134 | Inactivated, partially purified B/Massachusetts/02/2012 virus (ca. 35µgHA/ml) |
| Serum-Tubes | S-Monovette, Sardstedt | 01.1601.100 | For serum extraction with clotting activator |
| Turkey RBC (10%) | Cedarlane | CLC1180 | 10% suspension of turkey red blood cells in Alsever's solution |
| Phosphate Buffered Saline (PBS) | Gibco |

