Transplantation of Induced Pluripotent Stem Cell-derived Mesoangioblast-like Myogenic Progenitors in Mouse Models of Muscle Regeneration
Summary
Induced pluripotent stem cell (iPSC)-derived myogenic progenitors are promising candidates for cell therapy strategies to treat muscular dystrophies. This protocol describes transplantation and functional measurements required to evaluate the engraftment and differentiation of iPSC-derived mesoangioblasts (a type of muscle progenitors) in mouse models of acute and chronic muscle regeneration.
Abstract
Patient-derived iPSCs could be an invaluable source of cells for future autologous cell therapy protocols. iPSC-derived myogenic stem/progenitor cells similar to pericyte-derived mesoangioblasts (iPSC-derived mesoangioblast-like stem/progenitor cells: IDEMs) can be established from iPSCs generated from patients affected by different forms of muscular dystrophy. Patient-specific IDEMs can be genetically corrected with different strategies (e.g. lentiviral vectors, human artificial chromosomes) and enhanced in their myogenic differentiation potential upon overexpression of the myogenesis regulator MyoD. This myogenic potential is then assessed in vitro with specific differentiation assays and analyzed by immunofluorescence. The regenerative potential of IDEMs is further evaluated in vivo, upon intramuscular and intra-arterial transplantation in two representative mouse models displaying acute and chronic muscle regeneration. The contribution of IDEMs to the host skeletal muscle is then confirmed by different functional tests in transplanted mice. In particular, the amelioration of the motor capacity of the animals is studied with treadmill tests. Cell engraftment and differentiation are then assessed by a number of histological and immunofluorescence assays on transplanted muscles. Overall, this paper describes the assays and tools currently utilized to evaluate the differentiation capacity of IDEMs, focusing on the transplantation methods and subsequent outcome measures to analyze the efficacy of cell transplantation.
Introduction
Mesoangioblasts (MABs) are vessel-associated muscle progenitors derived from a subset of pericytes1-3. The main advantage of MABs over canonical muscle progenitors such as satellite cells4 resides in their ability to cross the vessel wall when delivered intra-arterial and hence contribute to skeletal muscle regeneration in cell therapy protocols. This feature has been evaluated and confirmed in both murine and canine models of muscular dystrophy1,5,6. These preclinical studies have built the foundations for a first-in-man phase I/II clinical trial based upon intra-arterial transplantation of donor HLA-identical MABs in children with Duchenne muscular dystrophy (EudraCT no. 2011-000176-33; currently on going at San Raffaele Hospital of Milan, Italy). One of the main hurdles of cell therapy approaches, where billions of cells are needed to treat the muscle of an entire body, is the limited proliferative potential of the "medicinal product" (the cells). Moreover it has been recently demonstrated that it is not possible to obtain MABs from patients affected by some forms of muscular dystrophies, as it occurs in limb-girdle muscular dystrophy 2D (LGMD2D; OMIM #608099)7.
To overcome these limitations, a protocol for deriving MAB-like stem/progenitor cells from human and murine iPSCs has been recently established7. This procedure generates easily expandable cell populations with a vascular gene signature and immunophenotype highly similar to adult muscle-derived MABs. Upon expression of the myogenic regulatory factor MyoD, both murine and human IDEMs (respectively MIDEMs and HIDEMs) undergo terminal skeletal muscle differentiation. To achieve this aim IDEMs can be transduced with vectors able to drive MyoD expression, either constitutively or in an inducible fashion8-10. A lentiviral vector containing MyoD cDNA fused with the estrogen receptor (MyoD-ER) is used, thus allowing its nuclear translocation upon tamoxifen (an estrogen analogue) administration11. This strategy results in the formation of hypertrophic multinucleated myotubes, with high efficiency7. Notably, IDEMs are nontumorigenic, can engraft and differentiate inside host muscles upon transplantation and can be genetically corrected using different vectors (e.g. lentiviruses or human artificial chromosomes), paving the way for future autologous therapeutic strategies. The derivation, characterization and transplantation of IDEMs have been described in the above publication7. This protocol paper details the assays performed to evaluate the in vitro myogenic differentiation of IDEMs and the subsequent outcome measurements to test the efficacy of their transplantation in mouse models of muscle regeneration.
Protocol
1. Assessment of Myogenic and Engraftment Potential
- In vitro: MyoD-induced differentiation
- Generate a stable cell line of IDEMs transduced with the tamoxifen-inducible MyoD-ER lentiviral vector, titrating the multiplicity of infection (MOI; e.g. 1, 5 and 50) using the staining described in 1.1.9 (MyHC) as an outcome of the efficiency of the procedure.
- Coat a 3.5 cm dish with 1 ml of 1% Matrigel and incubate for 30 min at 37 °C.
- Wash the plate twice with medium, seed 1 x 105 MyoD-ER transduced IDEMs in the 3.5 cm tissue culture dish and incubate at 37 °C in growth medium.
- Expect that cells reach confluence in one or two days and then add 1 μM 4OH-tamoxifen into the growth medium (1st dose).
- After 24 hr, replace the growth medium with differentiation medium supplemented with 1 μM 4OH-tamoxifen (2nd and last dose).
- Replace half of the medium with fresh differentiation medium every other day.
- Examine daily the cultures for myotube formation.
- After one week (5 days in differentiation medium), wash the plates gently with PBS and fix with 4 % paraformaldehyde for 5 min at RT.
- Perform an immunofluorescence staining with antibodies against myosin heavy chain (MyHC) to confirm the presence of myotubes. Counterstain with a nuclear dye (e.g. Hoechst).
The efficiency of differentiation is evaluated as the percentage of nuclei inside MyHC-positive cells: proceed to the next step if the efficiency is >50%.
- In vivo: engraftment in a model of acute muscle regeneration
To evaluate the in vivo contribution of MyoD-ER IDEMs to muscle regeneration, the cells are previously labeled with a vector encoding for the green fluorescent protein (GFP) that will enable to trace them inside the tissue. MyoD-ER GFP IDEMs are then injected into adult murine muscles, previously injured with a myotoxin (e.g. cardiotoxin). To avoid immune rejection against xenogeneic (such as HIDEMs) or genetically manipulated/corrected cells it is required to use either immunodeficient or immunosuppressed mice.- Pretreat the animals with an intra-peritoneal injection of 3.5 μl/g of 10 mg/ml tamoxifen (liposoluble form) 24 hr before transplantation and pretreat cells adding 1 μM 4OH-tamoxifen (aqueous form) into the growth medium overnight before the transplantation day.
- Administer anesthesia and analgesia to the mouse following the specific guidelines that regulate surgical procedures in the animal facility.
- Inject 25 μl of 100 μM cardiotoxin (CTX; from Naja mossambica mossambica; CAUTION: potentially harmful substance) in the tibialis anterior (TA) muscles.
- 24 hr after treating the animals with tamoxifen and CTX, detach the cells by trypsinization and count.
- Centrifuge the cells at 232 x g for 5 min.
- Wash the cell pellet in Ca2+– and Mg2+-free PBS, centrifuge and then gently resuspend the cell pellet in Ca2+– and Mg2+-free PBS to a final concentration of 106 cells/30 μl, which will be the final volume of each injection.
- Inject 30 μl of cell suspension into the previously injured muscles using a syringe with 29 or 30 G needle. Pay particular attention while removing the needle from the muscle. Do it slowly, avoiding spilling the cell suspension through the needle's track. Do not transplant the contralateral TA and use it as control, injecting 30 μl of Ca2+– and Mg2+-free PBS to replicate the conditions of the transplanted one.
- Treat the animals with tamoxifen for six additional days and administer analgesia (e.g. Carprofen) for two additional days.
- Explant the muscles from 14 days after transplantation onwards. Process and analyze the samples as detailed in Protocols 4 and 5.
2. Transplantation in Mouse Models of Muscular Dystrophy
This transplantation assay allows evaluating the extent of engraftment of IDEMs in mouse models of muscular dystrophy. The animals, treated as follows, can also be assessed for functional amelioration of the disease phenotype. Functional tests can be performed starting from two weeks after transplantation. In order to enhance engraftment consider performing pretransplantation treadmill exercise (as described in Protocol 3) and/or serial cell injections every three weeks for 3x (i.e. for a total of 3 injections/muscle).
- Intramuscular transplantation
- Pretreat the animals with an intra-peritoneal injection of 3.5 μl/g of 10 mg/ml tamoxifen (liposoluble formulation) 24 hr before transplantation and pretreat cells adding 1 μM 4OH-tamoxifen (aqueous formulation) into the growth medium overnight before the transplantation day.
- Detach by trypsinization, count and centrifuge the cells at 232 x g for 5 min.
- Wash the cell pellet in Ca2+– and Mg2+-free PBS, centrifuge and then resuspend the pellet in Ca2+– and Mg2+-free PBS to a concentration of 106 cells/30 μl.
- Administer analgesia and disinfect the skin of the animal (optional) with a povidone iodine- or chlorexidine-based disinfectant. This will also help to localize the tibialis anterior (TA), gastrocnemius (GC), and quadriceps femoris (QC; specifically the vastus intermedius) muscles.
- Inject 30 μl of cell suspension into the muscles using a syringe with 29 or 30 G needle. For the TA, insert 5 mm of the needle 2 mm below the insertion of the proximal tendon (craniocaudal direction) with a 15° inclination relative to the tibia and slowly inject the cell suspension while retracting the needle (empty the syringe with 2 mm of the needle still inside the muscle). For the GC and QC, repeat the same procedure as detailed for the TA, with the main difference being the caudocranial insertion of the needle 2 mm above the myotendinous junction of the Achilles tendon for the GC and 2 mm above the distal tendon for the QC (15° inclination with respect to the femur). Pay attention while removing the needle from the muscle in order to avoid spilling the cell suspension through the needle's track.
TROUBLESHOOTING: In case of low engraftment consider injecting juvenile (1-2 weeks old) mice7 with 3 x 105 cells/10 μl (note that in this case tamoxifen needs to be administered subcutaneously).
- Intra-arterial transplantation
This part of the protocol allows the evaluation of the ability of MIDEMs and HIDEMs to be delivered in the arterial circulation, cross the vessel wall and contribute to skeletal muscle regeneration in the muscles downstream to the injection site.- Pretreat animals and cells as described above in step 2.1.1.
- Detach by trypsinization and filter with a 40 μm cell strainer (to remove clusters from the cell suspension in the unlikely event that overnight exposure to tamoxifen could drive fusion and formation of myotubes from adjacent cells) count and centrifuge the cells at 232 x g for 5 min
- Wash the cell pellet in Ca2+– and Mg2+-free PBS, centrifuge and then resuspend the pellet in Ca2+– and Mg2+-free PBS with 0.2 International
Units of sodium heparin (optional) to a final cell concentration of 106 cells/50 μl. Add 10% Patent blue dye (final concentration: 1.25 mg/ml in normal saline or Ca2+– and Mg2+-free PBS) to the solution in order to visualize the distribution of the cell suspension. - Administer anesthesia and analgesia to the mouse according to the guidelines regulating surgical procedures in the specific institutional animal facility.
- Shave the inguinal region (also know as femoral or Scarpa's triangle) and disinfect the skin with a povidone iodine- or chlorhexidine-based disinfectant.
- Make a 5-7 mm incision and localize the femoral bundle: vein, artery and nerve (the nerve lays laterally to the artery and the vein medially).
- Gently remove the connective fascia that covers the bundle with forceps.
- Separate the femoral vein and nerve from the artery by gently introducing the tip of a forceps (or a 30 G needle) in between them and by progressively enlarging the hole.
TROUBLESHOOTING: The femoral vein is fragile: pay attention not to pinch it with the forceps during its detachment from the femoral artery. In case of hemorrhage, drain the blood with sterile gauze and use a micro cauterizer to facilitate hemostasis. - Lift the artery with one tip of the forceps and clamp the artery with the other tip.
- Puncture the artery with a syringe equipped with a 30 G needle. Inject 50 μl of cell suspension downstream of the clamped area, at an infusion speed of approximately 5 μl/sec.
TROUBLESHOOTING: Carefully resuspend the cells in the syringe before the injection: it is vital to avoid precipitation of cells and air bubble formation inside the syringe. The diameter of the femoral artery is slightly smaller than a 30 G needle: be careful not to truncate the artery while inserting the needle. Patent blue dye allows recognizing the effectiveness of the injection: if correctly injected, the whole limb will rapidly turn light blue. - Slowly remove the needle and the forceps from the artery to restore bloodstream in the limb.
- Apply pressure with sterile gauze to avoid bleeding and/or cauterize as required.
- Suture the wound and monitor the animals until recovery from anesthesia.
- Administer analgesia for 3 days and inspect the wound daily (in case of wound infection discuss this with the animal facility personnel and administer antibiotics as required).
3. Outcome Measures on Transplanted Dystrophic Animals: Treadmill Test
Starting from two weeks after cell transplantation, it is possible to evaluate functional amelioration of the motor capacity of treated mice with the treadmill tests. This test allows the evaluation of exercise tolerance/endurance of treated mice. A mouse is considered fatigued when lays in the resting area for more than 5 sec, without attempting to reengage the treadmill after a series of 3 consecutive mechanical stimuli (one every 5 sec). Baseline measurements start approximately one month before treatment and are used to evaluate the improvement of each single tested animal. This test can be followed by additional assays to monitor fiber fragility, force improvement, engraftment, differentiation of transplanted cells, and morphological amelioration of the transplanted muscles (see Discussion).
- Acclimatize the animals (usually three groups: treated, untreated, and wild type/non dystrophic controls) to the exercise before the first measurement: set the treadmill at a speed of 6 m/min for 10 min. Repeat the procedure every other day for one week.
TROUBLESHOOTING: Record all the measurements at the same hour of the day to avoid biases owing to the circadian cycles. - Place the animals into the treadmill, which has been previously set up with an inclination of 10°.
- Turn on the treadmill, with a starting speed of 6 m/min (provide a gentle mechanical stimulus in case the animals are not willing to start the exercise).
- Start the timer and increase the speed 2 m/min every 2 min.
- As soon as an animal lies in the resting area for more than 5 sec without attempting to reengage the treadmill, gently touch it with a stick to stimulate the restart of the exercise (see above).
- Record the performance (time and or distance) of each animal.
- Repeat the measurements weekly or every 10 days for at least 3x.
- Repeat the same procedure after transplantation.
- Analyze performance data comparing the measurement of each animal to its baseline performance. We suggest to plot the values in a graph as a percentage of the average motor capacity relative to baseline and to analyze them by a one- or two-way ANOVA test followed by appropriate post-test to compare the groups.
TROUBLESHOOTING: use a minimum of 5 age-, genotype-, and sex-matched animals/group and repeat the measurements for at least 3x after transplantation.
4. Evaluation of Cell Engraftment and Differentiation in Transplanted Muscles
Transplanted and control muscles are harvested at the appropriate time point (<2 days for short-term engraftment analysis, 2-3 weeks for mid-term and >1 month for long-term analysis). If the transplanted cells are labeled with GFP, the engraftment in freshly isolated muscles can be assessed by direct fluorescence under an UV-equipped stereomicroscope.
- Lay and orient along the vertical axis freshly isolated muscles in tragachanth gum (6% w/v)
- Dehydrate the samples in prechilled isopentane for one minute, freeze them in liquid nitrogen for at least 2 min and place them immediately at -80 °C for storage.
- Process the samples with a cryostat to obtain 7 μm thick sections on polarized slides. Sample the majority of the muscle on the slides, collecting approximately 8-10 slides with 30-40 sections/slide. It is advisable to collect a series of sections into a 1.5 ml tube to perform molecular biology/biochemistry assays.
- Evaluate cell engraftment with different immunofluorescent staining, depending on the experimental setup. For example, in case of HIDEM transplantation in Sgca-null/scid/bg mice, stain sections with: a) an antibody against Lamin A/C to detect grafted human cell nuclei; b) an antibody against Laminin to visualize the overall structure of the muscle; and c) an antibody against Sgca to detect donor-derived restoration of the protein absent in the dystrophic animal.
- Quantify, using a fluorescence microscope, the number of donor nuclei per muscle section inside and outside muscle fibers and the number of donor-derived skeletal myofibers per section.
5. Muscle Histopathology
Histopathological analyses allow the evaluation of the morphological structure of the transplanted muscle. Architectural improvement in the tissue structure is expected as an outcome of the cell therapy approach.
- Fix the freshly isolated muscles with 4% paraformaldehyde for 1 hr at 4 °C.
- Dehydrate the samples with an ascending sucrose gradient (e.g. 7.5-15-30% w/v).
- Leave the muscles overnight in the highest sucrose solution.
- Embed the samples in Tissue Tek OCT, place them in prechilled isopentane until the OCT becomes solid (avoiding complete immersion of the samples), freeze them in liquid nitrogen for at least 2 min and place them immediately at -80 °C for storage.
- Process the samples with a cryostat to obtain 7 μm thick sections as described above.
- Stain the sections with hematoxylin and eosin or Masson's trichrome according to standard manufacturer's protocols.
Hematoxylin and eosin staining enables calculating hallmarks of regenerating muscle, such as: a) the number of myofibers; b) cross sectional area; c) the number of myofibers containing a central nucleus. Masson's trichrome is used to calculate the fibrotic index, done by subtracting the total area occupied by the skeletal myofibers from the total area of the image: the resulting area mainly reflects the connective and fat infiltrate of the muscle. All the analyses on the images could be performed using ImageJ software (NIH) with the measurement tool and cell counter plugin.
Representative Results
The reported representative results follow the main in vitro/in vivo assays depicted in the workflow in Figure 1. 48 hr after 4OH-tamoxifen administration MyoD-positive nuclei are identifiable within MyoD-ER transduced IDEMs in culture (Figure 2A). The cells then fuse and differentiate into multinucleated myotubes (Figure 2B). When transplanted intramuscularly into a murine model of acute muscle injury, IDEMs contribute to tissue regeneration (Figure 3). The efficacy of IDEMs in a gene- and cell-therapy setting for murine models of muscular dystrophy was assessed by the treadmill exercise tolerance test: Figure 4 shows the results obtained after transplantation of wild-type MIDEMs into Sgca-null/scid/beige mice, displaying an amelioration of the motor capacity in treated mice7. Ex vivo analyses of transplanted muscles show GFP-positive areas representing the extent of colonization of IDEMs into the host tissue (Figures 5A-C), thus demonstrating that donor cells engraft into dystrophic muscle. Importantly, transplanted cells are able to differentiate in vivo, forming new skeletal myofibers. Indeed Figure 5 shows Sgca expression from genetically corrected HIDEMs into Sgca-null/scid/beige mice (Figures 5D and 5E). Structural amelioration in the architecture of transplanted muscles can be assessed through Masson's trichrome staining: Figure 5F shows a decrease in the amount of fibrotic tissue in treated muscle.
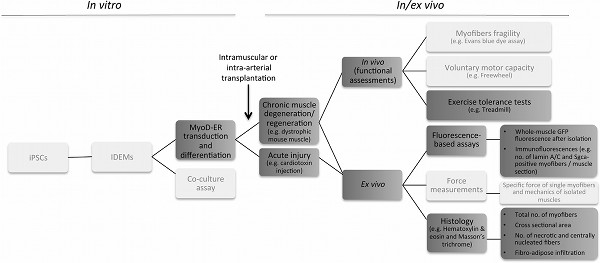
Figure 1. Protocol flow chart. The scheme provides an overview of the IDEM-based strategy, from preliminary in vitro differentiation assays (left) to the various steps necessary to assess engraftment, myogenic potential and functional amelioration in vivo and ex vivo (right). Dark grey boxes contain the various steps described in the protocol; light grey boxes contain parts of the method not detailed in this article. Click here to view larger figure.
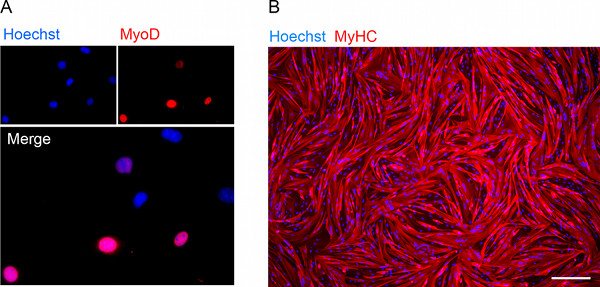
Figure 2. Assessment of myogenic potential in vitro. (A) Immunofluorescence showing nuclear MyoD expression in 4 out of 7 MyoD-ER transduced MIDEM nuclei after 48 hr of exposure to 4OH-tamoxifen. (B) Immunofluorescence staining for myosin heavy chain (MyHC) on 4OH-tamoxifen-induced HIDEM-derived myotubes after one week in differentiation medium (Scale bar, 200 μm). Click here to view larger figure.
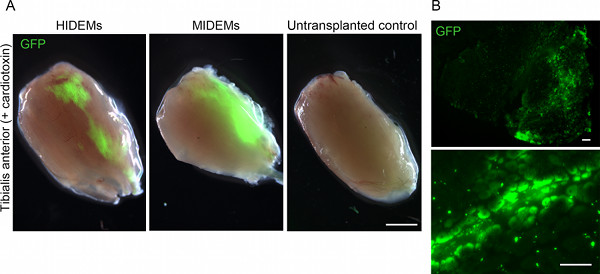
Figure 3. In vivo assessment of cell engraftment in a model of acute muscle regeneration. (A) Stereomicroscopic GFP fluorescence images of freshly isolated cardiotoxin-injured tibialis anterior muscles explanted 2 weeks after intramuscular injection of 106 GFP-HIDEMs (left) and GFP-MIDEMs (center). Scale bar, 2 mm. (B) Low (top) and high (bottom) magnification pictures of the muscle transplanted with MIDEMs shown in (A) displaying GFP-positive myofibers. Scale bar, 200 μm. Click here to view larger figure.
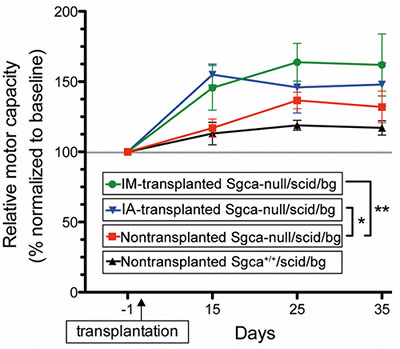
Figure 4. Treadmill exercise tolerance test. Representative treadmill test for transplanted (IM = intramuscular; IA = intra-arterial). Sgca-null/scid/ beige mice (106 cell/injection) versus nontransplanted dystrophic and nondystrophic control immunodeficient mice. The plot shows functional amelioration of dystrophic mice transplanted with MIDEMs (12-22% more than nontransplanted animals 35 days after transplantation). Data are shown as average motor capacity relative to baseline performances (i.e. 100% represents the baseline performance of each group and only treated mice significantly improve it upon repeated measurements). *P < 0.05; **P < 0.005, one-way ANOVA. From previously published work of the authors7. Click here to view larger figure.
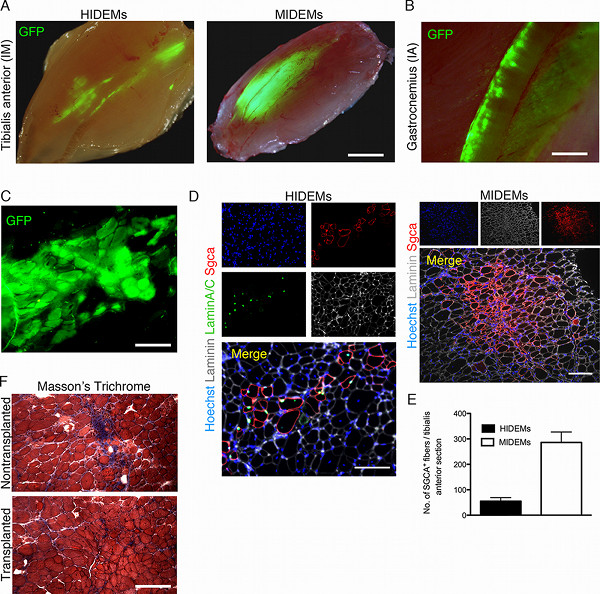
Figure 5. In vivo assessment of engraftment and myogenic potential in mouse models of muscular dystrophy. (A) Stereomicroscopic GFP fluorescence images of freshly isolated tibialis anterior muscles of Sgca-null/scid/beige mice explanted 3-4 weeks after intramuscular injection of 106 human (HIDEMs, left; transplantation in juvenile mice) and murine (MIDEMs; right) GFP-IDEMs. Scale bar, 2 mm. (B) Stereomicroscopic GFP fluorescence image of a freshly isolated gastrocnemius muscle explanted 3 weeks after intra-arterial injection of 106 GFP-MIDEMs. Scale bar, 1 mm. (C) Fresh frozen transverse section of the muscle transplanted with MIDEMs shown in (A) displaying a cluster of GFP-positive myofibers. Scale bar, 200 μm. (D) Immunofluorescence staining on sections of intra-muscularly transplanted muscles (as in A) showing clusters of genetically-corrected fibers, originated from grafted IDEMs. Scale bar, 150 μm. (E) Quantification of α-sarcoglycan (Sgca)-positive myofibers one month after intramuscular transplantation of genetically-corrected IDEMs into Sgca-null/scid/beige mice. (F) Masson trichrome staining of tibialis anterior muscles from transplanted and control Sgca-null/scid/beige mice (red: muscle fibers; blue: fibrosis) highlighting the reduction of the fibrotic infiltrate in treated muscle. Scale bar, 200 μm. Click here to view larger figure.
Discussion
iPSCs can be expanded indefinitely while preserving their self-renewal potential and thus be directed to differentiate into a broad range of cell lineages12. For this and other reasons iPSC-derived stem/progenitor cells are considered a promising source for autologous gene and cell therapy approaches13. A previously published work from the Authors reported the generation of mouse and human myogenic progenitors from iPSCs with a phenotype similar to that of pericyte-derived MABs (IDEMs) and their efficient contribution to muscle regeneration in a mouse model of muscular dystrophy7.
A prerequisite to maximize IDEM myogenic potential is the expression of the myogenic factor MyoD in the cells. The transduction with a tamoxifen-inducible MyoD-ER cassette enabled to have a precise temporal control over its expression with the advantage of synchronizing its activation in the whole culture and subsequently in vivo. A transplantation strategy based upon a single administration of cells per muscle (or per femoral artery) has been described in this article. However, repeated cell injections have been shown to be efficacious in increasing MAB engraftment in different cell therapy protocols8. Therefore, in case of suboptimal results with IDEMs, it is worth performing repeated transplantations to increase cell dosage and hence, contribution to host muscle.
Outcome measurements allow evaluation of the contribution of donor cells in the functional amelioration of the phenotype of treated animals. In addition to the exercise tolerance/endurance test performed with the treadmill, further tests to analyze voluntary motor capacity (e.g. freewheel test), fiber fragility (e.g. Evans blue dye uptake assay) and specific force (e.g. single fibers or whole muscle mechanics) can be considered8. Additional methods to test functional phenotype amelioration are available as standard operating procedures in the Treat-NMD website (http://www.treat-nmd.eu/research/preclinical/dmd-sops/).
To gather significant information from the transplanted animals, the functional amelioration must be followed and validated by immunohistochemical and histological analyses, to evaluate cell engraftment and tissue architecture/remodeling. In particular, it is important to evaluate the morphometric structure of the grafted muscles for hallmarks of regeneration and fibrosis, such as assessing the number of fibers with a central nucleus, the myofibers cross sectional area and the fibrotic index. The latter analyses, together with the functional outcomes give an exhaustive overview on the effectiveness of the strategy. Moreover, monitoring the lineage adopted by transplanted cells in terms of contribution to the tissue (i.e. myofibers) and maintenance of the stem cell compartment is important for the long-term efficacy of all cell therapy strategies. In this case, it has been already reported the donor-derived contribution in vivo to the pericyte compartment from which mesoangioblasts are derived7; additionally, preliminary results indicate functional contribution from IDEMs also to the satellite cell pool (Gerli and Tedesco, unpublished results). Molecular analyses to demonstrate engraftment and expression of the corrected gene (i.e. quantitative PCR and western blot) are also critical, but are beyond the scope of this paper and would require a dedicated article.
Although this protocol has been mainly developed utilizing Sgca-null/scid/beige mice7 (a recently published immunodeficient model of LGMD2D), it is expected that it could be easily applicable to other models of muscle disease. In the case of Sgca-null/scid/beige, mice we did not use cardiotoxin, since the muscles of these animals are severely compromised and chronically subject to degeneration and regeneration cycles (thus an additional damage from cardiotoxin is not necessary). However, we could not exclude that a pretreatment with cardiotoxin could facilitate engraftment in milder models of muscular dystrophy (e.g. mdx mice). Ameliorations of our strategy may include methods to enhance the immunodeficiency of the mouse host (which in the current models is still suboptimal) and the efficacy of donor cell engraftment, in particular upon intra-arterial delivery of xenogeneic cells. Although multiple cell transplantation and the use of juvenile mice contribute to the increase in cell engraftment7,8, the mouse adhesion molecules encountered by human cells during extravasation and/or migration might pose additional hurdles to the limited immunodeficiency of the current models. Future directions for the translation of this gene and cell therapy approach in a clinical setting may include pharmacological strategies to enhance cell extravasation and the use of nonintegrating vectors (e.g. plasmids, mRNAs, human artificial chromosomes, or nonintegrating viral vectors) to reprogram and genetically correct the cells, thus avoiding the risk of insertional mutagenesis.
Divulgations
The authors have nothing to disclose.
Acknowledgements
The authors thank Giulio Cossu, Martina Ragazzi and the whole laboratory for helpful discussion and support, and Jeff Chamberlain for kindly providing MyoD-ER vector. All the experiments involving live animals were completed in accordance and compliance with all relevant regulatory and institutional agencies, regulations and guidelines. Work in the authors laboratory is supported by UK Medical Research Council, European Community 7th Framework projects Optistem and Biodesign and the Italian Duchenne Parent Project.
Materials
| REAGENTS | |||
| MegaCell DMEM | Sigma | M3942 | |
| DMEM | Sigma | D5671 | |
| IMDM | Sigma | I3390 | |
| Horse serum | Euroclone | ECS0090L | |
| Foetal Bovine Serum | Lonza | DE14801F | |
| PBS Calcium/Magnesium free | Lonza | BE17-516F | |
| L-Glutammine | Sigma | G7513 | |
| Penicilline/Streptomicin | Sigma | P0781 | |
| 2-Mercaptoethanol | Gibco | 31350-010 | |
| ITS (Insulin-Transferrin-Selenium) | Gibco | 51500-056 | |
| Non-essential amino acid solution | Sigma | M7145 | |
| Fer-In-Sol | Mead Johnson | ||
| Ferlixit | Aventis | ||
| Oleic Acid | Sigma | 01257-10 mg | |
| Linoleic Acid | Sigma | L5900-10 mg | |
| Human bFGF | Gibco | AA 10-155 | |
| Grow factors-reduced Matrigel | Becton Dickinson | 356230 | |
| Trypsin | Sigma | T3924 | |
| Sodium heparin | Mayne Pharma | ||
| Trypan blue solution | Sigma | T8154 | HARMFUL |
| Patent blue dye | Sigma | 19, 821-8 | |
| EDTA | Sigma | E-4884 | |
| Paraformaldehyde | TAAB | P001 | HARMFUL |
| Tamoxifen | Sigma | T5648 | |
| 4-OH Tamoxifen | Sigma | H7904 | |
| pLv-CMV-MyoD-ER(T) | Addgene | 26809 | |
| Cardiotoxin | Sigma | C9759 | HARMFUL |
| Povidone iodine | |||
| Tragachant gum | MP biomedicals | 104792 | |
| Isopenthane | VWR | 24,872,323 | |
| Tissue-tek OCT | Sakura | 4583 | |
| Sucrose | VWR | 27,480,294 | |
| Polarized glass slides | Thermo | J1800AMNZ | |
| Eosin Y | Sigma | E4382 | |
| Hematoxylin | Sigma | HHS32 | |
| Masson's trichrome | Bio-Optica | 04-010802 | |
| Mouse anti Myosin Heavy Chain antibody | DSHB | MF20 | |
| Mouse anti Lamin A/C antibody | Novocastra | NLC-LAM-A/C | |
| 4/11/13 | Cappel | 559762 | |
| Hoechst 33342 | Sigma fluka | B2261 | |
| Rabbit anti Laminin antibody | Sigma | L9393 | |
| MATERIALS AND EQUIPMENT | |||
| Adsorbable antibacteric suture 4-0 | Ethicon | vcp310h | |
| 30G needle syringe | BD | 324826 | |
| Treadmill | Columbus instrument | ||
| Steromicroscope | Nikon | SMZ800 | |
| Inverted microscope | Leica | DMIL LED | |
| Isoflurane unit | Harvad Apparatus | ||
| Fiber optics | Euromecs (Holland) | EK1 | |
| Heating pad | Vet Tech | C17A1 | |
| Scalpels | Swann-Morton | 11REF050 | |
| Surgical forceps | Fine Scientific Tools | 5/45 | |
| High temperature cauteriser | Bovie Medical | AA01 | |
| MEDIA COMPOSITION | |||
| Media composition is detailed below. HIDEMs growth medium:
|
|||
Table 1. List of Reagents, Materials, Equipment, and Media.
References
- Dellavalle, A., et al. Pericytes of human skeletal muscle are myogenic precursors distinct from satellite cells. Nat. Cell Biol. 9, 255-267 (2007).
- Minasi, M. G., et al. The meso-angioblast: a multipotent, self-renewing cell that originates from the dorsal aorta and differentiates into most mesodermal tissues. Development. 129, 2773-2783 (2002).
- Dellavalle, A., et al. Pericytes resident in postnatal skeletal muscle differentiate into muscle fibres and generate satellite cells. Nat. Commun. 2, 499 (2011).
- Tedesco, F. S., Dellavalle, A., Diaz-Manera, J., Messina, G., Cossu, G. Repairing skeletal muscle: regenerative potential of skeletal muscle stem cells. J. Clin. Invest. 120, 11-19 (2010).
- Sampaolesi, M., et al. Cell therapy of alpha-sarcoglycan null dystrophic mice through intra-arterial delivery of mesoangioblasts. Science. 301, 487-492 (2003).
- Sampaolesi, M., et al. Mesoangioblast stem cells ameliorate muscle function in dystrophic dogs. Nature. 444, 574-579 (2006).
- Tedesco, F. S., et al. Transplantation of Genetically Corrected Human iPSC-Derived Progenitors in Mice with Limb-Girdle Muscular Dystrophy. Sci. Transl. Med. 4, (2012).
- Tedesco, F. S., et al. Stem cell-mediated transfer of a human artificial chromosome ameliorates muscular dystrophy. Sci. Transl. Med. 3, (2011).
- Lattanzi, L., et al. High efficiency myogenic conversion of human fibroblasts by adenoviral vector-mediated MyoD gene transfer. An alternative strategy for ex vivo gene therapy of primary myopathies. J. Clin. Invest. 101, 2119-2128 (1998).
- Chaouch, S., et al. Immortalized skin fibroblasts expressing conditional MyoD as a renewable and reliable source of converted human muscle cells to assess therapeutic strategies for muscular dystrophies: validation of an exon-skipping approach to restore dystrophin in Duchenne muscular dystrophy cells. Hum. Gene Ther. 20, 784-790 (2009).
- Kimura, E., et al. Cell-lineage regulated myogenesis for dystrophin replacement: a novel therapeutic approach for treatment of muscular dystrophy. Hum. Mol. Genet. 17, 2507-2517 (2008).
- Yamanaka, S. Induced pluripotent stem cells: past, present, and future. Cell Stem Cell. 10, 678-684 (2012).
- Wu, S. M., Hochedlinger, K. Harnessing the potential of induced pluripotent stem cells for regenerative medicine. Nat. Cell Biol. 13, 497-505 (2011).

