Visualization of Streptococcus pneumoniae within Cardiac Microlesions and Subsequent Cardiac Remodeling
Summary
Streptococcus pneumoniae forms discrete non-purulent microscopic lesions in the heart. Outlined is the protocol for a murine model of cardiac microlesion formation. Instruction is provided on microlesion visualization using microscopy, discrimination between early and late microlesions, and methods to detect cardiac remodeling in hearts of convalescent animals.
Abstract
During bacteremia Streptococcus pneumoniae can translocate across the vascular endothelium into the myocardium and form discrete bacteria-filled microscopic lesions (microlesions) that are remarkable due to the absence of infiltrating immune cells. Due to their release of cardiotoxic products, S. pneumoniae within microlesions are thought to contribute to the heart failure that is frequently observed during fulminate invasive pneumococcal disease in adults. Herein is demonstrated a protocol for experimental mouse infection that leads to reproducible cardiac microlesion formation within 30 hr. Instruction is provided on microlesion identification in hematoxylin & eosin stained heart sections and the morphological distinctions between early and late microlesions are highlighted. Instruction is provided on a protocol for verification of S. pneumoniae within microlesions using antibodies against pneumococcal capsular polysaccharide and immunofluorescent microscopy. Last, a protocol for antibiotic intervention that rescues infected mice and for the detection and assessment of scar formation in the hearts of convalescent mice is provided. Together, these protocols will facilitate the investigation of the molecular mechanisms underlying pneumococcal cardiac invasion, cardiomyocyte death, cardiac remodeling as a result of exposure to S. pneumoniae, and the immune response to the pneumococci in the heart.
Introduction
Hospitalization of adults for community-acquired pneumonia (CAP) carries a documented risk for adverse cardiac events that contributes to mortality1-4. In a recent study by Corrales-Medina et al. cardiac complications were found to be associated with and/or responsible for 27% of pneumonia-associated deaths3. Streptococcus pneumoniae (the pneumococcus), the most common cause of CAP and sepsis5, has been directly associated with adverse cardiac events in as many as 19% of admitted adult patients6. Adverse cardiac events associated with pneumonia include new or worsened congestive heart failure, arrhythmias, and myocardial infarction in order of decreasing frequency6.
In a recent PLoS Pathogens article by Brown et al., S. pneumoniae was found to be capable of translocation through the cardiac vascular endothelium, entry into the myocardium, and formation of discrete non-purulent microscopic lesions (microlesions) filled with bacteria in the ventricles during invasive pneumococcal disease (IPD)7. Evidence of cardiac microlesion formation was observed in cardiac samples from non-human primates and individuals who had succumbed to pneumococcal infection. Likewise, experimentally infected mice reproducibly developed cardiac microlesions. In mice, microlesion size and number was positively correlated with the duration and severity of bacteremia, levels of cardiac troponin in sera, and aberrant cardiac electrophysiology. Bacterial translocation into the heart was found to occur through the same mechanisms responsible for translocation of the pneumococcus across the blood-brain barrier and development of pneumococcal meningitis, i.e., Choline-binding protein A mediated invasion of vascular endothelial cells in a Laminin receptor and Platelet-activating factor receptor dependent manner8. Microlesion formation also required the pneumococcal pore-forming toxin pneumolysin that was found to kill cardiomyocytes7.
Pneumococcal cardiac microlesions are distinct from the purulent soft-tissue and cardiac abscesses that are caused by other Gram-positive bacteria including Staphylococcus aureus. These are characterized by a single foci of bacteria surrounded by neutrophils and fibrin deposition9,10. Microlesions formed by S. pneumoniae are smaller in size, distributed throughout an affected heart, and lack immune cell infiltrates. During the early stages of infection, microlesions caused by S. pneumoniae appear as areas of damaged or inflamed tissue reminiscent of the pathological signs of cardiomyopathy. Some monocytes may be observed during this time, however their presence is short lived and lesions rapidly become necrotic in appearance and filled with bacteria while simultaneously continuing to grow in size until the death of the animal or antimicrobial intervention. Importantly, within 3 days of antibiotic intervention, profuse immune cell infiltration is observed at the former lesion site and this is accompanied by robust collagen deposition. Similar cardiac remodeling has been reported to occur following infarction along with lasting consequences on cardiac function11-15. Thus, microlesions are a potential explanation for the adverse cardiac events that occur during IPD and possibly the increased incidence of cardiac-related mortality in convalescent individuals who have survived the disease episode.
Herein, instruction is provided on the experimental mouse model of IPD and cardiac lesion formation and visualization of cardiac microlesions at early and late stages of infection. The protocol for detection of collagen deposition in animals that have been saved by antimicrobial intervention is demonstrated. The goal of this article is to facilitate the research of other investigators on this important and novel pneumococcal pathology.
Protocol
NOTE: All mouse experiments were reviewed and approved by the Institutional Animal Care and Use Committees at The University of Texas Health Science Center at San Antonio (protocol #13032-34-01C). Animal care and experimental protocols adhered to Public Law 89-544 (Animal Welfare Act) and its amendments, Public Health Services guidelines, and the Guide for the Care and Use of Laboratory Animals (U.S. Department of Health & Human Services).
1. Infection
- Obtain BALB/c mice of both sexes between the ages of 10-12 weeks of age.
NOTE: S. pneumoniae serotype 4 strain TIGR4 is a virulent clinical isolate that has been sequenced16. - Grow TIGR4 in Todd-Hewitt broth at 37 °C in 5% CO2 until reaching an optical density (OD)620 of 0.5 (corresponding to 1.0 x 108 colony forming units [CFU]/ml). Pellet the pneumococcal culture by centrifugation for 10 min at 3,500 x g and remove the supernatant using a vacuum line. Suspend and dilute bacteria with sterile phosphate-buffered saline (PBS) to a final concentration of 1.0 x 104 CFU/ml.
- Using an induction chamber, anesthetize mice with 2.5% vaporized isoflurane in oxygen. Confirm anesthetized state by gentle toe pinch with blunt tweezers.
- Holding the anesthetized mouse by its scruff and upright with one hand, inject each mouse intraperitoneally (i.p.) with 100 µl of the S. pneumoniae suspension using a syringe with a 27-30 gauge needle (corresponding to a challenge dose of 1.0 x 103 CFU). Place the mouse back in its cage. Mice typically awaken 30-40 sec after injection.
- Allow the infection to proceed for at least 24 (early) or 30 (advanced) hr.
- Euthanize the mice by CO2 asphyxiation. Perform cervical dislocation to ensure mouse has deceased. Euthanasia is further confirmed by removal of the heart in Step 1.6.
- Disinfect the tail with an alcohol swab and snip a 2-3 mm section. Collect 2 µl of blood and serially dilute the blood 10-fold in PBS containing sodium heparin 1 U/ml five times. Plate serial dilutions on a tryptic soy blood agar plate and incubate overnight at 37 °C in 5% CO2. The next day extrapolate from colony counts the level of bacteremia.
- Immobilize the mouse in a supine position on a surgical platform. Spray the chest with 70% ethanol and pat dry. Using surgical scissors and forceps open the chest cavity, remove the rib cage, and transect the diaphragm to expose the heart and lungs. Using scissors cut the blood vessels connected to the heart and gently excise the heart with care to avoid bruising with forceps.
- Rinse the heart with PBS and then place into tissue specimen collection cassettes such that coronal sections would be obtained during tissue sectioning. For paraffin embedding, place the cassettes in 10% buffered-formalin solution and the next day send for paraffin embedding.
- Alternatively flash freeze using O.C.T. Compound within a cryomold that has been placed on dry ice.
2. Visualization of Cardiac Lesions and Pneumococci within Lesions
- Cut down paraffin-embedded heart sections such that the 4 chambers of the heart are visible in each tissue section. Cut slides at thickness of 5 µm.
- Deparaffinize and stain slides with hematoxylin & eosin (H&E) using standard methods17.
- View H&E stained cardiac sections using a light microscope at low (100X, 200X) and high (oil immersion: 400X, 1,000X) magnifications. Characterize hearts for the presence of microlesions in the myocardium. Here, acquire H&E images using a Zeiss Axioskop 2 microscope equipped with a 100X 1.3 numerical aperture Plan-NEOFLUAR objective.
NOTE: At early stages cardiac lesions are discriminated by their differential colored appearance and in some instances the presence of immune cells (monocytes and granulocytes) what appear to be monocytes. In advanced lesions, particularly those seen at 30 hr, immune cells are absent and large vacuole-like lesions are observed in their place. It is important to note that while immune cell influx during the early stages of cardiac lesion development can occur it does not appear to be a requirement.
3. Immunofluorescent Microscopy for Pneumococci within Microlesions
- Section heart in cryomolds with microtome to a thickness of 5 μm and place sections onto positively charged glass slides. Allow frozen cardiac sections to thaw and air dry. Fix sections for 10 min in 10% neutral buffered formalin (pH 6.8-7.2 at 25 °C).
- Wash slides (3x) in PBS for 5 min, permeabilize for 15 min in 0.2% Triton X-100 in PBS, then wash again in PBS.
- Block tissue section on slides with 10% goat serum in PBS for 1 hour.
- Rinse slides with PBS, cover and incubate cardiac sections with rabbit anti-serotype 4 pneumococcus antiserum at 1:1,000 in PBS with 10% goat serum for 2 hr at 37 °C.
NOTE: For negative controls investigators should use naive rabbit antiserum - After washing with PBS, cover and incubate sections with 10% goat serum-PBS with goat anti-rabbit FITC conjugated antibody at 1:2,000 for 30 min at 37 °C.
- Following the manufacturer’s instruction, use DAPI (4',6- Diamidino-2-Phenylindole) at 5 mg/ml for visualization of eukaryotic nuclei. Wash and mount tissue sections with FluorSave.
- Acquire fluorescent images using a confocal microscope system at both low and high magnification. Adjust the numerical aperture accordingly to obtain optimal results. Here, acquire fluorescent images using a confocal system with a 60X 1.42 numerical aperture objective.
4. Antibiotic Rescue of Septic Mice
- Begin with mice infected i.p. with 1.0 x 103 CFU of TIGR4 as previously indicated in step 1.
- Beginning at 30 hr post-infection, administer pharmaceutical grade ampicillin i.p. (80 mg/kg body weight) in 100 µl saline every 12 hr for 36 hr. Collect hearts at the desired time point and process as described in step 1.6 for paraffin embedding and tissue, sectioning.
5. Collagen Deposition Detection
- Using xylene and graded alcohol washes with deionized water, deparaffinize and hydrate tissue sections using standard methods17. Treat sections with aqueous 0.2% phosphomolybdic acid for 5 min and rinse with deionized water for 5 min.
- Stain sections with 0.1% Sirius Red in picric acid for 2 hr at room temperature.
- Wash sections in 0.01 N hydrochloric acid for 3 min and rinse using 70% ethanol for 1 min.
- Dehydrate the sections using the identical but reverse order of washes to that outlined in 5.1, dehydrate cardiac sections in increasing and graded concentrations of ethanol then xylene.
- Mount tissue sections with a mounting solution for microscopic evaluation.
- To identify regions of collagen deposition, examine stained tissue sections for areas of deep read staining under low and high magnification using a light microscope. Any area within the ventricles of the heart that is red is an area where collagen has been deposited. Areas without this red stain are sites of healthy tissue lacking collagen deposition. Herein, acquire Picro Sirius Red stained images using a microscope equipped with a 20X 0.5 numerical aperture objective.
NOTE: The atrium stains red in healthy tissues.
Representative Results
Visualization of Microlesions by H&E
Cardiac microlesions were observed in the ventricles of H&E stained cardiac sections from mice with IPD following their i.p. challenge with TIGR4. Figure 1 demonstrates the increase in size of these lesions between 24 and 30 hr post-infection (hpi), with greater number of hematoxylin-stained (i.e., purple-blue) pneumococci visible within the lesion. Microlesions were characterized by a vacuole-like morphology, denser eosin (red) staining of cardiomyocytes in the cells immediately adjacent to the lesion site, diplococci within the lesions, and a general absence of immune cells. Approximately half the lesions were adjacent to visible blood vessels. Between 24 and 30 hpi microlesions became more numerous and substantially larger. At 30 hpi extracellular bacteria could often be seen streaming from a lesion between cells.
Visualization of S. pneumoniae Using Immunofluorescent Microscopy
To visualize and confirm that S. pneumoniae were indeed within lesions, immunofluorescent staining for serotype 4 capsular polysaccharide that is produced by TIGR4 (Figure 2) was used. Images captured showed the presence of tightly packed and dense aggregates of S. pneumoniae within microlesion sites.
Evidence of Cardiac Scarring
Picro Sirius Red staining was performed on cardiac sections from mice that had been rescued with ampicillin and uninfected controls. In antibiotic rescued convalescent mice, there was a dramatic increase in the presence and intensity of the Picro Sirius Red stain with bands streaming from what appear to be former lesion sites in the ventricles (Figure 3). In contrast, the ventricles in control sections were unstained indicating that collagen deposition was absent (not shown).
Together these results demonstrate the ability to visualize cardiac microlesions in mice with severe IPD and to access resultant scar formation in the ventricles using standard histological procedures. Importantly, cardiac microlesions are invariably linked to high titers of S. pneumoniae and time needed to mediate their formation.
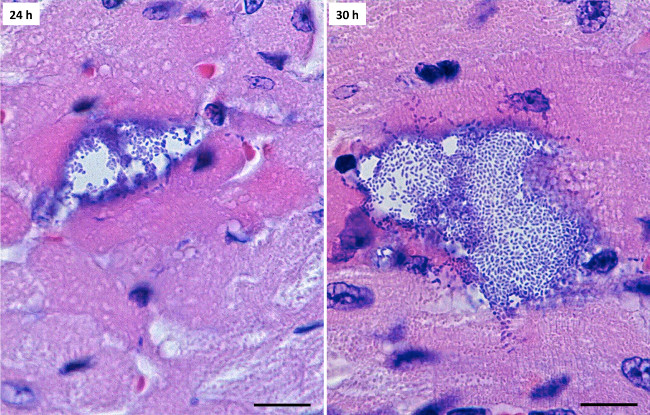
Figure 1: Detection of microlesions in H&E stained cardiac sections. Representative images (1,000X) of cardiac microlesions observed at 24 hor and 30 hr post infection. Black scale bars correspond to 10 µm. Please click here to view a larger version of this figure.
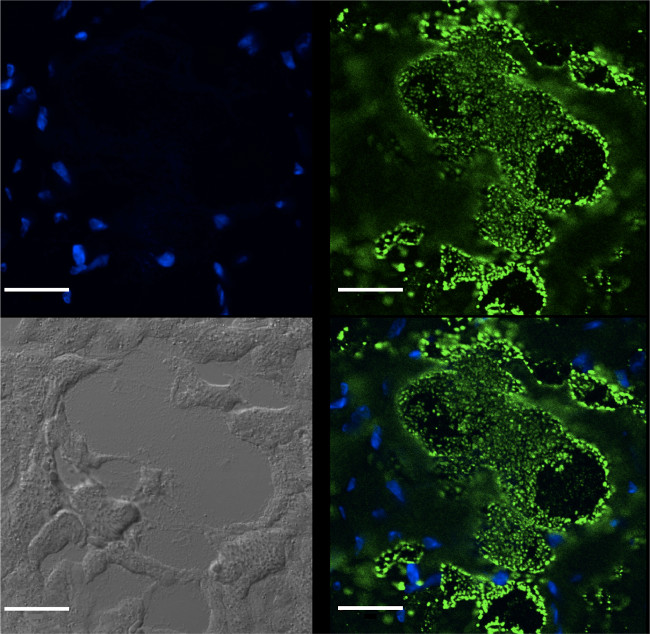
Figure 2: Immunofluorescent detection of S. pneumoniae within cardiac microlesions. Immunofluorescent microscopy using antibody against serotype 4 capsular polysaccharide (green) confirmed the presence of S. pneumoniae within cardiac microlesions. The nuclei were stained blue with DAPI. White scale bar correspond to 20 µm. Please click here to view a larger version of this figure.
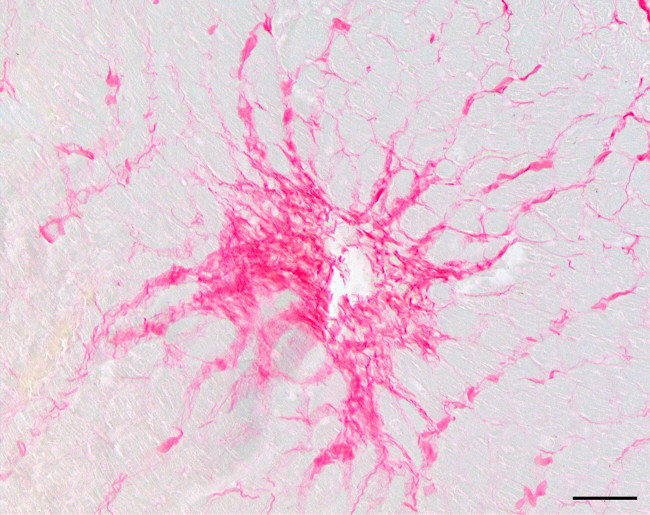
Figure 3: Collagen deposition at a former cardiac microlesion site in a convalescent mouse. Representative image (200X) showing a Picro Sirius Red stained cardiac microlesion in the ventricle of a mouse 5 days after antibiotic intervention for invasive pneumococcal disease. Red indicates collagen deposition. Black scale bar correspond to 20 µm. Please click here to view a larger version of this figure.
Discussion
In this report, a highly reproducible method to induce S. pneumoniae-mediated cardiac microlesions in mice during IPD and the techniques for their visualization is demonstrated. Mice are infected with a dose of bacteria that surpasses the immune system’s threshold for clearance, leading to high-grade bacteremia, similar to that which occurs during human sepsis, and leads to eventual bacterial translocation into the myocardium. For yet unknown reasons, S. pneumoniae in the heart is able to replicate undeterred by the host immune response. This is the subject of ongoing investigation and this publication may facilitate other investigators in their efforts to participate.
H&E staining allows for microlesion visualization using standard light microscopy since cardiomyocytes stain pink to red, whereas cardiac microlesions and the bacteria within stain blue to deep purple. This sharp contrast enables easy detection of the microlesions at later stages of infection when the lesions are larger. In order to verify the presence of S. pneumoniae within the cardiac microlesions, immunofluorescent microscopy using antiserum against serotype 4 capsular polysaccharide that is carried by TIGR4 was performed. Importantly, the specific anti-capsular antibody that should be used depends on the serotype of the pneumococcal strain that is used for experimental challenge. Alternatively, investigators can also use commercially available monoclonal antibodies against pneumolysin, the pore-forming toxin of S. pneumoniae, or that detect components of the pneumococcal cell wall (i.e., TEPC-15). While these antibodies detect pneumococci within the heart, past experience suggests that antibodies against capsular polysaccharide provide the most compelling images7.
S. pneumoniae mediated cardiac microlesion formation requires the interaction of the pneumococcal adhesin Choline binding protein A with the host protein Laminin receptor for bacterial adhesion to the vascular endothelial cells and interaction of bacteria cell wall phosphorylcholine with host Platelet-activating factor receptor for bacterial transmigration through the cell7,8. It is therefore important to note that microlesion formation is tightly linked to infectious burden and time, which increases the frequency and duration of these interactions and allows time for the bacteria to replicate and visible lesions to develop. We have previously determined that an i.p. infectious dose greater than 1.0 x 103 CFU of S. pneumoniae strain TIGR4 results in too rapid death of the mouse and as a result poor lesion formation. Below this infectious dose spontaneous clearance occurs in some mice and in others delayed onset of severe disease is observed. Yet for mice challenged with 1.0 x 103 CFU of TIGR4 between 24 and 36 hr, lesions rapidly accumulate in number and grow in size continuously until the mouse becomes moribund. Thus, the infectious dose of the infecting strain needs to be titrated to replicate a similar state of disease with a moribund state developing in no less than 30 hpi. Importantly, we have repeatedly observed cardiac lesion formation in mice that were infected intranasal with 1.0 x 107 CFU and intratracheally with 1.0 x 105 CFU of TIGR4. Following these challenge routes development of severe disease occurred at later time points. We also observed microlesion formation with other virulent strains of S. pneumoniae, such as serotype 2 isolate D39, albeit at different frequencies. Thus, lesions are not a result of the i.p. infection route or specific to TIGR4, but instead tied to duration and severity of the disease episode. Herein we advise to infect mice i.p. since this challenge route offers the highest degree of reproducibility. If a different strain of S. pneumoniae is used or a different infectious route is desired, investigators should aim for bacteremia at levels >105 CFU ml of blood for at least 24 hr.
Antimicrobial intervention allows the study of events that occur during convalescence, modeling a human in the Intensive Care Unit setting. It is worth noting that not all mice survive despite antimicrobial treatment and confirmed bacterial clearance. Investigators should expect survival rates between 50-60% and as such should adjust the cohort size accordingly to deal with the loss of animals. Regular monitoring of animals after 24 hr is highly recommended to ensure compliance with Institutional approved animal protocols. Picro Sirius Red staining is a simple and standard technique used to visualize collagen deposition. Herein, it is demonstrated how this stain can be used to examine cardiac remodeling in the ventricles of the heart following a traumatic infectious episode. Picro Sirius Red was chosen over other commonly used techniques to visualize collagen deposition, such as “Masson’s Trichrome Stain”, because of the sharp contrast it offered between the deposited collagen and surrounding tissue. This has proven to be tremendously useful when assessing the extent of myocardial collagen deposition following antibiotic in a quantitative fashion using software such as ImageJ.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This work was supported by grant 13IRG14560023 from the American Heart Association and NIH grants AI078972 and HL108054 to CJO. Support for AOB was from the NIH National Center for Advancing Translational Sciences NIH ULTR001120, and F31 A110417701.
Materials
| Name of Reagent/ Equipment | Company | Catalog Number | Comments/Description |
| Todd-Hewitt broth | Neogen | 7161A | |
| O.C.T Compound | Tissue-Tek (Sakura Finetek) | 4583 | |
| Triton X-100 | Fisher Scientific (Acros) | 9002-93-1 | |
| Normal Goat Serum | Abcam | ab7481 | |
| anti-serotype 4 pneumococcus antiserum | Statens Serum Institut | 16747 | |
| goat anti-rabbit FITC conjugated antibody | Jackson ImmunoResearch | 111-096-144 | |
| DAPI | Invitrogen | D1306 | |
| Fluorsave | Millipore | 345789 | |
| Permount | Fisher Scientific | S70104 | |
| Ampicillin | Sigma | A9393 | |
| phosphomolybdic acid 0.2% | Electron Microscopy Sciences | 26357-01 | |
| 0.1% Sirius Red in picric acid | Electron Microscopy Sciences | 26357-02 | |
| 0.01 N hydrochloric acid | Electron Microscopy Sciences | 26357-03 | |
| Tissue Tack Microscope Slides | Polysciences, Inc | 24216 | |
| Paralube Vet Ointment | Dechra | 12920060 | |
| Hematoxylin and Eosin Staining Kit | unspeciified | Standard |
References
- Corrales-Medina, V. F., et al. Cardiac complications in patients with community-acquired pneumonia: incidence, timing, risk factors, and association with short-term mortality. Circulation. 125, 773-781 (2012).
- Corrales-Medina, V. F., et al. Acute bacterial pneumonia is associated with the occurrence of acute coronary syndromes. Medicine (Baltimore). 88, 154-159 (2009).
- Corrales-Medina, V. F., et al. Cardiac complications in patients with community-acquired pneumonia: a systematic review and meta-analysis of observational studies. PLoS Med. 8, e1001048 (2011).
- Corrales-Medina, V. F., et al. Risk stratification for cardiac complications in patients hospitalized for community-acquired pneumonia. Mayo Clin. Proc. 89, 60-68 (2014).
- Kumar, S., et al. Detection of 11 common viral and bacterial pathogens causing community-acquired pneumonia or sepsis in asymptomatic patients by using a multiplex reverse transcription-PCR assay with manual (enzyme hybridization) or automated (electronic microarray) detection. Journal Of Clinical Microbiology. 46, 3063-3072 (2008).
- Musher, D. M., Rueda, A. M., Kaka, A. S., Mapara, S. M. The association between pneumococcal pneumonia and acute cardiac events. Clin Infect Dis. 45, 158-165 (2007).
- Brown, A. O., et al. Streptococcus pneumoniae translocates into the myocardium and forms unique microlesions that disrupt cardiac function. PLoS Pathogens. In press, (2014).
- Orihuela, C. J., et al. Laminin receptor initiates bacterial contact with the blood brain barrier in experimental meningitis models. The Journal of Clinical Investigation. 119, 1638-1646 (2009).
- Flick, M. J., et al. Genetic elimination of the binding motif on fibrinogen for the S. aureus virulence factor ClfA improves host survival in septicemia. Blood. 121, 1783-1794 (2013).
- Cheng, A. G., DeDent, A. C., Schneewind, O., Missiakas, D. A play in four acts: Staphylococcus aureus abscess formation. Trends Microbiol. 19, 225-232 (2011).
- Arenal, A., et al. Do the spatial characteristics of myocardial scar tissue determine the risk of ventricular arrhythmias. Cardiovasc Res. 94, 324-332 (2012).
- Deneke, T., et al. Human histopathology of electroanatomic mapping after cooled-tip radiofrequency ablation to treat ventricular tachycardia in remote myocardial infarction. J Cardiovasc Electrophysiol. 16, 1246-1251 (2005).
- Bakker, J. M., et al. Reentry as a cause of ventricular tachycardia in patients with chronic ischemic heart disease: electrophysiologic and anatomic correlation. Circulation. 77, 589-606 (1988).
- Verma, A., et al. Relationship between successful ablation sites and the scar border zone defined by substrate mapping for ventricular tachycardia post-myocardial infarction. J Cardiovasc Electrophysiol. 16, 465-471 (2005).
- Wu, K. C. Assessing risk for ventricular tachyarrhythmias and sudden cardiac death: is there a role for cardiac MRI. Circ Cardiovasc Imaging. 5, 2-5 (2012).
- Tettelin, H., et al. Complete genome sequence of a virulent isolate of Streptococcus pneumoniae. Science. 293, 498-506 (2001).
- Fischer, A. H., Jacobson, K. A., Rose, J., Zeller, R. Hematoxylin and eosin staining of tissue and cell sections. CSH Protoc. 2008, (2008).

