Patch Angioplasty in the Rat Aorta or Inferior Vena Cava
Summary
We have established a model of pericardial patch angioplasty that can be used in either small-diameter veins or arteries. This model can be used to compare venous and arterial neointimal hyperplasia formation.
Abstract
Pericardial patches are commonly used in vascular surgery to close vessels. To facilitate studies of the neointimal hyperplasia that forms on the patch, we developed a rat model of patch angioplasty that can be used in either a vein or an artery, creating a patch venoplasty or a patch arterioplasty, respectively. Technical aspects of this model are discussed. The infra-renal IVC or aorta are dissected and then clamped proximally and distally. A 3 mm venotomy or arteriotomy is performed in the infrarenal inferior vena cava or aorta of 6 to 8 week-old Wistar rats. A bovine pericardial patch (3 mm x 1.5 mm x 0.6 mm) is then used to close the site using a 10-0 nylon suture. Compared to arterial patches, venous patches show increased neointimal thickness on postoperative day 7. This novel model of pericardial patch angioplasty can be used to examine neointimal hyperplasia on vascular biomaterials, as well as to compare the differences between the arterial and venous environments.
Introduction
Vascular surgeons commonly close blood vessels with pericardial patches, either bovine or porcine, to reduce the risk of restenosis1,2. In arterial surgery, pericardial patch angioplasty reduces the rate of restenosis compared to a primary closure3,4. Patch venoplasty is also similarly performed to close veins, although less frequently when compared to arterial procedures5,6. Patches typically develop neointimal hyperplasia on their luminal surfaces; however, the mechanism of neointimal formation remains unclear, especially in the comparison of differences in neointimal formation between the arterial and venous environments. We previously showed that pericardial patches attract progenitor cells in both the arterial and the venous environments and that venous patch angioplasty shows more aggressive neointimal formation as compared to the arterial patches7,8.
In this study, we describe the rat model of pericardial patch angioplasty that is common to closing both veins and arteries and that may be used to study neointimal formation; this method is a small-animal model of an operative technique commonly used in human patients. The rat model has a larger diameter than the mouse model, facilitating technical aspects of the surgery and improving the learning curve for the model.
Protocol
Approval by the appropriate Institutional Animal Care and Use Committee must be obtained, indicating that your protocol follows the guidelines of your institution's animal research ethics committee.
1. Anesthesia and Pre-operative Procedures
- Anesthetize male Wistar rats, weighing around 200 g, with vaporized 3% isoflurane and 1 L/min oxygen administered into an acrylic induction chamber.
- Confirm adequate anesthesia through a lack of reaction to a toe and tail pinch. Place ointment on the eyes to prevent dryness while the animals are under anesthesia. Position the each rat supine on the plastic operation plate and position a silicone mask to deliver vaporized 2 – 3% isoflurane by continuous inhalation. Do not leave an animal unattended until it has regained sufficient consciousness to maintain sternal recumbency.
- While wearing sterile gloves, remove the ventral abdomen hair using a hair remover. Buprenorphine, either subcutaneously or intraperitoneally, is frequently given at this point.
2. Operative Procedures
- Patch venoplasty
- Before the surgery, trim the bovine pericardial patch using a microscissor to a 3 mm x 1.5 mm x 0.6 mm size. Soak the patch in 10 mL of sterilized saline for 5 min in a cell culture dish.
- Prepare the incision site with a topical antiseptic according to your institutional protocol and apply a surgical drape. Use sterile gloves and instruments to maintain aseptic technique throughout the surgery. Local anesthesia can be injected into the site of the planned skin incision.
- Using a scalpel, make a midline abdominal incision extending from the level of the xiphoid to just above the pubis.
- Insert a retractor and eviscerate all bowels from the abdominal cavity towards the right side. Wrap the bowels in gauze soaked with saline.
- Expose the infrarenal vena cava (IVC). The site of patch implantation is approximately 2 mm below the level of the origin of the renal veins; dissect the IVC free at this site. Ligate and divide all lumbar and tributary veins at this level using 6-0 nylon sutures.
- Place two microsurgery clips across both the proximal and distal infarenal IVC. Perform a venotomy with a 25 G needle by puncturing the anterior wall of the IVC, and then extend it to around 3 mm using scissors (Figure 1A).
- Close the venotomy with the patch using running or interrupted 10-0 nylon sutures (Figure 1B).
- Complete the venoplasty closure. Remove the distal clamp first to vent the patch and then remove the proximal clamp to restore blood flow to the IVC.
- Confirm hemostasis by observing for 30 s without compression. Return the bowels to their natural position. Close the abdomen with a running suture according to approved animal protocols.
- Patch arterioplasty
- Before the surgery, trim the bovine pericardial patch using a microscissor to a 3 mm x 1.5 mm x 0.6 mm size. Soak it in sterilized saline.
- Prepare the incision site with a topical antiseptic and apply a surgical drape. Use sterile gloves and instruments to maintain aseptic technique throughout the surgery.
- Using a scalpel, make a midline abdominal incision extending from the level of the xiphoid to just above the pubis.
- Insert a retractor and eviscerate all bowels from the abdominal cavity towards the right side. Wrap the bowels in gauze soaked with saline.
- Expose the aorta. The site of patch implantation is approximately 2 mm below the level of the origin of the renal arteries; dissect the aorta free at this site. Ligate and divide all lumbar arteries at this level using 6-0 nylon sutures.
- Clamp the infrarenal aorta. Use a 25 G needle to perform an arteriotomy by puncturing the anterior wall of the aorta, and then extend it to around 3 mm using scissors (Figure 1C).
- Close the arteriotomy with the patch using running 10-0 nylon sutures (Figure 1D). Complete the arterioplasty closure. Remove the distal clamp first to vent the patch and then remove the proximal clamp to restore blood flow to the aorta.
- Confirm hemostasis by observing for 30 s without compression. Return the bowels to their natural position. Close the abdomen with a running suture according to approved animal protocols.
3. Post-operative Procedures
- After the closure of the abdomen, discontinue the anesthesia. Apply post-operative care, including analgesia and wound care, in accordance with instructions recommended by the Institutional Animal Care and Use Committee.
- For analgesia, use buprenorphine at 0.1 mg/kg intramuscularly no less than every 12 h for 24 h following the surgical procedures. Check the status of the animal every day in the animal room, ensuring proper recovery from the peri-operative period as well as adequate treatment of post-surgical pain. Do not return an animal that has undergone surgery to the company of other animals until it has fully recovered.
Representative Results
After the patch angioplasty, there was no thrombosis in either the venous (n = 42) or arterial (n = 24) patch groups. Survival at day 7 was 100% in both groups with either a venous or arterial patch.
The patches were harvested on day 7 and stained with hematoxylin and eosin, and the neointimal thickness was compared as previously described7. Because of the distinct morphological difference between the patch and the neointima, it is very easy to discriminate the neointima. In the venous patch, there was a distinct layer of thick neointima on the luminal side of the patch, with more cells accumulated in the neointima (Figure 2A, 2C); in the arterial patch, there was a much thinner neointimal layer, with only a few cells attached to the luminal side of the patch (Figure 2B, 2D). The neointima was significantly thicker in the venous patch compared to the artery patch (Figure 2E). Similar results were obtained on day 14 (data not shown). These results show that the patch venoplasty model is superior over other models to acquire a thick neointima in a short period of time.
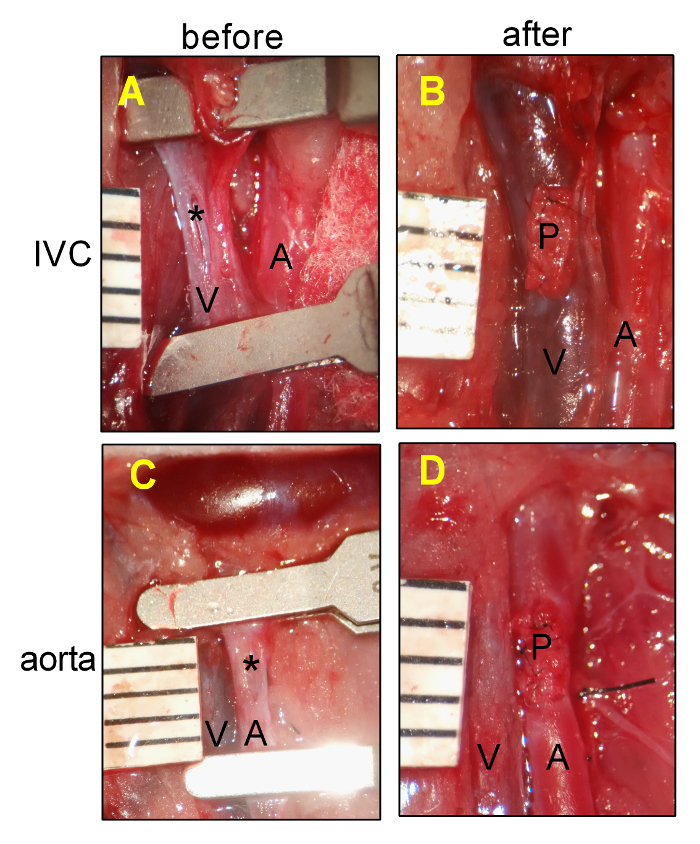
Figure 1: Rat Pericardial Patch Angioplasty. (A) Representative photograph showing the IVC venotomy. (B) Representative photograph showing the IVC after patch placement. (C) Representative photograph showing the aortic arteriotomy. (D) Representative photograph showing the aorta after patch placement. A, aorta; IVC, inferior vena cava; P, pericardial patch; *, posterior intima of IVC or aorta; ruler markings are 1 mm. Please click here to view a larger version of this figure.
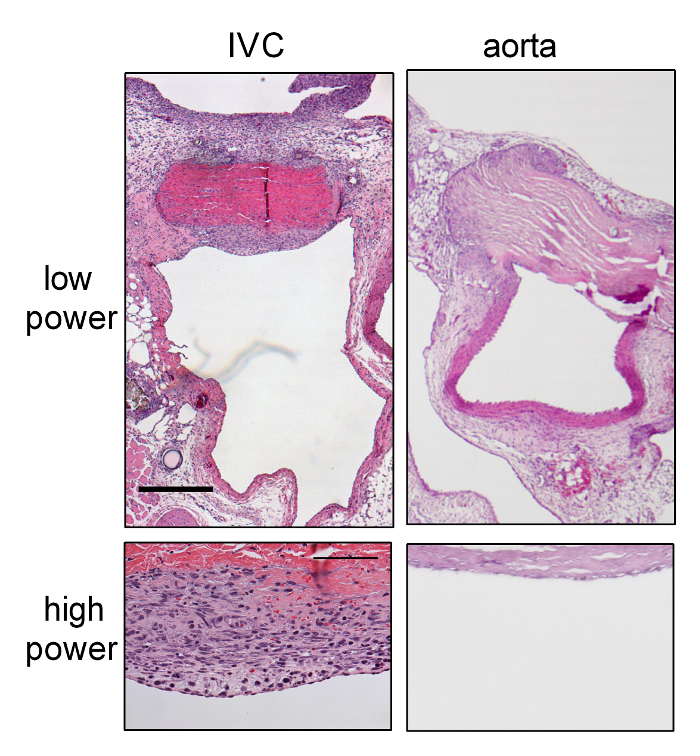
Figure 2: Aggressive Neointimal Hyperplasia in Patch Venoplasty Compared to Patch Arterioplasty at Day 7.
(A) Representative lower-power photograph showing the patch venoplasty on day 7, which was stained with H&E. (B) Representative lower-power photograph showing the patch angioplasty at day 7, which was stained with H&E. (C) Representative high-power photograph showing the neointimal formation on the patch venoplasty. (D) Representative high-power photograph showing the neointimal formation on the patch arterioplasty. Scale bar = 0.5 mm; high power = 100 µm. P, patch; L, IVC or aorta lumen; N, neointima. (E) Bar graph showing increased neointimal thickness in the venoplasty compared to arterioplasty. The error bar denotes the SEM; *P = 0.0015, t-test; n = 4. Please click here to view a larger version of this figure.
Discussion
We describe a patch angioplasty model in the rat that can be used in either veins or arteries, creating a patch venoplasty or patch arterioplasty, respectively. Using this pericardial patch model, we showed aggressive and stable neointimal hyperplasia in the rat IVC venoplasty compared to the aortic arterioplasty (Figure 2)7,8.
Several technical aspects are notable. In the aortic arterioplasty, it is important that the sutures are tight and adequate in number and/or spacing to prevent leakage of blood in the higher pressure of the arterial environment. In the IVC venoplasty, it is important to perform all of the operative maneuvers with particular care and gentleness, as the IVC wall is extremely thin and fragile. In addition, lumbar and small tributary veins around the implantation site must be carefully dissected and ligated to prevent bleeding. In both arterial and venous procedures, the distal clamp is removed first to vent any luminal air and to avoid a pulmonary air embolism. The proximal clamp is then removed to restore the distal blood flow. Lastly, when puncturing the aorta or IVC wall, do not puncture the posterior wall; if the posterior wall is punctured, a 10-0 suture should be used to close the hole.
A disadvantage of the patch angioplasty in the aorta is the development of pseudoaneurysms at later times (e.g., post-operative day 30). However, there were rarely any pseudoaneurysms observed on post-operative days 7 or 14, suggesting the utility of this model during the first 2 weeks after the procedure to study neointimal formation in either the venous or the arterial environment7,9.
Small-animal patch angioplasty has several advantages compared to interposition graft models. First, the host vessel does not need to be transected, as only an anterior wall incision is made; this reduces surgical time and minimizes the risk of bleeding. Second, the interposition graft requires two anastomoses, increasing the operative time and risk for complications, as well as extending the learning curve. Patch angioplasty requires only a single anastomosis, reducing the operative time and the risk of complications. Third, patch angioplasty intrinsically reduces the risk of stenosis at the anastomotic site, potentially allowing an increased time for postoperative observation10.
In conclusion, we describe a method to perform patch angioplasty that uses pericardial patches to close small-diameter vessels; the procedure is equally applicable to both veins and arteries. Pericardial patch venoplasty shows more aggressive neointimal formation compared to the arterioplasty model at early postoperative times. This result suggests the utility of this model in determining the differences in interactions between the venous and the arterial environments with implantable biomaterials. In addition, different types of patch compositions can be examined to test the response to these different materials.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This work was supported in part by the National Institutes of Health Grants R56-HL095498, R01-HL-095498 (to AD), R01-HL128406, the Merit Review Award I01-BX002336 from the United States Department of Veterans Affairs Biomedical Laboratory Research and Development Program (to AD), a Yale Department of Surgery Ohse award (to AD), a Sarnoff Cardiovascular Foundation Fellowship (to JMS), and through the resources and the use of facilities at the Veterans Affairs Connecticut Healthcare System (West Haven, CT). Bovine pericardial patches were generously provided by LeMaitre Vascular Inc. (Burlington, MA).
Materials
| Bovine pericardial patch | LeMaitre Vascular | |
| Vascular clamp | Roboz Surgical Instrument Co. | RS-5424 |
| Clamp applying forceps | Roboz Surgical Instrument Co | RS-5410 |
| 10-0 Prolene suture | Arosurgical Co. | T05A10N10-13 |
References
- Muto, A., Nishibe, T., Dardik, H., Dardik, A. Patches for carotid artery endarterectomy: current materials and prospects. J Vasc Surg. 50 (1), 206-213 (2009).
- Li, X., et al. Current usage and future directions for the bovine pericardial patch. Ann Vasc Surg. 25 (4), 561-568 (2011).
- Biasi, G. M., Sternjakob, S., Mingazzini, P. M., Ferrari, S. A. Nine-year experience of bovine pericardium patch angioplasty during carotid endarterectomy. J Vasc Surg. 36 (2), 271-277 (2002).
- Papakostas, J. C., et al. Use of the vascu-guard bovine pericardium patch for arteriotomy closure in carotid endarterectomy. Early and long-term results. Ann Vasc Surg. 28 (5), 1213-1218 (2014).
- Ohwada, S., et al. Glutaraldehyde-fixed heterologous pericardium for vena cava grafting following hepatectomy. Hepatogastroenterology. 46 (26), 855-858 (1999).
- Mori, A., et al. Standard hepatic vein reconstruction with patch plasty using the native portal vein in adult living donor liver transplantation. Liver Transpl. 18 (5), 602-607 (2012).
- Bai, H., et al. Pericardial patch venoplasty heals via attraction of venous progenitor cells. Physiological Reports. 4 (12), (2016).
- Li, X., et al. Pericardial patch angioplasty heals via an Ephrin-B2 and CD34 positive cell mediated mechanism. PLoS One. 7 (6), 38844 (2012).
- Bai, H., et al. Pretreatment of pericardial patches with antibiotics does not alter patch healing in vivo. J Vasc Surg. 63 (4), 1063-1073 (2016).
- Muto, A., et al. Eph-B4 prevents venous adaptive remodeling in the adult arterial environment. J Exp Med. 208 (3), 561-575 (2011).

