Live-cell Imaging of Platelet Degranulation and Secretion Under Flow
Summary
This work describes a fluorescence microscopy-based method for the study of platelet adhesion, spreading, and secretion under flow. This versatile platform enables the investigation of platelet function for mechanistic research on thrombosis and hemostasis.
Abstract
Blood platelets are essential players in hemostasis, the formation of thrombi to seal vascular breaches. They are also involved in thrombosis, the formation of thrombi that occlude the vasculature and injure organs, with life-threatening consequences. This motivates scientific research on platelet function and the development of methods to track cell-biological processes as they occur under flow conditions.
A variety of flow models are available for the study of platelet adhesion and aggregation, two key phenomena in platelet biology. This work describes a method to study real-time platelet degranulation under flow during activation. The method makes use of a flow chamber coupled to a syringe-pump setup that is placed under a wide-field, inverted, LED-based fluorescence microscope. The setup described here allows for the simultaneous excitation of multiple fluorophores that are delivered by fluorescently labeled antibodies or fluorescent dyes. After live-cell imaging experiments, the cover glasses can be further processed and analyzed using static microscopy (i.e., confocal microscopy or scanning electron microscopy).
Introduction
Platelets are anucleate cells that circulate in the blood stream. Their main function is to seal vascular breaches at sites of injury and to prevent blood loss. At these sites of injury, subendothelial collagen fibers become exposed and are subsequently covered by the multimeric protein, von Willebrand factor (VWF). VWF interacts with the platelets in circulation in a mechanism that depends upon the glycoprotein Ibα-IX-V complex on the cell surface1, slowing down the speed of the platelets. This is particularly important at high shear rates. The platelets subsequently undergo morphological changes while receiving activating impulses from collagen. This leads to irreversible spreading and eventually to platelet aggregation. Both processes depend upon the secretion of granule contents to facilitate platelet-platelet crosstalk. Amongst others, platelet α-granules contain fibrinogen and VWF to assist platelet adhesion and to bridge platelets together in an integrin-dependent manner. The platelet-dense granules contain inorganic compounds2, including calcium and adenosine diphosphate (ADP), which help to reinforce platelet activation. Furthermore, platelets contain mediators of (allergic) inflammation3, complement-controlling proteins4, and angiogenesis factors5,6, raising the questions of whether and how these contents are differentially released under varying conditions.
Since the 1980s, the study of platelet function in flow models has been valuable to the investigation of thrombotic mechanisms7. Since then, much technical progress has been made, and flow models that include fibrin formation are currently developed to assay the hemostatic potential of therapeutic platelet concentrates ex vivo8 or to investigate the influence of disturbances in shear rates on thrombus morphology9. The differences in the molecular and cell-biological mechanisms that drive stable adhesion and physiological thrombus formation (hemostasis) versus pathological thrombus formation (thrombosis) may be very subtle and motivate the development of flow models that allow for the real-time visualization of these subcellular processes.
An example of a process for which such a setup would be valuable is the (re)distribution of intracellular polyphosphate and the recruitment of clotting factors to uncover the time-dependent impact that this has on fibrin ultrastructure10. Studies are often limited to end-point analyses. The main aim of the described method is to enable the real-time visual investigation of dynamic subcellular processes that take place during platelet activation under flow.
Protocol
The local Medical Ethical Committee of the University Medical Center Utrecht approved the drawing of blood for ex vivo research purposes, including those of this study.
1. Solution Preparation
- Prepare a 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) Tyrode's buffer by dissolving 10 mM HEPES, 0.5 mM Na2HPO4, 145 mM NaCl, 5 mM KCl, 1 mM MgSO4, and 5.55 mM D-glucose in distilled water.
- Make two variants of the buffer: set the pH levels at 6.5 and at 7.4, respectively. Make fresh buffer for each day of experiments. Supplement with 10 ng/mL prostacyclin where indicated.
- Prepare TRIS-buffered saline (TBS) by dissolving 50 mM Tris and 150 mM NaCl in distilled water and adjust the pH to 9.0.
- Prepare acid citrate dextrose (ACD) by dissolving 85 mM Tri-sodium citrate, 71 mM citric acid, and 111 mM D-glucose in distilled water.
- Prepare a fixative solution by adding 2% (v/v) paraformaldehyde (PFA) to HEPES Tyrode's buffer, pH 7.4. Heat to dissolve (50 – 70 °C) and set the pH to 7.4. Aliquot and store at -20 °C. Thaw at 37 °C before use.
- Prepare blocking buffer by dissolving 1% (m/v) human serum albumin (HSA) in HEPES Tyrode's buffer, pH 7.4
- Make a polyvinyl alcohol solution by adding 4.8 g of polyvinyl alcohol to 12 g of glycerol and mix well. Add 12 mL of distilled water and leave it on a rotator at room temperature overnight.
- Add 24 mL of 0.2 M Tris-HCL, pH 8.5. Heat to 50 °C while mixing for 30 min. Add 1.25 g of 1,4-diazabicyclo[2.2.2]octane (DABCO) and mix well. Centrifuge at 1,500 x g for 5 min. Aliquot the supernatant (1 mL is sufficient for 15-20 slides) and store at -20 °C. Use the aliquots once.
2. Cover Glass Preparation
- Degrease cover glasses (25 x 50 mm2) by incubating overnight in undiluted chromosulfuric acid. Rinse the cover glasses with distilled water and dry them overnight at 60 °C in an oven.
- Adhere a strip of a paraffin film (10 x 15 cm2) on top of a laboratory bench with a drop of water and remove the air by smoothing the paraffin film. Pipette 80 µL drops of 10 µg/mL VWF or 100 µg/mL fibrinogen onto the paraffin film. Place the glasses onto the drops; the protein solution will spread between the two surfaces to cover the whole back side of the glass. Incubate for 1.5 h at room temperature (RT).
- Remove the paraffin film and block the cover glasses by placing them, with the coated side up in a 4-well plate with blocking buffer. Incubate for 1 h at 37 °C or overnight at 4 °C. As a control, block a cover glass that has not been coated with VWF or fibrinogen.
3. Preparation of Platelet-rich Plasma (PRP)
- Collect citrated whole blood (final concentration: 0.32% sodium citrate (M/V)) by venipuncture.
- Centrifuge the blood for 15 min at 160 x g and RT without brake. Collect the supernatant (i.e., the PRP) with a plastic Pasteur pipette.
NOTE: After centrifugation, three layers can be observed (from top to bottom): PRP, white blood cells, and red blood cells. Make sure not to include the white and red cells. Typically, 3 mL of PRP can be collected after centrifuging of a 9-mL blood tube. - Continue to section 4 if the experiment will be performed with washed platelets. If PRP is desired, continue with the next step.
- After PRP collection, centrifuge the remainder of the blood (containing the red cell pellet) again for 10 min at 2,000 x g without brake. Collect the supernatant containing the platelet-poor plasma (PPP).
NOTE: Typically, 2 mL can be collected after this second centrifugation step. - Count the number of the platelets in the PRP on a cell analyzer and dilute it with the PPP (step 3.4) to a concentration of 150,000 platelets/µL.
NOTE: The total volume of diluted PRP depends on the platelet count of the donor. Platelet counts vary widely between donors, and PRP dilution requires assessment on an individual basis. Platelet counts are considered normal when within 150,000 and 450,000 platelets/µL. - Continue to section 5.
4. Preparation of Washed Platelets
- Determine the mean platelet volume (MPV) in the PRP using a cell analyzer.
NOTE: This is an indicator of platelet pre-activation11. A normal MPV for resting platelets is 7.5-11.5 fL11. The MPV should not increase by more than 1.5 fL during the platelet washing procedure. - Add a 1/10 volume of ACD to PRP to prevent platelet pre-activation and centrifuge for 15 min at 400 x g and RT without brake. Remove the supernatant and re-suspend the pellet carefully in HEPES Tyrode's buffer, pH 6.5, to the original volume of PRP.
- Thaw an aliquot of prostacyclin by adding TBS to a concentration of 10 µg/mL; keep it on ice.
- To prevent platelet activation, add prostacyclin at a final concentration of 10 ng/mL to the re-suspended platelets and immediately centrifuge for 15 min at 400 x g and RT without brake. Remove the supernatant and re-suspend the pellet carefully in HEPES Tyrode's buffer, pH 7.4.
- Count the number of platelets (step 3.5) and determine the change in MPV (step 4.1) (the MPV should not change more than 1.5 fL).
- Adjust the number of platelets with HEPES Tyrode's buffer, pH 7.4, to a concentration of 150,000 platelets/µL. Leave the platelets to recover for 30 min at RT prior to the experiments. Use washed platelets for experiments within 4 h after isolation.
5. Flow Chamber Assembly
NOTE: These experiments make use of an in-house developed flow chamber12. A variety of commercially produced flow chambers can be used, as long as they can hold a cover glass, which preferentially is removable for further analyses. The flow chamber used for the described experiments is made from poly(methyl methacrylate) to precisely fit a microscope insert stage. It contains an inlet and outlet (Figure 1A–C; 1, 2), as well as a vacuum connection (Figure 1A and C; 3). A customized silicone sheet (Figure 1A and D; 4) is placed on top. Two cut-outs form vacuum channels (Figure 1A and D; 5) to attach the cover glass (Figure 1A; 6) tightly to the chamber. A central cut-out forms the flow channel (Figure 1A and D; 7; 2.0 mm width and 0.125 mm height). The inlet and outlet are connected to the flow channel, and the vacuum is connected to the vacuum channel.
- Cover the top of the flow chamber with distilled water and place the customized silicone sheet with the smooth side down on the water. Float the sheet into position by removing excess water while keeping the sheet in position.
NOTE: This silicone sheet is re-usable (silicone is known for its inert properties); generally, platelets have not been observed to attach to it. The sheets can be cleaned with detergent and re-used until the material becomes damaged (i.e., tearing, especially in the area between channels). Generally, a sheet can be used for 20 days with multiple experiments per day. - Incubate the flow chamber for 1 h at 60 °C to evaporate the residual water to firmly adhere the silicone sheet to the flow chamber.
- With a plastic Pasteur pipette, add a drop of HEPES Tyrode's buffer, pH 7.4, onto the flow channel. Place the cover glass (see section 2) with the coated side down, onto the flow channel. Attach the vacuum pump to the vacuum connection (Figures 1A and C; 3). Apply ~10 kPa pressure.
6. Pre-staining of the Platelets and Preparation of the Antibodies
- Platelet staining
- Incubate the washed platelets with 10 µg/mL DAPI for 30 min at 37 °C in the dark. To wash away unbound DAPI and to prevent platelet activation, add prostacyclin at a final concentration of 10 ng/mL and immediately centrifuge for 15 min at 400 x g and RT without brake.
- Reconstitute the platelet pellet in HEPES Tyrode's buffer, pH 7.4, to a platelet concentration of 150,000 platelets/µL.
- Preparation of antibodies
- Mix biotin-labeled antibody with Alexa-labeled streptavidin at a molar ratio of 1:1. Incubate for a minimum of 10 min at room temperature before use (step 7.6).
NOTE: Biotin labeling efficiency can vary between different batches. Should there be problems with the visualization of target molecules, control experiments should be performed to confirm successful biotinylation, as well the target-binding properties of the specific biotinylated antibody.
- Mix biotin-labeled antibody with Alexa-labeled streptavidin at a molar ratio of 1:1. Incubate for a minimum of 10 min at room temperature before use (step 7.6).
7. Perfusion
- Start the fluorescence microscope, as well as the microscope software. Use the microscope settings listed in Table 1.
NOTE: Initiate the microscope and recording settings for the live-cell visualization and recording of the platelets and the chosen fluorescently labeled antibodies and/or dyes by loading an appropriate experiment in the microscope software. Perform flow experiments at RT. - Block the inlet tubing by connecting a 10-mL syringe to the tubing and aspirating 1% HSA blocking buffer into the inlet tubing. Incubate for 15 min at RT. Rinse the inlet tubing by aspirating HEPES Tyrode's buffer, pH 7.4, into the inlet tubing.
- Connect the blocked inlet tubing and the untreated outlet tubing, both with a diameter of 1.02 mm and a ~20-30 cm length to the inlet and outlet of the chamber. Connect a 10-mL syringe (or lower volume for more accuracy) to the outlet tubing. Power on the syringe pump.
NOTE: The diameter of the syringe, in combination with the flow rate set on the pump, determine the volumetric flow rate. Together with the surface area of the flow channel, the shear rate can be calculated according to the formula13 below. Hence, it is possible to manipulate the shear rate by changing the flow rate of the syringe pump or altering the dimensions of the flow channel. A shear rate of 800-1,600 s-1 typically models for arterial blood flow, while 25 – 100s-1 models for venous blood flow. For this setup, a flow rate of 7.5 µL/min results in a shear rate of 25 s-1.
Shear rate = 6Q / h2w in s-1
Wherein: Q = volumetric flow rate (mL/s), h = height channel (cm) (0.0125 cm; this is the thickness of the silicone sheet), w = width channel (cm) (0.2 cm in this chamber). - Place a drop of immersion oil on a 100X objective (total amplification of 1,000X) and mount the chamber on the microscope with the glass surface down.
- Place the inlet tubing into a tube containing HEPES Tyrode's buffer, pH 7.4. Manually pull the plunger of the syringe connected to the outlet tubing and pull the solution through the chamber to remove the air from the system. Place the syringe into the syringe pump (Figure 1B and C; 8) and run the pump briefly to tighten the plunger and ensure stable flow.
- Add antibodies and/or dyes to the platelet suspension or PRP, carefully mix with a plastic pipette, and transfer the mixture to a 1.5-mL tube.
NOTE: For a typical 30-min perfusion at shear rate 25s-1 (7.5 µL/min), a minimum of 225 µL of platelet suspension is needed. The antibodies used for the representative results are shown in Table 2. - Move the inlet tubing, while squeezing, from the HEPES Tyrode's buffer, pH 7.4, to a 1.5-mL tube containing either PRP or the washed platelets and the antibodies and/or dyes (Figure 1B and C; 9).
NOTE: It is important that, when transferring the tubing, the tubing is squeezed (pressing out the air) to prevent air from entering the tubing. It is possible to treat adherent platelets with a variety of reagents in a subsequent step by moving the inlet tubing to another tube in a similar manner. A list of potentially useful reagents is provided in Table 3. - Select the "live visualization" option in the software, focus the microscope on the surface of the coated cover glass, and start the pump at a shear rate of 1,600s-1 (484 µL/min) until the platelets reach the flow chamber. At this moment, reduce the shear rate back to 25s-1 by changing the settings of the syringe pump to a flow rate of 7.5 µL/min.
- Monitor the surface of the coated side of the cover glass, wait until the first platelet starts to adhere, focus the microscope on this platelet, and start recording by initiating the experiment in the software. See Table 1 for the recording settings used here.
- Follow the live recording visually. After 30 min, stop the syringe pump.
NOTE: Ensure that the platelets stay in focus during the course of the experiment. Typically, 20-30 platelets attach in the field of view at a magnification of 1,000X; this is about 2,000-3,000 platelets in the whole flow channel. - Fixation using the flow chamber (optional).
- When the flow has stopped after the syringe pump is stopped, transfer the inlet tubing (air-free) to the fixative solution. Restart the pump and flow the fixation buffer through the channel for a minimum of 10 min at the same flow rate as mentioned in step 7.8.
- Remove the chamber from the microscope, clean the immersion oil from the glass, disconnect the vacuum, and carefully remove the cover glass from the chamber with an 18 G needle.
NOTE: Prevent the cover glass from breaking and damaging the silicon sheet. - Place the cover slide (with the cells directed upwards) in a 4-well plate on top of a tissue, pre-wetted with distilled water.
NOTE: This prevents the cover glass from adhering to the well surface and prevents drying. - Pipette 750 µL of fixative solution on top of the cover glass and incubate for 1 h at room temperature. In case the samples cannot be analyzed directly, store them in 0.5% PFA (diluted by HEPES Tyrode's buffer, pH 7.4) at 4 °C to ensure stable fixation. Proceed to section 8 to process the cover glass for imaging.
- After use, rinse the chamber, tubing, and sheet with distilled water and incubate overnight in a detergent solution. Before the re-use of the chamber and other components, rinse with distilled water.
- Save the microscope raw data files containing all the metadata. When needed, export the movies to .MOV, h.264 compressed, or .AVI uncompressed. Save separate frames as JPEG or TIF files.
8. Sample Preparation for Confocal Fluorescence Microscopy
- Transfer the cover glasses to new 4-well plates and rinse 5 times with 3 mL of HEPES Tyrode's buffer, pH 7.4. Add 3 mL/well of blocking buffer and incubate for 1 h at RT.
- Remove the blocking buffer; add 3 mL/well of 0.15% glycine (M/V) in HEPES Tyrode's buffer, pH 7.4; and incubate for 15 min at RT.
- Remove glycine solution and wash 5 times in 3 mL/well of blocking buffer. Optionally, add fluorophore-conjugated antibodies diluted in blocking buffer for additional staining. Incubate for 1 h at RT in the dark.
- Wash 5 times with blocking buffer and 2 times with distilled water. Dry the glass by removing excess liquid with a tissue (from the edges inward), without touching the cells.
- Pipette 60 µL of polyvinyl alcohol solution onto a microscope slide. Place the cover glass, with the cells down, on top of the drop and let the polyvinyl alcohol spread between the two surfaces. Incubate overnight at RT in the dark.
- Analyze the cells by confocal microscopy14.
- Save the raw data files of the recorded stack data containing all the metadata. When needed, process the raw data files into MOV, h.264 compressed, or .AVI uncompressed files. Save separate stacks as JPEG or TIF files.
Representative Results
Figure 1 shows images of the flow chamber and experimental setup; the position and dimensions of the silicon sheet; and tubing connections. Figure 2 provides details on the dimensions of the flow chamber. Figure 3 and Movie 1 show a time-series of images of platelet adhesion and spreading on immobilized VWF. CD63 is a transmembrane protein that is inserted into the membrane of intracellular dense granules of resting platelets15. Its time-dependent mobilization onto the platelet surface is shown in green. Similarly, P-selectin (Psel) is a transmembrane protein inserted in the membrane of intracellular α-granules of resting platelets. Its time-dependent mobilization onto the platelet surface is shown in orange. Figure 3B shows a representative image at a 45° angle, acquired by confocal fluorescence microscopy after a flow experiment. Movie 2 shows a tilt series of the same experiment. Note that CD63 is mainly located at the central granulomere, while P-selectin is distributed over the entire cell body and appears concentrated at the edges. Figure 4A and Movie 3 show the time-dependent mobilization of CD63 onto the platelet surface (green). In these experiments, platelets (which do not have nuclear DNA) were pre-incubated with DAPI (blue), which stains polyphosphate in dense granules16. Remarkably, despite clear signs of dense granule release (single-cell analyses are shown in Figure 4B and Movie 4), polyphosphate is retained within or on the outside of these platelets. Figure 5 and Movie 5 show time-dependent mobilization (or recruitment) of VWF, a thrombogenic multimeric protein17 stored in α-granules, to the platelet surface (green). Similar to polyphosphate, VWF remains associated with the surface of the activated platelets.
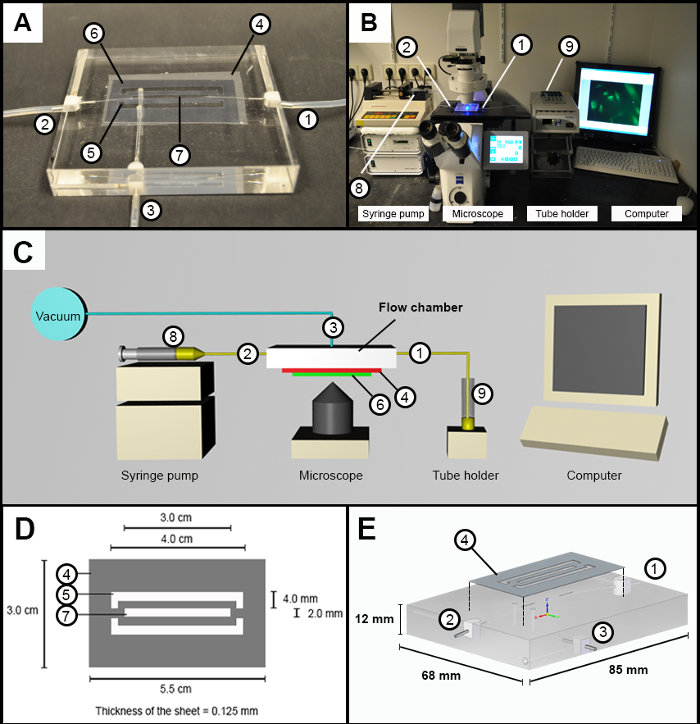
Figure 1: Flow Chamber and Experimental Setup. (A) Image of flow chamber design with an inlet and outlet (1 and 2), a vacuum connection (3) on which the silicone sheet (4) is placed. Two outer cutouts (5) in the silicon sheet form the vacuum channels to attach the cover glass (6). A central cutout (7) forms the flow channel (B) Image of the experimental setup. Inlet and outlet (1 and 2), syringe pump (8), and sample holder (9). (C) Schematic overview of the experimental setup; the numbers are identical to those in A and B. (D) Schematic overview with the dimension of the customized cut silicone sheet. (E) General schematic overview of the poly(methyl methacrylate) flow chamber. Please click here to view a larger version of this figure.
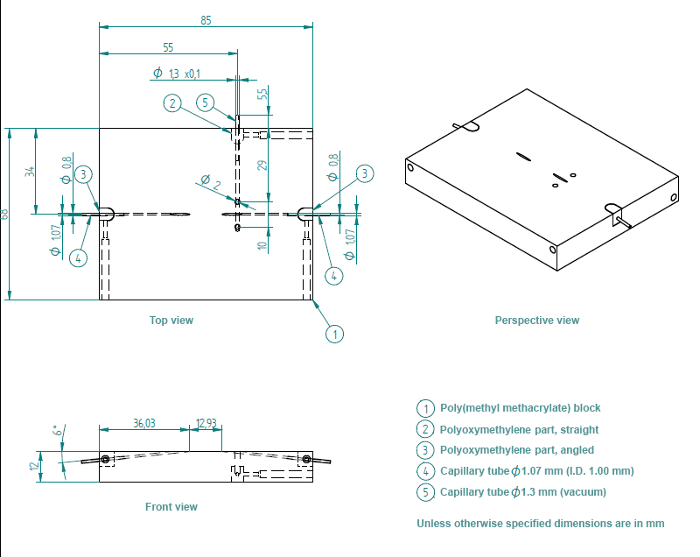
Figure 2: Detailed Blueprint of Flow Chamber. The flow chamber consists of a poly(methyl methacrylate) block (1), one straight polyoxymethylene insert for the vacuum inlet (2), and two angled polyoxymethylene inserts for the sample inlet and outlet (3). The angled polyoxymethylene inserts contain metal capillary tubes, with diameters of 1.07 mm. The straight insert contains a capillary tube with a diameter of 1.3 mm. Please click here to view a larger version of this figure.
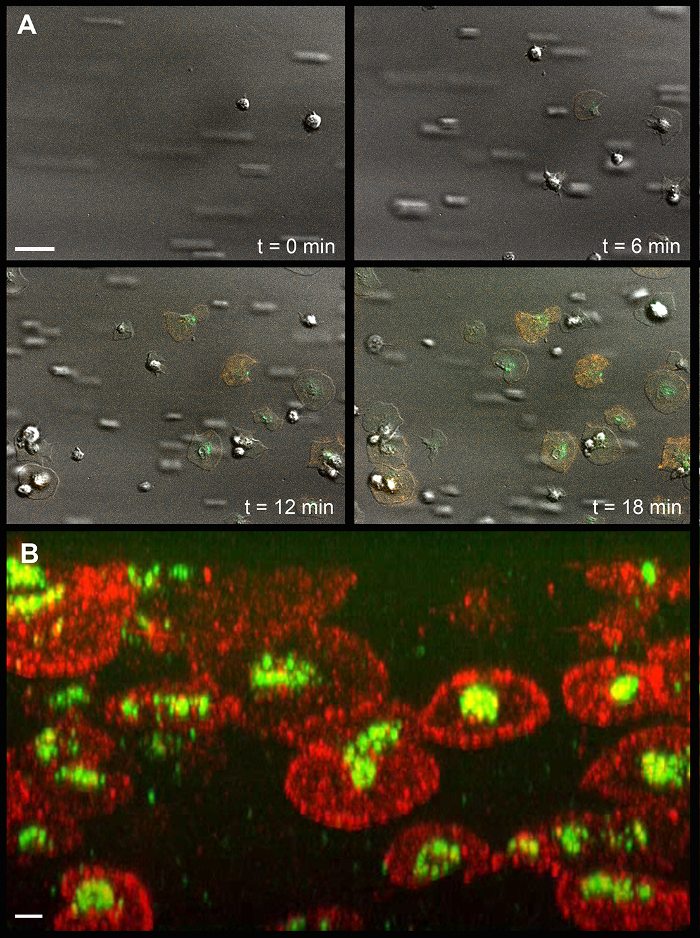
Figure 3: Platelet Spreading and Degranulation on Immobilized von Willebrand Factor. (A) Time-dependent mobilization of CD63 and P-selectin onto the surface of spreading and degranulating platelets. The white scale bar (upper left) indicates 10 µm. (B) Representative confocal fluorescence microscopy image at a 45° angle. The white scale bar (lower left) indicates 2 µm. Green, CD63; Orange, P-selectin. Please click here to view a larger version of this figure.
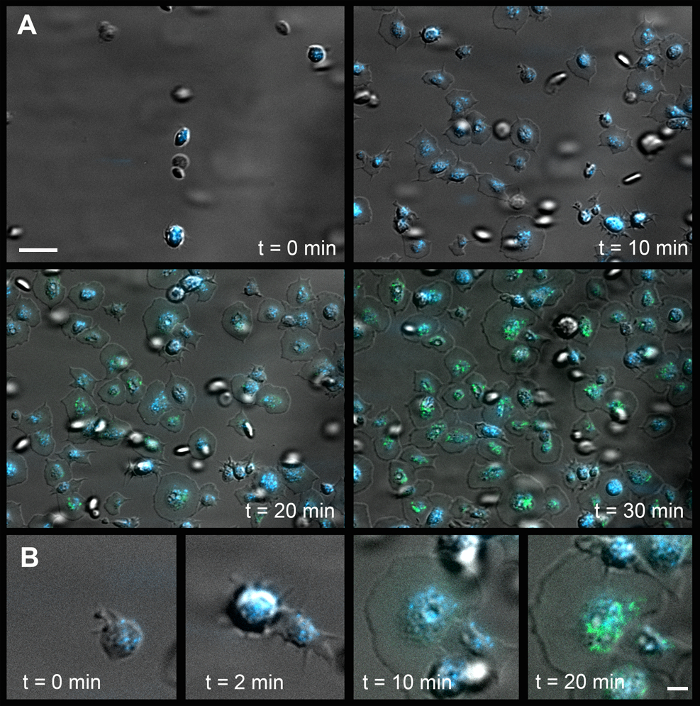
Figure 4: Platelet Spreading, Degranulation, and Mobility of Dense Granules on Immobilized Fibrinogen. (A) Time-dependent mobilization of CD63 and polyphosphate onto the platelet surface. The white scale bar (upper left) indicates 10 µm. (B) Time series of a single platelet (t = 0 represents the first adhesion to surface). The white scale bar (lower right) indicates 2 µm. Green, CD63; Blue, DAPI. Please click here to view a larger version of this figure.
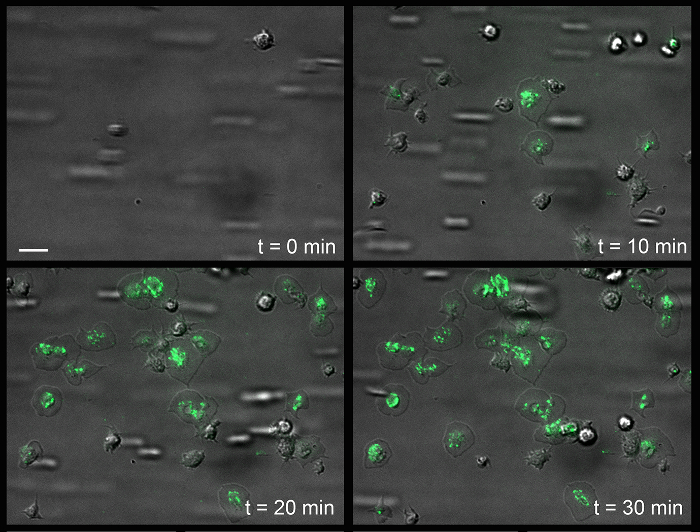
Figure 5: Surface Association of von Willebrand Factor onto the Platelet Surface of Immobilized Fibrinogen. Time-dependent mobilization (or recruitment) of VWF to the platelet surface. The white scale bar (upper left) indicates 5 µm. Green, VWF. Please click here to view a larger version of this figure.
| Setting | Value |
| Exposure time FITC (Alexa488) | 200 ms / 300 ms |
| LED 488 intensity FITC (Alexa488) | 50% |
| Exposure time DIC | 30 ms |
| Voltage halogen lamp | 4.5 Volt |
| Exposure time TRITC (Alexa 546) | 400 ms |
| LED 555 intensity FITC (Alexa546) | 50% |
| Exposure time DAPI | 500 ms |
| LED 365 intensity | 50% |
| Filterset FITC | Filterset 10 |
| Filterset TRITC | Filterset 20 |
| Filterset DAPI | Filterset 01 |
| Objective | Alpha Plan-Fluar 100x/1.45 Oil |
| Frame rate, recording time | 1 frame / 10 s, 30 min |
| File compression movies | MOV (h.264), AVI(uncompressed) |
| File compression pictures | JPEG, TIF |
Table 1: Microscope settings. Settings used for the representative experiments.
| Target | Antibody / Dye | Concentration |
| CD63 | Anti-CD63-biotin *) | 325 ng/mL |
| P-selectin | Anti-CD62P-biotin **) | 90 ng/mL |
| DNA | DAPI | 10 µg/mL |
| VWF | Anti-human VWF- FITC | 5 µg/mL |
| *) anti-CD63-biotin antibody is mixed with streptavidin-Alexa488 in a molar ratio of 1:1 before use | ||
| **) anti-CD62P-biotin antibody is mixed with streptavidin-Alexa546 in a molar ratio of 1:1 before use | ||
Table 2: Antibodies and Dyes. Recommended antibody and dye concentrations for the shown representative experiments.
| Reagent category | Example compounds |
| Platelet agonists | Thrombin, ADP, PAR-1 or -4 activating peptides, U46619, Thromboxane A2, ADP |
| Enzyme inhibitors | Hirudin, PPACK, heparinoids (indirect), corn trypsin inhibitor, soy bean trypsin inhibitor |
| Receptor antagonists | anti-Glyprotein 1bα, anti-Glycoprotein VI; clopidogrel, (cyclic) RGD-containing peptides |
| Activation inhibitors | Aspirin pretreatment, fixation |
Table 3: Potentially useful reagents.
Supplemental Movies: Movie 1: Time-series of images of platelet (see Figure 1), Movie 2: tilt series of Figure 3B, Movie 3: Time-dependent mobilization of CD63 onto the platelet surface (see Figure 4), Movie 4: Time series of single-cell analyses (see Figure 4B), Movie 5: Time-dependent mobilization of VWF (see Figure 5).
Please click here to download Movie 1.
Please click here to download Movie 2.
Please click here to download Movie 3.
Please click here to download Movie 4.
Please click here to download Movie 5.
Discussion
Worldwide, thrombosis is a leading cause of death and morbidity, and platelets play a central role in its development. This work describes a method for the live-cell imaging of platelet degranulation under flow. It is generally assumed that, when platelets become activated, all granular contents are directly released into solution. The accompanying results suggest that this is not necessarily the case. During adhesion and degranulation, platelets retain a significant amount of polyphosphate (Figure 4). Additionally, the multimeric protein VWF is associated with the platelet surface after degranulation (Figure 5). This indicates that the molecular mechanisms that control platelet granule degranulation are subtler than generally assumed.
To enable the live-cell imaging of platelet degranulation under flow, the experimental conditions were selected to keep platelet aggregation to a minimum. Platelet counts of 150,000 platelets/µL were used. This is at the lower end of the normal range (150,000-450,000 platelets/µL). Furthermore, platelet adhesion and spreading were studied on immobilized VWF or fibrinogen, which led to mild platelet activation (in contrast to, for example, collagen). In studies with washed platelets, increased shear rates move these cells to the center of the flow. This interferes with adhesion and subsequent spreading and complicates the investigation of degranulation.
In physiological hemostasis, platelet margination is important. This process is driven by erythrocytes, explaining why low anemia is sometimes accompanied by a bleeding diathesis. A limitation of the presented technique is that the visualization of platelet degranulation (with the presented materials and reagents) is disturbed by red blood cells. Platelet function is influenced by shear stress, which is a function of the viscosity of the fluid (µ), as well as of the shear rate. Hence, it is recommended to study platelet function in flow models in solutions with comparable viscosities (i.e., washed platelets, reconstituted blood, PRP, or whole blood).
There are several practical factors to take into account when performing these experiments. First, fluorescence signals are strongly influenced by the focus area. If the microscope is not in focus, the signal is easily blurred or even lost. Prior to the arrival of platelets into the flow channel, it is best to focus on the channel itself (begin at the edge of the silicon sheet/flow channel wall). When platelets start to adhere, focus on this adhesion event. A second point of concern is the bleaching of certain fluorophores (e.g., FITC). Repeated excitation will lead to a loss of signal, disturbing the investigation. These obstacles can be overcome by using more stable fluorophores, reducing the intensity of the exciting LED light, shortening the exposure times, and/or lowering the frame rate.
The model described here is very useful for performing a detailed study of platelet responses to a wide variety of immobilized proteins and at different flow rates. Potentially, this method can also be used to study their interaction with activated endothelial cells18. The technical advances that allow for the flexible designs of the special flow chambers are currently opening avenues for biocompatibility studies19. The live-cell imaging of platelet reactivity under flow will reveal valuable insights on the complexity of the platelet adhesion response. Ultimately, these experiences should lead to the development experimental flow models that allow for the combined investigation of platelet function and influence on the coagulation system.
Divulgations
The authors have nothing to disclose.
Acknowledgements
CM acknowledges financial support from the International Patient Organization for C1-Inhibitor Deficiencies (HAEi), Stichting Vrienden van Het UMC Utrecht, and the Landsteiner Foundation for Blood Transfusion Research (LSBR).
Materials
| 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) | VWR | 441476L | |
| Na2HPO4 | Sigma | S-0876 | |
| NaCl | Sigma | 31434 | |
| KCl | Sigma | 31248 | |
| MgSO4 | Merck KGaA | 1.05886 | |
| D-glucose | Merck KGaA | 1.04074 | |
| Prostacyclin | Cayman Chemical | 18220 | |
| Tri-sodium citrate | Merck KGaA | 1.06448 | |
| Citric acid | Merck KGaA | 1.00244 | |
| Cover glasses | Menzel-Gläser | BBAD02400500#A | 24x50mm, No. 1 = 0.13-0.16 mm thickness. |
| Chromosulfuric acid (2% CrO3) | Riedel de Haen | 07404 | CAS [65272-70-0]. |
| Von Willebrand factor (VWF) | in-house purified | ||
| Fibrinogen | Enzyme Research Laboratories | FIB3L | |
| 4 well dish, non-treated | Thermo Scientific | 267061 | |
| Human Serum Albumin Fraction V | Haem Technologies Inc. | 823022 | |
| Blood collection tubes, 9 ml, 9NC Coagulation Sodium Citrate 3.2% | Greiner Bio-One | 455322 | |
| Cell analyser | Abbott Diagnostics | CELL-DYN hematology analyzer | |
| Paraformaldehyde | Sigma | 30525-89-4 | |
| Syringe pump | Harvard Apparatus, Holliston, MA | Harvard apparatus 22 | |
| 10 mL syringe with 14.5 mm diameter | BD biosciences | 305959 | Luer-Lok syringe |
| Anti-CD63-biotin | Abcam | AB134331 | |
| Anti-CD62P-biotin | R&D Systems | Dy137 | |
| 4’,6-Diamidino-2-phenylindole dihydrochloride (DAPI) | Polysciences Inc. | 9224 | |
| Streptavidin, Alexa Fluor 488 conjugate | Thermo Scientific | S11223 | |
| Immersion oil | Zeiss | 444963-0000-000 | |
| Detergent solution | Unilever, Biotex | ||
| Glycine | Sigma | 56-40-6 | |
| Polyvinyl alcohol | Sigma | 9002-89-5 | Mowiol 40-88. |
| Tris hydrochloride | Sigma | 1185-53-1 | |
| 1,4-Diazabicyclo[2.2.2]octane (DABCO) | Sigma | 280-57-9 | |
| Sheep Anti-hVWF pAb | Abcam | AB9378 | |
| Alexa fluor 488-NHS | Thermo Scientific | A20000 | |
| Glycerol | Sigma-Aldrich | 15523-1L-R | |
| Parafinn film | Bemis | PM-996 | 4 in. x 125 ft. Roll. |
| Silicone sheet non-reinforced | Nagor | NA 500-1 | 200mmx150mmx0.125mm. |
| Customized cut silicone sheet with perfusion and vacuum channels | in-house made | Made of Silicone sheet non-reinforced (Nagor, NA 500-1) | |
| 1.5 mL tubes | Eppendorf AG | T9661-1000AE | |
| Fluorescent microscope | Zeiss Observer Z1 | Equiped with LED excitation lights. | |
| Microscope software | Zeiss ZEN 2 | blue edition | |
| 18 G needle (18 G x 1 1/2") | BD biosciences | 305196 | |
| NaCl | Riedel de Haen | 31248375 | |
| Tris | Roche | 10708976 | |
| Plastic pasteur pipet | VWR | 612-1681 | 7 ml non sterile, graduated up to 3ml. |
| Silicone tubing | VWR | 228-0656 | Inner diamete. x Outer diameter x Wall thickness = 1.02 x 2.16 x 0.57 mm. |
| Microscope slides | Thermo Scientific | ABAA000001##12E | 76 x 26 x 1 mm, ground edges 45°, frosted end. |
References
- Springer, T. A. von Willebrand factor, Jedi knight of the bloodstream. Blood. 124 (9), 1412-1425 (2014).
- Fitch-Tewfik, J. L., Flaumenhaft, R. Platelet granule exocytosis: a comparison with chromaffin cells. Front Endocrinol (Lausanne). 4, 11 (2013).
- May, B., Menkens, I., Westermann, E. Differential release of serotonin and histamine from blood platelets of the rabbit by aliphatic and aromatic amines. Life Sci. 6 (19), 2079-2085 (1967).
- Schmaier, A. H., Smith, P. M., Colman, R. W. Platelet C1- inhibitor. A secreted alpha-granule protein. J. Clin. Invest. 75 (1), 242-250 (1985).
- Battinelli, E. M., Markens, B. A., Italiano, J. E., et al. Release of angiogenesis regulatory proteins from platelet alpha granules: modulation of physiologic and pathologic angiogenesis. Blood. 118 (5), 1359-1369 (2011).
- Kamykowski, J., Carlton, P., Sehgal, S., Storrie, B. Quantitative immunofluorescence mapping reveals little functional coclustering of proteins within platelet α-granules. Blood. 118 (5), 1370-1373 (2011).
- Sakariassen, K. S., Aarts, P. A., de Groot, P. G., Houdijk, W. P., Sixma, J. J. A perfusion chamber developed to investigate platelet interaction in flowing blood with human vessel wall cells, their extracellular matrix, and purified components. J Lab Clin Med. 102 (4), 522-535 (1983).
- Van Aelst, B., Feys, H. B., Devloo, R., Vandekerckhove, P., Compernolle, V. Microfluidic Flow Chambers Using Reconstituted Blood to Model Hemostasis and Platelet Transfusion In Vitro. J Vis Exp. (109), (2016).
- Nesbitt, W. S., Westein, E., et al. A shear gradient-dependent platelet aggregation mechanism drives thrombus formation. Nat Med. 15 (6), 665-673 (2009).
- Mitchell, J. L., Lionikiene, A. S., et al. Polyphosphate colocalizes with factor XII on platelet-bound fibrin and augments its plasminogen activator activity. Blood. 128 (24), 2834-2845 (2016).
- Park, Y., Schoene, N., Harris, W. Mean platelet volume as an indicator of platelet activation: methodological issues. Platelets. 13 (5-6), 301-306 (2002).
- Sixma, J. J., de Groot, P. G., van Zanten, H., IJsseldijk, M. A new perfusion chamber to detect platelet adhesion using a small volume of blood. Thromb Res. 92, S43-S46 (1998).
- Slack, S. M., Turitto, V. T. Flow chambers and their standardization for use in studies of thrombosis. On behalf of the Subcommittee on Rheology of the Scientific and Standardization Committee of the ISTH. Thromb Haemost. 72 (5), 777-781 (1994).
- Oreopoulos, J., Berman, R., Browne, M. Spinning-disk confocal microscopy: present technology and future trends. Methods Cell Biol. 123, 153-175 (2014).
- Nishibori, M., Cham, B., McNicol, A., Shalev, A., Jain, N., Gerrard, J. M. The protein CD63 is in platelet dense granules, is deficient in a patient with Hermansky-Pudlak syndrome, and appears identical to granulophysin. J. Clin. Invest. 91 (4), 1775-1782 (1993).
- Ruiz, F. A., Lea, C. R., Oldfield, E., Docampo, R. Human platelet dense granules contain polyphosphate and are similar to acidocalcisomes of bacteria and unicellular eukaryotes. J. Biol. Chem. 279 (43), 44250-44257 (2004).
- Tersteeg, C., Fijnheer, R., et al. Keeping von Willebrand Factor under Control: Alternatives for ADAMTS13. Semin Thromb Hemost. 42 (1), 9-17 (2016).
- Tersteeg, C., de Maat, S., et al. Plasmin cleavage of von Willebrand factor as an emergency bypass for ADAMTS13 deficiency in thrombotic microangiopathy. Circulation. 129 (12), 1320-1331 (2014).
- Basir, A., de Groot, P., et al. In Vitro Hemocompatibility Testing of Dyneema Purity Fibers in Blood Contact. Innovations (Phila). 10 (3), 195-201 (2015).

