Isolation of Murine Adipose Tissue-derived Microvascular Fragments as Vascularization Units for Tissue Engineering
Summary
We present a protocol to isolate adipose tissue-derived microvascular fragments that represent promising vascularization units. They can be rapidly isolated, do not require in vitro processing and, thus, may be used for one-step prevascularization in different fields of tissue engineering.
Abstract
A functional microvascular network is of pivotal importance for the survival and integration of engineered tissue constructs. For this purpose, several angiogenic and prevascularization strategies have been established. However, most cell-based approaches include time-consuming in vitro steps for the formation of a microvascular network. Hence, they are not suitable for intraoperative one-step procedures. Adipose tissue-derived microvascular fragments (ad-MVF) represent promising vascularization units. They can be easily isolated from fat tissue and exhibit a functional microvessel morphology. Moreover, they rapidly reassemble into new microvascular networks after in vivo implantation. In addition, ad-MVF have been shown to induce lymphangiogenesis. Finally, they are a rich source of mesenchymal stem cells, which may further contribute to their high vascularization potential. In previous studies we have demonstrated the remarkable vascularization capacity of ad-MVF in engineered bone and skin substitutes. In the present study, we report on a standardized protocol for the enzymatic isolation of ad-MVF from murine fat tissue.
Introduction
Tissue engineering focuses on the fabrication of tissue and organ substitutes that maintain, restore or augment the function of inoperable in vivo counterparts1,2. The fate of engineered tissue constructs crucially depends on an adequate vascularization3. Microvascular networks within these constructs should be hierarchically organized with arterioles, capillaries, and venules to allow efficient blood perfusion after inosculation to the recipient's vasculature4. The generation of such networks is among the key challenges in tissue engineering. For this purpose, a broad spectrum of experimental vascularization strategies has been introduced over the last two decades5,6.
Angiogenic approaches stimulate the ingrowth of recipient microvessels into engineered tissues by means of structural or physicochemical scaffold modification, such as the incorporation of growth factors7. However, for the vascularization of large three-dimensional constructs, angiogenesis-dependent strategies are markedly limited by slow growth rates of developing microvessels8.
In contrast, the concept of prevascularization aims for the generation of functional microvascular networks within tissue constructs prior to their implantation9. Conventional prevascularization involves the co-culture of vessel-forming cells, such as endothelial cells, mural cells or stem cells10, within scaffolds. After microvascular network formation, the prevascularized constructs can then be implanted into tissue defects. Noteworthy, this prevascularization approach is difficult to apply in a clinical setting, because it is based on complex and time-consuming in vitro procedures, which are restricted by major regulatory hurdles9. Accordingly, there is still a need for the development of novel prevascularization strategies that are more suitable for a broad clinical application.
Such a prevascularization strategy may be the application of adipose tissue-derived microvascular fragments (ad-MVF). ad-MVF represent potent vascularization units that can be harvested in large amounts from the fat tissue of rats11,12 and mice13. They consist of arteriolar, capillary, and venular vessel segments, which exhibit a physiological microvessel morphology with a lumen and stabilizing perivascular cells14,15. This unique feature allows the immediate implantation of ad-MVF-seeded scaffolds into tissue defects without precultivation. There, the ad-MVF rapidly reassemble into functional microvascular networks. Furthermore, ad-MVF represent a rich source of mesenchymal stem cells16, which may additionally contribute to their striking regenerative capacity. Accordingly, ad-MVF are increasingly used in different fields of tissue engineering14,15,17,18,19,20,21.
The isolation of ad-MVF has originally been established in rats11,12. Herein, we describe a protocol, which allows the standardized isolation of murine ad-MVF from epididymal fat pads. This may provide further insights into molecular mechanisms underlying ad-MVF function by using transgenic mouse models.
Protocol
All procedures were performed according to the National Institute of Health guidelines for the use of experimental animals and followed institutional guidelines (Landesamt für Soziales, Gesundheit und Verbraucherschutz, Abt. Lebensmittel- und Veterinärwesen, Zentralstelle, Saarbrücken, Germany).
1. Preparation of Surgical Instruments
- Keep ready the dissection scissors, surgical forceps, small preparation scissors, fine forceps and a sterile Petri dish with 15 mL Dulbecco's modified Eagle medium (DMEM; 10% fetal calf serum (FCS), 100 U/mL penicillin, 0.1 mg/mL streptomycin) for harvesting epididymal fat pads.
- Expose the surgical instruments to a disinfecting solution for 5 min. Alternatively, sterilize them (steam sterilization; 121 °C, 20 min).
2. Animals and Anesthesia
- Choose carefully the strain of the mice as indicated for the study and the subject under investigation.
NOTE: In this study we used male wild-type C57BL/6 mice as donors for the harvesting of epididymal fat. Of interest, ad-MVF may be also isolated from transgenic green fluorescent protein (GFP)-positive donor animals (C57BL/6-Tg(CAG-EGFP)1Osb/J)22. This bears the major advantage that the fragments are easily detectable by immunohistochemical staining of GFP after implantation into GFP-negative wild-type mice14. - Anesthetize the animals with an intraperitoneal injection of xylazine (15 mg/kg) and ketamine (75 mg/kg). Make sure that the animals are deeply anesthetized by performing a toe pinch with no response. Eye lubricant is not indicated as the donor animals are sacrificed after fat harvesting.
NOTE: Ensure that analgesia and surgical sterility are in agreement with the respective guidelines of the country and institution where the experiments are planned.
3. Harvesting of Epididymal Fat Pads
- Transfer the animal to an operation table. Place the animal in a supine position under a surgical stereomicroscope and confirm deep anesthesia using toe pinch.
- Immobilize the paws by taping them to a surgical drape and disinfect the abdomen with disinfecting solution.
- Separate the abdominal skin free of the underlying muscle layer with the dissection scissors.
- Perform a midline laparotomy with the dissection scissors and laterally unfold the flaps of the abdominal wall.
- Bilaterally identify testis, epididymis and the epididymal fat pad using the fine forceps (Figure 1). Do not harm the intestinal structures to prevent fecal contamination of the fat pads.
- Harvest the epididymal fat pads with the small preparation scissors and the fine forceps under the stereomicroscope. Keep a safety margin of several mm between the epididymis and the fat to reduce the risk of accidental epididymal harvesting.
NOTE: This procedure can also be performed without a stereomicroscope. However, this may further increase the risk of accidental injury of the epididymis and spermatic cords. - Transfer the epididymal fat pads into a Petri dish containing 15 mL of DMEM pre-heated at 37 °C for the transport to the cell laboratory.
- Sacrifice the animal by incision of the abdominal aorta or cervical dislocation.
4. Isolation of ad-MVF
- Prepare three sterile Petri dishes with 15 mL of phosphate-buffered saline (PBS), sterile 14-mL polypropylene (PP) tubes, a sterile 50-mL Erlenmeyer flask, sterile 1.5-mL conical microcentrifuge tubes and sterilized fine scissors.
- Wash the fat pads thrice in Petri dishes with 15 mL of PBS under a laminar flow hood.
- Transfer the fat into a 14-mL PP tube. Determine the volume of harvested fat tissue (in mL) by means of the tube scale. Mince the fat tissue mechanically with the fine scissors until a homogeneous tissue suspension is obtained.
- Transfer the minced tissue with two volumes of collagenase NB4G (0.5 U/mL PBS) into a 50-mL Erlenmeyer flask by means of 10-mL measuring pipette. The total volume becomes thrice the volume of fat tissue measured in step 4.3. Perform tissue digestion in an incubator for 10 min under vigorous stirring by means of an automated stirrer (size of magnetic stir bar: 25 mm) at 37 °C and humidified atmospheric conditions with 5% CO2.
- Observe a small fraction (10 µL) of the digested tissue under a microscope to judge whether the digestion can be stopped. Ascertain that the digestate mainly contains "free" ad-MVF next to single cells, indicating the appropriate point to stop the enzymatic digestion process (Figure 2).
NOTE: This step requires experience with the procedure and is critical for the quality of isolated ad-MVF. Prolonged fat digestion results in a single-cell suspension without ad-MVF. - Neutralize the enzyme with two volumes of PBS/20% FCS. The total volume becomes thrice the volume of the cell-vessel suspension in step 4.4. Transfer the cell-vessel suspension back into new PP tubes.
- Incubate the suspension for 5 min at 37 °C to separate ad-MVF from remaining fat by gravity. Then carefully remove the main fat supernatant with a 1-mL precision pipette. Repeat this cycle several times with a 100-µL precision pipette until the suspension appears to be fat-free.
- Put a 500-µm filter on top of a 50-mL conical centrifuge tube. Transfer the cell-vessel suspension with a 10-mL measuring pipette from the 14-mL PP tubes onto the filter membrane to remove remaining fat clots. Transfer the filtered suspension into new 14-mL PP tubes according to the number of individual ad-MVF isolates for the planned experiments.
NOTE: For in vitro assays focusing on microvascular network formation, it may be beneficial to further purify the cell-vessel suspension to improve the imaging quality during microscopic analyses. For this purpose, the suspension may be additionally filtered once with a 20-µm filter to remove single cells from the ad-MVF collected on the filter. - Centrifuge the cell-vessel suspension (600 x g, 5 min, room temperature) to obtain a pellet containing ad-MVF.
- After centrifugation, remove the supernatant until 1 mL is left. Resuspend the pellet with ad-MVF in this 1 mL and transfer the suspension into a 1.5-mL conical microcentrifuge tube.
- Centrifuge the ad-MVF suspension in the microcentrifuge tube (600 x g, 5 min) to obtain a pellet.
- Remove the supernatant to resuspend the pellet in the required final volume of PBS / 20% FCS.
NOTE: The number of ad-MVF per isolate can be assessed by microscopic counting. For this purpose, 1/10 of the final cell-vessel suspension is diluted 1:10 in PBS and 100 µL of this dilution are transferred into a well of a 96-well plate. The entire number of ad-MVF exhibiting a vessel-like morphology is then counted and extrapolated to the whole isolate. The required ad-MVF concentration and purification can be individually adapted to the respective in vitro or in vivo application of the ad-MVF. This can include the embedding of ad-MVF in collagen gels for the in vitro analysis of microvascular network formation or the seeding of ad-MVF on scaffolds for in vivo implantation into tissue defects.
Representative Results
In the present study we performed six ad-MVF isolation procedures with fat tissue from 7- to 12-month-old male wild-type C57BL/6 mice (mean body weight: 35 ± 1 g). Figure 1 illustrates the harvesting of murine epididymal fat pads with subsequent mechanical and enzymatic ad-MVF isolation. The time required for the harvesting of fat was 30 min and for the isolation of ad-MVF was 120 min. In total, the procedure took 150 min.
We harvested 1.2 ± 0.1 mL of adipose tissue per donor animal. This amount of fat allowed the isolation of 42,000 ± 2,000 ad-MVF per mL. The mean length of the isolated ad-MVF was 42 ± 1 µm (Figure 2A). The duration of enzymatic digestion was determined by means of microscopic control as shown in Figure 2B. ad-MVF exhibited a typical microvessel morphology with hierarchical vessel segments (Figure 2C).
We additionally characterized the ad-MVF by means of flow cytometry. For this purpose, ad-MVF were further digested in cell detachment solution for 30 min into single cells15. The flow cytometric analyses revealed that ad-MVF contain 26 ± 2% CD31-positive endothelial cells, 17 ± 2% α-smooth muscle actin (SMA)-positive perivascular cells, and 9 ± 1% cells positive for the mesenchymal stem cell marker CD117 (Figure 3A-C). After seeding on a dermal skin substitute, which was fixed and sectioned immediately for histological analyses, larger ad-MVF were mainly localized on the implant's surface (Figure 3D). However, capillary vessel segments could also be detected within its pores. Immunohistochemical staining of seeded ad-MVF further revealed physiological microvessel configuration with arteriolar, venular and capillary-like fragments (Figure 3E).
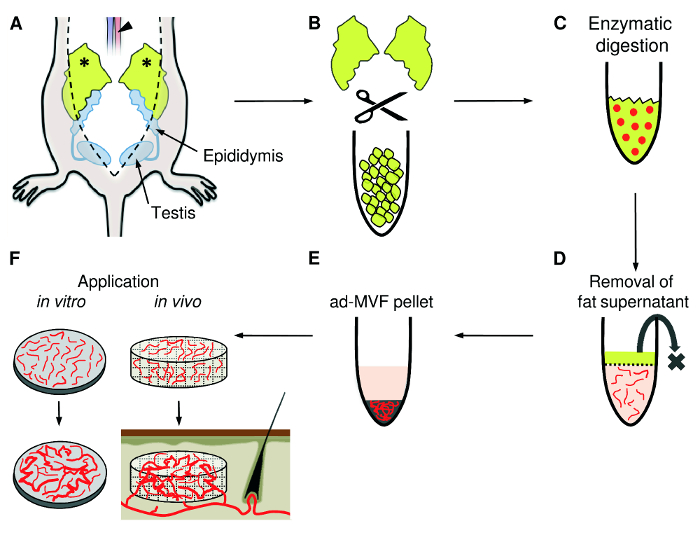
Figure 1: ad-MVF Isolation. A) Following midline laparotomy, the epididymal fat pads (asterisks) are identified. The adipose tissue is harvested with a small preparation scissors and the animal is sacrificed by incision of the abdominal aorta (arrowhead). B) Mechanical mincing of the fat pads with fine scissors until the tissue suspension appears homogeneous. C) For enzymatic digestion, collagenase is added to the tissue suspension. D) The cell-vessel suspension is incubated and the supernatant fat layer is discarded. E) After centrifugation, the pellet containing ad-MVF is resuspended and can be used for different applications, such as the in vitro analysis of microvascular network formation or the seeding of ad-MVF on scaffolds for in vivo implantation into tissue defects (F). Please click here to view a larger version of this figure.
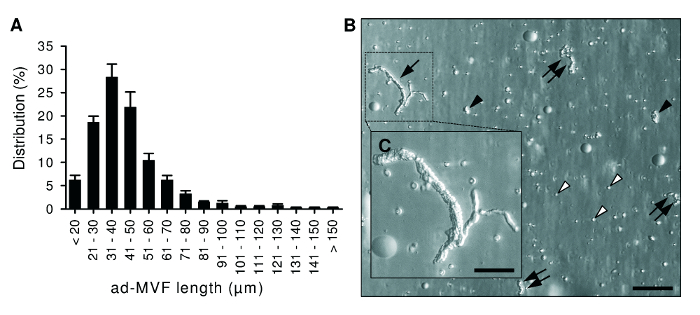
Figure 2: Morphological Characteristics of ad-MVF. A) Length distribution of freshly isolated ad-MVF. Mean ± standard error of the mean (n = 6). B) Microscopic image of a cell-vessel suspension smear with large (arrow), medium-sized (double arrows) and small (black arrowheads) ad-MVF as well as single cells (white arrowheads). Scale bar = 110 µm. Higher magnification reveals that ad-MVF exhibit a mature microvessel morphology with hierarchical microvessel segments (C, inset of B). Scale bar: C = 50 µm. Please click here to view a larger version of this figure.

Figure 3: Flow Cytometric, Histological and Immunohistochemical Characteristics of ad-MVF. A-C) Flow cytometric analyses of freshly isolated ad-MVF illustrating CD31-positive endothelial cell (A), α-SMA-positive perivascular cell (B) and CD117-positive mesenchymal stem cell (C) fractions. Appropriate isotype-identical controls were used to adjust threshold levels. Mean ± standard error of the mean (n = 6). D) Hematoxylin and eosin-stained section of a seeded dermal skin substitute. A few capillary vessel segments are located in the implant's pores (arrowheads). Larger microvessels with venular (double arrow) or arteriolar (arrow) morphology are localized on its surface. Scale bar = 40 µm. Broken line = implant border. E) Characterization of ad-MVF by means of immunohistochemical detection of the endothelial cell marker CD31 (red) and the perivascular cell marker α-SMA (green). Cell nuclei were stained with a DNA-binding fluorescent dye (blue). Arteriolar ad-MVF (arrow) are characterized by a thicker α-SMA-positive cell layer when compared to venular fragments with a larger lumen (double arrow). Arrowheads = capillary ad-MVF. Scale bar = 25 µm. Please click here to view a larger version of this figure.
Discussion
In this study we present a well-established protocol for the isolation of ad-MVF. Obtaining ad-MVF from murine adipose tissue is a straightforward procedure with a few critical steps. Mice exhibit different subcutaneous and intraabdominal fat deposits. As previously described for rats, the most suitable fat source for the isolation of ad-MVF are the epididymal fat pads due to their size, homogeneous structure and minimal contamination with larger blood vessels11,12. In contrast, subcutaneous fat deposits in mice are much smaller and more adherent to the surrounding tissue. However, the excision of epididymal fat pads bears the risk of accidental injury of the epididymis and spermatic cords, resulting in contamination of the harvested tissue with sperms. Therefore, the identification of epididymis, testis and spermatic cords is of major importance in this procedure. Moreover, the fat pads should be rapidly processed after harvesting to minimize ex vivo tissue damage. Finally, it should be considered that there are major age-dependent variations in the vascularization of murine epididymal fat pads23. In our experience, the use of 6- to 12-month-old male donor mice provides sufficient amounts of ad-MVF for in vitro and in vivo studies. However, it may also be possible to isolate ad-MVF from younger mice, which exhibit smaller but highly vascularized fat pads24.
The enzymatic digestion of the harvested epididymal fat pads crucially determines the final quality of isolated ad-MVF. Too short exposition to collagenase leads to incomplete separation of ad-MVF from surrounding adipocytes. On the other hand, prolonged digestion results in the destruction of ad-MVF to a single cell suspension, the so-called stromal vascular fraction (SVF). The SVF is a heterogeneous cell population containing mesenchymal stem cells, endothelial cells, pericytes, macrophages and many other cell types with regenerative potential through paracrine and differentiation mechanisms25. The use of ad-MVF exhibits considerable advantages when compared to SVF-based approaches. The fully functional microvessel morphology of ad-MVF allows their immediate implantation for in vivo experiments without time-consuming and complex in vitro cultivation. Moreover, the isolation of the SVF usually requires the digestion of the tissue with collagenase up to 90 min26. In contrast, tissue degradation should not exceed 10 min for the isolation of ad-MVF. This supports the idea of intraoperative one-step applications of ad-MVF9. For this purpose, ad-MVF may be automatically isolated from autologous lipoaspirates and transferred back into a tissue defect of the patient without leaving the operation theatre. This is a realistic goal considering the fact that the automated isolation of SVF from lipoaspirates is already a clinically established procedure in plastic surgery27,28,29.
Recently, we have demonstrated a strong effect of ad-MVF on the in vivo vascularization of bone14 and skin15 substitutes. Of interest, we found that it is beneficial not to remove single cells from the ad-MVF isolate, because they may provide a more physiological environment that contributes to the high vascularization capacity of ad-MVF13. Therefore, we only used a 500-µm filter during the isolation of ad-MVF for the removal of larger non-digested fat clots. Moreover, we found that ad-MVF induce lymphangiogenesis in implanted skin substitutes15. Hence, ad-MVF may also be promising building blocks for lymphatic tissue engineering and lymphedema therapy.
For the successful translation of these experimental findings into clinical practice, the next logical step is to clarify whether human adipose tissue is equally suited for the isolation of ad-MVF as rodent fat. In this context, a critical issue is the amount of fat required to isolate large amounts of ad-MVF. The herein described protocol resulted in approximately 40,000 ad-MVF per mL fat tissue. Accordingly, 3 mL fat tissue would be necessary to prevascularize 1 cm2 of dermal skin substitute15. Hence, this approach may not be feasible for the treatment of larger wounds or severely ill patients due to a limited amount of available fat tissue for ad-MVF isolation.
Beside a broad spectrum of potential in vivo applications in tissue engineering and regenerative medicine, ad-MVF are also suitable for in vitro modeling of angiogenic processes. For instance, rat ad-MVF have been cultured in three-dimensional collagen hydrogels to study the formation of new microvascular networks30. Similar approaches have been used to evaluate sophisticated imaging techniques for the visualization of neovessel growth13 or interstitial matrix remodeling during angiogenesis31.
Taken together, these findings indicate that ad-MVF are not only promising vascularization and lymphangiogenic units for future therapeutic strategies, but also for basic research in the field of angiogenesis and vascular physiology.
Divulgations
The authors have nothing to disclose.
Acknowledgements
We are grateful for the excellent technical assistance of Janine Becker, Caroline Bickelmann and Ruth Nickels. This study was funded by a grant of the Deutsche Forschungsgemeinschaft (DFG – German Research Foundation) – LA 2682/7-1.
Materials
| 1.5-mL conical microcentrifuge tube | VWR, Kelsterbach, Germany | 700-5239 | |
| 100-µL precision pipette | Eppendorf, Hamburg, Germany | 4920000059 | |
| 10-mL measuring pipette | Costar, Corning Inc., New York, USA | 4488 | |
| 14-mL PP tubes | Greiner bio-one, Frickenhausen, Germany | 187261 | |
| 1-mL precision pipette | Eppendorf, Hamburg, Germany | 4920000083 | |
| 500-µm filter (pluriStrainer 500 µm) | HISS Diagnostics, Freiburg, Germany | 43-50500-03 | |
| 50-mL conical centrifuge tube | Greiner bio-one, Frickenhausen, Germany | 227261 | |
| 50-mL Erlenmeyer flask | VWR, Kelsterbach, Germany | 214-0211 | |
| 96-well plate | Greiner bio-one, Frickenhausen, Germany | 65518 | |
| cell detachment solution (Accutase) | eBioscience, San Diego, CA USA | 00-4555-56 | |
| C57BL/6 mice | Charles River, Cologne, Germany | 027 | |
| C57BL/6-Tg(CAG-EGFP)1Osb/J mice | The Jackson Laboratory, Bar Harbor, USA | 003291 | |
| CD117-FITC | BD Biosciences, Heidelberg, Germany | 553373 | |
| CD31-PE | BD Biosciences, Heidelberg, Germany | 553354 | |
| Collagenase NB4G | Serva Electrophoresis GmbH, Heidelberg, Germany | 17465.02 | Lot tested by manufacturer for enzymatic activity |
| Dissection scissors | Braun Aesculap AG &CoKG, Melsungen, Germany | BC 601 | |
| DNA-binding dye (Bisbenzimide H33342) | Sigma-Aldrich, Taufkirchen, Germany | B2261 | |
| Dulbecco's modified Eagle medium (DMEM) | PAN Biotech, Rickenbach, Germany | P04-03600 | |
| Fetal calf serum (FCS) | Biochrom GmbH, Berlin, Germany | S0615 | |
| Fine forceps | S&T AG, Neuhausen, Switzerland | FRS-15 RM-8 | |
| Fine scissors | World Precision Instrumets, Sarasota, FL, USA | 503261 | |
| Dermal skin substitute (Integra) | Integra Life Sciences, Sain Priest, France | 62021 | |
| Ketamine | Serumwerk Bernburg AG, Bernburg, Germany | 7005294 | |
| M-IgG2akAL488 | eBioscience, San Diego, CA USA | 53-4724-80 | |
| Octeniderm (disinfecting solution) | Schülke & Mayer, Norderstedt, Germany | 118211 | |
| Penicillin/Streptomycin | Biochrom, Berlin, Germany | A2213 | |
| Petri dish | Greiner bio-one, Frickenhausen, Germany | 664160 | |
| Phosphate-buffered saline (PBS) | Lonza Group, Basel, Switzerland | 17-516F | |
| pluriStrainer 20-µm (20 µm filter) | HISS Diagnostics, Freiburg, Germany | 43-50020-03 | |
| Rat-IgG2akFITC | BD Biosciences, Heidelberg, Germany | 553988 | |
| Rat-IgG2akPE | BD Biosciences, Heidelberg, Germany | 553930 | |
| Small preparation scissors | S&T AG, Neuhausen, Switzerland | SDC-15 R-8S | |
| Surgical forceps | Braun Aesculap AG &CoKG, Melsungen, Germany | BD510R | |
| Tape (Heftpflaster Seide) 1.25 cm | Fink & Walter GmbH, Mechweiler, Germany | 1671801 | |
| Xylazine | Bayer Vital GmbH, Leverkusen, Germany | 1320422 | |
| α-SMA-AL488 | eBioscience, San Diego, CA USA | 53-9760-82 | Intracellular labeling additionally requires Cytofix/Cytoperm (BD Biosciences, Heidelberg, Germany; #554722) |
References
- Langer, R., Vacanti, J. P. Tissue engineering. Science. 260 (5110), 920-926 (1993).
- Khademhosseini, A., Langer, R. A decade of progress in tissue engineering. Nat Protoc. 11 (10), 1775-1781 (2016).
- Novosel, E. C., Kleinhans, C., Kluger, P. J. Vascularization is the key challenge in tissue engineering. Adv Drug Deliv Rev. 63 (4-5), 300-311 (2011).
- Rouwkema, J., Khademhosseini, A. Vascularization and Angiogenesis in Tissue Engineering: Beyond Creating Static Networks. Trends Biotechnol. 34 (9), 733-745 (2016).
- Laschke, M. W., Menger, M. D. Vascularization in tissue engineering: angiogenesis versus inosculation. Eur Surg Res. 48 (2), 85-92 (2012).
- Sarker, M., Chen, X. B., Schreyer, D. J. Experimental approaches to vascularisation within tissue engineering constructs. J Biomater Sci Polym Ed. 26 (12), 683-734 (2015).
- Frueh, F. S., Menger, M. D., Lindenblatt, N., Giovanoli, P., Laschke, M. W. Current and emerging vascularization strategies in skin tissue engineering. Crit Rev Biotechnol. 20, 1-13 (2016).
- Utzinger, U., Baggett, B., Weiss, J. A., Hoying, J. B., Edgar, L. T. Large-scale time series microscopy of neovessel growth during angiogenesis. Angiogenesis. 18 (3), 219-232 (2015).
- Laschke, M. W., Menger, M. D. Prevascularization in tissue engineering: Current concepts and future directions. Biotechnol Adv. 34 (2), 112-121 (2016).
- Baiguera, S., Ribatti, D. Endothelialization approaches for viable engineered tissues. Angiogenesis. 16 (1), 1-14 (2013).
- Wagner, R. C., Kreiner, P., Barrnett, R. J., Bitensky, M. W. Biochemical characterization and cytochemical localization of a catecholamine-sensitive adenylate cyclase in isolated capillary endothelium. Proc Natl Acad Sci U S A. 69 (11), 3175-3179 (1972).
- Wagner, R. C., Matthews, M. A. The isolation and culture of capillary endothelium from epididymal fat. Microvasc Res. 10 (3), 286-297 (1975).
- Laschke, M. W., Menger, M. D. Adipose tissue-derived microvascular fragments: natural vascularization units for regenerative medicine. Trends Biotechnol. 33 (8), 442-448 (2015).
- Laschke, M. W., et al. Vascularisation of porous scaffolds is improved by incorporation of adipose tissue-derived microvascular fragments. Eur Cell Mater. 24, 266-277 (2012).
- Frueh, F. S., et al. Adipose tissue-derived microvascular fragments improve vascularization, lymphangiogenesis and integration of dermal skin substitutes. J Invest Dermatol. 137 (1), 217-227 (2017).
- McDaniel, J. S., Pilia, M., Ward, C. L., Pollot, B. E., Rathbone, C. R. Characterization and multilineage potential of cells derived from isolated microvascular fragments. J Surg Res. 192 (1), 214-222 (2014).
- Nakano, M., et al. Effect of autotransplantation of microvessel fragments on experimental random-pattern flaps in the rat. Eur Surg Res. 30 (3), 149-160 (1998).
- Nakano, M., et al. Successful autotransplantation of microvessel fragments into the rat heart. Eur Surg Res. 31 (3), 240-248 (1999).
- Shepherd, B. R., Hoying, J. B., Williams, S. K. Microvascular transplantation after acute myocardial infarction. Tissue Eng. 13 (12), 2871-2879 (2007).
- Pilia, M., et al. Transplantation and perfusion of microvascular fragments in a rodent model of volumetric muscle loss injury. Eur Cell Mater. 28, 11-23 (2014).
- Laschke, M. W., et al. Adipose tissue-derived microvascular fragments from aged donors exhibit an impaired vascularisation capacity. Eur Cell Mater. 28, 287-298 (2015).
- Okabe, M., Ikawa, M., Kominami, K., Nakanishi, T., Nishimune, Y. ‘Green mice’ as a source of ubiquitous green cells. FEBS Lett. 407 (3), 313-319 (1997).
- Honek, J., et al. Modulation of age-related insulin sensitivity by VEGF-dependent vascular plasticity in adipose tissues. Proc Natl Acad Sci U S A. 111 (41), 14906-14911 (2014).
- Cho, C. H., et al. Angiogenic role of LYVE-1-positive macrophages in adipose tissue. Circ Res. 100 (4), e47-e57 (2007).
- Han, S., Sun, H. M., Hwang, K. C., Kim, S. W. Adipose-Derived Stromal Vascular Fraction Cells: Update on Clinical Utility and Efficacy. Crit Rev Eukaryot Gene Expr. 25 (2), 145-152 (2015).
- Chen, Y. J., et al. Isolation and Differentiation of Adipose-Derived Stem Cells from Porcine Subcutaneous Adipose Tissues. J Vis Exp. (109), e53886 (2016).
- Guillaume-Jugnot, P., et al. Autologous adipose-derived stromal vascular fraction in patients with systemic sclerosis: 12-month follow-up. Rheumatology (Oxford). 55 (2), 301-306 (2016).
- Tissiani, L. A., Alonso, N. A Prospective and Controlled Clinical Trial on Stromal Vascular Fraction Enriched Fat Grafts in Secondary Breast Reconstruction. Stem Cells Int. , 2636454 (2016).
- Calcagni, M., et al. The novel treatment of SVF-enriched fat grafting for painful end-neuromas of superficial radial nerve. Microsurgery. , (2016).
- Hoying, J. B., Boswell, C. A., Williams, S. K. Angiogenic potential of microvessel fragments established in three-dimensional collagen gels. In Vitro Cell Dev Biol Anim. 32 (7), 409-419 (1996).
- Kirkpatrick, N. D., Andreou, S., Hoying, J. B., Utzinger, U. Live imaging of collagen remodeling during angiogenesis. Am J Physiol Heart Circ Physiol. 292 (6), H3198-H3206 (2007).

