Plant Promoter Analysis: Identification and Characterization of Root Nodule Specific Promoter in the Common Bean
Summary
Promoter expression analyses are crucial to improving the understanding of gene regulation and the spatiotemporal expression of target genes. Herein we present a protocol to identify, isolate, and clone a plant promoter. Further, we describe the characterization of the nodule-specific promoter in the common bean hairy roots.
Abstract
The upstream sequences of gene coding sequences are termed as promoter sequences. Studying the expression patterns of promoters are very significant in understanding the gene regulation and spatiotemporal expression patterns of target genes. On the other hand, it is also critical to establish promoter evaluation tools and genetic transformation techniques that are fast, efficient, and reproducible. In this study, we investigated the spatiotemporal expression pattern of the rhizobial symbiosis-specific nodule inception (NIN) promoter of Phaseolus vulgaris in the transgenic hairy roots. Using plant genome databases and analysis tools we identified, isolated, and cloned the P. vulgaris NIN promoter in a transcriptional fusion to the chimeric reporter β-glucuronidase (GUS) GUS-enhanced::GFP. Further, this protocol describes a rapid and versatile system of genetic transformation in the P. vulgaris using Agrobacterium rhizogenes induced hairy roots. This system generates ≥2 cm hairy roots at 10 to 12 days after transformation. Next, we assessed the spatiotemporal expression of NIN promoter in Rhizobium inoculated hairy roots at periodic intervals of post-inoculation. Our results depicted by GUS activity show that the NIN promoter was active during the process of nodulation. Together, the present protocol demonstrates how to identify, isolate, clone, and characterize a plant promoter in the common bean hairy roots. Moreover, this protocol is easy to use in non-specialized laboratories.
Introduction
Promoters are important molecular biological tools which play a crucial role in understanding the regulation of the expression of genes of interest. Promoters are DNA sequences located upstream of the translation initiation codon of gene sequences and they carry the central regulatory information of genes; therefore, their correct annotation and characterization are vital to understanding gene function. Depending on the expression patterns, plant promoters are classified as constitutive, tissue-specific, or development-stage-specific and inducible1. Advances in transcriptomic technologies, improvements in computer modeling, and the availability of increasing numbers of genome sequences for different plant species have facilitated the large-scale prediction of promoter sequences2.
On the other hand, it is also critical to establish promoter evaluation tools and genetic transformation techniques that are fast, efficient, and reproducible. Unlike the other model plants, the functional characterization of common bean legume (P. vulgaris) genes is impeded chiefly due to the recalcitrant nature of Phaseolus sp. for stable genetic transformation. Transient transformation systems serve as an alternative for rapid gene functional characterization studies3. In legume symbiosis research, the interaction between the legume host plant and rhizobial bacterium is one of the most tractable model systems for the functional analysis of nodule-specific genes and promoter studies. So far, several legume promoters related to these symbioses have been characterized, viz., Medicago truncatula PT44, SWEET115, Lotus japonicus Cyclops, UBQ6, VAG17, Glycine max PT58, Exo70J9, P. vulgaris RbohB10,11,12, TRE113, PI3K14, TOR15, etc.Cis elements directly influence gene regulation. The transcription factor ENBP1A binds to a Cis regulatory region (−692 bp) of early nodulin VfENOD12, and this facilitates the expression of a reporter gene in nodule primordia of Vicia faba16. Replacement of Cis regulatory regions (−161 to −48 bp) of the nodule-specific promoter leghemoglobin GLB3 with the heterologous truncated promoters δ-p35S and δ-pNOS, resulted in a loss of nodule specificity and reduced promoter activity17.
Previous reports show that the transcription factor NIN is required for the initiation of rhizobial infection in the root hair cells and is also essential for nodule organogenesis in L. japonicus18. In the present study, we describe a protocol for identification, isolation, cloning, and characterization of nodule-specific promoter in the common bean hairy roots. To achieve this, we selected a rhizobial symbiosis-specific NIN promoter of P. vulgaris and cloned in a transcriptional fusion to the chimeric reporter GUS-enhanced::GFP. Further, this protocol describes a rapid and versatile system of genetic transformation in the P. vulgaris using A. rhizogenes induced hairy roots. This system generates hairy roots in less than 2 weeks after transformation. Finally, we assessed the spatiotemporal expression of NIN promoter in rhizobia colonized root nodules by GUS staining.
The procedure described here may be useful not only for the study of nodulation and mycorrhization11 of legume plants, but also for the study of promoter expression patterns in roots19. Moreover, this protocol is easy to use in non-specialized laboratories.
Protocol
1. Identification, Isolation, and Cloning of P. Vulgaris NIN Promoter
- Identify the promoter sequence for the gene of interest. There are several genome databases and analysis tools available for plants such as Phytozome, Ensembl Plants, NCBI, etc. A legume promoter P. vulgaris NIN (PvNIN; Phvul.009G115800) was used in this study20.
- Design oligos either for Gateway or traditional restriction enzyme cloning depending on the vector system used (Figure 1A).
NOTE: Standard (25-35 bp), desalted primers work fine. Reverse oligo sequences that flank between the promoter and gene are advisable. - Amplify the PvNIN promoter region (or promoter region of the gene of interest) from freshly isolated P. vulgaris genomic DNA using the promoter-specific oligo set. Use a high-fidelity polymerase with the following PCR conditions: 95 °C for 5 min; 30 cycles (95 °C for 30 s, 55 °C for 30 s, 72 °C for 1 min); and 72 °C for 5 min.
- Run the amplified fragment on 1% agarose gel at 60 V on a 7 x 10 cm gel tray, and elute the corresponding amplicon (700 bp, Figure 1B) and clone into the pENTR/D-TOPO vector according to the manufacturer's instructions.
- Transform 2 µL of the reaction into competent Escherichia coli cells using the manufacturer's instructions and plate 150 µL of the transformation mixture on lysogeny brother (LB) supplemented with 50 mg/L kanamycin (Kan50). Grow the plated cells at 37 °C overnight.
- Pick 3-6 colonies and culture them overnight in LB medium containing Kan50. Isolate plasmid DNA using the method of choice and analyze the plasmid by PCR using the vector specific M13 oligos. For PCR, use 100 ng of plasmid, 0.3 pm of oligos, 10X PCR buffer, 0.5 U Taq DNA polymerase, and 10 mM dNTPs.
- Use the following PCR amplification conditions; 95 °C for 3 min; 30 cycles (95 °C for 30 s, 58 °C for 30 s, 72 °C for 75 s); and 72 °C for 7 min. Visualize the PCR products on a 1% agarose gel. Ensure that the correct insertions have a band at 1,024 bp and vectors without inserts will have a 324 bp band (Figure 1C).
- Perform the LR reaction between an entry vector (pENTR/D-TOPO-PvNIN) and destination vector pBGWFS7.021 using a commercial enzyme mix according to the manufacturer's instructions.
- Transform 2 µL of LR reaction into competent E. coli cells using standard techniques as described in the manufacturer's instructions and plate 150 µL of transformation mixture on LB supplemented with 100 mg/L of spectinomycin (Spe100). Grow the plated cells at 37 °C overnight.
- Pick approximately 5 colonies and culture them overnight in LB medium containing Spe100. Isolate the plasmid DNA using a method of choice and analyze the plasmid by PCR using the promoter-specific oligos.
- Use the PCR conditions in step 1.3. Visualize the PCR products on a 1% agarose gel to confirm amplification. Amplicons are 700 bp.
- Sequence the positive plasmid pBGWFS7.0/PvNIN::GUS-GFP (Figure 1D) with the promoter-specific oligos to verify the authenticity of the insert.
- Transform the plasmids into A. rhizogenes strain K599 (however, any other strains of A. rhizogenes can be used). Add 2 µL of the plasmid prep to 50 µL of electrocompetent cells and electroporate in a 1 mm cuvette with the following settings: 1.8 kV, 25 µF, and 200 Ω.
- Recover cells in ~ 500 µL of SOC medium and shake at 28 °C for 2 h. Plate on LB-Spe100 and grow at 28 °C for 2 days.
- Perform colony screening as mentioned in step 1.7 and use the PCR conditions as in step 1.3. Make glycerol stocks from positive clones and store them in -80 °C.
- Use A. rhizogenes culture harboring empty pBGWFS7.0 vector as the negative control.
2. A. Rhizogenes Transformed Hairy Roots of the Common Bean
- Sterilize bean (P. vulgaris cv. Negro Jamapa) seeds (approximately 20) in 50 mL of 96% ethanol for 1 min and discard the ethanol; then add 50 mL of 20% bleach (sodium hypochlorite) for 10 min, with constant mixing in a laminar flow hood. Remove the bleach and wash in excess sterile water 3 times.
NOTE: Other P. vulgaris cultivars can be used for hairy root induction. - Germinate the sterilized seeds on sterile filter papers moistened with Broughton & Dilworth (B&D) solution22 (Table 1) for 2 days in the dark at 28 °C.
- The day before transformation prepare the following:
- Streak out 150 µL of A. rhizogenes culture harboring the binary vector (prepared in steps 1.13 and 1.14) on solid LB Spe100 and grow at 28 °C overnight.
- Fill a 15 mL tube with B&D solution and replace the screw cap with double layered aluminum foil. Assemble this into a 22 cm glass tube containing 10 mL of B&D solution (Figure 2C) and autoclave it.
- On the day of transformation, assemble the following sterile materials in a laminar flow hood: germinated seeds from step 2.2, culture plate from step 2.3.1, tubes from step 2.3.2, sterile double distilled water (10 mL), pipettes and pipette tips, sterile forceps, and 3 mL syringes.
- Use a bent 200 µL tip to scrape A. rhizogenes cells from the plate into a microcentrifuge tube and resuspend in 1 mL of the double sterile water. Similarly, prepare vector control cells separately.
- Mix the cells gently to resuspend. Do not vortex.
- Make a 3 mm hole on the aluminum foil of the tubes from step 2.3.2 using a sterile pipette tip.
- Collect the cells (either promoter or vector control) from step 2.5 with the syringe and slightly prick the hypocotyls of the germinated seeds with the needle tip (Figure 2G).
NOTE: Make sure the needle penetrates (4-6 times) the vascular tissue and inject small (~ 5 µL) drops of the cells on the wound at different positions around the hypocotyl. - Carefully insert the roots of the injected seedlings into the 3 mm hole of the 15 mL tube containing B&D solution. Return the entire setup into the 22 mL glass tubes and seal them to maintain humidity.
- Incubate the tubes in a growth chamber maintained at 28 °C with 16 h light:8 h dark photoperiod.
Notes: 3-4 days after incubation, the plants grow up to the rim of the 22 mL glass tubes. At this stage, remove the caps and seal the rim with parafilm around the stem as shown in Figure 2I. - Observe the callus at the wounding sites at 5-7 days, and find ~ 2 cm hairy roots by 10-12 days after transformation.
NOTE: It is important to maintain the B&D solution constantly in both plastic and glass tubes. - After 2 weeks, remove the primary root by cutting the stem 2 cm below the hairy roots and transplant the primary roots into pots containing sterile vermiculite. Maintain the plants in a growth chamber (16 h light:8 h dark at 28 °C) and irrigate with B&D solution.
3. Promoter Analysis: NIN Promoter Expression in Rhizobial Nodules of the Common Bean
- For induction of root nodules, inoculate Rhizobium tropici or Rhizobium etli (that are compatible to the common bean) in 100 mL of PY medium (0.5 g peptone, 0.3 g yeast extract) supplemented with 700 mM CaCl2 and 20 mg mL−1 nalidixic acid. Incubate at 30 °C for 24 h with shaking at 300 rpm.
Note: Any specific antibiotics could be used in the case of transgenic Rhizobium. - Pellet the cells in a centrifuge for 3 min at 2,800 x g at room temperature and resuspend in 10 mL of 10 mM MgSO4. Adjust the OD of cells to 0.6 at OD600 by dilution with 10 mM MgSO4.
- Inoculate 1-2 mL of culture (prepared from step 3.2) per plant (both promoter and vector control) to induce nodule growth on the hairy roots. Irrigate the plants with B&D solution with limited nitrogen (2 mM KNO3) to promote nodulation.
- Depending on the experimental need, collect the hairy roots at different time points. Collect samples for studying infection threads between 3-5 days post inoculation (dpi).
Note: Primordial nodule and young nodules can be studied at 6-9 dpi and 10-12 days dpi, respectively. Finally, mature and functional nodules can be studied at 14-21 dpi, and senesced nodules at > 28 dpi. - Process the root/nodules for the activity of reporter genes (GUS or GFP). Analyze GUS activity according to the protocol described by Jefferson23. Observe GFP under a fluorescence microscope. Excite GFP fluorescence with a blue argon ion laser (488 nm), and collect the emitted fluorescence from 510 to 540 nm.
Representative Results
The objective of this study was to assess the spatiotemporal expression pattern of nodule-specific P. vulgaris NIN. To do this, a 700 bp region upstream of the translation initiation codon of the NIN gene was selected and a set of oligos was designed as depicted in Figure 1A. Using a high-fidelity polymerase, the NIN promoter fragment was amplified, isolated (Figure 1B), and cloned into the destination binary vector pBGWFS7.0 (Figure 1C) through a Gateway approach to generate a transcriptional fusion to the chimeric reporter GUS-enhanced GFP (pBGWFS7.0/PvNIN::GUS-GFP). The pBGWFS7.0/PvNIN::GUS-GFP vector construct typically generates hundreds of independent clones. Colony screening by PCR with gene-specific oligos easily identifies the correct clones (Figure 2D). The PCR positive plasmid was Sanger sequenced with the promoter-specific oligos to verify the authenticity of the insert.
Maintaining high humidity throughout the experiment is necessary for the induction of callus and hairy roots. At the site of wounding, calli are usually observed at 5 to 7 days (Figure 2J) and ≥ 2 cm hairy roots are observed at 10 to 12 days after transformation. The majority of A. rhizogenes transformed plants successfully to produce hairy roots by 2 weeks (Figure 2K). However, the number and length of hairy roots can be variable. Therefore, select the most vigorously growing roots for analysis and further experiments. Excise the primary root by cutting the stem 2 cm below the hairy roots and transplant them into pots containing sterile vermiculite (Figure 2L) or in a hydroponics system.
To study the spatiotemporal expression of the nodule-specific NIN promoter, hairy roots were inoculated with R. tropici, and GUS activity was observed at periodic intervals post-inoculation depending on the experimental need. Inoculation with R. tropici induced a strong GUS expression in the transgenic roots, indicating that rhizobia induces the NIN expression in the common bean (Figure 3A). Further, sampling at 6-9 dpi showed GUS expression in Rhizobium infected root hair cells (Figure 3B). Phaseolus nodule primordium, and young and mature nodules also demonstrated NIN expression (Figure 3C). At all stages of nodulation, no such GUS expression was seen in the vector control roots (Figure 3D, 3E, 3F).
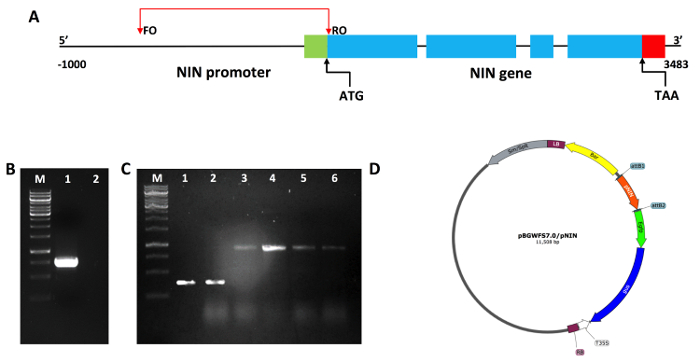
Figure 1: Outline of P. vulgaris NIN gene structure and cloning. (A) A schematic representation of the NIN gene structure showing 5'UTR (green), 4 exons, 3 introns, and 3'UTR (red) on chromosome number 9 of P. vulgaris. Upstream to the NIN translation initiation codon ATG, shows a promoter region on which oligos were designed. FO, forward oligo; RO, reverse oligo; ATG, start codon; TAA, stop codon. (B) PvNIN promoter region was amplified from freshly isolated P. vulgaris genomic DNA using the promoter-specific oligo set. M, 1 Kb ladder; 1, PCR reaction with gDNA showing amplicon size of 700 bp; 2, PCR reaction without gDNA (-ve control). (C) Screening of the pENTR/D-TOPO-PvNIN plasmids by PCR using the vector specific M13 oligos. Correct insertions have a band at 1,024 bp (lane 3-6) and vectors without inserts will have a 324 bp band (lane 1-2). (D) Promoter expression binary vector map used in the present study, pBGWFS7.0 vector showing PvNIN promoter in fusion with GUS and GFP. Please click here to view a larger version of this figure.
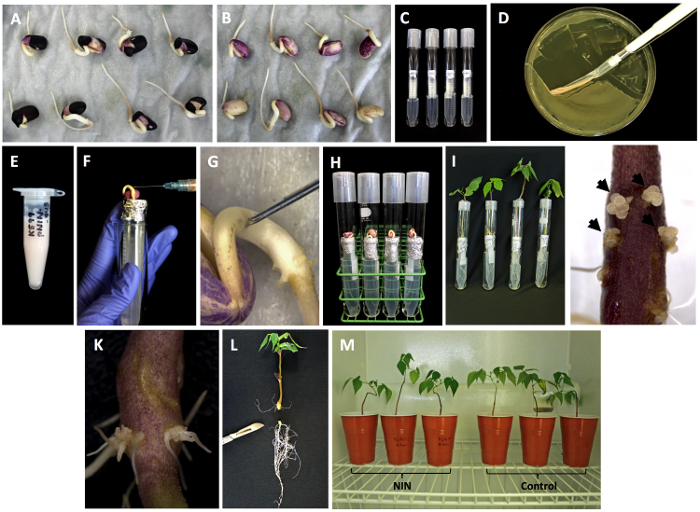
Figure 2: A. rhizogenes transformed hairy roots of the common bean. (A) The sterilized seeds of P. vulgaris cv. Negro Jamapa germinating on sterile filter paper. (B) Seeds, coat removed. (C) Tube setup for hairy root induction. (D) Scraping out A. rhizogenes culture (pNIN & control). (E) A. rhizogenes culture resuspended in sterile distilled water. (F, G) Injecting bacterial cells into hypocotyles. (H) Tube setup with Phaseolus seedlings injected with bacterial cells. (I, J) Calli formed at wounded site 5-7 dpi. (K) Hairy roots 15 dpi (L) excising the tap roots 2 cm below the site of hairy root induction. (M) Composite plants transplanted into sterile vermiculite inoculated with R. tropici. Please click here to view a larger version of this figure.
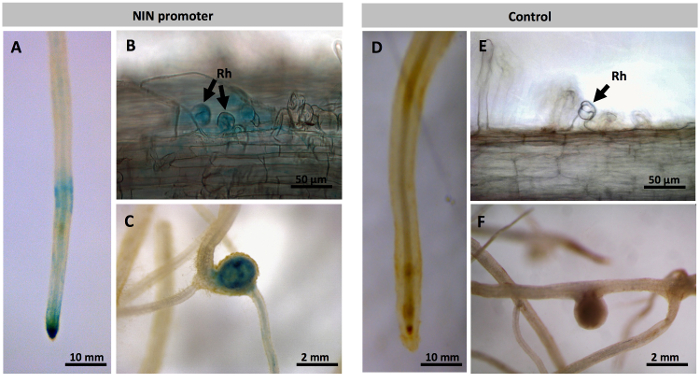
Figure 3: Promoter analysis of P. vulgaris NIN in transgenic P. vulgaris roots. Spatial and temporal pattern of NIN expression revealed by a promoterNIN::GUS-GFP construct in transgenic hairy roots incubated with GUS as a substrate. Optical microscope images of PvNIN:GUS : (A) Roots inoculated with R. tropici, (B) GUS expression in curled root hairs, and (C) young nodules. Images of control (empty vector) showing no such GUS expression in (D) roots inoculated with R. tropici, (E) curled root hairs, and (F) young nodules. Rh, root hair. Please click here to view a larger version of this figure.
Discussion
During functional analysis of genes, the study of gene expression patterns plays a crucial role in understanding the spatial and temporal regulation of the genes in vivo. A well-known method to study gene expression patterns is to clone the gene promoter region of interest, upstream to reporter genes such as fluorescent marker genes (GFP, RFP, etc.) or β-glucuronidase. Herein, we have selected a promoter region of the root nodule symbiosis (RNS) specific gene, NIN, to study the spatio-temporal expression patterns in P. vulgaris during RNS. The selected promoter sequence has been cloned upstream to GFP::GUS reporters using the Gateway method into a promoter expression vector.
We have described the expression of the symbiosis specific promotor in the hairy root system of P. vulgaris. The hairy root system has been widely used for functional characterization of genes, including RNAi mediated gene knock down, ectopic constitutive expression of genes, promoter expression, and protein localization in recalcitrant crops. The main advantages of the system are that it is easy, reproducible, reliable, and at the same time not labor intensive. The hairy root system has been successfully adopted in other crop legumes such as G. max24, M. truncatula25, L. japonicus26,27, tomato28, etc., with the necessary modifications. Further, the tool can be adopted in other legume and non-legume species when optimized for the suitable A. rhizogenes strain, method of inoculation, and growth conditions.
There are many critical parameters. While selecting the promoter region, care should be taken to design the oligos which would amplify the necessary promoter elements, beginning from the upstream region of the translation start sitein the protein coding genes; promoter analysis should be carried out using the available software to collect information on the transcription factors binding to the regulatory region. Take care while inducing hairy roots: inject seedlings at the appropriate stage; during injection, ensure that the needle does not pass through the hypocotyle; and maintain high humid conditions. Further, while studying promoter expression profiles in hairy roots, the hairy roots produced by A. rhizogenes are not always transformed, and hence it is important to confirm the transformed nature of the roots using GFP or GUS reporters prior to performing promoter expression studies. Suitable strains of Rhizobium should be chosen and the concentrations of Rhizobium should be adjusted as indicated in the protocol. Sampling at the appropriate stages post inoculation is crucial for documenting spatio-temporal expression of the promoter at different stages of RNS.
A. rhizogenes transformed hairy root production is one of the fastest and efficient alternative methods to generate composite plants in P. vulgaris. These composite plants cannot produce transgenic seeds, but can produce transgenic roots within a few days. In this protocol, unlike the other methods29, the closed tubes maintain constant high humidity to promote hairy roots faster at 10 days dpi.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This work was partially supported by the Dirección General de Asuntos del Personal Académico, DGAPA/PAPIIT-UNAM (grant no. IN219916 to M.L and IA205117 to M-K.A) and the Consejo Nacional de Ciencia y Tecnològia (CONACYT grant no. 240614 to M.L.).
Materials
| Primers for qRT-PCR assay | |||
| pNIN Forward | CACC ATA GCT CCC CAA AAT GGT AT | ||
| pNIN Reverse | CAT CTT CCT TCC ACT AAC TAA C | ||
| M13 Forward | GTA AAA CGA CGG CCA G | ||
| M13 Reverse | CAG GAA ACA GCT ATG AC | ||
| Name | Company | Catalog Number | Comments |
| REAGENTS | |||
| pENTR/D-TOPO Cloning Kit | Invitrogen | K243520 | |
| Gateway LR Clonase II Enzyme Mix | Invitrogen | 11791100 | |
| pBGWFS7.0 | Plant systems biology | https://gateway.psb.ugent.be/vector/show/pBGWFS7/search/index/ | |
| Platinum Taq DNA Polymerase | ThermoFisher Scientific | 10966018 | |
| DNeasy Plant Mini Kit | Qiagen | 69104 | |
| PureLink Quick Gel Extraction Kit | ThermoFisher Scientific | K210012 | |
| Platinum Pfx DNA Polymerase | Invitrogen | 11708013 | |
| Certified Molecular Biology Agarose | Bio-Rad | 1613102 | |
| One Shot TOP10 Chemically Competent E. coli | Invitrogen | C404006 | |
| Nacl | Sigma-Aldrich | S7653 | |
| Tryptone | Sigma-Aldrich | T7293-250G | |
| Yeast extract | Sigma-Aldrich | Y1625-250G | |
| Bacteriological agar | Sigma-Aldrich | A5306-1KG | |
| Kanamycin sulfate | Sigma-Aldrich | 60615-25G | |
| Spectinomycin sulfate | Sigma-Aldrich | PHR1441 | |
| Ethyl alcohol | Sigma-Aldrich | E7023 | |
| Bacteriological peptone | Sigma-Aldrich | P0556 | |
| Calcium chloride | Sigma-Aldrich | C1016 | |
| Nalidixic acid | Sigma-Aldrich | N8878 | |
| Magnesium sulfate | Sigma-Aldrich | M7506 | |
| Gel loading solution | Sigma-Aldrich | G7654 | |
| Name | Company | Catalog Number | Comments |
| EQUIPMENT | |||
| Thermocycler | Veriti Thermal Cycler | 4375786 | |
| Centrifuge | Sigma | Sigma 1-14K | |
| Gel documentation unit | Carestream | Gel Logic 212 PRO | |
| MaxQ SHKE6000 6000 Shaking Incubator – 115VAC | Thermo scientific | EW-51708-70 | |
| Plant growth chamber | MRC | PGI-550RH | |
| Horizantal laminarair flow cabinate | Lumistell | LH-120 | |
| Fluorescent microscope | Leica | DM4500 B | |
| Petridish | sym laboratorios | 90X15 | |
| Scalpel Blade | Fisher scientific | 53223 | |
| Falcon 15mL Conical Centrifuge Tubes | Fisher scientific | 14-959-53A | |
| 22 mL glass tubes | Thomas scientific | 45048-16150 |
References
- Hernández-Garcia, C. M., Finer, J. J. Identification and validation of promoters and cis-actingregulatory elements. Plant Science. 217-218, 109-119 (2014).
- Dhanapal, A. P., Govindaraj, M. Unlimited Thirst for Genome Sequencing, Data Interpretation, and Database Usage in Genomic Era: The Road towards Fast-Track Crop Plant Improvement. Genetics Research International. , 684321 (2015).
- Nanjareddy, N., Arthikala, M. K., Blanco, L., Arellano, E. S., Lara, M. Protoplast isolation, transient transformation of leaf mesophyll protoplasts and improved Agrobacterium-mediated leaf disc infiltration of Phaseolus vulgaris: Tools for rapid gene expression analysis. BMC Biotechnol. 16 (1), 53 (2016).
- Pumplin, N., Zhang, X., Noar, R. D., Harrison, M. J. Polar localization of a symbiosis-specific phosphate transporter is mediated by a transient reorientation of secretion. Proc Natl Acad Sci U S A. 109 (11), E665-E672 (2012).
- Kryvoruchko, I. S., et al. MtSWEET11, a Nodule-Specific Sucrose Transporter of Medicago truncatula. Plant Physiol. 171 (1), 554-565 (2016).
- Suzaki, T., Yano, K., Ito, M., Umehara, Y., Suganuma, N., Kawaguchi, M. Positive and negative regulation of cortical cell division during root nodule development in Lotus japonicus is accompanied by auxin response. Development. 139 (21), 3997-4006 (2012).
- Suzaki, T., et al. Endoreduplication-mediated initiation of symbiotic organ development in Lotus japonicus. Development. 141 (12), 2441-2445 (2014).
- Qin, L., et al. The high-affinity phosphate transporter GmPT5 regulates phosphate transport to nodules and nodulation in soybean. Plant Physiol. 159 (4), 1634-1643 (2012).
- Wang, Z., Panfeng, L., Yan, Y., Chi, Y., Fan, B., Chen, Z. Expression and Functional Analysis of a Novel Group of Legume-specific WRKY and Exo70 Protein Variants from Soybean. Sci Rep. 6, 32090 (2016).
- Montiel, J., et al. A Phaseolus vulgaris NADPH oxidase gene is required for root infection by Rhizobia. Plant Cell Physiol. 53 (10), 1751-1767 (2012).
- Arthikala, M. K., et al. PvRbohB negatively regulates Rhizophagus irregularis colonization in Phaseolus vulgaris. Plant Cell Physiol. 54 (8), 1391-1402 (2013).
- Arthikala, M. K., Sánchez-López, R., Nava, N., Santana, O., Cárdenas, L., Quinto, C. RbohB a Phaseolus vulgaris NADPH oxidase gene, enhances symbiosome number, bacteroid size, and nitrogen fixation in nodules and impairs mycorrhizal colonization. New Phytol. 202 (3), 886-900 (2014).
- Barraza, A., et al. Down-regulation of PvTRE1 enhances nodule biomass and bacteroid number in the common bean. New Phytol. 197 (1), 194-206 (2013).
- Estrada-Navarrete, G., et al. An autophagy-related kinase is essential for the symbiotic relationship between Phaseolus vulgaris and both rhizobia and arbuscular mycorrhizal fungi. Plant Cell. 28 (9), 2326-2341 (2016).
- Nanjareddy, K., et al. A Legume TOR Protein Kinase Regulates Rhizobium Symbiosis and Is Essential for Infection and Nodule Development. Plant Physiol. 172 (3), 2002-2020 (2016).
- Frühling, M., Schröder, G., Hohnjec, N., Pühler, A., Perlick, A. M., Küster, H. The promoter of the Vicia faba L. gene VfEnod12 encoding an early nodulin is active in cortical cells and nodule primordia of transgenic hairy roots of Vicia hirsuta as well as in the prefixing zone II of mature transgenic V. hirsuta root nodules. Plant Science. 160 (1), 67-75 (2000).
- Szabados, L., Ratet, P., Grunenberg, B., de Bruijn, F. J. Functional analysis of the Sesbania rostrata leghemoglobin glb3 gene 5′-upstream region in transgenic Lotus corniculatus and Nicotiana tabacum plants. Plant Cell Online. 2 (10), 973-986 (1990).
- Madsen, L. H., Tirichine, L., Jurkiewicz, A., Sullivan, J. T., Heckmann, A. B., Bek, A. S., Ronson, C. W., James, E. K., Stougaard, J. The molecular network governing nodule organogenesis and infection in the model legume Lotus japonicus. Nat Commun. 12, 1-10 (2010).
- Montiel, J., Arthikala, M. K., Quinto, C. Phaseolus vulgaris RbohB functions in lateral root development. Plant Signal Behav. 8 (1), 1-3 (2013).
- Nanjareddy, K., Arthikala, M. K., Gómez, B. M., Blanco, L., Lara, M. Differentially expressed genes in mycorrhized and nodulated roots of common bean are associated with defense, cell wall architecture, N metabolism, and P metabolism. PLoS ONE. 12 (8), e0182328 (2017).
- Karimi, M., Inzé, D., Depicker, A. Gateway vectors for Agrobacterium-mediated plant transformation. Trends Plant Sci. 7 (5), 193-195 (2002).
- Broughton, W. J., Dilworth, M. J. Control of leghemoglobin synthesis in snake beans. Biochem J. 125 (4), 1075-1080 (1971).
- Jefferson, R. A. Assaying chimeric genes in plants, the GUS gene fusion system. Plant Mol Biol Rep. 5 (4), 387-405 (1987).
- Cho, H. J., Farrand, S. K., Noel, G. R., Widholm, J. M. High-efficiency induction of soybean hairy roots and propagation of the soybean cyst nematode. Planta. 210 (2), 195-204 (2000).
- Deng, Y., Mao, G., Stutz, W., Yu, O. Generation of Composite Plants in Medicago truncatula used for Nodulation Assays. J. Vis. Exp. (49), e2633 (2011).
- Kumagai, H., Kouchi, H. Gene silencing by expression of hairpin RNA in Lotus japonicus roots and root nodules. Mol Plant Microbe Interact. 16 (8), 663-668 (2003).
- Okamoto, S., Yoro, E., Suzaki, T., Kawaguchi, M. Hairy Root Transformation in Lotus japonicus. Bio-protocol. 3 (12), e795 (2013).
- Jacobs, T. B., Martin, G. B. High-throughput CRISPR Vector Construction and Characterization of DNA Modifications by Generation of Tomato Hairy Roots. J. Vis. Exp. (110), e53843 (2016).
- Estrada-Navarrete, G., et al. Agrobacterium rhizogenes-transformation of the Phaseolus spp.: a tool for functional genomics. Mol Plant Microbe Interact. 19 (12), 1385-1393 (2006).

