Three-Dimensional Bone Extracellular Matrix Model for Osteosarcoma
Summary
The bone extracellular matrix (BEM) model for osteosarcoma (OS) is well established and shown here. It can be used as a suitable scaffold for mimicking primary tumor growth in vitro and providing an ideal model for studying the histologic and cytogenic heterogeneity of OS.
Abstract
Osteosarcoma (OS) is the most common and a highly aggressive primary bone tumor. It is characterized with anatomic and histologic variations along with diagnostic or prognostic difficulties. OS comprises genotypically and phenotypically heterogeneous cancer cells. Bone microenvironment elements are proved to account for tumor heterogeneity and disease progression. Bone extracellular matrix (BEM) retains the microstructural matrices and biochemical components of native extracellular matrix. This tissue-specific niche provides a favorable and long-term scaffold for OS cell seeding and proliferation. This article provides a protocol for the preparation of BEM model and its further experimental application. OS cells can grow and differentiate into multiple phenotypes consistent with the histopathological complexity of OS clinical specimens. The model also allows visualization of diverse morphologies and their association with genetic alterations and underlying regulatory mechanisms. As homologous to human OS, this BEM-OS model can be developed and applied to the pathology and clinical research of OS.
Introduction
Osteosarcoma (OS) usually occurs in actively growing areas, the metaphysis of long bones, during adolescence. More than 80% of the OS-affected sites have preference for the metaphysis of proximal tibia and proximal humerus as well as both distal and proximal femur, corresponding to the location of the growth plate1. OS comprises multiple cell subtypes with mesenchymal properties and considerable diversity in histologic features and grade. Evidences support mesenchymal stem cells (MSCs), osteoblasts committed precursors and pericytes as the cells of origin2,3,4,5. These cells can accumulate genetic or epigenetic alterations and give rise to OS under the influence of certain bone microenvironmental signals. Both intrinsic and extrinsic mechanisms result in the genomic instability and heterogeneity of OS, with multiple morphological and clinical phenotypes6,7. For individualized therapies or screening of new drugs, novel models need to be generated to against heterogeneity or other clinical disorders.
OS is an intra-osseous malignant solid tumor. The complexity and activity of surrounding microenvironment elements confer phenotypic and functional differences upon OS cells in different locations of a tumor. Bone extracellular matrix (BEM) provides a structural and biochemical scaffold for mineral deposition and bone remodeling. The organic portion of extracellular matrix (ECM) mainly consists of type I collagen secreted by osteoblastic lineage cells, while its mineralized portion is composed of calcium phosphate in the form of hydroxyapatite8. The dynamic role of ECM networks is to regulate cell adhesion, differentiation, cross-talk and tissue function maintenance9.
Demineralized BEM and ECM hydrogels have been successfully used in cell culture and can enhance cell proliferation10,11. Synthesized bone-like ECM can regulate the pool size, fate decisions and lineage progression of MSCs12,13,14. Moreover, results evidence its clinical significance to provide osteogenic activity by stimulating cellular processes during bone formation and regeneration15,16,17.
In this article, our group establishes a modified model and favorable alternative for three-dimensional long-term culture. OS cells injected into the tissue-derived BEM present a heterogeneously mesenchymal phenotype readily as compared to plastic two-dimensional cultures. BEM derived from site-specific homologous tissue show its dramatic advantage as being a native niche for OS cells in vitro and has great potential in OS theoretical and clinical research. This characterized BEM platform is simple but efficient for in vitro research and may be extended in modeling multiple cancers.
Protocol
Animal care and use are conducted according to the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH publication NO.80-23, revised in 1996) after approval from the Animal Ethics Committee of Sun Yat-sen University.
1. Bone preparation
- Obtain 4 to 6-week-old BALB/c mice (without sex-specific requirement). Euthanize a mouse aseptically by cervical dislocation and cut off fresh fibula, tibia and femur from a hindlimb with sterile surgical scissors. Peel off the epithelial tissue and then remove as much of the soft tissue as possible using scissors and tweezers.
- Rinse the leg bones with sterile 10 mM phosphate buffered saline (PBS) twice to remove blood in a 6 cm dish. Immerse the bones in 75% ethanol for 3 min, then rinse with PBS twice. The clean bones can be stored in a sterile 50 mL centrifuge tube with sterile PBS at -80 °C for months until required.
NOTE: PBS used in all the following steps has 10 mM PO43−.
2. Bone demineralization and decellularization
- Thaw the frozen bones at room temperature, and then freeze again at -80 °C for 1 h. Subject the bones to more than 2–3 freeze-thaw cycles for cell lysis and tissue breakdown.
- Incubate the bones in a sterile 50 mL centrifuge tube with 0.5 N HCl overnight at room temperature on a rocking platform or orbital shaker with gentle rocking/shaking to ensure complete and even coverage of bones.
NOTE: Make sure the bones are entirely immersed during motion in the acid and do not settle during the process. The volume of HCl solution should be more than ten times as that of the bones. - After decalcification, decant the HCl solution completely and rinse under running water for 1 h. Then, wash the bones twice for 15 min per wash with distilled water on a rocking platform or orbital shaker.
NOTE: Make sure to completely remove the solution or water between washes and after the final wash with distilled water. - Extract the lipids in the demineralized bones with a 1:1 mixture of methanol and chloroform in a 50 mL centrifuge tube wrapped with tin foil for 1 h at room temperature10.
- Then, transfer the bones into another tube of methanol wrapped with tin foil for 30 min. Remove the methanol completely and rinse with distilled water twice for 15 min with gently shaking. Decant final wash water and proceed with the following steps under sterile condition.
NOTE: During the extraction step, light must be avoided to prevent chloroform decomposition. The mixture can be stored in light-resistant container or a centrifuge tube wrapped with tin foil. Perform all treatment and wash steps under modest rotation or rocking motion. - Rinse the bones in a 6 cm dish with sterile PBS for 3 min, and then transfer the bones into a new 50 mL centrifuge tube. Add 40 mL sterile 0.05% trypsin-EDTA (TE) into the tube and incubate bones for 23 h in a CO2 incubator at 37 °C18.
- Discard the TE solution and rinse twice with sterile PBS supplemented with 90 μg/mL ampicillin and 90 μg/mL kanamycin. After decanting the final wash PBS completely, replenish with 40 mL sterile PBS. Wash thoroughly for 24 h at room temperature with gentle rocking or shaking for antibiotic soak.
NOTE: All the sterile PBS used in this and the following steps contains 90 μg/mL ampicillin and 90 μg/mL kanamycin. Overnight wash under rotation or rocking motion are performed for long periods thorough immersion with antibiotics to achieve effective sterilization of pore spaces. - Remove the PBS and transfer the bones into a fresh 50 mL centrifuge tube filled with sterile PBS. The prepared demineralized and decellularized bones are called bone extracellular matrix (BEM) and can be stored at 4 °C for 2 months until required.
3. Cell seeding and culture
- Take out the BEM from 4 °C refrigerator and immerse it in 75% ethanol for 30 s, then rinse with PBS twice. Transfer the BEM onto a clean 6-well cell culture plate. Add 2 mL complete culture medium (Dulbecco’s modified Eagle’s medium/F12 (DF12) containing 5% fetal bovine serum, 90 μg/mL ampicillin and 90 μg/mL kanamycin). Incubate the BEM overnight in a CO2 incubator at 37 °C.
- Obtain human OS cell lines (MNNG/HOS and MG-63). Suspend approximately 1.0 x 105 OS cells with 100 μL PBS containing phenol red as indicator.
NOTE: To better track and observe multi-layer cells within the three-dimensional BEM model, MNNG/HOS and MG-63 are infected with lentiviral vector expressing fluorescent mCherry and green fluorescent protein (GFP). - After the BEM is fully soaked in the medium, inject OS cells into BEM from proximal or distal epiphysis when the needle reaches the medullary cavity of BEM. Incubate the OS-BEM model for a minimum of 2 h in a humidified 5% CO2 atmosphere at 37 °C to ensure the injected cells firmly adhere to BEM.
NOTE: Pre-warm all the media used for cell culture. The incubator used for cell culture has a humidified 5% CO2 atmosphere at 37 °C. - Add 1 mL complete culture medium into the plate to completely coat the surface of the BEM culture overnight in a CO2 incubator at 37 °C.
- Gently transfer the OS-BEM model into a new well of 6-well plate with a sterile tweezer and refeed 1 mL fresh culture medium. Culture the model for 14 days in a CO2 incubator at 37 °C and refresh the culture medium according to the proliferation situation of OS cells.
- Keep monitoring medium color and cell status under the inverted fluorescence microscope during the culture process. When OS cells expand to plate, gently transfer the OS-BEM model to another new well with sterile tweezer.
NOTE: The culture medium is bright red at pH 7.4, which is the optimum pH value for most mammalian cell culture. If the medium turns into orange or even yellow, immediately refresh the medium to maintain a healthy environment for OS cells. - Transfer the OS-BEM model into a new well with tweezers and gently rinse with PBS to remove the culture medium. Then, transfer into a 15 mL centrifuge tube and fix with 10% buffered formalin for histological identification.
Representative Results
After demineralization and decellularization, BEM appears to be translucent with stronger resilience and tenacity compared to native mouse bone. A little muscle residue and the space of medullary cavity can be clearly observed (Figure 1A, B). To determine the effective decellularization of BEM, BEM is embedded in paraffin after fixation, and then sliced into 3–5 μm sections for hematoxylin-eosin (H&E) staining. The thorough removal of cell nuclei is shown by bright-field imaging. The natural porous structure and collagen network arrangement is well maintained in decellularized BEM (Figure 1C, D). Additionally, immunohistochemical (IHC) staining of predominant organic components of bone matrix, such as collagen I and collagen IV demonstrate no damage on ECM components in decellularized BEM compared to the native bone (Figure 1E). Therefore, BEM provides a suitable and promising scaffold with great biocompatibility for OS cell seeding and proliferation.
MNNG/HOS cells exhibit a highly atypical morphology with finely vacuolated cytoplasm, while MG-63 cells have fibroblast-like spindled shapes in monolayer culture (Figure 2A, B). The histological section from an OS patient displays significant cellular pleomorphism with rounded or polygonal cells, anisonucleosis and multiple mitoses (Figure 2C). To verify the viability and quality of the BEM model, both cell lines are injected into the medullary cavity of BEM and monitored via fluorescence imaging during the 14-day culture (Figure 3A, B). Histological sections with H&E staining reveal that OS cells attach to muscle residues and grow into thick piles or adhere along bone matrix and proliferate. Both periosteum and endosteum are infiltrated by the expansion of OS cells. Strikingly, the cell growth patterns of OS-BEM model differ from two-dimensional plate culture. As illustrated in Figure 3C, OS cells on the decellularized BEM show highly heterogeneous morphology similar to the cytopathologic features of an OS section. Some OS cells locating in cancellous bone and medullary cavity are spherical and partly spread out, whereas the cells resting along the periosteum and endosteum are extremely spread out into elongated cells accompanied by nuclear pleomorphism. Cell activity is determined using Ki67 immunostaining, which also shows great advantages in long-term cultures. Also, OS cells in BEM culture highly express bone matrix glycoprotein—secreted protein acidic and rich in cysteine (SPARC/osteonectin)—which is specific for osteoid matrix (Figure 3D).
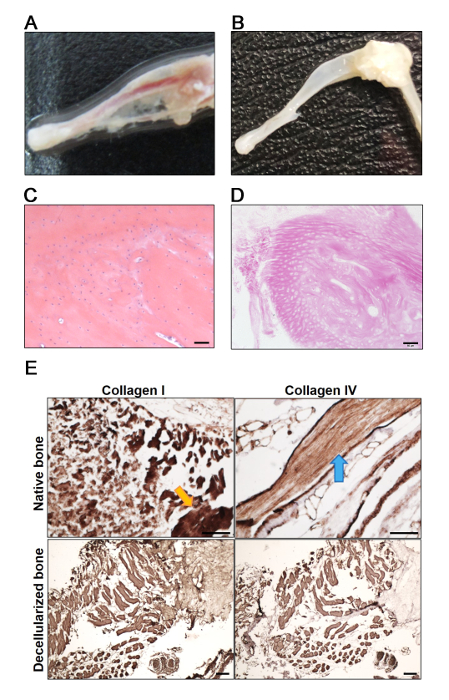
Figure 1: The structural characteristics of mouse decellularized BEM. (A, B) Overview of mouse native (A) and decellularized (B) bone. (C, D) Decellularization was assessed by H&E staining of mouse native tibia (C) and decellularized bone (D). Nuclei stained with hematoxylin could be observed in native mouse tibia, but not in the BEM. (E) IHC staining for collagen I and collagen IV to detect the main components of ECM that are preserved in mouse tibia after decellularization. Yellow arrow points out the abundant collagen I within cancellous bone and blue arrow points out the abundant collagen IV within compact bone. Scale bars = 50 μm. Please click here to view a larger version of this figure.
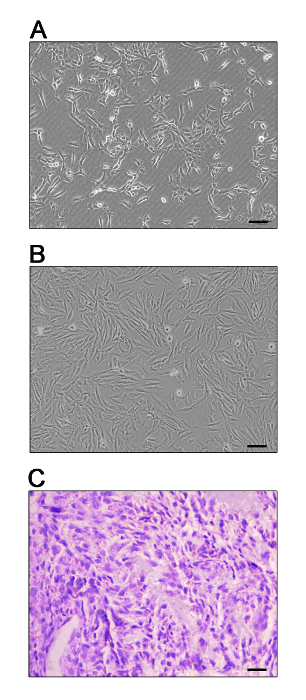
Figure 2: The cytomorphological characteristics of OS. (A, B) Human OS cell lines MNNG/HOS (A) and MG-63 (B) expanded in plastic flask culture. Scale bar = 100 μm. (C) Histopathologic section with H&E staining of OS patient. Scale bar = 50 μm. Please click here to view a larger version of this figure.
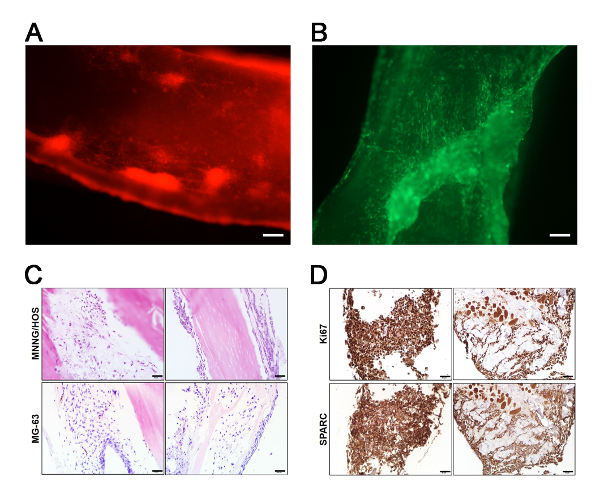
Figure 3: Characterization of OS cells in decellularized bone extracellular matrix model. (A, B) Representative mCherry expression (red) image of MNNG/HOS (A) and GFP expression (green) image of MG-63 (B) by fluorescence microscopy after seeding and culturing in BEM. Scale bar = 100 μm. (C) H&E analysis showing typical morphology of the injected MNNG/HOS and MG-63 cells after culturing in BEM. (D) IHC analysis on Ki67 and SPARC expression level of MNNG/HOS cells after culturing in BEM model for 14 days. The representative images are two sets of serial sections stained with Ki67 and SPARC. Scale bar = 50 μm. Please click here to view a larger version of this figure.
Discussion
Generally, OS can be classified as osteoblastic, chondroblastic, and fibroblastic subtypes depending on its dominant histologic component. Its prognosis is dependent not only on histologic parameters but also on its anatomic site. It may occur inside the bones (in the intramedullary or intracortical compartment), on the surfaces of bones, and in extraosseous sites19. The emergence and heterogeneity of OS can be elucidated as a conjugation of oncogenic events and an adequate microenvironmental boost, followed by increasing development and migration to distant organs20,21,22,23. Mystery during OS evolution might be deciphered with a proper model to outline clinical implications targeting the OS environment and niche.
Cultivation either on plastic dishes or in flasks in vitro can hardly recapitulate the dynamic and intricate biological microenvironment. Great strides of various pre-clinical models (e.g., mouse, zebrafish and dog) mimicking the osteosarcoma have been declared and applied to pathogenesis investigation and drug development4,24,25,26,27,28. Still, researchers have concern for experimental animals due to their discomfort and suffering during experiments. In vitro three-dimensional models like our decellularized BEM model has the advantage due to its convenience, quick operation and cost saving; it provides long-term and easy maintenance of viable cells or tissues, and is also closer to the native biological situation than plastic culture. It has been used in our research demonstrating the phenotypic heterogeneity and regulatory mechanism of OS dedifferentiation with success29.
This protocol clarified the feasibility to generate a tissue-derived BEM from mouse and might be used for tissues from human, rat and dog. The most critical steps for successful establishment and application of BEM are: (i) complete removal of cell debris; (ii) maintenance of a sterile, healthy culture condition; (iii) manual dexterity and gentle pipetting during injection, transfer and culture of OS-BEM model.
Other reported protocols generally employ pressurization or a combination of chemical and enzymatic treatments, such as Triton X-100, sodium dodecyl sulfate (SDS) and DNase/RNase solution to achieve potent decellularization30,31. The cartilage tissue that undergoes decellularization with detergents has been shown to remove ECM components including glycosaminoglycans32. To recapitulate a more intact BEM to the greatest extent, a moderate yet powerful decellularization method is performed here to avoid the dissolution and damage of key components and native architecture of the bone environment.
However, it is not to be neglected that this OS-BEM model rested in a plate without flowing medium, consequently leading to an uneven distribution of oxygen and nutrients. Vascular network and other cell subtypes that help regulating the communication and interaction of microenvironmental signals and bone homeostasis need to be taken into consideration24, 33,34,35. Hopefully, this model will be combined with other high-tech engineering techniques to shed light on OS research and guide precision medicine.
Divulgations
The authors have nothing to disclose.
Acknowledgements
The authors value the support of Liuying Chen for her administrative assistance and Long Zhao for his excellent technical assistance during the construction of bone extracellular matrix scaffolds. This study is supported by grants from the National Natural Science Foundation of China (31871413).
Materials
| 15 mL centrifuge tube | Greiner | 188271 | |
| 50 mL centrifuge tube | Greiner | 227270 | |
| 6 cm cell culture dish | Greiner | 628160 | |
| 6-well plate | Greiner | 657160 | |
| Ampicillin | Sigma-Aldrich | A9393 | |
| C57-BL/6J mouse | Sun Yat-sen University Laboratory Animal Center | ||
| CO2 incubator | SHEL LAB | SCO5A | |
| Dibasic sodium phosphate | Guangzhou Chemical Reagent Factory | BE14-GR-500G | |
| DMEM/F12 | Sigma-Aldrich | D0547 | |
| Fetal bovine serum | Hyclone | SH30084.03 | |
| Hemocytometer | BLAU | 717805 | |
| Kanamycin | Sigma-Aldrich | PHR1487 | |
| MG-63 | Chinese Academy of Science, Shanghai Cell Bank | Human osteosarcoma cell line | |
| MNNG/HOS | Chinese Academy of Science, Shanghai Cell Bank | Human osteosarcoma cell line | |
| Phenol red | Sigma-Aldrich | P4633 | A solution of phenol red is used as a pH indicator: its color exhibits a gradual transition from yellow to red over the pH range 6.6 to 8.0. |
| Potassium chloride | Sangon Biotech | A100395 | |
| Potassium Phosphate Monobasic | Sangon Biotech | A501211 | |
| Sodium chloride | Sangon Biotech | A501218 |
References
- Longhi, A., Errani, C., De Paolis, M., Mercuri, M., Bacci, G. Primary bone osteosarcoma in the pediatric age: State of the art. Cancer Treatment Reviews. 32, 423-436 (2006).
- Mohseny, A. B., et al. Osteosarcoma originates from mesenchymal stem cells in consequence of aneuploidization and genomic loss of Cdkn2. Journal of Pathology. 219, 294-305 (2009).
- Mutsaers, A. J., Walkley, C. R. Cells of origin in osteosarcoma: mesenchymal stem cells or osteoblast committed cells. Bone. 62, 56-63 (2014).
- Sato, S., et al. Mesenchymal tumors can derive from Ng2/Cspg4-Expressing pericytes with β-Catenin modulating the neoplastic phenotype. Cell Reports. 16, 917-927 (2016).
- Patane, S., et al. MET overexpression turns human primary osteoblasts into osteosarcomas. Recherche en cancérologie. 66, 4750-4757 (2006).
- Poos, K., et al. Genomic heterogeneity of osteosarcoma – shift from single candidates to functional modules. PLoS One. 10, 123082 (2015).
- Martin, J. W., Squire, J. A., Zielenska, M. The genetics of osteosarcoma. Sarcoma. 2012, 1-11 (2012).
- Alfranca, A., et al. Bone microenvironment signals in osteosarcoma development. Cellular and Molecular Life Sciences. 72, 3097-3113 (2015).
- Alford, A. I., Kozloff, K. M., Hankenson, K. D. Extracellular matrix networks in bone remodeling. The International Journal of Biochemistry & Cell Biology. 65, 20-31 (2015).
- Sawkins, M. J., et al. Hydrogels derived from demineralized and decellularized bone extracellular matrix. Acta Biomaterialia. 9, 7865-7873 (2013).
- Alom, N., Peto, H., Kirkham, G. R., Shakesheff, K. M., Bone White, L. J. Bone extracellular matrix hydrogel enhances osteogenic differentiation of C2C12 myoblasts and mouse primary calvarial cells. Journal of Biomedical Materials Research Part B-Applied Biomaterials. 106, 900-908 (2018).
- Datta, N., Holtorf, H. L., Sikavitsas, V. I., Jansen, J. A., Mikos, A. G. Effect of bone extracellular matrix synthesized in vitro on the osteoblastic differentiation of marrow stromal cells. Biomaterials. 26, 971-977 (2005).
- Rubio, R., et al. Bone environment is essential for osteosarcoma development from transformed mesenchymal stem cells. Stem Cells. 32, 1136-1148 (2014).
- Sadr, N., et al. Enhancing the biological performance of synthetic polymeric materials by decoration with engineered, decellularized extracellular matrix. Biomaterials. 33, 5085-5093 (2012).
- Gautschi, O. P., Frey, S. P., Zellweger, R. Bone morphogenetic proteins in clinical applications. Anz Journal of Surgery. 77, 626-631 (2007).
- Rochet, N., et al. Modification of gene expression induced in human osteogenic and osteosarcoma cells by culture on a biphasic calcium phosphate bone substitute. Bone. 32, 602-610 (2003).
- Spang, M. T., Christman, K. L. Extracellular matrix hydrogel therapies: in vivo applications and development. Acta Biomaterialia. 68, 1-14 (2018).
- Schenke-Layland, K., et al. Impact of decellularization of xenogeneic tissue on extracellular matrix integrity for tissue engineering of heart valves. Journal of Structural Biology. 143, 201-208 (2003).
- Klein, M. J., Siegal, G. P. Osteosarcoma: anatomic and histologic variants. American Journal of Clinical Pathology. 125, 555-581 (2006).
- Lipinski, K. A., et al. Cancer evolution and the limits of predictability in precision cancer medicine. Trends in Cancer. 2, 49-63 (2016).
- McGranahan, N., Swanton, C. Clonal heterogeneity and tumor evolution: past, present, and the future. Cell. 168, 613-628 (2017).
- Brown, H. K., Schiavone, K., Gouin, F., Heymann, M., Heymann, D. Biology of bone sarcomas and new therapeutic developments. Calcified Tissue International. 102, 174-195 (2018).
- Abarrategi, A., et al. Osteosarcoma: cells-of-origin, cancer stem cells, and targeted therapies. Stem Cells International. 2016, 1-13 (2016).
- Tsukamoto, S., et al. Mesenchymal stem cells promote tumor engraftment and metastatic colonization in rat osteosarcoma model. International Journal of Oncology. 40, 163-169 (2012).
- Rodriguez, C. J., et al. Aerosol gemcitabine: preclinical safety and in vivo antitumor activity in osteosarcoma-bearing dogs. Journal of Aerosol Medicine and Pulmonary Drug Delivery. 23, 197-206 (2010).
- Rodriguez, C. J. Using canine osteosarcoma as a model to assess efficacy of novel therapies: Can old dogs teach us new tricks. Advances in Experimental Medicine and Biology. 804, 237-256 (2014).
- Mohseny, A. B., et al. An osteosarcoma zebrafish model implicates Mmp-19 and Ets-1 as well as reduced host immune response in angiogenesis and migration. Journal of Pathology. 227, 245-253 (2012).
- Saalfrank, A., et al. A porcine model of osteosarcoma. Oncogenesis. 5, 210 (2016).
- Zhang, Y., Pan, Y., Xie, C., Zhang, Y. MiR-34a exerts as a key regulator in the dedifferentiation of osteosarcoma via PAI-1–Sox2 axis. Cell Death & Disease. 9, (2018).
- Hashimoto, Y., et al. The effect of decellularized bone/bone marrow produced by high-hydrostatic pressurization on the osteogenic differentiation of mesenchymal stem cells. Biomaterials. 32, 7060-7067 (2011).
- Benders, K. E. M., et al. Extracellular matrix scaffolds for cartilage and bone regeneration. Trends in Biotechnology. 31, 169-176 (2013).
- Grayson, W. L., et al. Effects of initial seeding density and fluid perfusion rate on formation of tissue-engineered bone. Tissue Engineering Part A. 14, 1809-1820 (2008).
- Mikulic, D., et al. Tumor angiogenesis and outcome in osteosarcoma. Pediatric Hematology and Oncology. 21, 611-619 (2004).
- Ren, K., et al. Vasculogenic mimicry: a new prognostic sign of human osteosarcoma. Human Pathology. 45, 2120-2129 (2014).
- Bonuccelli, G., et al. Role of mesenchymal stem cells in osteosarcoma and metabolic reprogramming of tumor cells. Oncotarget. 5, 7575-7588 (2014).

