Nucleofection and In Vivo Propagation of Chicken Eimeria Parasites
Summary
Here, we provided a method to achieve stable transfection of chicken Eimeria parasites by nucleofecting sporozoites or second-generation merozoites. Genetically modified eimerian parasites expressing heterologous antigenic genes could be used as vaccine delivery vehicles.
Abstract
Transfection is a technical process through which genetic material, such as DNA and double-stranded RNA, are delivered into cells to modify the gene of interest. Currently, transgenic technology is becoming an indispensable tool for the study of Eimeria, the causative agents of coccidiosis in poultry and livestock. This protocol provides a detailed description of stable transfection in eimerian parasites: purification and nucleofection of sporozoites or second-generation merozoites, and in vivo propagation of transfected parasites. Using this protocol, we achieved transfection in several species of Eimeria. Taken together, nucleofection is a useful tool to facilitate genetic manipulation in eimerian parasites.
Introduction
Eimeria spp. causes coccidiosis, which leads to substantial economic losses in the livestock and poultry industry. Although anticoccidial drugs, and to an extent, attenuated anticoccidial vaccines, have been used widely for the control of coccidiosis, there are still shortcomings regarding their drug resistance, drug residues, and the potential diffusion of vaccine strains that regain virulence1. With the development of molecular biology, transfection has become a vital tool for studying gene functions, developing novel vaccines, and screening new drug targets for Eimeria.
In the last decades, transfection has been applied successfully for apicomplexan parasites such as Plasmodium and Toxoplasma gondii2,3,4,5,6. A study using β-gal as a reporter for the transfection in E. tenella piloted such work in Eimeria7. The transfection of E. tenella8,9, E. mitis10, and E. acervulina (Zhang et al., unpublished data) was successful in chickens. Recently, we achieved transfection using merozoites of E. necatrix through nucleofection11.
Studies showed that Eimeria expressing a heterologous antigen has the potential to be developed as a recombinant vaccine, such as those expressing Campylobacter jejuni antigen A (CjaA) or chicken interleukin 2 (chIL-2)12,13. Therefore, this protocol describes a nucleofection study of Eimeria spp. in chickens. The procedure describes purification of sporozoites or merozoites, nucleofection with plasmid DNA, cloacal inoculation/intravenous injection and in vivo propagation to help researchers starting studies on transgenic Eimeria parasites.
Protocol
Chickens for all animal experiments were housed and maintained according to the China Agricultural University Institutional Animal Care and Use Committee guidelines and followed the International Guiding Principles for Biomedical Research Involving Animals. The experiments were approved by the Beijing Administration Committee of Laboratory Animals.
1. Extraction and purification of sporozoites of Eimeria spp. (e.g., E. tenella)
- Release of sporocysts
- Centrifuge 1 x 107sporulated oocysts in potassium dichromate solution (2.5%, m/v) at 2,300 x g for 5 min. Wash them with PBS (phosphate buffer solution) three times.
- Resuspend the pellets with 1 mL of PBS and transfer to a 15 mL tube. Add an equal volume of glass beads (1 mm x 1 mm diameter range) and oscillate the oocyst suspension using a vortex mixer to release the sporocysts.
- Monitor the release of sporocysts by microscopy every minute. Stop vortexing when more than 90% of oocysts are broken.
NOTE: Most of the oocysts (such as E. tenella, E. necatrix, and E. acervulina) were broken after 1 min using the vortex mixer. - Transfer the sporocyst suspension to new 1.5 mL tubes and centrifuge at 1,600 x g for 5 min.
- Resuspend the precipitate with 1 mL of 50% density gradient solution, combine in a 1.5 mL tube, and centrifuge at 10,000 x g for 1 min.
NOTE: For the density gradient composition, refer to Table 1. The density gradient is a silica-based colloidal medium, consisting of colloidal silica particles of 15-30 nm diameter (23% w/w in water), which have been coated using polyvinylpyrrolidone (PVP).
- Release of sporozoites
- Resuspend the precipitate with the excystation buffer (Table 1) and incubate in a 42 °C water bath for 40-60 min to release the sporozoites. Stop incubating when more than 90% of sporozoites are released. Then centrifuge at 600 x g for 10 min.
NOTE: Shake the tubes once every 5 minutes during excystation. - Resuspend the precipitation with 1 mL of 55% density gradient solution and centrifuge at 10,000 x g for 1 min.
- Resuspend the precipitation with 1 mL of PBS and count the sporozoites using a hemocytometer.
- Resuspend the precipitate with the excystation buffer (Table 1) and incubate in a 42 °C water bath for 40-60 min to release the sporozoites. Stop incubating when more than 90% of sporozoites are released. Then centrifuge at 600 x g for 10 min.
2. Collection and purification of merozoites of E. necatrix
NOTE: Use Arbor Acre (AA) broilers aged 7-14 d in the experiment. Coccidia-free chickens (n=3) were inoculated with 2 x 105 oocysts of E. necatrix. At 109 h post-infection, the birds were sacrificed by cervical dislocation. The intestine was removed for the collection of the 2nd generation merozoites. For different Eimeria species, there was a different time for collection of the 2nd generation merozoites: E. necatrix at 109 h, and E. tenella at 112 h post-inoculation. For the transfection of E. necatrix, merozoites are the optimal choice as the second merozoites are easy to purify.
- Collection of the second-generation merozoites of E. necatrix
- Cut the chicken intestine longitudinally, from the yolk stalk(the middle of the small intestine) to the ileocecal orifice, and wash it with PBS or HBSS (Hank's Balanced Salt Solution) gently three times in a Petri dish.
- Cut the intestine into 0.5 cm x 0.5 cm pieces and place it in a conical flask with a digestion buffer (Table 1). Place the flask on a magnetic mixer at 37 °C with a stirring bar for 30-60 min to release merozoites. After 30 min of incubation, monitor the release of merozoites by microscopic examination every 5 min.
- Purify the merozoites by filtration and centrifugation.
- Filter the suspension containing digested merozoites using four layers of gauze14, and centrifuge at 600 x g for 10 min.
- After centrifugation, discard the supernatant containing intestine debris. Transfer the precipitation with purified merozoites to 1.5 mL tubes.
- Resuspend the precipitation with 1 mL of PBS and count the merozoites using a hemocytometer.
3. Nucleofection of merozoites or sporozoites
- Preparation before nucleofection of parasites
- Prepare about 107 merozoites or sporozoites in one tube. If transfecting merozoites, prepare 3-4 tubes.
- Prepare an amount of plasmid DNA or purified PCR fragment that is greater than or equal to 10 µg.
NOTE: The plasmid used in this study contains 2 genes: enhanced yellow fluorescent protein (EYFP) and dihydrofolate reductase thymidylate synthase derived from Toxoplasma gondii (TgDHFR-TS)15. - Prepare 25 U of restriction enzyme. If plasmids are linearized, the restriction enzyme can improve the transfection efficiency. If the plasmids are circular, omit the restriction enzyme.
- Prepare 85 µL of nucleofection buffer: mix 20 µL of nucleofection buffer I and 1 mL of nucleofection buffer II, and use a part of the solution. The volume of the total buffer is 100 µL.
- Nucleofection
- Centrifuge the sporozoite or merozoite suspension at 600 x g for 10 min. Then discard the supernatant.
- In the following order, add 85 µL of nuclear transfection buffer, 10 µg of plasmid (PCR fragment), and 25 U of restriction enzyme (usually 5 µL) into the 1.5 mL tube containing sporozoites or merozoites.
- Transfer the suspension to a nuclear transfection cup. Put the cup into a nuclear transfer groove.
- Turn on the nucleofection device by using the power button and select the transfection procedure U-033. If the nucleofection device starts in the Free Program Choice mode, exit this mode by pressing the X button.
- When the program finishes, press the X button of the nucleofection device, and the screen should display OK, indicating that the nucleofection is successful.
- Add 0.5-1 mL of Dulbecco's Modified Eagle's Medium (DMEM)8 to the nucleofection cup to stop the reaction and transfer the suspension to 1.5 mL tube after mixing gently.
4. Cloacal inoculation or intravenous injection
- Inoculate the nucleofected parasites into 7-day-old chickens. Inoculate the merozoites of E. necatrix or the sporozoites of E. tenella via the cloacal route, but inoculate E. acervulina sporozoites via intravenous injection. Inoculate about 2 x 107 million sporozoites into each chicken, and incoluate merozoites 107 for each bird.
5. Propagation and FACS sorting
- Collect oocysts from feces 5-9 days post-inoculation with transfected sporozoites. Collect the oocysts on the third day after inoculating with transfected merozoites.
- Use fluorescence-activated cell sorting (FACS) and 150 mg/kg pyrimethamine8 to successively increase the transgenic population ratio.
NOTE: Use pyrimethamine by adding it directly in the feed. For more convenient use, prepare water-soluble pyrimethamine. Dissolve 1 g of pyrimethamine in 0.2 mL pf H2SO4 and 9.8 mL of N-methyl pyrrolidone (NMP), and then add 1.5 mL of this stock solution into 1 L of drinking water for birds.
6. Optional column purification
NOTE: If more pure sporozoites or merozoites are needed, there is an optional method that purifies them through a diethylaminoethyl-52 cellulose (DE-52 cellulose) column.
- Prepare the DE-52 cellulose column at least one day in advance.
- Prepare glycine eluent buffer (Table 1). Adjust the pH of glycine eluent buffer from 7.6 to 8.0 and prewarmed to 41 °C.
- Add 2.5 g of DE-52 cellulose to the column. Add water and soak overnight. Discard the supernatant.
- Add water and soak for 1 h. Discard the supernatant.
- Add 0.1 M NaOH and soak for at least 2 hours. Repeat this step.
- Replace the supernatant with water. After the cellulose completely settles to the bottom (about half an hour), repeat this step.
- Discard the supernatant, add 0.1 M HCl, and soak for at least 2 hours. Repeat this step.
- Discard the supernatant, and soak the cellulose twice with glycine eluent buffer.
- Measure and adjust pH from 7.6 to 8.0 by adding 0.1 M HCl or 0.1 M NaOH.
NOTE: In this part, the liquid has at least 5x more volume than that of DE-52 cellulose.
- Adjust the flow rate between 40-50 r/min.
- When the sedimentation of cellulose is completed, add the sporozoite or merozoite suspension to the chromatographic column. Adjust the flow rate to 30-40 r/min.
NOTE: Resuspend the sporozoite or merozoite precipitation with glycine eluent buffer before adding the chromatographic column. - Collect with glycine eluent buffer into 50 mL tubes. Stop the collection according to the results of the microscopic examination of sporozoites or merozoites during the elution process.
- Centrifuge the glycine eluent buffer collected at 600 x g for 10 min. Transfer the sporozoite or merozoite precipitation to new 1.5 mL tubes.
- Count the sporozoites or merozoites using a hemocytometer.
Representative Results
This protocol has been used to transfect eimerian parasites. In this study, the 2nd generation meronts and merozoites of E. necatrix were shown in Figure 2A and Figure 2B, while Figure 2C and Figure 2D showed the sporocysts and sporozoites of E. tenella after using the density gradient solutions. The oocysts of E. necatrix (Figure 3A) and E. tenella (Figure 3B) after nucleofecting the merozoites or sporozoites were also shown. The transfection efficiency of first-generation oocysts after nucleofecting the sporozoites is about 3-10%, in general. However, after nucleofecting the merozoites, the transfection efficiency of second-generation oocysts is only a few thousandths (Transfection efficiency of first-generation oocysts could not be calculated).
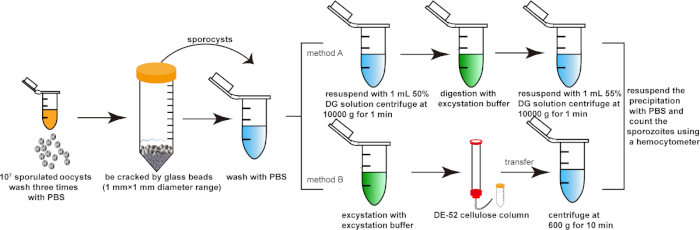
Figure 1: Purification of sporozoites. (A) Purify sporozoites through the density-gradient centrifugation with density gradient solutions. (B) Purify sporozoites through the DE-52-cellulose column. Please click here to view a larger version of this figure.
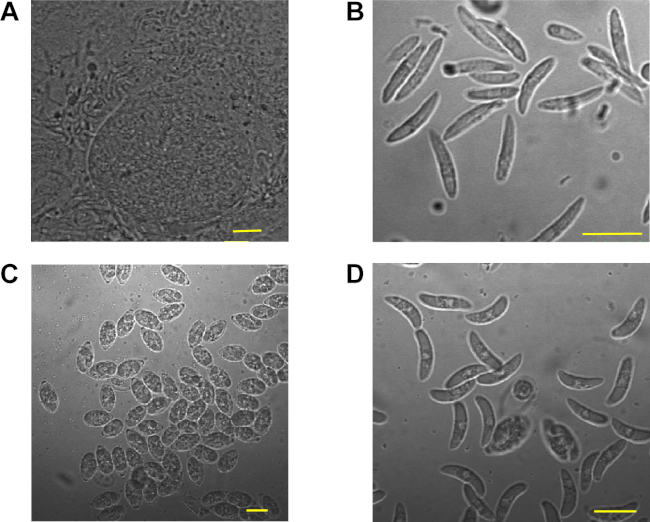
Figure 2: The 2nd generation meronts and merozoites of E. necatrix along with the sporocysts and sporozoites of E. tenella. (A) Mature 2nd generation meronts of E. necatrix. (B) The released 2nd generation merozoites of E. necatrix. (C) Sporocysts of E. tenella after purification. (D) Sporozoites E. tenella after purification. The scale bar is 10 µm. Please click here to view a larger version of this figure.
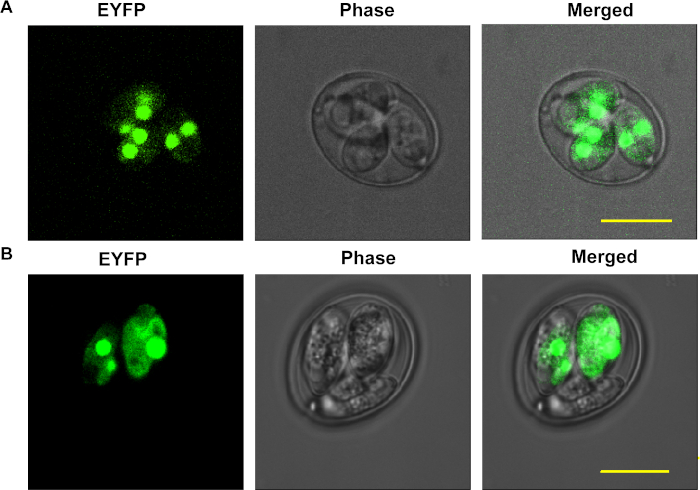
Figure 3: The oocysts obtained after infecting with the nucleofected merozoites or sporozoites. (A) The oocysts of E. necatrix after infecting with the nucleofected merozoites. (B) The oocysts of E. tenella after infecting with nucleofected sporozoites. The scale bar is 10 µm. Please click here to view a larger version of this figure.
| Buffer | Composition |
| Digestion buffer | 0.25 g trypsin, 0.5 g sodium taurodeoxycholate hydrate, 100 ml PBS or HBSS |
| 0.1 M NaOH | 2 g NaOH, 500 ml water |
| 0.1 M HCl | 4.3 ml concentrated hydrochloric acid, 500 ml water |
| Glycine eluent buffer | 0.75 g glycine, 7.9 g NaCl, 500 ml water |
| Excystation buffer | 0.75% trypsin, 10% chicken bile, PBS |
| 1 × DG gradient stock solution(Percoll) | 90% 1 × DG gradient stock solution(Percoll), 10% PBS (10 X) |
| Nucleofection buffer I | ATP-disodium 2 g, MgCl2-6H2O 1.2 g, 10 ml water |
| Nucleofection buffer II | KH2PO4 6 g, NaHCO3 0.6 g, glucose 0.2 g, 500 ml water |
| *water: distilled water | |
Table 1: Composition of Buffers.
Discussion
In the 1990s, a transfection system was developed for apicomplexan parasites, and it was used for studies on eimerian parasites. Recently, stable transfection was conducted in E. tenella8,9 and E. nieschulzi15. We achieved the stable transfection of E. necatrix by transfecting second-generation merozoites11. Inoculation of transfected sporozoites of E. acervulina through the wing vein resolved the the inability of sporozoites of E. acervulina to be inoculated via the cloacal route (Zhang et al., unpublished data). Here, we described a detailed transfection procedure to help researchers nucleofect eimerian parasites.
Previous studies showed that it was feasible to inject transfected sporozoites into the intestinal lumen of rabbits in a laparotomy for in vivo stable transfection of E. magna16 and E. intestinalis17. According to our experience, there was higher efficiency when sorting sporocysts by FACS instead of oocysts. There were also reports about transfection of unsporulated oocysts of E. maxima using a gene gun system or successful electroporation of sporulated oocysts with eGFP-Ham-OTU RNA18,19. Thus, our future studies explore transfection of oocysts or sporocysts to simplify the transfection procedures in Eimeria parasites.
The transfection success in eimerian parasites could enable genetically modified Eimeria to be used as vaccine vehicles to carry heterologous antigens, such as CjaA from C. jejuni13. Although transfection efficiency in Eimeria has been significantly improved, gene editing technology continues to have limitations in eimerian parasites. With the development of transfection in Eimeria, CRISPR/CAS9 technology in Eimeria (Hu et al., unpublished data) could lead to genetic manipulation of Eimeria.
In conclusion, this protocol provides a detailed procedure for nucleofection in chicken Eimeria. The transfection of sporozoites or merozoites is valuable for the study of gene function in Eimeria.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2017YFD0501200) and the National Natural Science Foundation of China (31572507, 31772728 and 31873007).
Materials
| ATP-disodium | Sigma | A26209 | |
| Cellulose DE-52 | Solarbio | C8350 | |
| Constant Flow Pump | SHANGHAI JINGKE INDUSTRIAL CO., LTD. | HL-2B | |
| DMEM | MACGENE | CM15019 | |
| Glass beads | Sigma | Z250473-1PAK | |
| Glucose | Sigma | No. V900116 | |
| Glycine | Biotopped | G6200 | |
| HBSS | MACGENE | CC016 | |
| KH2PO4 | Sigma | No. V900041 | |
| Low Speed Centrifuge | BEIJING ERA BEILI CENTRIFUGE CO., LTD. | DT5-2 | |
| Magnetic Mixer | SCILOGEX | MS-H280-Pro | |
| MgCl2 | Sigma | 449164 | |
| MoFlo cell sorter | BeckMan Coulter, US | 201309995 | |
| NaHCO3 | Sigma | 144-55-8 | |
| Nucleofection device | LONZA/amaxa | 90900012 (Nucleofector II) | |
| PBS | Solarbio | P1010 | |
| Percoll (DG gradient stock solution) | GE Healthcare | 17-0891-09 | |
| Sodium taurodeoxycholate hydrate | Sigma | T0875 | |
| Sorvall Legend Micro 17 Microcentrifuge | ThermoFisher Scientific | 75002430 | |
| The composition of DMEM: 4.5 g/L glucose with sodium pyruvate, L-glutamine, and 25 mM HEPES. | |||
| Trypsin | Solarbio | T8150 | |
| Vortex Mixer | Beijing North TZ-Biotech Develop.co. | HQ-60-II | |
| Water Bath Thermostat | Grant Instruments (Cambridge), Ltd. | GD120,GM0815010 |
References
- Suo, X., et al. The efficacy and economic benefits of Supercox, a live anticoccidial vaccine in a commercial trial in broiler chickens in China. Veterinary Parasitology. 142 (1-2), 63-70 (2006).
- Kim, K., Soldati, D., Boothroyd, J. C. Gene replacement in Toxoplasma gondii with chloramphenicol acetyltransferase as selectable marker. Science. 262 (5135), 911-914 (1993).
- Sibley, L. D., Messina, M., Niesman, I. R. Stable DNA transformation in the obligate intracellular parasite Toxoplasma gondii by complementation of tryptophan auxotrophy. Proceedings of the National Academy of Sciences of the United States of America. 91 (12), 5508-5512 (1994).
- Donald, R. G., Roos, D. S. Stable molecular transformation of Toxoplasma gondii: a selectable dihydrofolate reductase-thymidylate synthase marker based on drug-resistance mutations in malaria. Proceedings of the National Academy of Sciences of the United States of America. 90 (24), 11703-11707 (1993).
- Soldati, D., Boothroyd, J. C. Transient transfection and expression in the obligate intracellular parasite Toxoplasma gondii. Science. 260 (5106), 349-352 (1993).
- Goonewardene, R., Daily, J., et al. Transfection of the malaria parasite and expression of firefly luciferase. Proceedings of the National Academy of Sciences of the United States of America. 90 (11), 5234-5236 (1993).
- Kelleher, M., Tomley, F. M. Transient expression of beta-galactosidase in differentiating sporozoites of Eimeria tenella. Molecular and Biochemical Parasitology. 97 (1-2), 21-31 (1998).
- Clark, J. D., et al. A toolbox facilitating stable transfection of Eimeria species. Molecular and Biochemical Parasitology. 162 (1), 77-86 (2008).
- Yan, W. C., et al. Stable transfection of Eimeria tenella: Constitutive expression of the YFP-YFP molecule throughout the life cycle. International Journal for Parasitology. 39 (1), 109-117 (2009).
- Qin, M., et al. Transfection of Eimeria mitis with Yellow Fluorescent Protein as Reporter and the Endogenous Development of the Transgenic Parasite. PloS One. 9 (12), e114188 (2014).
- Duan, C. H., et al. Stable transfection of Eimeria necatrix through nucleofection of second generation merozoites. Molecular and Biochemical Parasitology. , 1-5 (2019).
- Li, Z. R., et al. Transgenic Eimeria mitis expressing chicken interleukin 2 stimulated higher cellular immune response in chickens compared with the wild-type parasites. Frontiers in Microbiology. 6, 533 (2015).
- Clark, J. D., et al. Eimeria species parasites as novel vaccine delivery vectors: anti-Campylobacter jejuni protective immunity induced by Eimeria tenella-delivered CjaA. Vaccine. 30 (16), 2683-2688 (2012).
- Eckert, J., Braun, R., Shirley, M. W., Coudert, P. Eimeria species and strains of chickens. Biotechnology: Guidelines on techniques in coccidiosis research. Part. I: Eimeria and Isospora, 1-24 (1995).
- Kurth, M., Entzeroth, R. Reporter gene expression in cell culture stages and oocysts of Eimeria nieschulzi (Coccidia, Apicomplexa). Parasitology Research. 104 (2), 303-310 (2009).
- Tao, G. R., et al. Transgenic Eimeria magna Perard, 1925 Displays Similar Parasitological Properties to the Wild-type Strain and Induces an Exogenous Protein-Specific Immune Response in Rabbits (Oryctolagus cuniculus L.). Frontiers in Immunology. 8, 2 (2017).
- Shi, T. Y., et al. Stable Transfection of Eimeria intestinalis and Investigation of Its Life Cycle, Reproduction and Immunogenicity. Frontiers in Microbiology. 7, 807 (2016).
- Wang, P., et al. A novel telomerase-interacting OTU protein of Eimeria tenella and its telomerase-regulating activity. Acta Biochimica et Biophysica Sinica. 49 (8), 744-745 (2017).
- Li, J. N., Zou, J., Yin, G. W., Liu, X. Y., Suo, X. Plasmid DNA could be delivered into Eimeria maxima unsporulated oocyst with gene gun system. Acta Polytechnica Hungarica. 60 (4), 431-440 (2012).

