A High-Throughput Image-Guided Stereotactic Neuronavigation and Focused Ultrasound System for Blood-Brain Barrier Opening in Rodents
Summary
The blood-brain barrier (BBB) can be temporarily disrupted with microbubble-mediated focused ultrasound (FUS). Here, we describe a step-by-step protocol for high-throughput BBB opening in vivo using a modular FUS system accessible for non-ultrasound experts.
Abstract
The blood-brain barrier (BBB) has been a major hurdle for the treatment of various brain diseases. Endothelial cells, connected by tight junctions, form a physiological barrier preventing large molecules (>500 Da) from entering the brain tissue. Microbubble-mediated focused ultrasound (FUS) can be used to induce a transient local BBB opening, allowing larger drugs to enter the brain parenchyma.
In addition to large-scale clinical devices for clinical translation, preclinical research for therapy response assessment of drug candidates requires dedicated small animal ultrasound setups for targeted BBB opening. Preferably, these systems allow high-throughput workflows with both high-spatial precision as well as integrated cavitation monitoring, while still being cost effective in both initial investment and running costs.
Here, we present a bioluminescence and X-ray guided stereotactic small animal FUS system that is based on commercially available components and fulfills the aforementioned requirements. A particular emphasis has been placed on a high degree of automation facilitating the challenges typically encountered in high-volume preclinical drug evaluation studies. Examples of these challenges are the need for standardization in order to ensure data reproducibility, reduce intra-group variability, reduce sample size and thus comply with ethical requirements and decrease unnecessary workload. The proposed BBB system has been validated in the scope of BBB opening facilitated drug delivery trials on patient-derived xenograft models of glioblastoma multiforme and diffuse midline glioma.
Introduction
The blood-brain barrier (BBB) is a major obstacle for drug delivery into the brain parenchyma. Most therapeutic drugs that have been developed do not cross the BBB due to their physicochemical parameters (e.g., lipophilicity, molecular weight, hydrogen bond acceptors and donors) or are not retained due to their affinity for efflux transporters in the brain1,2. The small group of drugs that can cross the BBB are typically small lipophilic molecules, which are only effective in a limited number of brain diseases1,2. As a consequence, for the majority of brain diseases, pharmacological treatment options are limited and new drug delivery strategies are needed3,4.
Therapeutic ultrasound is an emerging technique that can be used for different neurological applications such as BBB disruption (BBBD), neuromodulation, and ablation4,5,6,7. In order to achieve a BBB opening with an extracorporeal ultrasound emitter through the cranium, focused ultrasound (FUS) is combined with microbubbles. Microbubble-mediated FUS results in increased bioavailability of drugs in the brain parenchyma5,8,9. In the presence of sound waves, microbubbles start to oscillate initiating transcytosis and disruption of the tight junctions between the endothelial cells of the BBB, enabling paracellular transport of larger molecules10. Previous studies confirmed the correlation between the intensity of the acoustic emission and the biological impact on the BBB opening11,12,13,14. FUS in combination with microbubbles has already been used in clinical trials for the treatment of glioblastoma using temozolomide or liposomal doxorubicin as the chemotherapeutic agent, or for therapy of Alzheimer’s disease and amyotrophic lateral sclerosis5,9,15,16.
Since ultrasound mediated BBB opening results in entirely new possibilities for pharmacotherapy, preclinical research for clinical translation is needed to assess the therapy response of selected drug candidates. This typically requires a high-throughput workflow with both high-spatial precision and preferably an integrated cavitation detection for monitoring of targeted BBB opening with a high reproducibility. If possible, these systems need to be cost effective in both initial investment and running costs in order to be scalable according to the study size. Most preclinical FUS systems are combined with MRI for image-guidance and treatment planning15,17,18,19. Although MRI gives detailed information about the tumor anatomy and volume, it is an expensive technique, which is generally performed by trained/skilled operators. In addition, high-resolution MRI may not always be available for researchers in preclinical facilities and requires long scanning times per animal, making it less suitable for high-throughput pharmacological studies. Noteworthy is that, for preclinical research in the field of neuro-oncology, in particular infiltrative tumor models, the possibility to visualize and target the tumor is essential for treatment success20. Currently, this requirement is only fulfilled by MRI or by tumors transduced with a photoprotein, enabling visualization with bioluminescence imaging (BLI) in combination with administration of the photoprotein substrate.
MRI-guided FUS systems often use a water bath to ensure ultrasound wave propagation for transcranial applications, whereby the head of the animal is partly submerged in the water, the so called ‘’bottom-up’’ systems15,17,18. While these designs work generally well in smaller animal studies, they are a compromise between animal preparation times, portability and realistically maintainable hygienic standards during usage. As an alternative to MRI, other guidance methods for stereotactic navigation encompass the use of a rodent anatomical atlas21,22,23, laser pointer assisted visual sighting24, pinhole-assisted mechanical scanning device25, or BLI26. Most of these designs are “top-down” systems in which the transducer is placed on top of the animal’s head, with the animal in a natural position. The ‘’top-down’’ workflow consists either of a water bath22,25,26 or a water-filled cone21,24. The benefit of using a transducer inside a closed cone is the more compact footprint, shorter setup time and straight-forward decontamination possibilities simplifying the entire workflow.
The interaction of the acoustic field with the microbubbles is pressure dependent and ranges from low-amplitude oscillations (referred to as stable cavitation) to transient bubble collapse (referred to as inertial cavitation)27,28. There is an established consensus that ultrasound-BBBD requires an acoustic pressure well above the stable cavitation threshold to achieve successful BBBD, but below the inertial cavitation threshold, which is generally associated with vascular/neuronal damage29. The most common form of monitoring and control is the analysis of the (back-)scattered acoustic signal using passive cavitation detection (PCD), as suggested by McDannold et al.12. PCD relies on the analysis of the Fourier spectra of microbubble emission signals, in which the strength and appearance of stable cavitation hallmarks (harmonics, subharmonics, and ultraharmonics) and inertial cavitation markers (broadband response) can be measured in real-time.
A “one size fits all” PCD-analysis for precise pressure control is complicated due to the polydispersity of the microbubble formulation (the oscillation amplitude depends strongly on the bubble diameter), the differences in bubble shell properties between brands, and the acoustic oscillation, which depends strongly on frequency and pressure30,31,32. As a consequence, many different PCD detection protocols have been suggested, which have been adapted to particular combinations of all these parameters and have been used in various application scenarios (ranging from in vitro experimentation over small animal protocols to PCD for clinical usage) for robust cavitation detection and even for retroactive feedback control of the pressure11,14,30,31,32,33,34,35. The PCD protocol employed in the scope of this study is derived directly from McDannold et al.12 and monitors the harmonic emission for the presence of stable cavitation and broadband noise for inertial cavitation detection.
We have developed an image-guided neuronavigation FUS system for transient opening of the BBB to increase drug delivery into the brain parenchyma. The system is based on commercially available components and can be easily adapted to several different imaging modalities, depending on the available imaging techniques in the animal facility. Since we require a high-throughput workflow, we have opted to use X-ray and BLI for image-guidance and treatment planning. Tumor cells transduced with a photoprotein (e.g., luciferase) are suitable for BLI imaging20. After administration of the photoprotein substrate, tumor cells can be monitored in vivo and tumor growth and location can be determined20,36. BLI is a low-cost imaging modality, it enables to follow the tumor growth over time, it has fast scanning times and it correlates well with tumor growth measured with MRI36,37. We have opted to replace the water bath with a water-filled cone attached to the transducer to enable flexibility to freely move the platform on which the rodent is mounted8,24. The design is based on a detachable platform equipped with integration of (I) small-animal stereotactic platform (II) fiducial markers with both X-ray and optical-image compatibility (III) rapid-detachable anesthesia mask, and (IV) integrated temperature regulated animal heating system. After the initial induction of anesthesia, the animal is mounted in a precise position on the platform where it remains during the entire procedure. Consequently, the entire platform passes all stations of the workflow of the entire intervention, while maintaining an accurate and reproducible positioning and sustained anesthesia. The control software allows the automatic detection of the fiducial markers and automatically registers all types of images and image modalities (i.e., micro-CT, X-ray, BLI and fluorescence imaging) into the frame of reference of the stereotactic platform. With help of an automatic calibration procedure, the focal length of the ultrasound transducer is precisely known within, which enables the automatic fusion of interventional planning, acoustic delivery and follow-up imaging analysis. As shown in Figure 1 and Figure 2, this setup provides a high degree of flexibility to design dedicated experimental workflows and allows interleaved handling of the animal at different stations, which in-turn facilitates high-throughput experiments. We have used this technique for successful drug delivery in mouse xenografts of high-grade glioma such as diffuse midline glioma.
Protocol
All in vivo experiments were approved by the Dutch ethical committee (license permit number AVD114002017841) and the Animal Welfare Body of the Vrije Universiteit Amsterdam, the Netherlands. The investigators were trained in the basics of the FUS system in order to minimize the discomfort of the animals.
1. Focused ultrasound system
NOTE: The described setup is an inhouse built BBB disruption system based on commercially available components and includes a 3D-printed custom-made cone and detachable stereotactic platform. The system is designed modular, which facilitates modifications according to available equipment and specific use. The protocol describes the procedure for the sonoporation of a larger area in the pontine region of the mouse brain. By adjusting the target location, different parts of the brain could be targeted. In this study a 1 MHz mono-element transducer with a focal length of 75 mm, an aperture of 60 mm and a focal area of 1.5 x 1.5 x 5 mm (FWHM of peak pressure) was used. The focal plane of the transducer is positioned through the cranium of the animal in the horizontal plane intersecting with the ear bars.
- Select an appropriate transducer for BBB opening in rodents.
NOTE: Based on the properties of the microbubbles and the employed frequency, the acoustic settings, in particular the mechanical index (MI), are subject to change13,38. - Place the transducer in the 3D-printed cone.
- Employ an acoustically transparent mylar membrane at the bottom-end of the cone to achieve acoustic coupling of the beam propagation path, and fill the cone with degassed water.
- Mount the transducer above the animal on a motorized linear stage as shown in Figure 1 allowing automatic vertical positioning of the transducer.
- Design a detachable stereotactic platform based on the requirements of the study, which includes temperature regulated heating, bite and ear bars, anesthesia and multi-modality fiducial markers, as shown in Figure 1 and Figure 2. The mounting of the stereotactic platform consists of a 2D linear stage system, which allows precise automatic positioning (< 0.1 mm) of the animal under the beam.
- Connect the transducer to the acoustic emission chain shown in Figure 1 consisting of a transducer, a function generator and a power amplifier.
- Devise an image-processing pipeline to detect the multi-modality fiducial markers that allows precise sonoporation targeting of the brain area of interest and collection of the cavitation data detected by the needle hydrophone.
- Calibrate the system and determine the focus point of the transducer in correspondence to vertical positioning of the animal on the stereotactic platform.
2. Animal preparation
NOTE: The following protocol is specified for mice but can be adapted for rats. For these experiments female athymic nude Foxn1-/- mice (6-8 week old) were used.
- Allow the animal to acclimatize for at least one week in the animal facility and weigh the animal regularly.
- Administer buprenorphine (0.05 mg/kg) via subcutaneous (s.c.) injection 30 min prior to FUS treatment to start analgesic treatment.
- Anesthetize the animal with 3% isoflurane, 2 L/min O2 and verify that the animal is deeply anesthetized. Keep the animals anesthetized during the whole procedure and monitor the breathing frequency and heart rate to adjust the concentration of isoflurane as required.
- Apply eye ointment to prevent dry eyes and avoid possible injury.
- Remove hair on the top of the head with a razor and depilatory cream and wash afterwards with water to remove any residues to avoid irritation to the skin.
- For experiments with BLI tumor models, inject 150 µL of D-luciferin (30 mg/mL) intraperitoneal (i.p.) with a 29 G insulin syringe for BLI image-guidance.
- Insert a 26-30 G tail vein catheter and flush the catheter and vein with a small volume of heparin solution (5 UI/mL). Fill the catheter with heparin solution to avoid blood clotting.
NOTE: Good catheterization is seen when there is a reflux of blood into the catheter. Avoid air bubbles in the catheter to prevent emboli. To avoid excessive injection pressure, make sure the length of the catheter is as short as possible. - Place the animal on the temperature regulated stereotactic platform to avoid hypothermia.
NOTE: Hypothermia reduces blood circulation, which can affect the injection/circulation of microbubbles and the pharmacokinetics of the drugs39. - Immobilize and fix the head of the animal on the stereotactic platform using ear bars and a bite bar. Fixate the body with a strap and tape the tail of the animal to the platform.
3. In vivo image-guided focused ultrasound
NOTE: For this protocol a 1 MHz mono-element transducer with a tone-burst pulse with a 10 ms duration, a MI of 0.4 and a pulse repetition frequency of 1.6 Hz with 40 cycles for 240 s was used. The protocol is optimized for microbubbles stabilized by phospholipids containing sulphur hexafluoride (SF6) as an innocuous gas, whereby the mean bubble diameter is 2.5 μm and more than 90% of the bubbles are smaller than 8 μm.
- Place the stereotactic platform with the mounted animal in the imaging modality (e.g., BLI or X-ray) and take image(s) of the animal.
- Use the multi-modality fiducial markers in combination with the image-processing pipeline to mark the position of the animal according to the focus point of the transducer.
- Determine the target area by placing a brain outline over the acquired X-ray image or using BLI images to determine the center of the tumor (Figure 2). The position of specific parts of the brain are specified in the Paxinos Brain Atlas40 using the skull markings bregma and lambda as reference points. For example the pons is located x=-1.0, y=-0.8 and z=-4.5 from lambda.
- Shield the animal’s nostrils and mouth with adhesive tape to prevent ultrasound gel interfering with breathing.
- Apply ultrasound gel on top of the animal’s head.
- Retract the skin of the animals’ neck, lubricate the needle hydrophone with ultrasound gel and place the needle hydrophone in the direct vicinity of the occipital bone.
- Guide the transducer to the correct position using the image-processing pipeline and the focus point.
- Apply the preconfigured settings to all attached devices and target the brain region of interest.
NOTE: Depending on the research question, tumor or brain regions can be sonoporated as a single focal point or as volumetric shape, as shown in Figure 2. - Activate microbubbles as described by the manufacturer. Inject one bolus of 120 µL (5.4 µg) of microbubbles.
- Flush the tail vein catheter with saline to check the opening of the catheter.
- Inject the microbubbles and start the insonation.
- Record microbubble cavitation with the needle hydrophone.
- Administer an intravascular contrast agent or drug after sonoporation. The dose, timing and planning are dependent on the purpose of the study and the drug.
NOTE: Evans blue is a common color agent to assess BBB opening41. - Monitor the animal until the predetermined time point or before the humane endpoint.
4. Analysis of microbubble cavitation
NOTE: Here the applied procedure is described, which is suitable for in vivo experimentation for SF6-phospholipid microbubbles with an average diameter of 2.5 µm (80% of the bubbles below 8 µm) excited with a burst-tone pulse of 10 ms duration at a frequency of 1 MHz, as originally suggested by McDannold et al.12.
- Fourier-transform the recorded PCD signal from the time-domain into the frequency domain.
- Integrate the resulting spectral power for stable cavitation detection around the 2nd and 3rd harmonic (± 50 kHz), as shown in Figure 3 (green box at 2 and 3 MHz).
- Integrate the spectral power for inertial cavitation detection, between principal frequency, the 2nd, 3rd harmonic, the 1st and 2nd ultraharmonic and the first sub-harmonic (± 150 kHz), as shown in Figure 3 (red boxes).
- Integrate the spectral power around the principle frequency (1 MHz ± 50 kHz) for the normalization of both previously obtained PCD signals.
NOTE: The PCD signal, for SF6-phospholipid microbubbles in vivo experiments at 1 MHz, does not display ultraharmonics or subharmonics before inertial cavitation sets in, as shown in Figure 3.
Representative Results
The described FUS system (Figure 1 and Figure 2) and the associated workflow have been used in over a 100 animals and produced reproducible data on both healthy and tumor bearing mice. Based on the recorded cavitation and the spectral density at the harmonics at the peak moment of the microbubble bolus injection, the spectral power of each frequency can be calculated using the Fourier analysis as explained in step 4 of the Protocol. Based on the acoustic protocol (1 MHz, 10 ms pulse duration) with a MI of 0.4 in combination with microbubbles, the normalized integrated power spectrum at the 2nd and 3rd harmonics normalized the integrated power spectrum of the excitation frequency observed in Figure 3. This provided a very sensitive and reliable means of stable cavitation detection, in comparison to no detection of subharmonics when no microbubbles were injected or the observation of inertial cavitation when a MI of 0.6 was applied. In case of inertial cavitation, an increased broad-band noise floor of up to 25 dB was detected as well as the appearance of ultra-harmonics and subharmonics. Although an acoustic pressure of an MI of 0.4 and 0.6 resulted in no macroscopic damage, microscopic damage was evidenced histologically at a MI of 0.6, as shown in Figure 4. A further increase of the pressure amplitude up to a MI of 0.8 resulted in a macroscopic brain hemorrhage of larger vessels and wide-spread tissue lysis with the extravasation of erythrocytes. The histological findings corresponded to the acoustic data from the passive cavitation sensor, as shown in Figure 3, confirming the damaging properties of inertial cavitation of the brain tissue. As a consequence, a MI of 0.4 was chosen as the safe pressure amplitude that provided very reproducible BBB-opening, while providing a safe margin to the inertial cavitation regime, as observed before11.
Intravenous Evans blue was injected to validate the opening of the BBB in the pontine region. The strong albumin-binding of Evans blue leads to a large molecule of more than 66 kDa42. At the level of the pons and partly the cerebellum, extravasation of Evans blue-conjugated albumin was observed in the mouse treated with FUS and microbubbles in contrast to the mouse without microbubbles (Figure 5). This emphasizes the precise targeting of the region of interest based on image-guided stereotactic navigation with the in-house build FUS system and the described protocol.

Figure 1: Focused ultrasound setup.
(A) Schematic representation of the focused ultrasound set up. (B) Picture of the focused ultrasound setup. The system consists of a top-down mounted transducer on a 1D linear stage over a second 2D stage for automatic 3D positioning. The transducer is built in a water filled beam-cone, closed at the bottom with an acoustically transparent mylar membrane, which conducts the sound to the cranium of the animal. The transducer is connected to a power amplifier, which is in-turn connected to an arbitrary waveform generator (AWG) for signal generation. For cavitation detection a detachable hydrophone in combination with a low-noise voltage amplifier is used. The hydrophone is placed in the direct vicinity of the occipital bone. The external hydrophone has a 2 mm active surface and is acoustically coupled with ultrasound gel. Both the high-voltage signal of the excitation pulse as well as the recorded cavitation signal are digitalized by a standard 200 MHz oscilloscope and relayed to a control computer (not shown) for on-the-fly processing and real-time control. Please click here to view a larger version of this figure.
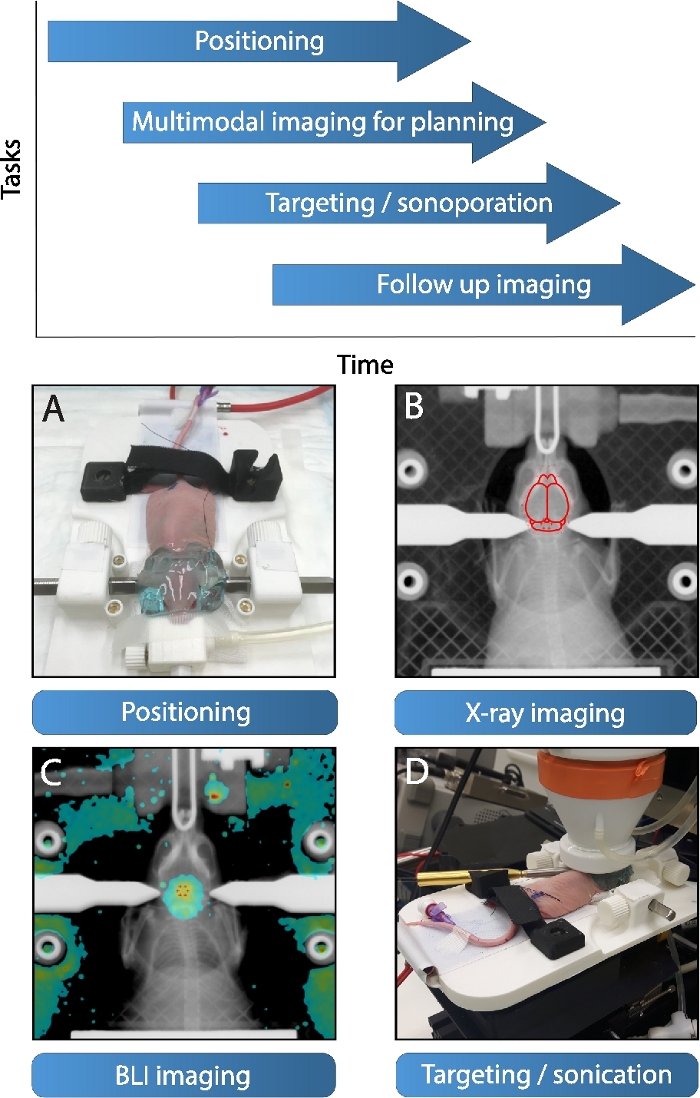
Figure 2: Focused ultrasound workflow.
The proposed workflow of the focused ultrasound system starts with (A) the initial positioning of animal on a detachable stereotactic platform, note the application of the acoustic coupling gel (applied post BLI/X-ray). Simultaneously multimodal imaging can be conducted for targeting. (B) At first X-ray imaging is a possibility, whereas a region of interest can be targeted with the help of an outline of the brain (which in turn is referenced to the mouse brain atlas40, adapted to the size and posture of the skull). (C) Alternatively, a BLI image of a luciferase transfected diffuse midline glioma tumor overlaid on an X-ray maximum intensity projection can be applied for targeting. (D) Subsequently, the stereotactic platform is mounted with the animal in therapy position with both hydrophone and transducer attached. The transducer automatically drives in therapy position and sonicates the chosen trajectory post bolus injection. The system is optimized for high-throughput experiments, whereby multiple platforms allow interleaved work, as shown on top. Please click here to view a larger version of this figure.
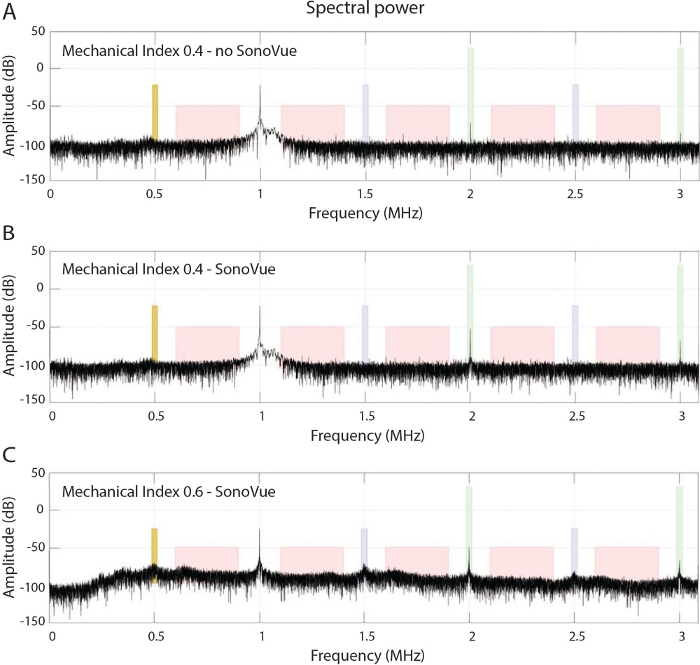
Figure 3: Cavitation monitoring.
(A) Frequency spectrum of an in vivo experiment in the absence of microbubble administation at a MI of 0.4 at 1 MHz. (B) Shown is the corresponding spectrum at peak-bolus after injection of microbubbles. Note the increase of the higher harmonics, which is indicative for stable cavitation of the microbubbles. (C) Corresponding spectrum observed at a higher MI of 0.6 in combination with microbubble injection, within the transition band to the onset of inertial cavitation, leading to an increase in noise floor up to 25 dB and the appearance of ultraharmonics and subharmonics. Please click here to view a larger version of this figure.
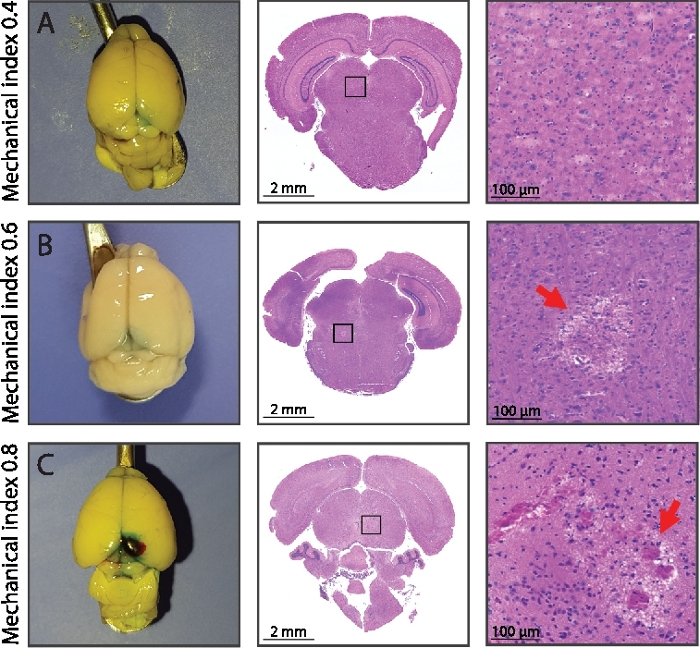
Figure 4: BBB opening and associated histology.
(A) Stable cavitation using an MI of 0.4 evidenced an intact brain parenchyma in both white light macroscopy and HE stained microscopy. (B) After a MI of 0.6 first signs of local irreversible tissue damage of the brain parenchyma is becoming apparent in the HE stained histological data. (C) For even higher mechanical pressure of MI 0.8, macroscopic hemorrhaging is apparent as well as wide-spread tissue lysis of the brain parenchyma and the extravasation of erythrocytes due to micro-hemorrhaging. The blue hue in the white light macroscopy is indicative for the extravasation of the co-injected intra-vascular contrast agent Evans blue indicating BBB opening (see Figure 5 for a sagittal view). Please click here to view a larger version of this figure.
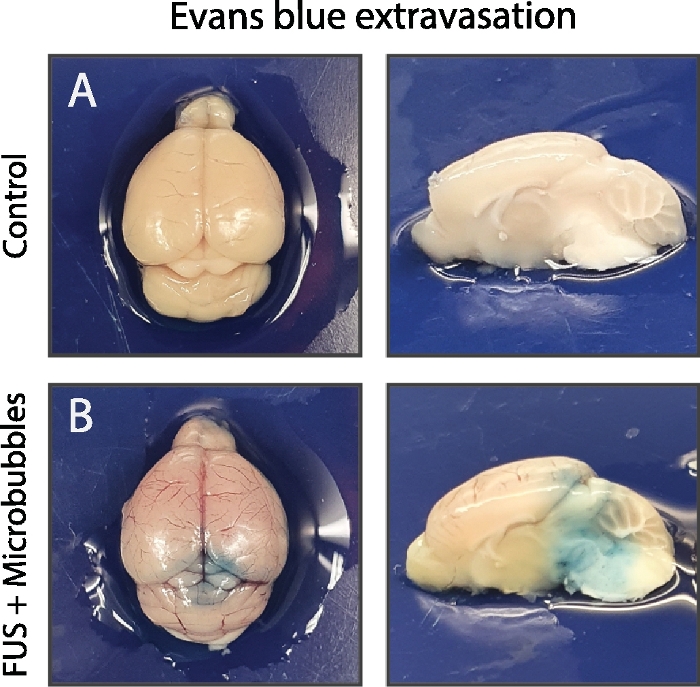
Figure 5: Validation of BBB opening.
Demonstration of successful BBB opening in the stable cavitation regime (B) compared to the control (A), no microbubbles injected. In this case Evans blue has been used as an intravascular contrast agent. The strong albumin-binding of Evans blue leads to a large molecule of more than 66 kDa. As a consequence, evidence of the Evans blue extravasation is indicative for paracellular transport across the BBB due to a (partial) opening of the tight junctions. Please click here to view a larger version of this figure.
Discussion
In this study, we developed a cost-effective image guided based FUS system for transient BBB disruption for increased drug delivery into the brain parenchyma. The system was largely built with commercially available components and in conjunction with X-ray and BLI. The modularity of the proposed design allows the use of several imaging modalities for planning and assessment in high-throughput workflows. The system can be combined with more comprehensive high-resolution 3D imaging modalities, for example high-resolution MRI or micro-CT, while for the bulk of the study 2D imaging modalities such as 2D X-ray and/or BLI are used. 2D X-ray and/or BLI are both considerably more cost effective as well as ideal for high-volume studies due to their respective short acquisition times. The transducer described here is well suited to produce BBBD in larger areas (on the scale of a mouse brain) in deeper parts of the brain (f number of 1.25). We have used the system for diffusely growing tumors in the pontine region43,44. For these regions a larger volume needs to be sonoporated that encompasses the entire tumor region in the pons. The modular system can easily be adjusted for other types of brain tumors in more supratentorial parts of the brain. In order to decide on the transducer type one should hold into account the f-number, focal length and frequency.
The overall design proposes thereby two refinements compared to previously suggested designs. (I) Frequently a water bath is used for ultrasound wave transmission of therapeutic systems. For transcranial applications in small animals this type of design results in larger and inverted setups, whereby the animal is partially submerged11,22,25. While these designs work generally very well in the scope of smaller animal studies, they are a compromise with respect to setup times, portability and realistically maintainable hygienic standards during usage. In particular the latter is of considerable importance in the scope studies encompassing immunocompromised animals and thus strict hygienic standards. As a consequence, in order to design a system with a more compact footprint, shorter setup time, easy decontamination possibilities and a natural position of the animal during the entire workflow, a “top-down” design was chosen. (II) The second design choice that differs from several previously described designs was to omit the direct integration of the acoustic delivery system into a medical imaging system such as an MRI or a micro-CT15,17,18,19,45. While fully integrated systems are ideal for longitudinal pharmacokinetic studies or explorative research on a limited number of animals, such setups are generally less suitable for high-volume pharmacological studies due to considerably increased complexity, high running-costs and need for trained/skilled operators. Furthermore, such systems are generally limited to only one imaging modality. As a consequence, the proposed design here relies on a modular detachable stereotactic platform, which is compatible with several imaging modalities (micro-CT, small animal MRI, a variety of BLI/fluorescence cameras, these with or without integrated X-ray imaging) and provides also multi-modality fiducial markers for automatic fusion of all image data in a common frame of reference for both interventional planning and the follow-up post BBB opening.
With respect to practical considerations, the most critical point of failure in the procedure is the stability of the microbubbles due to their limited lifetime and their fragile nature. We would like to emphasize that the following discussion concerns microbubbles stabilized by phospholipids and containing sulphur hexafluoride (SF6) as an innocuous gas46,47, while other microbubble formulations will generally display different properties.
Timing before microbubble injection: The advertised lifespan of commercially available microbubbles after re-hydration is between as 3 and 4 hours. While this is suitable for diagnostic ultrasound applications, it should be noted that during this entire period the microbubbles continuously lose gas and consequently the mean bubble diameter is subject to a continuous downward-drift from the initial average size of 2.5 μm. For therapeutic applications such as ultrasound-mediated BBBD this implies much stricter timing-imperatives, since the oscillation amplitude of stable cavitation (at a given frequency and pressure) and the onset-threshold of inertial cavitation are as a direct consequence also subject to a continuous drift. In our experience, we have observed that microbubbles are best used within 30 minutes after rehydration in order to obtain reproducible results, similar to previous reportings48.
Timing after microbubble injection: In larger primates, commercially available SF6-phospholipid microbubbles display a blood-plasma elimination half-life of about 6 minutes and more than 80% of the administered gas is exhaled via the lungs after only 11 minutes48. In small mammals such as mice and rats the blood-plasma elimination half-life of this type of microbubbles in vivo is with 90-120 seconds considerably shorter due to the higher heart rate20. As a consequence, the rapid dynamic of the microbubble concentration directly after bolus injection and the fast subsequent plasma elimination combined with the continuous gas volume loss of the bubbles imposes strict timing requirements on the sonication/injection protocol in order to obtain reproducible results within the short duration of 3-4 minutes post-injection. Longer procedures or more extensive volumes of BBBD require preferably a continuous administration of microbubbles. However, such an approach is complicated by the buoyancy of the bubbles in both the syringe and the feeding-system and also introduces a considerably increased dead volume by the required infusion tubing. In our experience the simpler solution of splitting the total injection volume into 2 to 3 smaller sub-doses provided a robust and reproducible results.
In addition, microbubbles are very pressure sensitive and high hydrostatic pressures during injection are therefore not recommended. Large needles (>19 G) are recommended for the transfer of microbubbles into a plastic tube or to draw up microbubbles with a syringe49. For i.v. injection in mice 26-30 G needles are recommended; since larger needles are more difficult to insert into the tail vein. The 26 G needle is recommended since the hydrostatic pressure is lower with this needle. However, in case of difficult venous access the 30 G needle is recommended.
The cranium of the mouse is an important attenuator of the pressure amplitude that significantly lowers the pressure amplitude at the focus. Attenuation is determined by the frequency of the transducer and the density of the medium the ultrasound wave propagates. Higher ultrasound frequencies and high tissue densities, like bone results in high attenuation. The pressure amplitude is partially absorbed by bone and some pressure amplitude is lost by reflection and scattering50. In our experiments we have determined in mouse cadavers that the attenuation at 1 MHz is 14.5 ± 1.3 dB/cm with an average skull thickness of 0.9 mm as shown before21,50. Cavitation monitoring is highly recommended since microbubbles reflect distinct acoustic emissions during stable cavitation and inertial cavitation. Wideband emission is a distinct acoustic emission for inertial cavitation12. Real-time monitoring makes it possible to detect inertial cavitation and lower the pressure amplitude accordingly to avoid tissue damage.
Previous reports described the influence of the type of anesthesia on the achieved BBB permeability11,31. For isoflurane based anesthesia, a vasodilation occurs shortly after anesthesia initiation, which is associated with a slight reduction of the cerebral blood flow. Furthermore, anesthesia over extended durations, in particular in absence of a temperature stabilization, leads to a reduced heart rate. Since both factors can potentially lead to a larger variance of the cerebral concentration of both microbubbles or co-administered drugs, a strict anesthesia protocol is advisable to achieve reproducible results51. Anesthesia with 1.5% v/v isoflurane in 2 L/min oxygen for 35 to 45 minutes was not problematic, as advised by Constantinides et al.51. In contrast to McDannold et al. who showed that this gas mixture in combination with the specific type of their microbubbles was problematic52, we have not observed noteworthy problems with this type of microbubbles. Alternatively, the animals can be anesthetized with a mix of ketamine/xylazine, which has no known vasoactive effects53.
In summary, the imaging-guided BBB-opening technique described here has been used for high-volume preclinical drug evaluation studies that demonstrated the efficiency of the suggested workflow. The system could thereby be operated by non-technical personnel after a short training due to the high degree of automation. This in combination with the simplicity of the setup resulted in a high degree of standardization, which in turn ensures experimental reproducibility, reduced intra-group variability and thus allows to reduce the required sample size.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This project was funded by the KWF-STW (Drug Delivery by Sonoporation in Childhood Diffuse Intrinsic Pontine Glioma and High-grade Glioma). We thank Ilya Skachkov and Charles Mougenot for their input in the development of the system.
Materials
| 1 mL luer-lock syringe | Becton Dickinson | 309628 | Plastipak |
| 19 G needle | Terumo Agani | 8AN1938R1 | |
| 23 G needle | Terumo Agani | 8AN2316R1 | |
| 3M Transpore surgical tape | Science applied to life | 7000032707 | or similar |
| Arbitrary waveform generator | Siglent | n.a. | SDG1025, 25 MHz, 125 Msa/s |
| Automated stereotact | in-house built | n.a. | Stereotact with all elements were in-house built |
| Bruker In-Vivo Xtreme | Bruker | n.a. | Includes software |
| Buffered NaCl solution | B. Braun Melsungen AG | 220/12257974/110 | |
| Buprenorfine hydrochloride | Indivior UK limitd | n.a. | 0.324 mg |
| Cage enrichment: paper-pulp smart home | Bio services | n.a. | |
| Carbon filter | Bickford | NC0111395 | Omnicon f/air |
| Ceramic spoon | n.a | n.a. | |
| Cotton swabs | n.a. | n.a. | |
| D-luciferin, potassium salt | Gold Biotechnology | LUCK-1 | |
| Ethanol | VUmc pharmacy | n.a. | 70% |
| Evans Blue | Sigma Aldrich | E2129 | |
| Fresenius NaCl 0.9% | Fresenius Kabi | n.a. | NaCl 0.9 %, 1000 mL |
| Histoacryl | Braun Surgical | n.a. | Histoacryl 0.5 mL |
| Hydrophone | Precision Acoustics | n.a. | |
| Insulin syringe | Becton Dickinson | 324825/324826 | 0.5 mL and 0.3 mL |
| Isoflurane | TEVA Pharmachemie BV | 8711218013196 | 250 mL |
| Ketamine | Alfasan | n.a. | 10 %, 10 mL |
| Mouse food: Teklad global 18% protein rodent diet | Envigo | 2918-11416M | |
| Neoflon catheter | Becton Dickinson | 391349 | 26 GA 0.6 x 19 mm |
| Oscilloscope | Keysight technologies | n.a. | InfiniiVision DSOX024A |
| Plastic tubes | Greiner bio-one | 210261 | 50 mL |
| Power amplifier | Electronics & Innovation Ltd | 210L | Model 210L |
| Preamplifier DC Coupler | Precision Acoustics | n.. | Serial number: DCPS94 |
| Scissors | Sigma Aldrich | S3146-1EA | or similar |
| Sedazine | AST Farma | n.a. | 2% |
| SonoVue microbubbles | Bracco | n.a. | 8 µl/ml |
| Sterile water | Fresenius Kabi | n.a. | 1000 mL |
| Syringe | n.a. | n.a. | various syringes can be used |
| Temgesic | Indivior UK limitd | n.a. | 0.3 mg/ml |
| Transducer | Precision Acoustics | n.a. | 1 MHz |
| Tweezers | Sigma Aldrich | F4142-1EA | or similar |
| Ultrasound gel | Parker Laboratories Inc. | 01-02 | Aquasonic 100 |
| Vidisic gel | Bausch + Lomb | n.a. | 10 g |
References
- Lipinski, C. A. Lead- and drug-like compounds: the rule-of-five revolution. Drug Discovery Today: Technologies. 1 (4), 337-341 (2004).
- Pardridge, W. M. Blood-brain barrier delivery. Drug Discovery Today. 12 (1-2), 54-61 (2007).
- Alli, S., et al. Brainstem blood brain barrier disruption using focused ultrasound: A demonstration of feasibility and enhanced doxorubicin delivery. Journal of Controlled Release. 281, 29-41 (2018).
- Burgess, A., Hynynen, K. Noninvasive and targeted drug delivery to the brain using focused ultrasound. ACS Chemical Neuroscience. 4 (4), 519-526 (2013).
- Meng, Y., et al. Safety and efficacy of focused ultrasound induced blood-brain barrier opening, an integrative review of animal and human studies. Journal of Controlled Release. 309, 25-36 (2019).
- Darrow, D. P. Focused Ultrasound for Neuromodulation. Neurotherapeutics. 16 (1), 88-99 (2019).
- Zhou, Y. F. High intensity focused ultrasound in clinical tumor ablation. World Journal of Clinical Oncology. 2 (1), 8-27 (2011).
- O’Reilly, M. A., Hough, O., Hynynen, K. Blood-Brain Barrier Closure Time After Controlled Ultrasound-Induced Opening Is Independent of Opening Volume. Journal of Ultrasound in Medicine. 36 (3), 475-483 (2017).
- Mainprize, T., et al. Blood-Brain Barrier Opening in Primary Brain Tumors with Non-invasive MR-Guided Focused Ultrasound: A Clinical Safety and Feasibility Study. Scientific Reports. 9 (1), 321 (2019).
- Dasgupta, A., et al. Ultrasound-mediated drug delivery to the brain: principles, progress and prospects. Drug Discovery Today: Technologies. 20, 41-48 (2016).
- O’Reilly, M. A., Waspe, A. C., Chopra, R., Hynynen, K. MRI-guided disruption of the blood-brain barrier using transcranial focused ultrasound in a rat model. Journal of Visualized Experiments. (61), (2012).
- McDannold, N., Vykhodtseva, N., Hynynen, K. Targeted disruption of the blood-brain barrier with focused ultrasound: association with cavitation activity. Physics in Medicine & Biology. 51 (4), 793 (2006).
- McDannold, N., Vykhodtseva, N., Hynynen, K. Blood-brain barrier disruption induced by focused ultrasound and circulating preformed microbubbles appears to be characterized by the mechanical index. Ultrasound in Medicine and Biology. 34 (5), 834-840 (2008).
- Sun, T., et al. Closed-loop control of targeted ultrasound drug delivery across the blood-brain/tumor barriers in a rat glioma model. Proceedings of the National Academy of Sciences. 114 (48), 10281-10290 (2017).
- Lipsman, N., et al. Blood-brain barrier opening in Alzheimer’s disease using MR-guided focused ultrasound. Nature Communications. 9 (1), 2336 (2018).
- Carpentier, A., et al. Clinical trial of blood-brain barrier disruption by pulsed ultrasound. Science Translational Medicine. 8 (343), 342 (2016).
- Chopra, R., Curiel, L., Staruch, R., Morrison, L., Hynynen, K. An MRI-compatible system for focused ultrasound experiments in small animal models. Medical Physics. 36 (5), 1867-1874 (2009).
- Kinoshita, M., McDannold, N., Jolesz, F. A., Hynynen, K. Targeted delivery of antibodies through the blood–brain barrier by MRI-guided focused ultrasound. Biochemical and Biophysical Research Communications. 340 (4), 1085-1090 (2006).
- Larrat, B., et al. MR-guided transcranial brain HIFU in small animal models. Physics in Medicine & Biology. 55 (2), 365 (2009).
- Contag, C. H., Jenkins, D., Contag, P. R., Negrin, R. S. Use of reporter genes for optical measurements of neoplastic disease in vivo. Neoplasia. 2 (1-2), 41 (2000).
- Choi, J. J., Pernot, M., Small, S. A., Konofagou, E. E. Noninvasive, transcranial and localized opening of the blood-brain barrier using focused ultrasound in mice. Ultrasound in Medicine & Biology. 33 (1), 95-104 (2007).
- Bing, C., et al. Trans-cranial opening of the blood-brain barrier in targeted regions using astereotaxic brain atlas and focused ultrasound energy. Journal of Therapeutic Ultrasound. 2 (1), 13 (2014).
- Marquet, F., et al. Real-time, transcranial monitoring of safe blood-brain barrier opening in non-human primates. PloS One. 9 (2), (2014).
- Anastasiadis, P., et al. characterization and evaluation of a laser-guided focused ultrasound system for preclinical investigations. Biomedical Engineering Online. 18 (1), 36 (2019).
- Liu, H. L., Pan, C. H., Ting, C. Y., Hsiao, M. J. Opening of the blood-brain barrier by low-frequency (28-kHz) ultrasound: a novel pinhole-assisted mechanical scanning device. Ultrasound in Medicine & Biology. 36 (2), 325-335 (2010).
- Zhu, L., et al. Focused ultrasound-enabled brain tumor liquid biopsy. Scientific Reports. 8 (1), 1-9 (2018).
- Bader, K. B., Holland, C. K. Gauging the likelihood of stable cavitation from ultrasound contrast agents. Physics in Medicine & Biology. 58 (1), 127 (2012).
- Neppiras, E. Acoustic cavitation series: part one: Acoustic cavitation: an introduction. Ultrasonics. 22 (1), 25-28 (1984).
- Aryal, M., Arvanitis, C. D., Alexander, P. M., McDannold, N. Ultrasound-mediated blood-brain barrier disruption for targeted drug delivery in the central nervous system. Advanced Drug Delivery Reviews. 72, 94-109 (2014).
- Tung, Y. S., Choi, J. J., Baseri, B., Konofagou, E. E. Identifying the inertial cavitation threshold and skull effects in a vessel phantom using focused ultrasound and microbubbles. Ultrasound in Medicine & Biology. 36 (5), 840-852 (2010).
- Arvanitis, C. D., Livingstone, M. S., Vykhodtseva, N., McDannold, N. Controlled ultrasound-induced blood-brain barrier disruption using passive acoustic emissions monitoring. PloS One. 7 (9), (2012).
- Tsai, C. H., Zhang, J. W., Liao, Y. Y., Liu, H. L. Real-time monitoring of focused ultrasound blood-brain barrier opening via subharmonic acoustic emission detection: implementation of confocal dual-frequency piezoelectric transducers. Physics in Medicine & Biology. 61 (7), 2926 (2016).
- Chen, W. S., Brayman, A. A., Matula, T. J., Crum, L. A. Inertial cavitation dose and hemolysis produced in vitro with or without Optison. Ultrasound in Medicine & Biology. 29 (5), 725-737 (2003).
- Qiu, Y., et al. The correlation between acoustic cavitation and sonoporation involved in ultrasound-mediated DNA transfection with polyethylenimine (PEI) in vitro. Journal of Controlled Release. 145 (1), 40-48 (2010).
- Sun, T., Jia, N., Zhang, D., Xu, D. Ambient pressure dependence of the ultra-harmonic response from contrast microbubbles. The Journal of the Acoustical Society of America. 131 (6), 4358-4364 (2012).
- Rehemtulla, A., et al. Rapid and quantitative assessment of cancer treatment response using in vivo bioluminescence imaging. Neoplasia. 2 (6), 491-495 (2000).
- Puaux, A. L., et al. A comparison of imaging techniques to monitor tumor growth and cancer progression in living animals. International Journal of Molecular Imaging. 2011, (2011).
- Wu, S. K., et al. Characterization of different microbubbles in assisting focused ultrasound-induced blood-brain barrier opening. Scientific Reports. 7, 46689 (2017).
- van den Broek, M. P., Groenendaal, F., Egberts, A. C., Rademaker, C. M. Effects of hypothermia on pharmacokinetics and pharmacodynamics. Clinical Pharmacokinetics. 49 (5), 277-294 (2010).
- Paxinos, G., Franklin, K. B. . Paxinos and Franklin’s the mouse brain in stereotaxic coordinates. , (2019).
- Saunders, N. R., Dziegielewska, K. M., Møllgård, K., Habgood, M. D. Markers for blood-brain barrier integrity: how appropriate is Evans blue in the twenty-first century and what are the alternatives. Frontiers in Neuroscience. 385, 385 (2015).
- Yao, L., Xue, X., Yu, P., Ni, Y., Chen, F. Evans blue dye: a revisit of its applications in biomedicine. Contrast Media & Molecular Imaging. 2018, (2018).
- Caretti, V., et al. Monitoring of tumor growth and post-irradiation recurrence in a diffuse intrinsic pontine glioma mouse model. Brain Pathology. 21 (4), 441-451 (2011).
- Yoshimura, J., Onda, K., Tanaka, R., Takahashi, H. Clinicopathological study of diffuse type brainstem gliomas: analysis of 40 autopsy cases. Neurologia Medico-Chirurgica. 43 (8), 375-382 (2003).
- Yang, F. Y., et al. Micro-SPECT/CT-based pharmacokinetic analysis of 99mTc-diethylenetriaminepentaacetic acid in rats with blood-brain barrier disruption induced by focused ultrasound. Journal of Nuclear Medicine. 52 (3), 478-484 (2011).
- Sirsi, S., Borden, M. Microbubble compositions, properties and biomedical applications. Bubble Science, Engineering & Technology. 1 (1-2), 3-17 (2009).
- Greis, C. Technology overview: SonoVue. European Radiology. 14, 11-15 (2004).
- Schneider, M. Characteristics of sonovue. Echocardiography. 16, 743-746 (1999).
- Talu, E., Powell, R. L., Longo, M. L., Dayton, P. A. Needle size and injection rate impact microbubble contrast agent population. Ultrasound in Medicine & Biology. 34 (7), 1182-1185 (2008).
- Pinton, G., et al. Attenuation, scattering, and absorption of ultrasound in the skull bone. Medical Physics. 39 (1), 299-307 (2012).
- Constantinides, C., Mean, R., Janssen, B. J. Effects of isoflurane anesthesia on the cardiovascular function of the C57BL/6 mouse. ILAR journal/National Research Council, Institute of Laboratory Animal Resources. 52, 21 (2011).
- McDannold, N., Zhang, Y., Vykhodtseva, N. The effects of oxygen on ultrasound-induced blood-brain barrier disruption in mice. Ultrasound in Medicine & Biology. 43 (2), 469-475 (2017).
- McDannold, N., Zhang, Y., Vykhodtseva, N. Blood-brain barrier disruption and vascular damage induced by ultrasound bursts combined with microbubbles can be influenced by choice of anesthesia protocol. Ultrasound in Medicine and Biology. 37 (8), 1259-1270 (2011).

