Fat-Covered Islet Transplantation Using Epididymal White Adipose Tissue
Summary
This fat-covered islet transplantation method is suitable for the detection of engrafted islets in the intraperitoneal cavity. Notably, it does not require the use of biobinding agents or suturing.
Abstract
Islet transplantation is a cellular replacement therapy for severe diabetes mellitus. The intraperitoneal cavity is typically the transplant site for this procedure. However, intraperitoneal islet transplantation has some limitations, including poor transplant efficacy, difficult graft detection ability, and a lack of graftectomy capability for post-transplant analysis. In this paper, “fat-covered islet transplantation”, an intraperitoneal islet transplantation method that utilizes epididymal white adipose tissue, is used to assess the therapeutic effects of bioengineered islets. The simplicity of the method lies in the seeding of islets onto epididymal white adipose tissue and using the tissue to cover the islets. While this method can be categorized as an intraperitoneal islet transplantation technique, it shares characteristics with intra-adipose tissue islet transplantation. The fat-covered islet transplantation method demonstrates more robust therapeutic effects than intra-adipose tissue islet transplantation, however, including the improvement of blood glucose and plasma insulin levels and the potential for graft removal. We recommend the adoption of this method for assessing the mechanisms of islet engraftment into white adipose tissue and the therapeutic effects of bioengineered islets.
Introduction
Islet transplantation is a cellular replacement therapy for patients with severe diabetes mellitus. Recent reports have shown that rates of insulin-independence at three years after transplantation improve up to 44%1 and that approximately 80% of recipients who receive more than 600,000 total islet equivalents achieve insulin independence2. Furthermore, in the most recent Collaborative Islet Transplant Registry report, it was revealed that fasting blood glucose levels were maintained at 60-140 mg/dL for over a period of 5 years in over 70% of patients who underwent islet transplant alone. The study also determined that around 90% of the patients who received islet transplant alone or islet transplantation after kidney transplant did not develop any severe hypoglycemic events for over 5 years3.
Although the clinical outcomes of this treatment have been improving, some limitations must still be addressed, including the necessity of establishing an optimal transplant site. The liver is a typical transplant site for clinical islet transplantation because it is the largest organ that can accommodate a high volume of islets. However, in some patients the liver is unavailable (e.g., due to portal hypertension, hepatitis, and/or cirrhosis4) and therefore other sites, including the renal subcapsular space5,6, omental pouch7,8,9,10, mesentery11, gastrointestinal tract12, skeletal muscle13, subcutaneous tissue13, bone marrow14, and spleen15,16,17, have been considered as alternative transplant sites.
Although intraperitoneal islet transplantation can be performed easily under local anesthesia, making the intraperitoneal cavity an appealing site for clinical islet transplantation, upon transplant, the islets are dispersed throughout the entire intraperitoneal cavity, making islet engraftment detection and successful engraftment confirmation difficult. Therefore, the intraperitoneal cavity is not widely recognized as an ideal clinical transplant site. Instead, it is frequently utilized as a control model for preclinical studies to investigate the effectiveness of transplanted encapsulated18 and bioengineered islets19. However, an exact comparison between bioengineered and control islets is difficult to achieve due to the challenges in performing an accurate engraftment assessment.
In contrast, the use of intraperitoneal white adipose tissue in the omental pouch8, mesentery, and other extrahepatic locations has been well reported10,20,21,22,23 and many of the studies investigating the function of bioengineered islets transplanted using white adipose tissue were able to report promising therapeutic outcomes20,24,25,26. As the use of epididymal adipose tissue facilitates the detection of transplanted islets, the "fat-covered islet transplantation method", utilizing epididymal adipose tissue, was developed to overcome the limitations of intraperitoneal islet transplantation. In this paper, fat-covered islet transplantation using epididymal adipose tissue is described.
Protocol
The following procedure is performed in three steps. The first step includes the induction of diabetes in the recipient mice and the isolation of donor islets. The second step involves the preparation of islets before transplantation. In the third step, islet transplantation onto epididymal adipose tissue and covering of the islets using the adipose tissue is performed. After that, the therapeutic effects were assessed. The handling of the mice and the experimental procedures performed in this study comply with the ''Principles of Laboratory Animal Care'' (Guide for the Care and Use of Laboratory Animals, National Institutes of Health publication 8th edition, 2011), and the experimental protocol was approved by the Animal Care and Use Committee of Fukuoka University (approval number: 186018).
1. Surgical preparation
- Induction of diabetes: Induce diabetes in 20-25 g body weight, 8-12-week-old recipient male mice through intravenous injection of 18 mg/mL streptozotocin solution prepared in 0.1M citrate buffer (180 mg/kg body weight). Mice with blood glucose levels exceeding 400 mg/dL are considered to be diabetic. Use diabetic mice within 1 week after diabetes induction before excessive atrophy of the epididymal white adipose tissue for covering islets.
- Islet isolation: Perform murine islet isolation one day before transplantation following Gotoh's method27 for islet isolation.
- In brief, digest pancreatic tissue using collagenase solution. Isolate islets by density gradient centrifugation using an appropriate cell separation solution. Then culture islets overnight in an incubator at 22 °C and 5% CO2 (culture at <37 °C has been reported to prevent islet death28,29,30,31).
NOTE: Handle the purified islet cultures in a safety cabinet. Filter-sterilize all solutions used for islet isolation and culture using a 0.22 µm filter.
2. Preparation of islets for transplantation
- Gather the appropriate instruments and materials as indicated in Figure 1A.
- As digestive enzymes such as amylase and lipase may result in injury to the isolated and transplanted islets and a loss of islets can occur from being trapped in contaminating fibrous tissues within the culture dish, prior to transplantation, use forceps to handpick any extra-islet components from the pancreas, including acinar and fibrous tissues (Figure 1B), under a dissecting microscope. After picking, use a cell strainer to filter out single acinar cells.
- Transfer the filtered islets to a new culture dish containing any appropriate culture medium or buffer solution (e.g., DMEM with low glucose, RPMI1640, CMRL1066, or HBSS) supplemented with bovine serum or albumin to prevent islet attachment to plastic and swirl the dish to position the islets in the center of the dish (Figure 1C). Using a P200 micropipette and the microscope, pick the individual islets into an appropriate collection tube (Figure 1D).
- Place a new, 40 μm cell strainer on top of a 50 mL plastic tube (Figure 1E left and center) and wash the filter with fresh medium (Figure 1E right).
- Use a 1000 μL pipette to add the islets to the strainer to separate the islets and single acinar cells (Figure 1F1 and Figure 1F2).
NOTE: The purified islets on cell strainer will be approximately 100% pure. - Use forceps to invert the strainer on a new 60- or 100-mm sized non-treated culture dish containing culture medium or an appropriate buffer solution supplemented with bovine serum or albumin (Figure 1F3 and Figure 1F4). Use fresh medium/buffer to flush the islets into a new culture dish. Then add enough medium/buffer to the culture dish to reach a total volume of approximately 20 mL.
- Count the islets under a microscope and divide the number of islets equally between individual 1.5 mL plastic centrifuge tubes according to the number of donor animals (Figure 1G). For example, two hundred, 100-200 µm islet equivalents (IEQ) from two mice would be added to each of two tubes.
- Centrifuge the islets at 2,100 x g within 1 minute at room temperature and discard the supernatant. Around 20-30 µL of residual solution will typically remain in the tube (Figure 1H).
3. Islet transplantation onto epididymal adipose tissue and covering with epididymal white adipose tissue
- Before the surgery, collect an anesthesia machine for small animals, stereo microscope, light source, 50-200 µL micropipette with 200 µL micropipette tips, cotton swabs, a 4-0 suturing set, and disinfected surgical instruments (Figure 2A). Autoclave the cooper scissors, ophthalmic scissors, Pean forceps, tweezers, and needle holders. After autoclaving, immerse the equipment in a 1% povidone-iodine solution (Figure 2A). Use cotton swabs for mobilization of the epididymal white adipose tissue and for hemostasis in cases of bleeding. Use a micropipette with a 50-200 µL tips for islet transplantation.
- Deliver anesthesia to the diabetic recipient mouse using an inhaled anesthetic agent (2% isoflurane in oxygen). Apply ophthalmic lubricant to both eyes to prevent drying. Then place the mouse in the supine position (Figure 2B left) and remove the hair from the abdomen to prevent infection using hair clippers and/or depilatory cream. Disinfect the abdomen and the inguinal region using at least three alternating rounds of a povidone-iodine solution followed by 70% ethanol (Figure 2B right). Before the surgery, confirm anesthesia depth via the absence of a toe pinch reflex. Provide intra-operative thermal support using a heating pad and use a surgical drape to secure the sterile surgical area.
- Incise the skin at the lower median area (Figure 2C left). A skin incision that is approximately 2 cm in length is recommended. Clamp the left abdominal wall with the Pean forceps (atraumatic forceps or retractor can also be used) and pull the tissue to the left side of the mouse to secure the surgical field (Figure 2C right). After laparotomy, decrease the percentage of isoflurane to 1.0-1.5% for anesthesia maintenance.
- Use a cotton swab to mobilize the small and large intestine to the right side of the mouse (i.e., left side of the operator). The left epididymal white adipose tissue in the abdominal cavity are located in the left inguinal area. Mobilize the epididymal white adipose tissue and the left testis to outside of the abdomen (Figure 2D left) and stretch out the tissue (2D right).
- Use a P200 micropipette equipped with a 200 µL pipette tip to collect the entire volume of islets from one 1.5 mL tube with gentle pipetting (Figure 2E left), taking care that no islets are left in the tube upon collection. Allow the collected islets to settle to the tip of the pipette by gravity (Figure 2E right).
- Place the micropipette tip lightly onto the distended adipose tissue. Taking care to prevent excessive flushing of the medium/buffer in the tip, carefully seed the islets onto the tissue (Figure 2F left). After seeding, confirm a correct placement of the islets under a dissecting microscope (Figure 2F right).
- Cover the islets with the epididymal white adipose tissue (Figure 2G). The use of sutures or biobinding agents is not needed.
- Place the left testis under the epididymal white adipose tissue and return the tissues to the intraperitoneal cavity (Figure 2H). Close the skin in two layers (peritoneum, then muscle and skin) using a 4-0 suture (any sutures such as nylon or absorbable sutures can be used) (Figure 2I). Inject acetylsalicylic acid (300 mg/kg; SQ) near the wound for postoperative analgesia. Then place the mouse under a heat lamp and monitor until full recovery.
4. Monitoring after Islet transplantation (Summary)
- Assess the therapeutic effects of islet transplantation by monitoring blood glucose, glucose tolerance test and histological assessment at postoperative day (POD) 28.
- Monitor the blood glucose, including the measurements of blood glucose at glucose tolerance test, using a small glucose meter.
- Collect the blood samples (a little microliters) from tail vein. Regarding histological assessment, murine insulin (for detecting engrafted islets) and von Willebrand factor (for detection of vessels, which is an evidence for islet engraftment) were detected in transplanted islets in the recovered epididymal adipose tissue by immunohistochemistry.
Representative Results
To compare the transplant efficacy of fat-covered islet transplantation to that after intraperitoneal islet transplantation, the same number of islets was implanted onto the peritoneum at the left paracolic space of control recipient diabetic animals. The blood glucose levels of mice with fat-covered islet transplantation were observed to gradually and significantly decrease compared to intraperitoneal islet transplanted mice (p = 0.0023; Figure 3A). One month after transplantation, the blood glucose in mice with fat-covered islet transplantation was maintained at lower levels than that observed in intraperitoneal islet transplanted mice as assessed by intraperitoneal glucose tolerance testing (p = 0.0046; Figure 3B). Furthermore, as we have previously, reported that plasma insulin levels also improved after fat-covered islet transplantation. Re-elevation of blood glucose levels was also confirmed. These data demonstrate that intraperitoneal fat-covered islet transplantation using 200 IEQs can significantly improve the diabetic conditions of recipient mice.
Histological examination was also performed to assess islet engraftment into the epididymal white adipose tissue. In fat-covered islet transplant recipient animals, hematoxylin-eosin staining reveals the presence of islets within the epididymal white adipose tissue (Figure 3C, top image). In addition, fluorescence-conjugated antibody staining of insulin-positive islets facilitated the detection of von Willebrand factor-positive microvessels within the epididymal white adipose tissue of all of the recipient mice (n = 6; Figure 3C). In contrast, in intraperitoneal islet transplanted mice, no engrafted islets were observed in either the epididymal white adipose tissue or the abdominal wall (data not shown).

Figure 1-ABC. Preparation of islets for transplantation onto epididymal adipose tissue and covering with epididymal white adipose tissue. (A) Preparation of instruments: a. pipette aid, b. 10 mL pipette tips, c. 50 mL plastic tubes, d. 15 mL plastic tubes, e. 50-200 µL (left) and 200-1000 µL (right) micropipettes, f. 1.5 mL plastic centrifuge tubes, g. 200 and 1000 µL micropipette tips, h. medium or buffer containing albumin or fetal bovine serum (i.e., DMEM with low glucose containing 10% fetal bovine serum and 100 U/mL penicillin + 100 U/mL streptomycin solution), and i. 40 µm cell strainer. (B) Isolated islets with acinar (left: indicated by arrow) and fibrous tissues (right). Scale bar = 200 µm. (C) Collected islets in the plastic tube. Left, dispersed islets in culture dish. Center, islets are collected in the center of the culture dish by swirling. Right, collected islets in center of dish. Scale bar = 200 µm. Please click here to view a larger version of this figure.
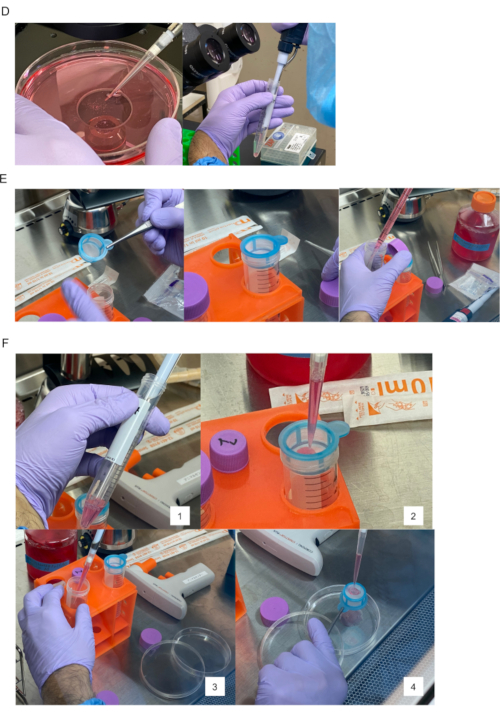
Figure 1-DEF. Preparation of islets for transplantation onto epididymal adipose tissue and covering with epididymal white adipose tissue. (D) Collected islets (left) are transferred into 15 mL plastic tubes (right). (E) The 40 µm cell strainer is set on top of the 50 mL plastic tube (left and center). Prepared medium/buffer added to other 50 mL plastic tube for flushing islets on the cell strainer into a new culture dish (right). (F) 1. Islets collected using a 200-1000 µL micropipette. 2. Islets poured into the cell strainer. 3 and 4. Medium/buffer used to flush islets onto strainer into new culture dish. Please click here to view a larger version of this figure.
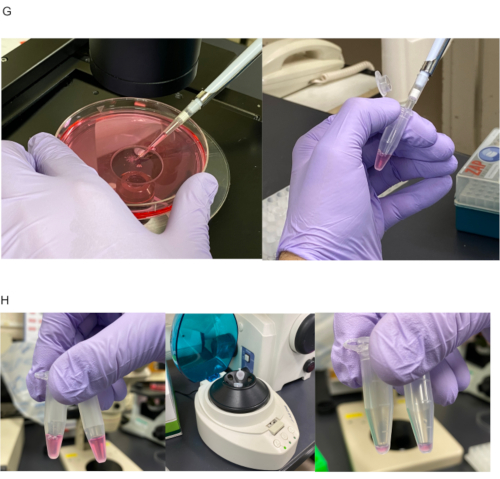
Figure 1-GH. Preparation of islets for transplantation onto epididymal adipose tissue and covering with epididymal white adipose tissue. (G) Islets divided into 1.5 mL plastic centrifuge tube according to number of recipient mice. Here, two hundred 100-200 µm islet equivalent (IEQ) were divided equally into each tube. (H) Islets divided equally in 1.5 mL tubes before centrifugation (left). Islets centrifuged to collect at tube bottom (center). 20-30 µL of supernatant remain in tube after discarding excess solution (right). Please click here to view a larger version of this figure.
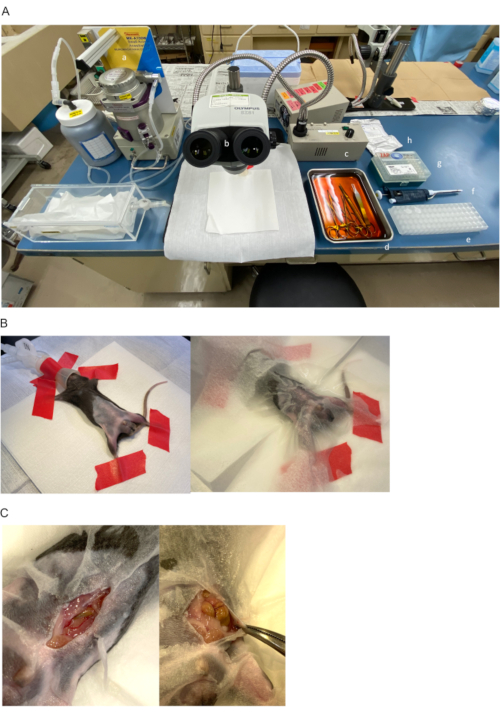
Figure 2-ABC. The procedure of islet transplantation onto epididymal adipose tissue and covering using epididymal white adipose tissue. (A) Preparation of instruments: a. anesthesia machine for small animals, b. stereo microscope, c. light source, d. disinfected surgical instruments, e. divided islets in 1.5 mL plastic tubes, f. 50-200 µL micropipettes, g. 200 µL micropipette tips, and h. 4-0 sutures. (B) Diabetic recipient mouse in a supine position under general anesthesia of 2% isoflurane (left). The abdomen and the inguinal region are disinfected using 70% ethanol and covered by a paper lab wipe (right). (C) Skin is incised at lower median position (left). Left abdominal wall is clamped by Pean forceps and pulled to left side of mouse to secure surgical field (right). Please click here to view a larger version of this figure.
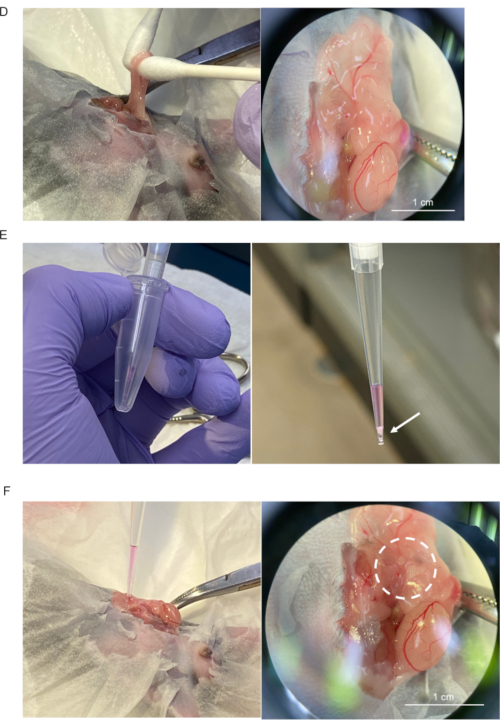
Figure 2-DEF. The procedure of islet transplantation onto epididymal adipose tissue and covering using epididymal white adipose tissue. (D) The left epididymal white adipose tissue and the left testis are mobilized outside of the abdomen (left) and distended (right). Scale bar = 1 cm. (E) Islets in 1.5 mL plastic tubes are completely collected using a micropipette with 200 µL pipette tip (left). Collected islets (indicated by arrow) allowed to completely sink by gravity to pipette tip (right). (F) Micropipette tip lightly placed onto the distended adipose tissue (left). Islet seeding (dotted circle) onto the tissue confirmed by dissecting microscope (right). Scale bar = 1 cm. Please click here to view a larger version of this figure.
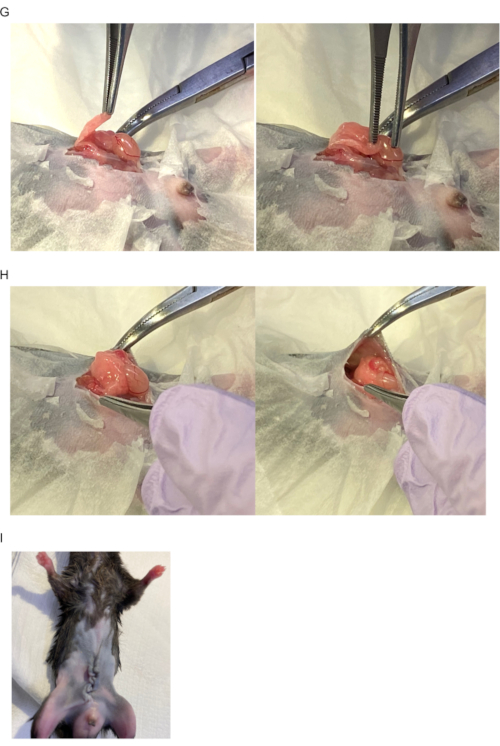
Figure 2-GHI. The procedure of islet transplantation onto epididymal adipose tissue and covering using epididymal white adipose tissue. (G) Islets are covered with epididymal white adipose tissue. (H) Left testis and epididymal white adipose tissue returned to intraperitoneal cavity. (I) Image after abdomen closure. Please click here to view a larger version of this figure.
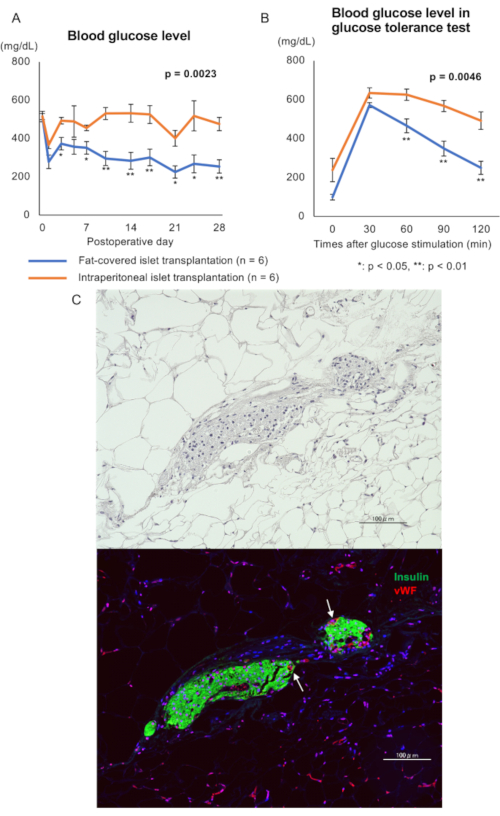
Figure 3. The therapeutic effect of fat-covered islet transplantation. (A) Blood glucose level. Blue line: fat-covered islet transplantation (n = 6); Orange line: intraperitoneal islet transplantation (n = 6) Statistical analysis was performed using repeated measures analysis of variance and a significant difference was defined as p < 0.05. (B) Blood glucose level from glucose tolerance test a month after transplantation. Blue line: fat-covered islet transplantation (n = 6); Orange line: intraperitoneal islet transplantation (n = 6). Statistical analysis was performed using repeated measures analysis of variance and a significant difference was defined as p < 0.05. (C) Histological image of engrafted islets one month after transplantation. Top image: hematoxylin-eosin staining; Bottom image: immunohistostaining for murine insulin (green) and von Willebrand factor (vWF: red, indicated by white arrow). Scale bar = 100 µm. Please click here to view a larger version of this figure.
Discussion
The fat-covered islet transplantation method incorporates techniques from two different transplant techniques: intraperitoneal islet transplantation and intra-adipose tissue islet transplantation. As the surface membrane of epididymal white adipose tissue is considered to be the white adipose tissue that is covered by the peritoneum and that is attached to the epididymis, the fat-covered islet transplantation method can be anatomically categorized as a type of intraperitoneal islet transplantation. The technique by which the islets are delivered to the recipient animal, however, are more similar to those utilized in intra-adipose tissue islet transplantation. Our data shows that the therapeutic effect of the fat-covered islet transplantation method is superior to that of intraperitoneal islet transplantation. Our previous study also showed that the transplant efficacy of this method is nearly equal to that of renal subcapsular islet transplantation, a method of islet transplantation with a supreme transplant efficacy23. It is speculated that the usefulness of this method may be due to the adhesion molecules present within white adipose tissues and adipocytokines and which are thought to contribute to islet engraftment10,23.
The size of the epididymal white adipose tissue allows a large volume of islets to be accommodated. In contrast, the rodent greater omentum is too small to easily contain and access many islets. The only physical limitation of epididymal white adipose tissue as a potential experimental islet transplantation site is that it does not provide a portal circulation of insulin10.
The method is unique because the islets are covered with adipose tissues, instead of being directly infused into the tissue. Intra-adipose tissue transplanted islets may suffer from the influence of lipotoxicity, as the impaired insulin function in diabetic animals can lead to excess free fatty acids32. This method also does not require biobonding agents or suturing to prevent implanted islet loss. As observed in Figure 3C, islets remain successfully engrafted within the adipose tissue up to 1 month after transplant and blood glucose level maintenance has been confirmed after graftectomy23. This phenomenon may be due to the ability of adipose tissue to trap the islets, which become difficult to peel off.
The advantages of this method are that it is technically easy to perform, allows a metabolic and histologic assessment of the therapeutic effects of the islets, and facilitates the evaluation of islet gene and/or protein expression after graftectomy. Compared to previous studies, the efficacy of this method is not inferior to that of islet transplantation into epididymal white adipose tissue10,33,34,35,36. It is important to note that a precise seeding of the islets onto the epididymal white adipose tissue without islets loss is necessary for a successful engraftment. To ensure success, the entire volume of islets must be aspirated gently into the micropipette tip from the 1.5 mL plastic tube, as rough and rapid pipetting may result in islet adhesion to the walls of the plastic tube, making dispensing difficult. Islet adhesion is a major reason for inadequate islet seeding. After allowing the islets to settle at the tip of the micropipette tip, the islets must be implanted onto the epididymal adipose tissue without flushing. It is important to minimize depression of the plunger button when aspirating and dispensing the islets to prevent excessive medium/buffer flushing of the adipose tissue. Therefore, it is essential to wait until the islets have aggregated completely at the tip of the micropipette tip before depressing the plunger button of the micropipette for implantation. It is sufficient to place the tip lightly onto the adipose tissue and to confirm the seeding of islets onto the tissue by light microscopy.
In conclusion, this method is very simple, despite requiring several critical steps for its success. We hope for the wide adoption of this method to further aid in the facilitation of islet engraftment onto white adipose tissue and for further evaluation of the therapeutic effects of bioengineered islets.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This study was funded by a Grant-in-Aid for Scientific Research (C) (19K09839, NS) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Materials
| 4-0 Nylon | Alfresa | ER2004NA45-KF2 | Closing abdomen |
| Alexa 488-conjugated donkey anti-guinea pig | Jackson Immunoresearch | 706-546-148 | Secondary antibody for insulin antibody |
| Alexa 647-conjugated donkey anti-rabbit | Jackson Immunoresearch | 711-606-152 | Secondary antibody for von Willebrand factor antibody |
| DMEM, low glucose, pyruvate | ThermoFisher Scientific | 11885084 | Culturing islets, transplanting islets |
| Eosin | Fujifilm Wako Chemicals | 051-06515 | Using for staining tissue by eosin |
| Eppendorf Safe-Lock Tubes, 1.5 mL | Eppendorf | 30120086 | Collecting islets |
| Falcon 15 mL Conical Centrifuge Tubes | Corning | 352095 | Collecting islets |
| Falcon 40 µm Cell Strainer | Falcon | 352340 | Using for separating islets from other pancreatic tissue |
| Falcon 50 mL Conical Centrifuge Tubes | Corning | 352070 | Discarding excessive medium/buffer |
| Guinea pig anti-insulin | Agilent Technologies Japan, Ltd. (Dako) | IR002 | Primary antibody for murine insulin |
| Hematoxylin | Muto Pure Chemicals Co., Ltd. | 30002 | Using for staining tissue by hematoxylin |
| Isodine solution 10% | Shionogi&Co., Ltd. | no catalog number | Using for disinfection |
| Isoflurane | Fujifilm Wako Chemicals | 095-06573 | Using for anesthesia |
| Labcon 1000 µL ZapSilk Low Retention Pipette Tips | Labcon | 1177-965-008 | Using for separating islets from other pancreatic tissue |
| Labcon 200 µL ZapSilk Low Retention Pipette Tips | Labcon | 1179-965-008 | Using for seeding islets onto epididymal white adipose tissue |
| Mintsensor | Sanwa Kagaku Kenkyusho Co. Ltd., | 8AEB02E | Using for monitoring blood glucose |
| Pipetteman P-1000 | Gilson | F123602 | Using for separating islets from other pancreatic tissue |
| Pipetteman P-200 | Gilson | F123601 | Using for seeding islets onto epididymal white adipose tissue |
| Rabbit anti-vWF | Abcam | ab6994 | Primary antibody for murine von Willebrand factor |
References
- Barton, F. B., et al. Improvement in outcomes of clinical islet transplantation: 1999-2010. Diabetes Care. 35 (7), 1436-1445 (2012).
- Balamurugan, A. N., et al. Islet product characteristics and factors related to successful human islet transplantation from the Collaborative Islet Transplant Registry (CITR) 1999-2010. American Journal of Transplantation. 14 (11), 2595-2606 (2014).
- Collaborative Islet Transplant Registry. . Collaborative Islet Transplant Registry. Annual Report. , (2017).
- Rajab, A., et al. Total Pancreatectomy and Islet Autotransplantation Following Treated Hepatitis C Infection. Cell Transplantation. 27 (10), 1569-1573 (2018).
- Mellgren, A., Schnell Landstrom, A. H., Petersson, B., Andersson, A. The renal subcapsular site offers better growth conditions for transplanted mouse pancreatic islet cells than the liver or spleen. Diabetologia. 29 (9), 670-672 (1986).
- Hiller, W. F., Klempnauer, J., Luck, R., Steiniger, B. Progressive deterioration of endocrine function after intraportal but not kidney subcapsular rat islet transplantation. Diabetes. 40 (1), 134-140 (1991).
- Yasunami, Y., Lacy, P. E., Finke, E. H. A new site for islet transplantation–a peritoneal-omental pouch. Transplantation. 36 (2), 181-182 (1983).
- Kin, T., Korbutt, G. S., Rajotte, R. V. Survival and metabolic function of syngeneic rat islet grafts transplanted in the omental pouch. American Journal of Transplantation. 3 (3), 281-285 (2003).
- Kasoju, N., et al. Bioengineering a pre-vascularized pouch for subsequent islet transplantation using VEGF-loaded polylactide capsules. Biomaterials Science. 8 (2), 631-647 (2020).
- Sakata, N., Yoshimatsu, G., Kodama, S. White Adipose Tissue as a Site for Islet Transplantation. Transplantology. 1 (2), 55-70 (2020).
- Osama Gaber, A., Chamsuddin, A., Fraga, D., Fisher, J., Lo, A. Insulin independence achieved using the transmesenteric approach to the portal vein for islet transplantation. Transplantation. 77 (2), 309-311 (2004).
- Fujita, M., et al. Technique of endoscopic biopsy of islet allografts transplanted into the gastric submucosal space in pigs. Cell Transplantation. 22 (12), 2335-2344 (2013).
- Sakata, N., et al. Strategy for clinical setting in intramuscular and subcutaneous islet transplantation. Diabetes/Metabolism Research and Reviews. 30 (1), 1-10 (2014).
- Cantarelli, E., et al. Transplant Site Influences the Immune Response After Islet Transplantation: Bone Marrow Versus Liver. Transplantation. 101 (5), 1046-1055 (2017).
- White, S. A., et al. The risks of total pancreatectomy and splenic islet autotransplantation. Cell Transplantation. 9 (1), 19-24 (2000).
- Itoh, T., Nishinakamura, H., Kumano, K., Takahashi, H., Kodama, S. The Spleen Is an Ideal Site for Inducing Transplanted Islet Graft Expansion in Mice. PLoS One. 12 (1), 0170899 (2017).
- Sakata, N., Yoshimatsu, G., Kodama, S. The Spleen as an Optimal Site for Islet Transplantation and a Source of Mesenchymal Stem Cells. International Journal of Molecular Sciences. 19 (5), (2018).
- Sakata, N., et al. Effect of rat-to-mouse bioartificial pancreas xenotransplantation on diabetic renal damage and survival. Pancreas. 32 (3), 249-257 (2006).
- Nagaya, M., et al. Effectiveness of bioengineered islet cell sheets for the treatment of diabetes mellitus. Journal of Surgical Research. 227, 119-129 (2018).
- Weaver, J. D., et al. Vasculogenic hydrogel enhances islet survival, engraftment, and function in leading extrahepatic sites. Science Advances. 3 (6), 1700184 (2017).
- Dufour, J. M., et al. Development of an ectopic site for islet transplantation, using biodegradable scaffolds. Tissue Engineering. 11 (9-10), 1323-1331 (2005).
- Chen, X., et al. The epididymal fat pad as a transplant site for minimal islet mass. Transplantation. 84 (1), 122-125 (2007).
- Sakata, N., et al. Mechanism of Transplanted Islet Engraftment in Visceral White Adipose Tissue. Transplantation. 104 (12), 2516-2527 (2020).
- Navarro-Requena, C., et al. PEG hydrogel containing calcium-releasing particles and mesenchymal stromal cells promote vessel maturation. Acta Biomaterialia. 67, 53-65 (2018).
- Phelps, E. A., Headen, D. M., Taylor, W. R., Thule, P. M., Garcia, A. J. Vasculogenic bio-synthetic hydrogel for enhancement of pancreatic islet engraftment and function in type 1 diabetes. Biomaterials. 34 (19), 4602-4611 (2013).
- Manzoli, V., et al. Immunoisolation of murine islet allografts in vascularized sites through conformal coating with polyethylene glycol. American Journal of Transplantation. 18 (3), 590-603 (2018).
- Gotoh, M., Maki, T., Kiyoizumi, T., Satomi, S., Monaco, A. P. An improved method for isolation of mouse pancreatic islets. Transplantation. 40 (4), 437-438 (1985).
- Brandhorst, D., Brandhorst, H., Hering, B. J., Bretzel, R. G. Long-term survival, morphology and in vitro function of isolated pig islets under different culture conditions. Transplantation. 67 (12), 1533-1541 (1999).
- Noguchi, H., et al. Low-temperature preservation of isolated islets is superior to conventional islet culture before islet transplantation. Transplantation. 89 (1), 47-54 (2010).
- Itoh, T., et al. Low temperature condition prevents hypoxia-induced islet cell damage and HMGB1 release in a mouse model. Cell Transplantation. 21 (7), 1361-1370 (2012).
- Komatsu, H., et al. Optimizing Temperature and Oxygen Supports Long-term Culture of Human Islets. Transplantation. 103 (2), 299-306 (2019).
- Unger, R. H. Lipid overload and overflow: metabolic trauma and the metabolic syndrome. Trends in Endocrinology, Metabolism. 14 (9), 398-403 (2003).
- Mao, D., et al. A macroporous heparin-releasing silk fibroin scaffold improves islet transplantation outcome by promoting islet revascularisation and survival. Acta Biomaterialia. 59, 210-220 (2017).
- Wang, K., Wang, X., Han, C. S., Chen, L. Y., Luo, Y. Scaffold-supported Transplantation of Islets in the Epididymal Fat Pad of Diabetic Mice. Journal of Visualized Experiments. (125), e54995 (2017).
- Wang, X., Wang, K., Zhang, W., Qiang, M., Luo, Y. A bilaminated decellularized scaffold for islet transplantation: Structure, properties and functions in diabetic mice. Biomaterials. 138, 80-90 (2017).
- Rios, P. D., Zhang, X., Luo, X., Shea, L. D. Mold-casted non-degradable, islet macro-encapsulating hydrogel devices for restoration of normoglycemia in diabetic mice. Biotechnology and Bioengineering. 113 (11), 2485-2495 (2016).

