Anterior Segment Organ Culture Platform for Tracking Open Globe Injuries and Therapeutic Performance
Summary
Open globe eye injuries may go untreated for multiple days in rural or military-relevant scenarios, resulting in blindness. Therapeutics are needed to minimize loss of vision. Here, we detail an organ culture open globe injury model. With this model, potential therapeutics for stabilizing these injuries can be properly evaluated.
Abstract
Open globe injuries have poor visual outcomes, often resulting in permanent loss of vision. This is partly due to an extended delay between injury and medical intervention in rural environments and military medicine applications where ophthalmic care is not readily available. Untreated injuries are susceptible to infection after the eye has lost its watertight seal, as well as loss of tissue viability due to intraocular hypotension. Therapeutics to temporarily seal open globe injuries, if properly developed, may be able to restore intraocular pressure and prevent infection until proper ophthalmic care is possible. To facilitate product development, detailed here is the use of an anterior segment organ culture open globe injury platform for tracking therapeutic performance for at least 72 h post-injury. Porcine anterior segment tissue can be maintained in custom-designed organ culture dishes and held at physiological intraocular pressure. Puncture injuries can be created with a pneumatic-powered system capable of generating injury sizes up to 4.5 mm in diameter, similar to military-relevant injury sizes. Loss of intraocular pressure can be observed for 72 h post-injury confirming proper injury induction and loss of the eye's watertight seal. Therapeutic performance can be tracked by application to the eye after injury induction and then tracking intraocular pressure for multiple days. Further, the anterior segment injury model is applicable to widely used methods for functionally and biologically tracking anterior segment physiology, such as assessing transparency, ocular mechanics, corneal epithelium health, and tissue viability. Overall, the method described here is a necessary next step toward developing biomaterial therapeutics for temporarily sealing open globe injuries when ophthalmic care is not readily available.
Introduction
Open globe (OG) injuries can result in permanent loss of vision when not treated or at least stabilized following injury1. Delays, however, are prevalent in remote areas where access to ophthalmic intervention is not readily available, such as in rural areas or on the battlefield in military scenarios. When treatment is not readily available, the current standard of care is to protect the eye with a rigid shield until medical intervention is possible. In military medicine, this delay is currently up to 24 h, but it is anticipated to increase up to 72 h in future combat operations in urban environments where air evacuation is not possible2,3,4. These delays can be even longer in rural, remote civilian applications where access to ophthalmic intervention is limited5,6. An untreated OG injury is highly susceptible to infection and loss of intraocular pressure (IOP) due to the watertight seal of the eye being compromised7,8. Loss of IOP can impact tissue viability, making any medical intervention unlikely to restore vision if the delay between injury and therapeutic is too long9.
To enable the development of easy-to-apply therapeutics for sealing OG injuries until an ophthalmic specialist can be reached, a benchtop OG injury model was previously developed10,11. With this model, high-speed injuries were created in whole porcine eyes while IOP was captured by pressure transducers. Therapeutics can then be applied to assess their ability to seal the OG injury site12. However, as this model uses whole porcine eyes, it can only assess immediate therapeutic performance with no way of tracking longer-term performance across the possible 72 h window in which the therapeutic must stabilize the injury site until the patient reaches specialty care. As a result, an anterior segment organ culture (ASOC) OG injury model was developed and detailed in this protocol as a platform for tracking long-term therapeutic performance13.
ASOC is a widely used technique for maintaining avascular tissue of the anterior segment, such as the cornea, for multiple weeks post-enucleation14,15,16,17. The anterior segment is maintained under physiological IOP by perfusing fluid at physiological flow rates and preserving the trabecular meshwork outflow region, the tissue responsible for regulating IOP, during ASOC setup18,19. The ASOC platform can maintain tissue physiologically, induce an OG injury using a pneumatic-powered device, apply a therapeutic, and track injury stabilization for at least 72 h post-injury13.
Here, the protocol provides a step-by-step methodology for using the ASOC platform. First it details how to set up and fabricate the ASOC platform. Next, the protocol details how to aseptically dissect the anterior segment and maintain the trabecular meshwork, followed by setting up anterior segment tissue in custom-built organ culture dishes. Then, it details how to create open globe injuries and apply therapeutic immediately following injury. Lastly, the protocol provides an overview on characterization parameters that are possible for use with this method that assesses functional, mechanical, and biological properties of the eye and how well the injury was stabilized. Overall, this model provides a much-needed platform to accelerate product development for stabilizing and treating open globe injuries and improve the poor vision prognosis following injury.
Protocol
Before performing this protocol, be aware that there are legal and ethical requirements in place for the use of animals in research and training. If live animals are used for the source of ocular tissue, seek approval by the local ethical or legal authority (IACUC or Ethics committee, etc.) before beginning. If there is any question in obtaining approval for the use of animals, do not proceed. We previously determined and reported that fresh porcine eyes obtained and used within 24 h post-mortem compared closest to in vivo physiology and fared well for these studies (Animal Technologies, Tyler, TX, USA)10,13. No live animals were used throughout this protocol, using a tissue vendor to obtain tissue within 24 h.
NOTE: Prior to tissue arrival, fabricate the organ culture dishes (Supplementary Protocol 1), clamping rings (Supplementary Protocol 1), dish stands (Supplementary Protocol 1), pressure transducer data collection setup (Supplementary Protocol 2), and pneumatic puncture platform (Supplementary Protocol 3). Sterilize the dishes, tools, and supplies and prepare the work areas. It's useful to have a non-sterile area to perform gross dissection on the eyes, as they usually come with connective, extra orbital tissue attached. Execute these first steps on an open, clean work surface, and then transfer the eyes aseptically into a BSC II cabinet for micro-dissection (cabinet #1). Optimally, the BSC II cabinet utilized for micro-dissection is separated from the dish assembly BSC II cabinet (cabinet #2) to minimize airflow and maximize workspace. Set up the micro-dissection cabinet with a dissecting microscope and a way to visualize the work surface (camera or eyepieces protruding from the cabinet).
1. Sterilization steps, supplies (see Table of Materials for more details), and setup
- Prepare and gas-sterilize the following items (1 kit for each eye): ASOC dish, clamping ring, two fluidic connectors with O-rings, two 18 G needle hubs, four screws, two lengths of PE-100 tubing (length of distance should be long enough to extend from the dish inside the incubator to the syringe pump and pressure transducer data collection setup), two 18 G 90° bent needle hubs, two 3-way valves.
- Prepare and autoclave the following kits.
- Prepare and autoclave the micro-dissection instrument kit, containing one pair of fine forceps, one pair of Vannas scissors, one pair of medium toothed forceps, one pair of large scissors, cotton swabs, and a razor blade or scalpel.
- Prepare and autoclave the assembly instrument kit, containing one pair of medium toothed forceps, one pair of surgical scissors, and one L-key.
- Prepare and autoclave the daily kit (quantity: one per day of culture), containing one L-key to tighten clamping rings to dishes each day as needed.
- Autoclave four 100 mL beakers for disinfecting and storing eyes and anterior segments.
- Autoclave the puncture objects.
- Gather the following sterile items: Petri dish (1 dish/eye), gauze (1-2/eye), dish stand, 20 mL syringes (1/eye), 10 mL syringes (1/eye), nylon syringe filters (1/eye).
- Prepare sterile media: DMEM with 4% FBS, 1x Glutamax, 1x Gentamicin, 1x Antibiotic-Antimycotic (AA; approximately 30-40 mL complete media/eye).
- Prepare AA-PBS: PBS with 1x AA (~500 mL).
- Prepare the gross dissection tool pack: clean and dry large surgical scissors and forceps.
- Set up the non-sterile dissection workspace: Gather supplies from gross dissection tool pack, enucleated porcine eyes submerged in PBS and on ice, surgical drape, 100 mL beaker with PBS. Lay out the surgical drape and items required for gross dissection.
- Set up the sterile dissection workspace: Gather supplies from micro-dissection instrument kit, sterile gauze, dissecting microscope, betadine solution, sterile PBS, sterile media, four sterilized 100 mL beakers, sterile Petri dish. Aseptically transfer to BSC II cabinet #1. Set up the cabinet for visualizing eyes on the dissecting microscope.
- Set up the ASOC assembly workspace: Gather gas-sterilized kits (dish kit and lid kit), assembly instrument kit, sterile media, dish stands, sterile Petri dishes, sterile syringes, and syringe filters. Aseptically transfer to BSC II cabinet #2. Set up the cabinet for dish assembly.
- When eyes are stabilized and ready for puncture (72 h post-setup), aseptically transfer them to a BSC II cabinet. Set up the OG injury induction workspace: Pneumatic powered injury induction device (assembly detailed in Supplemental 3) and Lab Jack and cross-tracking vise to hold the ASOC dish.
2. Dissection of tissue
- Prepare the porcine tissue using non-sterile dissection workspace.
- Procure enucleated porcine eyes from a local abattoir, animal studies, or vendor. Maintain the eyes on ice submerged in PBS during delivery and use immediately upon receiving.
- Cut away the extraorbital tissue and trim the conjunctiva leaving only the corneoscleral shell and optic nerve. Perform the dissection under non-sterile conditions with large surgical scissors and forceps in a gross dissection tool pack.
- Place the eyes back into fresh PBS on ice until all eyes required for experimental setup have been preliminary/gross dissected.
- Submerge the eyes in 10% betadine solution for 2 min in closed containers and transfer aseptically to BSC II cabinet #1. Perform all the subsequent works under sterile conditions to minimize contaminations during setup.
- Sterilely dissect anterior segments.
- After 2 min in betadine solution, transfer the eyes into three serial washes of sterile AA-PBS to remove excess betadine solution from the ocular surface while maintaining sterility of the ocular tissue. After three washes, maintain the tissue in AA-PBS until further use.
- Hemisect the eye using a razor blade/scalpel and curved scissors. Place the eye on an AA-PBS-soaked gauze and create an incision with a sterile razor blade or scalpel near the equator of the eye (60/40 split with 40 on the anterior side). Using curved surgical scissors, hemisect the eye to isolate the anterior eye (corneal half).
NOTE: The cut around the anterior segment needs to be continuous to prevent jagged, rough edges in the sclera that will create fluid leaks after setup in the organ culture. - Use microscissors as a shovel to scoop vitreous humor from the anterior segment. Remove the lens from the anterior segment using microscissors. Leave the anterior segments in AA-PBS until further dissection steps.
NOTE: All the eyes that will be dissected can be held at this step and one by one taken through the remainder of the dissection process. - With a dissecting microscope, cut back iris to iris root gradually, radially until trabecular meshwork (TM) is visible. The TM is a pigmented tissue that comprises fibers circumferentially oriented around the corneoscleral shell. Careful cuts into the iris toward the iris root will expose the depth of the TM under the tissue.
- Cut 360° around the iris at the same depth as the initial cut into the tissue to expose the entire TM region. Clean up any remaining residual iris covering TM as is necessary.
- Trim the ciliary body remnants posterior to the TM, leaving only a thin band of tissue posterior to the TM region (approximately 1 mm).
- Place the dissected anterior segment (AS) in media until further setup in ASOC in BSC II cabinet #2.
NOTE: All eyes can be held at this point prior to ASOC setup if a single user is performing dissection and organ culture dish assembly.
3. Setting up anterior segments in organ culture dishes
- Place a single AS in a Petri dish with AS inverted (cup up). Using a cotton swab, wet in the media and gently dab in the center of the cornea to remove any pigment. Using forceps to hold the eye and the same swab, wipe the cotton swab around the sclera to remove extra pigment.
- Invert the AS and place on top of bottom part of the dish over the elevated region, centering the cornea over the elevated region in the dish. Place the clamping ring on top of the newly placed AS.
- Place four screws into the corresponding holes to hold the ring in place with the AS under the ring. Gently hand-tighten the screws with the L-key.
NOTE: The tightening step will happen daily throughout the experiment, so the goal of the initial tightening here is to ensure the media does not leak while avoiding breaking the clamping ring. - With a sterile Petri dish set, place the top over the dish and invert the setup. Attach the dish stand. Attach the fluidic connectors with O-rings to threaded ports on the bottom of the dish.
- To one fluidic connector, attach an 18 G 90° bent needle hub, a length of tubing, an 18 G needle hub, a nylon syringe filter, a three-way valve, and a 20 mL syringe filled with media.
- To the second fluidic connector, attach an 18 G 90° degree bent needle hub, length of tubing, an 18 G needle hub, a three-way valve, and the barrel portion of a sterile 10 mL syringe (this will act as a reservoir to catch liquid and bubbles from the ASOC).
- With three-way valves open appropriately to the syringes, gently push media through the system using the fluidics connector port identified in step 3.5 to inflate the AS, fill media in the tubing, and eventually the reservoir.
NOTE: If media leaks into the ASOC dish, the AS is not secured tightly enough with the clamping ring. - Remove bubbles by gently pushing media into the dish and inverting the dish to push out the bubbles and into the reservoir.
- Place the dish and stand upright. Place the bottom portion of a Petri dish underneath the feet of the stand, careful not to ensnarl the tubing.
4. Starting anterior segment organ culture
- ASOC is now ready for incubation. Place the ASOC dish into the cell culture incubator (37 °C, 5% CO2). Ensure the height of the ASOC dish in the incubator above the pressure transducers is known and accounted for to calculate IOP accurately (Supplementary Protocol 4).
- Direct the tubing lines out through the bottom of the 37 °C, 5% CO2 incubator door so that they do not interfere with the opening and closing of the door. Attach the 20 mL syringe to the syringe pump set at 2.5 µL/min.
- Position the tubing line with the reservoir at the pressure transducer instrument. Connect the side 3-way valve to the pressure transducer setup while flowing PBS through the line to avoid air bubbles entering the tubing lines.
NOTE: Empty the PBS from the reservoirs after the system is set up to reduce the likelihood of reservoir contamination with microbial growth for the duration of the organ culture. - Initiate IOP data collection by first ensuring a microSD card is present for saving data files. Then, turn on the pressure transducer setup to begin data collection.
NOTE: Details for setting up the pressure transducer data collection device are provided in Supplementary Protocol 2.
5. Daily maintenance of ASOC
- After the ASOC has had 24 h to equilibrate, remove the dishes from the 37 °C, 5% CO2 incubator and place them into the BSC II cabinet.
NOTE: On pressure data acquisition, these time periods look like spikes as the ASOCs are removed from the incubator (height change) and adjusted in the cabinet. - Check for leaks under each dish on the Petri plate. If present, check for tight fluidic connections under the dish and re-tighten if necessary. Check for leaks in the dish top using a sterile L-key to tighten the screws in the clamping ring.
NOTE: The AS sclera tissue will compress and reduce thickness by 24 h, and the clamping ring will need to be tightened. - Aspirate the media from the dish well.
NOTE: The trabecular meshwork is filtering fluid from the media being pumped into the ASOC. Therefore, media will be present in the ASOC dish along the edges. - Repeat steps 3.7 and 3.8 to remove any trapped air bubbles.
- Refill syringes on the syringe pumps, ensure the syringe pumps are operating, and confirm the alignment of valves for perfusion into the ASOC. Return the ASOC dish to the 37 °C, 5% CO2 incubator.
NOTE: Optimally, these steps should be performed daily. However, the use of a 20 mL starting volume of media, the ASOC dish well volume, and a pump rate of 2.5 µL/min should be sufficient to let the system run for several days undisturbed.
6. OG injury induction with pneumatic-powered puncture device
NOTE: Construction of the pneumatic puncture device is detailed in Supplementary Protocol 3. OG injuries are induced after IOP has stabilized, which normally occurs after 3 days in culture. Acceptable IOP values are 5-20 mmHg based on physiological IOP, which can be determined by evaluating the IOP data files or setting LED indicators in the pressure measurement system as described in Supplementary Protocol 2.
- Prepare the BSC II cabinet for OG injury induction as detailed in step 1.10. Connect the puncture platform to a compressed air line. Attach the sterile puncture object to the chuck.
NOTE: An air compressor can be used to power the device, but tank compressed air or built-in laboratory lines can suffice if pressure is greater than 50 psi. - Set the pressure regulator on the puncture platform to 50 psi for adequate puncture force on objects up to 4.5 mm in diameter. Position the cross-tracking vise on the lab jack in front of the puncture platform to hold the ASOC dish during injury induction.
- Remove ASOC setup from 37 °C, 5% CO2 incubator, and place in the cross-tracking vise perpendicular to the puncture platform (Figure 2) after removing the lid and setting it aside. Keep the anterior segment perfusing but close off the 3-way valve port to the pressure transducer to prevent over-pressurization damage to the transducer.
- Extend the piston arm to its maximal distance and position the corneal apex within 1 mm of the puncture object. Retract the piston arm and bring the anterior segment 1 cm closer to the puncture object.
NOTE: This distance has been optimized for high-efficiency injury induction without hitting the ASOC dish. - Fire the puncture device by turning it on and opening the solenoid valve with the second switch on the device. To retract the device, press the second switch again to remove the puncture device from the eye. Verify proper injury induction by visual inspection and media leaking from the injury site.
- Remove ASOC dish from the vise; place the lid back onto the dish assembly and open the fluidic line to the pressure transducer. Place the ASOC back into the 37 °C, 5% CO2 incubator.
NOTE: At this point, therapeutic can be applied to the AS to assess its efficacy for sealing OG injuries.
7. Removing ASOC from culture
NOTE: Depending on endpoint analysis (see Representative Results for possible endpoint methods), the AS needs to remain in the ASOC dish inflated while other methods require AS tissue isolated from the culture chamber. The below methodology describes how to take AS out of the organ culture dishes and to remove the rest of the setup.
- Remove the ASOC dishes from the 37 °C, 5% CO2 incubator. Close the 3-way valve to the syringe and reservoir and disconnect the tubing from the system. Discard the syringe, reservoir, and filter. Place the 3-way valves, tubing, and needle hubs in a separate container for washing and sterilizing.
- Disconnect the needle hubs from the fluidic connections on the bottom of the dish. Unthread the fluidic connectors and o-rings. Place all items in a container for washing and sterilizing.
- Remove the four screws from the clamping ring using the L-key. Carefully remove the clamping ring.
- Using forceps, remove the AS from the dish and, depending on endpoint analysis and image, place in fixative or appropriate biohazard waste.
8. IOP data analysis
- Connect the microSD card to a computer to remove the .txt file containing data from the most recent experimental run.
NOTE: The file is named in the code controlling the microcontroller and should be updated for each experiment (see Supplementary Protocol 2). - Import the data into a spreadsheet.
- Organize the data into 12 columns: Time (in min) and mV signal for each of 11 pressure transducer channels. The first ten channels correspond to ten ASOC experimental setups. The final transducer signal is for a sensor open to air as a control channel to confirm mV signal did not alter due to changes in the input signal. Plot control channel versus time to confirm mV signal was consistent throughout.
- Convert the mV signals for ten channels into mmHg using slope-intercept equations generated from the initial calibration of each sensor (see Supplementary Protocol 4).
- Plot time (convert to days to simplify data interpretation) versus each of the ten channels to determine how the IOP fluctuated across the experimental time course.
- Determine average IOP values at key points in the data to more easily compare values between each and how they are altered before and after OG injury induction. Average 2-3 h of data for each 24 h interval to determine IOP at each day of ASOC.
NOTE: Representative IOP results are shown in Figure 4.
Representative Results
Images captured via Optical Coherence Tomography (OCT) are shown for OG injured eyes to illustrate how a successful injury induction looks. Figure 3 shows images for control and OG injured AS tissue immediately after injury and 72 h later. Two views are shown: cross-sectional images through the injury site and top-down maximum intensity projection (MIPs) to visualize the surface area of the image. Control eyes show no noticeable disruption in the cornea, while clear injuries can be located that cross the entire cornea after OG injury. From MIPs, it is evident that injuries are irregular in shape and size, but the injury size does decrease over 72 h. Previously, this effect has shown to be significant for a number of injury sizes tested13.
The primary data output for the OG injury model described in this protocol is intraocular pressure over the course of the experimental setup. Data is recorded in units of millivolts as an output from each pressure transducer which can be converted into mmHg via calibration (Supplementary Protocol 4). Example IOP data vs the experimental time course is provided for eyes that are considered acceptable and others that would not be considered usable (Figure 4A). From the pressure trace data, eyes were attached to sensors after 24 h in culture, but IOP continues to fluctuate over the first 72 h in culture. Physiological IOP for AS tissue in organ culture is approximately 8-10 mmHg, so 2x and ½x range was decided upon as a gate for usable IOP values after values have stabilized (5-20 mmHg). Only eyes that were in that range would be allowable for use with the remainder of the protocol. From prior experiments, we had a 90% success rate that was achieved in ASOC setup for eyes stabilizing in the required range (Figure 4B).
The results for how IOP changes due to OG injury and therapeutic intervention are also provided (Figure 4C,D). After OG injury induction, pressure should significantly drop and remain that way until the tissue is removed from ASOC (Figure 4C). If an eye after injury induction does not decrease in pressure, this indicates that a successful injury was not induced as IOP should be reduced if the watertight seal of the eye is compromised. However, smaller injury sizes may self-heal, which could result in IOP being restored. If therapeutic is applied to the eye after OG injury induction, restoration of IOP can be tracked during ASOC. This concept is demonstrated with data showing a Dermabond adhesive applied to 2.4 mm OG injuries (Figure 4D). Average results for five separate ASOC experiments with and without therapeutic are shown and it is evident the therapeutic is increasing IOP. This method can measure the efficacy of the therapeutic for restoring IOP and track whether that pressure is restored across the key 72 h post-OG injury.
Further, the ASOC protocol is adaptable for use with a wide range of characterization endpoints to meet the end user's experimental requirements. During culture, outflow media leaving the eye can be collected on a daily or even hourly basis which can be utilized for tracking protein level changes occurring during ASOC, after OG injury induction, or after therapeutic is applied. For instance, gelatin zymography has been previously performed to detect matrix metalloproteinase levels to track wound healing and tissue remodeling20. Further biological endpoints are possible after removing tissue from culture via traditional immunohistochemistry methods for assessing tissue viability21,22, tracking pathophysiological changes to the cornea23,24, or antibody-based staining for any protein of interest25,26.
Functional corneal metrics can also be obtained from eyes maintained in ASOC. Corneal epithelium integrity can be assessed via a fluorescein eye stain and image acquisition using a blue light source27,28. After removal from culture, corneal tissue can be assessed for transparency through simple image acquisition13. Traditional ocular imaging can also be performed to assess tissue structure with or without therapeutic intervention. OCT images, as shown in Figure 3, can create cross sectional images through the cornea and can be captured non-invasively, potentially allowing image collection while maintaining tissue in culture. Other imaging modalities such as slit-lamp microscopy, ultrasound, or in vivo confocal microscopy can also be adapted for acquiring further anatomical information.
Lastly, assessment of mechanical properties of the anterior segment can be captured to understand the effect of the OG injury or subsequent therapeutic on the underlying tissue. While IOP data collection alone highlights how the integrity of the watertight seal of the eye has been compromised, we have previously shown that additional test metrics can be measured to tease out additional mechanical features10,11. Ocular compliance, a lumped mechanical property describing how intraocular pressure changes due to inflation (change in volume/change in pressure), can be measured with a syringe pump to inject sudden small volumes of fluid into the eye and recording the resulting pressure increase with a pressure transducer. Higher compliance indicates the tissue is less stiff and can be used to track how therapeutic material properties differ from the underlying corneal tissue. Leak rate from the eye or a traditional outflow facility can be measured and calculated to determine the precise fluidic flow rate leaving the eye per unit of pressure20,29. Lastly, with regards to therapeutic testing, burst pressure can be measured to determine the maximum pressure the eye can hold prior to the therapeutic failing. This can be used to compare performance to uninjured eyes or to track changes in performance with time12,13.
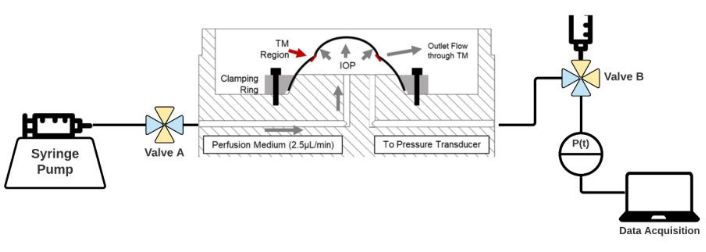
Figure 1: Diagram of the ASOC setup. Eyes are held in custom-built organ culture dishes and held in place with a clamping ring. ASOC media is infused via syringe pump through Valve A and connected to a pressure transducer, and subsequent data acquisition with Valve B. Open ports in each valve are highlighted in blue while yellow indicates closed channels. Please click here to view a larger version of this figure.

Figure 2: Overview of the OG injury setup. (A) Pneumatic powered injury device setup. From left to right, compressed air is introduced to the device via a compressed air line, which passes through a regulator to set pressure at 50 psi as measured by the pressure gauge. Two solenoid valves are connected to a linear actuator to direct expansion/retraction of the drill chuck holding the puncture object. Vise is positioned in front of the puncture device to hold the eye at the appropriate x, y, z positioning. (B) Representative ASOC is placed in front of the injury induction device. Further details of the device and its construction are detailed in Supplementary Protocol 3. Please click here to view a larger version of this figure.
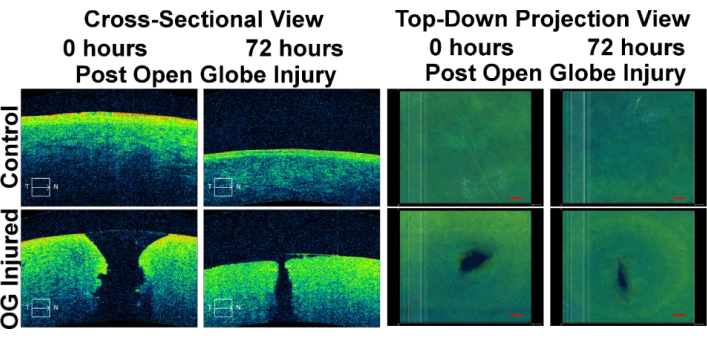
Figure 3: Optical Coherence Tomography Images of ASOC OG injury experiments. Images are shown for control eyes (uninjured) and OG injured eyes immediately post-injury and 72 h post-injury. Views are shown as cross-sections through the cornea (left side) and top-down maximum intensity projection views of the corneal surface (right side). The figure has been adapted with permission from Snider et al.13. Please click here to view a larger version of this figure.
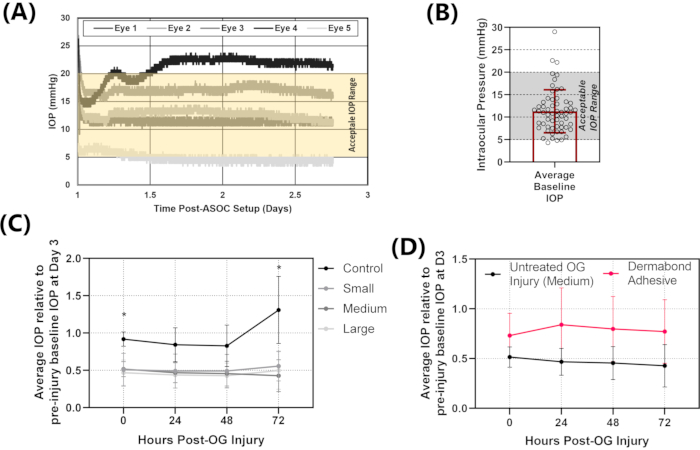
Figure 4: Representative IOP results for ASOC experiments. (A) Raw IOP data for the first 72 h of ASOC setup. Eyes are punctured at 72 h so the first 3 days of data are assessed to determine whether IOP stabilizes in the acceptable IOP range (5-20 mmHg). From the representative results, three of the five eyes fall within the acceptable IOP range, while one has IOP too high and one has IOP too low (falling outside of the highlighted yellow region on the plot). (B) Stabilized IOP for n = 50 ASOC setups from previous experiments to demonstrate the typical success rate with the ASOC method. (C) IOP for uninjured eyes compared to three different OG injury sizes after injury induction for 72 h. The loss of IOP is evident, with no signs of recovery. (D) Injured IOP results compared to injuries treated with a Dermabond adhesive. While the error rate is high due to some eyes being sealed and others not, the method can track changes to IOP over the 72 h period post-injury. The figure has been adapted with permission from Snider et al.13. Please click here to view a larger version of this figure.
Supplemental Files. Please click here to download these files.
Discussion
There are critical steps with the ASOC OG injury platform that should be highlighted to improve the likelihood of success when using the methodology. First, during the anterior segment dissection, preserving the trabecular meshwork is essential but challenging to do correctly. If the TM is disrupted, the eye will not maintain physiological pressure and will not meet eligibility criteria for experimental use. It is recommended to practice the dissection process under normal conditions first rather than introducing the additional aseptic technique challenges until proper dissections are obtained. Second, when setting the eyes in the ASOC dishes, it is imperative that they are tight enough to prevent fluid from leaking but loose enough to prevent damaging the ASOC dishes. If the eye is not secured tightly, fluid will leak out from the eye through non-physiological means resulting in little or no IOP. However, the clamping ring holding the eye down is plastic and can be easily broken if overtightened. It is essential to clamp the eyes down over 2 days as the scleral tissue under the ring will compress and loosen the tissue during the first 24 h. It is recommended to tighten the rings just until resistance to tightening is felt on day 1 and follow this up by re-tightening to similar levels after 24 h in culture for best results.
Third, it is critical to fully understand where fluid flow is directed at all times when using this model. Each ASOC dish is connected to multiple three-way valves to direct fluid flow from the syringe pump or 10 mL syringe reservoir and connect to pressure transducers. Different instances of the setup process require valves to be positioned in such a way so as to flush air bubbles from the eye or to protect pressure transducers from over-pressurization. Care should still be taken to understand what is open/close at all times prior to critical protocol steps. Lastly, maintaining sterility throughout the ASOC OG injury protocol is critical but easy to lose across the multi-step, multi-day process. Perfusion media contains high levels of antibiotics and antimycotics to prevent this, and eyes are submerged in betadine prior to set up to prevent contaminations, but there are still critical steps where mistakes are most likely. During the initial setup in the dish, avoid contact with the eyes while tightening clamping rings in place and keep lids on the dishes at all times when not in use. A more likely exposure step is during the day-to-day ASOC maintenance. It is important to do these routine steps in a biosafety cabinet, even if it seems they can be quickly accomplished without removing the eyes from the incubator. Carefully following the protocol and maintaining good aseptic technique should minimize contamination risks across the 6-day ASOC experiments.
Overall, the ASOC OG injury platform is unique from other methodologies looking at open globe injuries due to two key criteria. First is the injury induction method. The high-speed pneumatic injury device utilized induces injuries with a high force amount. This allows for inducing injuries with objects that are not especially sharp nor with a small diameter. This more closely mimics injuries that are irregular in shape; high-speed shrapnel injures resulting from explosive devices30,31. The pneumatic device can easily be fitted with irregular-shaped shrapnel mimicking objects to create injuries more challenging to heal compared to previous methods using lasers, needles, or scalpel blades to create clean, precise injury geometries32,33,34. Second, the ASOC methodology allows for tracking injury progress and therapeutic performance beyond initial injury induction. Being able to track out to 72 h was not possible in the previously developed benchtop OG injury platform10,11,12 and was the motivation behind developing this protocol. In fact, cell viability remained high in the corneal endothelium for at least 1 week in ASOC13. ASOC is the only means this long-term characterization can be accomplished without transitioning into costly in vivo experiments.
The main applications for the ASOC platform are two-fold. First, the model can be utilized for further characterizing open globe injuries, especially considering how they change with time. In the previous study, OG injuries were characterized in this manner and wound healing was observed over 72 h following injury13. Further tracking different injury sizes, shapes, locations for 72 h or even longer with regards to biological changes occurring will inform critical medical decisions that have to be made following OG injuries. Certain injury parameters may allow for self-healing by the cornea, or other parameters may be more severe if the intervention is not applied within the first 24 h. This information will be invaluable for triaging patients when limited medical supplies or evacuation resources are available.
Second, the ASOC OG platform can be used for developing and testing product development. For this application, the organ culture platform can fill a number of roles. During initial product development, shorter time frames can be tested with a range of product formulations to determine what is most effective. The organ culture system can be configured for even greater high-throughput for this application with additional syringe pumps to move beyond the ten simultaneous experiments possible with the system detailed here. For more refined products, longer time points can be evaluated to assess performance for 72 h or potentially even longer. Lastly, wound healing evaluation may be possible when evaluating biologically active products that may permanently treat OG injuries rather than temporary stabilization.
However, there are limitations with the ASOC OG platform that should be taken into consideration. First, while the model allows for longer-term assessment of therapeutics, it is missing all tissue of the eye outside of the corneoscleral shell, such as the iris and lens. These additional tissues are likely to be influenced by the OG injury and may play a role in injury progression. Similarly, an isolated anterior segment is missing immune response elements that would be included when transitioning from the ASOC model to subsequent animal testing. Next, the model is only suitable for creating corneal OG injures and potentially limbal OG injuries. Scleral or posterior OG injuries cannot be induced with this method. However, many of these injury types result in damage to the retina, making any temporary stabilization therapeutic unlikely to prevent loss of vision35,36. Lastly, injuries with the model out to 72 h post-injury were only tracked. ASOC has been utilized in other applications out to 2 weeks, so the model can likely be utilized for these applications, but it has not been tested at this time37,38,39.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This material is based upon work supported by the United States Department of Defense through an interagency agreement (#19-1006-IM) with the Temporary Corneal Repair acquisition program (United States Army Medical Materiel Development Agency).
Materials
| 10-32 Polycarbonate straight plug, male threaded pipe connector | McMaster-Carr | 51525K431 | |
| 10-32 Socket cap screw, ½" | McMaster-Carr | 92196A269 | |
| 10 mL syringe | BD | 302995 | |
| 20 mL syringe | BD | 302830 | |
| Anti-Anti | Gibco | 15240-096 | |
| Ball-End L key | McMaster-Carr | 5020A25 | |
| Betadine | Fisher Scientific | NC1696484 | |
| BD Intramedic PE 160 Tubing | Fisher Scientific | 14-170-12E | |
| Cotton swabs | Puritan | 25-8061WC | |
| DMEM media | ATCC | 30-2002 | |
| FBS | ATCC | 30-2020 | |
| Fine forceps | World Precision Instruments | 15914 | |
| Gauze | Covidien | 8044 | |
| Gentamicin | Gibco | 15710-064 | |
| Glutamax | Gibco | 35050-061 | |
| High temperature silicone O-ring, 2 mm wide, 4 mm ID | McMaster-Carr | 5233T47 | |
| Large forceps | World Precision Instruments | 500365 | |
| Large surgical scissors | World Precision Instruments | 503261 | |
| Medium toothed forceps | World Precision Instruments | 501217 | |
| Nail (puncture object) | McMaster-Carr | 97808A503 | |
| Nylon syringe filters | Fisher | 09-719C | |
| PBS | Gibco | 10010-023 | |
| Petri dish (100 mm) | Fisher | FB0875713 | |
| Polycarbonate, three-way, stopcock with male luer lock | Fisher | NC9593742 | |
| Razor blade | Fisher | 12-640 | |
| Stainless steel 18 G 90 degree angle dispensing needle | McMaster-Carr | 75165A81 | |
| Stainless steel 18 G straight ½'’ dispensing needle | McMaster-Carr | 75165A675 | |
| Sterile 100 mL beakers with lids | VWR | 15704-092 | |
| Vannas scissors | World Precision Instruments | WP5070 |
References
- Hilber, D., Mitchener, T. A., Stout, J., Hatch, B., Canham-Chervak, M. Eye injury surveillance in the US Department of Defense, 1996-2005. American Journal of Preventive Medicine. 38, 78-85 (2010).
- Linde, A. S., McGinnis, L. J., Thompson, D. M. Multi-Battle domain-perspective in military medical simulation trauma training. Journal of Trauma & Treatment. 06 (04), (2017).
- Riesberg, J., Powell, D., Loos, P. The loss of the golden hour. Special Warfare. , 49-51 (2017).
- Townsend, S., Lasher, W. . The US Army in Multi-Domain Operations 2028. (525-3-1), (2018).
- Blanch, R. J., Bishop, J., Javidi, H., Murray, P. I. Effect of time to primary repair on final visual outcome after open globe injury. The British Journal of Ophthalmology. 103 (10), 1491-1494 (2019).
- Lesniak, S. P., et al. Characteristics and outcomes of delayed open globe repair. Investigative Ophthalmology & Visual Science. 53 (14), 4954 (2012).
- Loporchio, D., Mukkamala, L., Gorukanti, K., Zarbin, M., Langer, P., Bhagat, N. Intraocular foreign bodies: A review. Survey of Ophthalmology. 61 (5), 582-596 (2016).
- Jonas, J. B., Budde, W. M. Early versus late removal of retained intraocular foreign bodies. Retina. 19 (3), 193-197 (1999).
- Watson, P. G., Jovanovik-Pandova, L. Prolonged ocular hypotension: would ciliary tissue transplantation help. Eye. 23 (10), 1916-1925 (2009).
- Snider, E. J., et al. Development and characterization of a benchtop corneal puncture injury model. Scientific Reports. 10 (1), 4218 (2020).
- Snider, E. J., et al. An open-globe porcine injury platform for assessing therapeutics and characterizing biological effects. Current Protocols in Toxicology. 86 (1), 98 (2020).
- Snider, E. J., Cornell, L. E., Gross, B., Zamora, D. O., Boice, E. N. Assessment of commercial off-the-shelf tissue adhesives for sealing military relevant corneal perforation injuries. Military Medicine. , (2021).
- Snider, E. J., Boice, E. N., Butler, J. J., Gross, B., Zamora, D. O. Characterization of an anterior segment organ culture model for open globe injuries. Scientific Reports. 11 (1), 8546 (2021).
- Erickson-Lamy, K., Rohen, J. W., Grant, W. M. Outflow facility studies in the perfused human ocular anterior segment. Experimental Eye Research. 52 (6), 723-731 (1991).
- Johnson, D. H., Tschumper, R. C. The effect of organ culture on human trabecular meshwork. Experimental Eye Research. 49 (1), 113-127 (1989).
- Johnson, D. H., Tschumper, R. C. Human trabecular meshwork organ culture. A new method. Investigative Ophthalmology & Visual Science. 28 (6), 945-953 (1987).
- Snider, E. J., et al. Improving stem cell delivery to the trabecular meshwork using magnetic nanoparticles. Scientific Reports. 8 (1), 12251 (2018).
- Llobet, A., Gasull, X., Gual, A. Understanding trabecular meshwork physiology: a key to the control of intraocular pressure. Physiology. 18 (5), 205-209 (2003).
- Goel, M., Picciani, R. G., Lee, R. K., Bhattacharya, S. K. Aqueous humor dynamics: A review. The Open Ophthalmology Journal. 4, 52-59 (2010).
- Snider, E. J., et al. Development of a porcine organ-culture glaucoma model mimicking trabecular meshwork damage. Investigative Ophthalmology & Visual Science. 62 (3), 18 (2021).
- Ren, H., Wilson, G. Apoptosis in the corneal epithelium. Investigative Ophthalmology & Visual Science. 37 (6), 1017-1025 (1996).
- Komuro, A., Hodge, D. O., Gores, G. J., Bourne, W. M. Cell death during corneal storage at 4°C. Investigative Ophthalmology & Visual Science. 40 (12), 2827-2832 (1999).
- Crespo-Moral, M., García-Posadas, L., López-García, A., Diebold, Y. Histological and immunohistochemical characterization of the porcine ocular surface. PLOS One. 15 (1), e0227732 (2020).
- Wilson, S. E., Medeiros, C. S., Santhiago, M. R. Pathophysiology of corneal scarring in persistent epithelial defects after prk and other corneal injuries. Journal of Refractive Surgery. 34 (1), 59-64 (2018).
- Auw-Haedrich, C., et al. Immunohistochemical expression of epithelial cell markers in corneas with congenital aniridia and ocular cicatrizing pemphigoid. Acta Ophthalmologica. 89 (1), 47-53 (2011).
- Lyngholm, M., et al. Immunohistochemical markers for corneal stem cells in the early developing human eye. Experimental Eye Research. 87 (2), 115-121 (2008).
- Bandamwar, K. L., Papas, E. B., Garrett, Q. Fluorescein staining and physiological state of corneal epithelial cells. Contact Lens & Anterior Eye: The Journal of the British Contact Lens Association. 37 (3), 213-223 (2014).
- Bandamwar, K. L., Garrett, Q., Papas, E. B. Sodium fluorescein staining of the corneal epithelium: What does it mean at a cellular level. Investigative Ophthalmology & Visual Science. 52 (14), 6496 (2011).
- Sherwood, J. M., Reina-Torres, E., Bertrand, J. A., Rowe, B., Overby, D. R. Measurement of outflow facility using iPerfusion. PLoS One. 11 (3), (2016).
- Weichel, E. D., Colyer, M. H., Ludlow, S. E., Bower, K. S., Eiseman, A. S. Combat ocular trauma visual outcomes during operations iraqi and enduring freedom. Ophthalmology. 115 (12), 2235-2245 (2008).
- Colyer, M. H., et al. Delayed intraocular foreign body removal without endophthalmitis during Operations Iraqi Freedom and Enduring Freedom. Ophthalmology. 114 (8), 1439-1447 (2007).
- Geggel, H. S., Maza, C. E. Anterior stromal puncture with the Nd:YAG laser. Investigative Ophthalmology & Visual Science. 31 (8), 1555-1559 (1990).
- Matthews, A., et al. Indentation and needle insertion properties of the human eye. Eye. 28 (7), 880-887 (2014).
- Rau, A., et al. The mechanics of corneal deformation and rupture for penetrating injury in the human eye. Injury. 49 (2), 230-235 (2018).
- Agrawal, R., Ho, S. W., Teoh, S. Pre-operative variables affecting final vision outcome with a critical review of ocular trauma classification for posterior open globe (zone III) injury. Indian Journal of Ophthalmology. 61 (10), 541 (2013).
- Knyazer, B., et al. Prognostic factors in posterior open globe injuries (zone-III injuries). Clinical & Experimental Ophthalmology. 36 (9), 836-841 (2008).
- Tan, J., et al. C3 Transferase-Expressing scAAV2 Transduces Ocular Anterior Segment Tissues and Lowers Intraocular Pressure in Mouse and Monkey. Molecular Therapy – Methods & Clinical Development. 17, 143-155 (2020).
- Bhattacharya, S. K., Gabelt, B. T., Ruiz, J., Picciani, R., Kaufman, P. L. Cochlin Expression in Anterior Segment Organ Culture Models after TGFβ2 Treatment. Investigative Ophthalmology & Visual Science. 50 (2), 551-559 (2009).
- Zhu, W., Godwin, C. R., Cheng, L., Scheetz, T. E., Kuehn, M. H. Transplantation of iPSC-TM stimulates division of trabecular meshwork cells in human eyes. Scientific Reports. 10 (1), 2905 (2020).

