Collection, Expansion, and Differentiation of Primary Human Nasal Epithelial Cell Models for Quantification of Cilia Beat Frequency
Summary
This protocol describes nasal epithelial cell collection, expansion, and differentiation to organotypic airway epithelial cell models and quantification of cilia beat frequency via live-cell imaging and custom-built scripts.
Abstract
Measurements of cilia function (beat frequency, pattern) have been established as diagnostic tools for respiratory diseases such as primary ciliary dyskinesia. However, the wider application of these techniques is limited by the extreme susceptibility of ciliary function to changes in environmental factors e.g., temperature, humidity, and pH. In the airway of patients with Cystic Fibrosis (CF), mucus accumulation impedes cilia beating. Cilia function has been investigated in primary airway cell models as an indicator of CF Transmembrane conductance Regulator (CFTR) channel activity. However, considerable patient-to-patient variability in cilia beating frequency has been found in response to CFTR-modulating drugs, even for patients with the same CFTR mutations. Furthermore, the impact of dysfunctional CFTR-regulated chloride secretion on ciliary function is poorly understood. There is currently no comprehensive protocol demonstrating sample preparation of in vitro airway models, image acquisition, and analysis of Cilia Beat Frequency (CBF). Standardized culture conditions and image acquisition performed in an environmentally controlled condition would enable consistent, reproducible quantification of CBF between individuals and in response to CFTR-modulating drugs. This protocol describes the quantification of CBF in three different airway epithelial cell model systems: 1) native epithelial sheets, 2) air-liquid interface models imaged on permeable support inserts, and 3) extracellular matrix-embedded three-dimensional organoids. The latter two replicate in vivo lung physiology, with beating cilia and production of mucus. The ciliary function is captured using a high-speed video camera in an environment-controlled chamber. Custom-built scripts are used for the analysis of CBF. Translation of CBF measurements to the clinic is envisioned to be an important clinical tool for predicting response to CFTR-modulating drugs on a per-patient basis.
Introduction
Measurements of Cilia Beat Frequency (CBF) and pattern have been established as diagnostic tools for respiratory diseases such as Primary Ciliary Dyskinesia (PCD)1. In Cystic Fibrosis (CF), dysfunction of the CF Transmembrane conductance Regulator (CFTR) chloride channel causes dehydration of the airway surface liquid and impaired mucociliary clearance2. Ciliary function has been investigated in vitro in primary airway cell models as an indicator of CFTR channel activity3. However, considerable patient-to-patient variability exists in CBF in response to CFTR-modulating drugs, even for patients with the same CFTR mutations3. Furthermore, the impact of dysfunctional CFTR-regulated chloride secretion on ciliary function is poorly understood. There is currently no comprehensive protocol demonstrating sample preparation of in vitro airway models, image acquisition, and analysis of CBF.
Nasal epithelial sheets isolated from nasal mucosal brushings are directly used for measurements of ciliary function for PCD diagnosis4. Yet, while there is no control over the size or quality of the nasal epithelial sheets obtained, CBF varies depending on whether it is measured on single cells or cell sheets and on epithelial sheet ciliated edges that are disrupted or undisrupted5. As such, secondary dyskinesias caused by damage to cells during the collection of nasal mucosal brushings may influence CBF. Primary cell culture of nasal epithelial cells and their differentiation at Air-Liquid Interface (ALI) or in three-dimensional basement membrane matrix into ciliated airway epithelial organoids give rise to cilia that are free from secondary dyskinesias4,6,7,8. Airway epithelial cells differentiated at ALI (henceforth termed ALI models) have been deemed an important secondary diagnostic aid that replicate the ciliary beat patterns and frequency of ex vivo nasal mucosal brushings6 and enable analysis of ciliary ultrastructure, beat pattern, and beat frequency while retaining patient-specific defects9. Yet, discrepancies exist in the methodologies used to create these pseudostratified, mucociliary differentiated cell models. Different culture expansion or differentiation protocols could induce distinct epithelial phenotypes (ciliated or secretory)10 and result in significant differences in CBF11. CBF has been quantified in nasal epithelial brushings4,6,12,13,14,15,16, airway epithelial organoids14,17,18 and ALI models3,4,6,13,19,20,21. Yet, amongst these protocols, there are large variabilities, and often many parameters are not controlled for. For example, in some studies, CBF is imaged in situ while the cells of the ALI model remain on the permeable support insert3,19,20,21, yet others scrape the cells from the permeable support insert and image them suspended in media4,6,13.
Furthermore, the wider application of techniques that measure ciliary function is limited by the extreme susceptibility of ciliary function to changes in environmental factors. Environmental factors such as temperature22, humidity23,24, and pH25,26 influence ciliary function and must be regulated to quantify CBF accurately. The various physiological parameters used across different laboratories and how they influence CBF has been reviewed previously27.
Various imaging technologies and approaches to CBF measurements are reported in the literature. For PCD diagnostics, video microscopy is used to measure ciliary function28,29. Recently, a video analysis algorithm based on differential dynamic microscopy was used to quantify both CBF and cilia coordination in airway epithelial cell ALI models3,30. This method enables the characterization of ciliary beating in airway epithelial cells in a fast and fully automated manner, without the need to segment or select regions. Various methods for imaging and quantification of CBF may add to the differences reported in CBF in the literature (Supplementary File 1).
A protocol from culture to quantification to streamline existing methods, standardization of culture conditions, and image acquisition, performed in strict environmentally controlled conditions, would enable consistent, reproducible quantification of CBF within and between individuals.
This protocol provides a complete description of the collection of epithelial cells, expansion and differentiation culture conditions, and quantification of CBF in three different airway epithelial cell model systems of nasal origin: 1) native epithelial sheets, 2) ALI models imaged on permeable support inserts and 3) Extracellular Matrix (ECM)-embedded three-dimensional organoids (Figure 1). Nasal epithelial cells obtained from nasal inferior turbinate brushings are used as representatives of the airway epithelium since they are an effective surrogate for bronchial epithelial cells31 while overcoming the invasive procedure associated with collecting bronchial brushings. The Conditional Reprogramming Cell (CRC) method is used to expand primary airway epithelial cells for the creation of ALI models and three-dimensional organoids. Conditional reprogramming of airway epithelial cells to a stem cell-like state is induced by co-culture with growth-arrested fibroblast feeder cell system and Rho-associated kinase (ROCK) inhibitor32. Importantly, the CRC method increases population doubling in airway epithelial cells while retaining their tissue-specific differentiation potential33,34. In all airway epithelial cell models, the ciliary function is captured in a temperature-controlled chamber using a high-speed video camera with standardized image acquisition settings. Custom-built scripts are employed for the quantification of CBF.
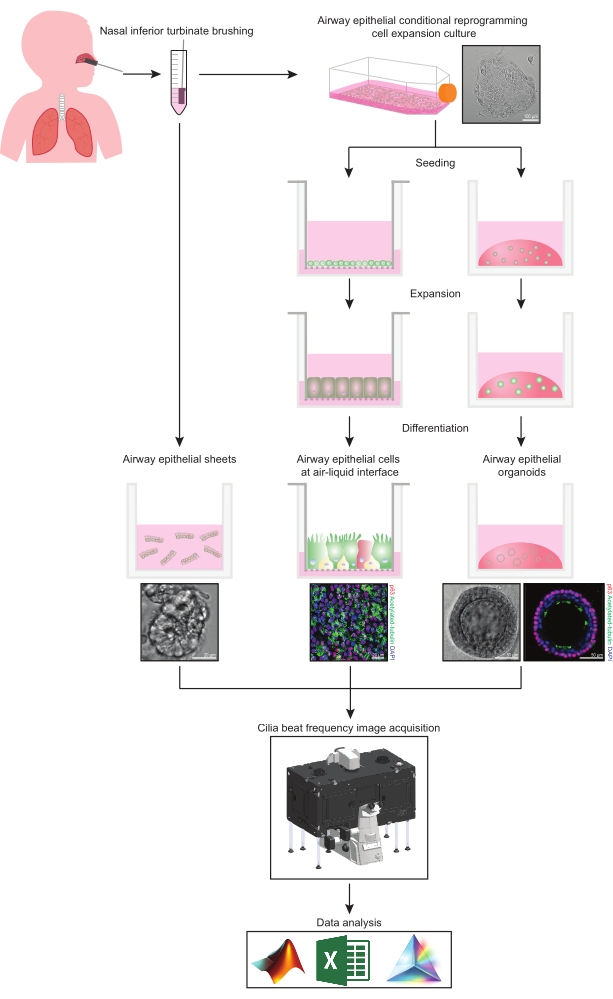
Figure 1: Schematic of workflow. Following brushing the participants' nasal inferior turbinate, airway epithelial cells are utilized in one of two ways. Either airway epithelial sheets are isolated, and cilia beat frequency is imaged immediately, or airway epithelial cells are expanded via the conditional reprogramming cell method. CRC-expanded airway epithelial cells are differentiated to establish airway epithelial cells at an air-liquid interface or airway epithelial organoid cultures. Imaging of ciliary beat frequency is acquired using a live-cell imaging microscope with a heating and humidity environmental chamber and a fast frame rate (>100Hz) scientific camera. Data analysis is performed using custom-built scripts. Please click here to view a larger version of this figure.
Protocol
Representative Results
Discussion
There are multiple factors that could obscure the quantification of CBF in nasal epithelial sheets. Epithelial sheets should be imaged within 3-9 hrs of sample collection since the ciliary function is most stable during this time37. Less red blood cells and debris are most optimal for imaging since these interfere with data acquisition. When selecting an ROI for imaging, it is important to select an epithelial sheet that edge has not been damaged or disrupted during the collection of the sample, a…
Divulgations
The authors have nothing to disclose.
Acknowledgements
We thank the study participants and their families for their contributions. We appreciate the assistance from Sydney Children's Hospitals (SCH) Randwick respiratory department in the organization and collection of patient biospecimens – special thanks to Dr. John Widger, Dr. Yvonne Belessis, Leanne Plush, Amanda Thompson, and Rhonda Bell. We acknowledge the assistance of Iveta Slapetova and Renee Whan from the Katharina Gaus Light Microscopy Facility within the Mark Wainwright Analytical Centre at UNSW Sydney. This work is supported by National Health and Medical Research Council (NHMRC) Australia (GNT1188987), CF Foundation Australia, and Sydney Children's Hospital Foundation. The authors would like to acknowledge Luminesce Alliance – Innovation for Children's Health for its contribution and support. Luminesce Alliance – Innovation for Children's Health is a not-for-profit cooperative joint venture between the Sydney Children's Hospitals Network, the Children's Medical Research Institute, and the Children's Cancer Institute. It has been established with the support of the NSW Government to coordinate and integrate pediatric research. Luminesce Alliance is also affiliated with the University of Sydney and the University of New South Wales Sydney. KMA is supported by an Australian Government Research Training Program Scholarship. LKF is supported by the Rotary Club of Sydney Cove/Sydney Children's Hospital Foundation and UNSW University postgraduate award scholarships.
Materials
| Adenine | Sigma-Aldrich | A2786 | 10 mg/mL |
| Advanced DMEM/F-12 | Thermo Fisher Scientific | 12634-010 | |
| Alanyl-glutamine | Sigma-Aldrich | G8541 | 200 mM |
| Andor Zyla 4.2 sCMOS | Oxford Instruments | Fast frame rate (>100 Hz) scientific camera | |
| Bottle-top vacuum filter system | Sigma-Aldrich | CLS431098 | |
| Ceftazidime hydrate | Sigma-Aldrich | A6987 | 50 mg/mL |
| Cell Culture Microscope | Olympus | CKX53 | |
| CFI S Plan Fluor ELWD 20XC | Nikon Instruments Inc. | MRH08230 | Long working distance objective lens. NA0.45 WD 8.2-6.9 |
| Cholera toxin | Sigma-Aldrich | C8052-1MG | 200 µg/mL |
| Corning Gel Strainer 40 UM | Sigma-Aldrich | CLS431750 | Pore size 40 μm |
| Corning Matrigel Matrix (Phenol red-free) | Corning | 356231 | Extracellular matrix (ECM) |
| Corning bottle-top vacuum filter system | Sigma-Aldrich | CLS431098 | |
| Corning CoolCell LX Cell Freezing Container | Sigma-Aldrich | CLS432002 | |
| Corning Transwell polyester membrane cell culture inserts | Sigma-Aldrich | CLS3470 | Permeable support inserts. 6.5 mm Transwell with 0.4 μm pore polyester membrane insert. |
| Countess Cell Counting Chamber Slides | Thermo Fisher Scientific | C10228 | |
| Countess II Automated Cell Counter | ThermoFisher Scientific | AMQAX1000 | Automated cell counter |
| Cytology brushes | McFarlane Medical | 33009 | |
| DMEM/F12-Ham | Thermo Fisher Scientific | 11330032 | |
| DMEM/F12-Ham | Thermo Fisher Scientific | 11330032 | |
| DMEM-High Glucose | Thermo Fisher Scientific | 11965-092 | |
| Dulbecco′s Phosphate Buffered Saline (PBS) | Sigma-Aldrich | D8537 | |
| Eclipse Ti2-E | Nikon | Live-cell imaging microscope. | |
| Fetal Bovine Serum, certified, heat inactivated, United States | Thermo Fisher Scientific | 10082147 | |
| Fungizone (Amphotericin B) | Thermo Fisher Scientific | 15290018 | 250 µg/mL |
| Gentamicin solution | Sigma-Aldrich | G1397 | 50 mg/mL |
| Graphpad Prism | Graphpad | Scientific analysis software | |
| Greiner Cryo.s vials | Sigma-Aldrich | V3135 | Cryogenic vials |
| HEPES solution | Sigma-Aldrich | H0887 | 1 M |
| HI-FBS | Thermo Fisher Scientific | 10082-147 | |
| Hydrocortisone | Sigma-Aldrich | H0888 | 3.6 mg/mL |
| Incubator NL Ti2 BLACK 2000 | PeCon | Microscope environmental chamber. Allows warm air incubation and local CO2 and O2 gassing | |
| Insulin | Sigma-Aldrich | I2643 | 2 mg/mL |
| Lab Armor 74220 706 Waterless Bead Bath 6L | John Morris Group | 74220 706 | Bead bath |
| Lab Armor Beads | Thermo Fisher Scientific | A1254302 | Thermal beads |
| MATLAB | MathWorks | Computing software | |
| Microsoft Excel | Microscoft | Spreadsheet software | |
| NIH/3T3 | American Type Culture Collection | CRL-1658 | Irradiated NIH-3T3 mouse embryonic feeder cells |
| NIS-Elements AR | Nikon Instruments Inc. | Image acquisition software | |
| Penicillin-Streptomycin | Sigma-Aldrich | P4333 | 10,000 units penicillin and 10 mg streptomycin/mL |
| Dulbecco′s Phosphate Buffered Saline (PBS) | Sigma-Aldrich | D8537 | |
| PneumaCult Airway Organoid Kit | StemCell Technologies | 5060 | Airway Organoid Kit |
| PneumaCult-ALI Medium | StemCell Technologies | 5001 | |
| PneumaCult-Ex Plus Medium | StemCell Technologies | 5040 | |
| PureCol-S | Advanced BioMatrix | 5015 | Type I Collagen solution |
| ReagentPack Subculture Reagents | Lonza | CC-5034 | |
| rhEGF (Epidermal Growth Factor, human) | Sigma-Aldrich | E9644 | 25 µg/mL |
| Y-27632 2HCl (ROCK inhibitor) | Selleckchem | S1049 | 10 mM |
| Tobramycin | Sigma-Aldrich | T4014 | 100 mg/mL |
| Trypan blue solution | Sigma-Aldrich | T8154 | 0.4% |
| UNO Stage Top Incubator | Okolab | Microscope incubator. Allows temperature, humidity and CO2 conditioning |
References
- Barbato, A., et al. Primary ciliary dyskinesia: a consensus statement on diagnostic and treatment approaches in children. European Respiratory Journal. 34 (6), 1264-1276 (2009).
- Cutting, G. R. Cystic fibrosis genetics: from molecular understanding to clinical application. Nature Reviews Genetics. 16 (1), 45-56 (2015).
- Chioccioli, M., Feriani, L., Kotar, J., Bratcher, P. E., Cicuta, P. Phenotyping ciliary dynamics and coordination in response to CFTR-modulators in Cystic Fibrosis respiratory epithelial cells. Nature Communications. 10 (1), 1763 (2019).
- Hirst, R. A., Rutman, A., Williams, G., O’Callaghan, C. Ciliated air-liquid cultures as an aid to diagnostic testing of primary ciliary dyskinesia. Chest. 138 (6), 1441-1447 (2010).
- Thomas, B., Rutman, A., O’Callaghan, C. Disrupted ciliated epithelium shows slower ciliary beat frequency and increased dyskinesia. European Respiratory Journal. 34 (2), 401-404 (2009).
- Coles, J. L., et al. A revised protocol for culture of airway epithelial cells as a diagnostic tool for primary ciliary dyskinesia. Journal of Clinical Medicine. 9 (11), (2020).
- Pifferi, M., et al. Simplified cell culture method for the diagnosis of atypical primary ciliary dyskinesia. Thorax. 64 (12), 1077-1081 (2009).
- Pifferi, M., et al. Rapid diagnosis of primary ciliary dyskinesia: cell culture and soft computing analysis. European Respiratory Journal. 41 (4), 960-965 (2013).
- Lee, D. D. H., et al. Higher throughput drug screening for rare respiratory diseases: Readthrough therapy in primary ciliary dyskinesia. European Respiratory Journal. 58 (4), 2000455 (2021).
- Saint-Criq, V., et al. Choice of differentiation media significantly impacts cell lineage and response to CFTR modulators in fully differentiated primary cultures of cystic fibrosis human airway epithelial cells. Cells. 9 (9), (2020).
- Awatade, N. T., et al. Significant functional differences in differentiated Conditionally Reprogrammed (CRC)- and Feeder-free Dual SMAD inhibited-expanded human nasal epithelial cells. Journal of Cystic Fibrosis. 20 (2), 364-371 (2021).
- Dabrowski, M., Bukowy-Bieryllo, Z., Jackson, C. L., Zietkiewicz, E. Properties of non-aminoglycoside compounds used to stimulate translational readthrough of PTC mutations in primary ciliary dyskinesia. International Journal of Molecular Sciences. 22 (9), (2021).
- Hirst, R. A., et al. Culture of primary ciliary dyskinesia epithelial cells at air-liquid interface can alter ciliary phenotype but remains a robust and informative diagnostic aid. PloS One. 9 (2), 89675 (2014).
- Marthin, J. K., Stevens, E. M., Larsen, L. A., Christensen, S. T., Nielsen, K. G. Patient-specific three-dimensional explant spheroids derived from human nasal airway epithelium: a simple methodological approach for ex vivo studies of primary ciliary dyskinesia. Cilia. 6, 3 (2017).
- Chilvers, M. A., O’Callaghan, C. Analysis of ciliary beat pattern and beat frequency using digital high speed imaging: comparison with the photomultiplier and photodiode methods. Thorax. 55 (4), 314-317 (2000).
- Chilvers, M. A., Rutman, A., O’Callaghan, C. Functional analysis of cilia and ciliated epithelial ultrastructure in healthy children and young adults. Thorax. 58 (4), 333-338 (2003).
- Castillon, N., et al. Polarized expression of cystic fibrosis transmembrane conductance regulator and associated epithelial proteins during the regeneration of human airway surface epithelium in three-dimensional culture. Laboratory Investigation. 82 (8), 989-998 (2002).
- Jorissen, M., Bessems, A. Normal ciliary beat frequency after ciliogenesis in nasal epithelial cells cultured sequentially as monolayer and in suspension. Acta Oto-Laryngologica. 115 (1), 66-70 (1995).
- Conger, B. T., et al. Comparison of cystic fibrosis transmembrane conductance regulator (CFTR) and ciliary beat frequency activation by the CFTR Modulators Genistein, VRT-532, and UCCF-152 in primary sinonasal epithelial cultures. JAMA Otolaryngology-Head & Neck Surgery. 139 (8), 822-827 (2013).
- Pique, N., De Servi, B. Rhinosectan((R)) spray (containing xyloglucan) on the ciliary function of the nasal respiratory epithelium; results of an in vitro study. Allergy, Asthma & Clinical Immunology. 14, 41 (2018).
- Chen, Q., et al. Host antiviral response suppresses ciliogenesis and motile ciliary functions in the nasal epithelium. Frontiers in Cell and Developmental Biology. 8, 581340 (2020).
- Clary-Meinesz, C. F., Cosson, J., Huitorel, P., Blaive, B. Temperature effect on the ciliary beat frequency of human nasal and tracheal ciliated cells. Biology of the Cell. 76 (3), 335-338 (1992).
- Ballenger, J. J., Orr, M. F. Quantitative measurement of human ciliary activity. Annals of Otology, Rhinology and Laryngology. 72, 31-39 (1963).
- Mercke, U. The influence of varying air humidity on mucociliary activity. Acta Oto-Laryngologica. 79 (1-2), 133-139 (1975).
- Sutto, Z., Conner, G. E., Salathe, M. Regulation of human airway ciliary beat frequency by intracellular pH. Journal of Physiology. 560, 519-532 (2004).
- Salathe, M. Regulation of mammalian ciliary beating. Annual Review of Physiology. 69, 401-422 (2007).
- Kempeneers, C., Seaton, C., Garcia Espinosa, B., Chilvers, M. A. Ciliary functional analysis: Beating a path towards standardization. Pediatric Pulmonology. 54 (10), 1627-1638 (2019).
- Kempeneers, C., Seaton, C., Chilvers, M. A. Variation of ciliary beat pattern in three different beating planes in healthy subjects. Chest. 151 (5), 993-1001 (2017).
- Jackson, C. L., et al. Accuracy of diagnostic testing in primary ciliary dyskinesia. European Respiratory Journal. 47 (3), 837-848 (2016).
- Feriani, L., et al. Assessing the collective dynamics of motile cilia in cultures of human airway cells by multiscale DDM. Biophysical Journal. 113 (1), 109-119 (2017).
- Brewington, J. J., et al. Brushed nasal epithelial cells are a surrogate for bronchial epithelial CFTR studies. JCI Insight. 3 (13), (2018).
- Liu, X., et al. ROCK inhibitor and feeder cells induce the conditional reprogramming of epithelial cells. The American Journal of Pathology. 180 (2), 599-607 (2012).
- Suprynowicz, F. A., et al. Conditionally reprogrammed cells represent a stem-like state of adult epithelial cells. Proceedings of the National Academy of Sciences of the United States of America. 109 (49), 20035-20040 (2012).
- Martinovich, K. M., et al. Conditionally reprogrammed primary airway epithelial cells maintain morphology, lineage and disease specific functional characteristics. Scientific Reports. 7 (1), 17971 (2017).
- Wong, J. Y., Rutman, A., O’Callaghan, C. Recovery of the ciliated epithelium following acute bronchiolitis in infancy. Thorax. 60 (7), 582-587 (2005).
- Gentzsch, M., et al. Pharmacological rescue of conditionally reprogrammed cystic fibrosis bronchial epithelial cells. American Journal of Respiratory Cell and Molecular Biology. 56 (5), 568-574 (2017).
- Sommer, J. U., Gross, S., Hormann, K., Stuck, B. A. Time-dependent changes in nasal ciliary beat frequency. European Archives of Oto-Rhino-Laryngology. 267 (9), 1383-1387 (2010).
- Ratjen, F., et al. Cystic fibrosis. Nature Reviews Disease Primers. 1, 15010 (2015).
- Delmotte, P., Sanderson, M. J. Ciliary beat frequency is maintained at a maximal rate in the small airways of mouse lung slices. American Journal of Respiratory Cell and Molecular Biology. 35 (1), 110-117 (2006).
- Smith, C. M., et al. Cooling of cilia allows functional analysis of the beat pattern for diagnostic testing. Chest. 140 (1), 186-190 (2011).
- Raidt, J., et al. Ciliary beat pattern and frequency in genetic variants of primary ciliary dyskinesia. European Respiratory Journal. 44 (6), 1579-1588 (2014).

