Biomolecular Imaging of Cellular Uptake of Nanoparticles using Multimodal Nonlinear Optical Microscopy
Summary
This article presents the integration of a spectral-focusing module and a dual-output pulse laser, enabling rapid hyperspectral imaging of gold nanoparticles and cancer cells. This work aims to demonstrate the details of multimodal nonlinear optical techniques on a standard laser scanning microscope.
Abstract
Probing gold nanoparticles (AuNPs) in living systems is essential to reveal the interaction between AuNPs and biological tissues. Moreover, by integrating nonlinear optical signals such as stimulated Raman scattering (SRS), two-photon excited fluorescence (TPEF), and transient absorption (TA) into an imaging platform, it can be used to reveal biomolecular contrast of cellular structures and AuNPs in a multimodal manner. This article presents a multimodal nonlinear optical microscopy and applies it to perform chemically specific imaging of AuNPs in cancer cells. This imaging platform provides a novel approach for developing more efficient functionalized AuNPs and determining whether they are within vasculatures surrounding the tumor, pericellular, or cellular spaces.
Introduction
Gold nanoparticles (AuNPs) have shown great potential as biocompatible imaging probes, for example, as effective surface-enhanced Raman spectroscopy (SERS) substrates in various biomedical applications. Major applications include fields such as biosensing, bioimaging, surface-enhanced spectroscopies, and photothermal therapy for cancer treatment1. Furthermore, probing AuNPs in living systems is crucial to assessing and understanding the interaction between AuNPs and biological systems. There are various analytical techniques, including Fourier transform infrared (FTIR) spectroscopy2, laser ablation inductively coupled mass spectrometry (LA-ICP-MS)3, and magnetic resonance imaging (MRI)4 that have been successfully used to investigate the distribution of AuNPs in tissues. Nevertheless, these methods suffer from several drawbacks such as being time-consuming and involving complex sample preparation3, requiring long acquisition times, or the lack of sub-micron spatial resolution2,4.
Compared to conventional imaging techniques, nonlinear optical microscopy offers several advantages for probing live cells and AuNPs: The nonlinear optical microscopy achieves deeper imaging depth and provides intrinsic 3D optical sectioning capability with the use of near-IR ultrafast lasers. With the significant improvement of imaging speed and detection sensitivity, two-photon excited fluorescence (TPEF)5,6,7 and second harmonic generation (SHG)8,9,10 microscopy have been demonstrated to further improve non-invasive imaging of endogenous biomolecules in living cells and tissues. Moreover, utilizing novel pump-probe nonlinear optical techniques such as transient absorption (TA)11,12,13,14 and stimulated Raman scattering (SRS)15,16,17,18, it is possible to derive label-free biochemical contrast of cellular structures and AuNPs. Visualizing AuNPs without the use of extrinsic labels is of great importance since chemical perturbations of the nanoparticles will modify their physical properties and hence their uptake in cells.
This protocol presents the implementation of a Spectral Focusing Timing and Recombination Unit (SF-TRU) module for a dual-wavelength pulse laser, enabling fast multimodal imaging of AuNPs and cancer cells. This work aims to demonstrate the details of integrated TPEF, TA, and SRS techniques on a laser scanning microscope.
Protocol
1. Switching on the laser system
- Switch on the interlock system and select arm laser before starting the system.
- Turn on the PC with the software to control the dual-output femtosecond laser.
- Load the software for the dual-output femtosecond laser; this software enables the laser to be powered on and off and directly controls the wavelength of the pump beam.
- Switch on the laser emission by holding down on the Power icon for a count of 3.
- Wait until the laser has warmed up and the laser-ready light is lit green in the software.
- The wavelength of the pump beam is directly controlled through the software; this can range from 680-1,300 nm. For cellular imaging in the C-H Raman vibrational region, select 802 nm.
- Open the shutters of the tunable pump beam and 1,045 nm Stokes beam by holding down on the aperture images for 3 s.
CAUTION: Laser safety guidance the dual-output femtosecond laser is a class IV infra-red pulsed laser and can cause permanent damage to eyesight. Therefore, all laser safety local rules must be followed while carrying out this procedure: (i) Only authorized users who have received laser safety training can carry out the procedure. (ii) Entry to the lab is via swipe card access with an interlock on the room door. (iii) For most of the operation, the laser beams are fully enclosed. (iv) When the laser beam is exposed, laser safety goggles must be worn (use the LG9 laser safety goggles, which give OD 7+ blocking of both the pump and Stokes beams).
2. Switching on the Spectral Focusing Timing and Recombination Unit (SF-TRU)
- Load the ATM software on the same PC with the laser software, which controls the delay stages and laser attenuators in the SF-TRU unit.
NOTE: This unit is responsible for making sure the pump and Stokes beams are spatially overlapped, controlling the dispersion in the pump and Stokes beams, and controlling the time delay between the pump and Stokes beams.The polarization of both pump and Stokes beams is vertical. Note that each beam is equipped with a half-wave plate to independently control polarizations before exiting the SF-TRU. - Ensure that both the pump and Stokes lasers are attenuated to <10% (pump) and <15% (Stokes beams). If beams are not attenuated, the laser power can cause damage to the system and will burn biological samples.
- For cellular imaging, set the optimal laser settings to 6% (pump) and 10% (Stokes), corresponding to 12 mW and 30 mW at the sample.
- The SF-TRU has two settings: femtosecond (fs) mode and picosecond (ps) mode.
- For fs mode, push in the knobs on either side of the box (this removes the dispersion gratings from the beam path).
- For ps mode, pull out the knobs on either side of the box (this places the dispersion gratings in the beam path).
- For hyperspectral imaging, select the ps mode.
- Ensure that the delay stage is in the correct position for the pump and Stokes beams to be temporally overlapped for C-H imaging on this system.
- Ensure that the dispersion settings of the pump and Stokes beam are correct; for an 802 nm pump, this is 5 mm for the pump beam and 30 mm for the Stokes beam.
3. Modulating the Stokes beam for SRS imaging
- For SRS detection, modulate the intensity of the Stokes beam.
- Switch on the signal generator for the beam modulation.
- Recall previous settings, which are as follows:
OUTPUT 1: Square wave: Frequency = 19.5 MHz, Amplitude = 1.4 V.
OUTPUT 2: Square wave: Frequency = 19.5 MHz, Amplitude = 300 mV. - Switch on Outputs 1 and 2.
- Connect Output 1 to an amplifier for the EOM in the SF-TRU box via a BNC cable.
- Connect Output 2 to the lock-in amplifier used for SRS detection via a BNC cable.
- Switch on the power supply to the amplifier on the gantry.
4. Switching on the lock-in amplifier
- For SRS imaging, use the SRS detection module that comprises a large area photodiode and a purpose-built lock-in amplifier.
- Switch on the power supply to the lock-in amplifier on the gantry.
- Load the software for the lock-in amplifier “SRS detection Module” on the PC.
- In the software, chose the following settings: Phase = 0°, Offset = -80 mW, Gain = 58 dB.
- Select the lock-in amplifier integration time constant in the software: for cell imaging the time constant of 2 µs gives good quality images.
5. Operating on the laser scanning microscope
- Switch on plugs of all the equipment except the remote-control box. Wait until standby is lit on the control box located on the top of the gantry.
- Switch on the remote-control box on the gantry and switch on the back of the box. Wait until the remote light goes blue (on the control box), and press Start Operation.
- Turn on the PC of the laser scanning microscope.
- Run the confocal microscope software.
- Check the light path of the microscope via the computer software.
NOTE: Some modifications have been made to the microscope to make it suitable for SRS imaging: (i) In the beam path, a 775 nm short pass has been added to the filter wheel where the laser beams enter the scan unit, this can be selected in the Lightpath tab in the computer software. (ii) Instead of using the confocal microscope’s inbuilt PMTs for detection, a bespoke detector has been added to the transmitted light path, which allows both SRS and CARS detection. (iii) The outputs from these detectors are connected to the Analog box on the gantry. CARS signal from the PMT is connected via a BNC cable to CD1 and SRS signal from the lock-in amplifier is connected via a BNC cable to CD2. - Control the focus through the touch screen or the remote-control knobs or the computer software.
- Select the desired image settings in the software; this includes zoom, the image size in pixels and pixel dwell time.
- Ensure that the imaging speed (pixel dwell time) is greater than the integration time set in the lock-in amplifier software.
NOTE: A NA 1.2 water immersion objective was used for imaging.
6. Mounting a sample in the microscope stage
- Prepare the cell samples for imaging as follows.
- Culture 4T1 mouse mammary carcinoma cells in RPMI-1640 supplemented with 10% Fetal Bovine Serum (FBS). Change the medium every 2 days and passage the cells at 70%–80% confluence.
NOTE: Passages 8–10 were used throughout the course of the experiments described here. - For imaging experiments, incubate 1 x 105 cells on square coverslips for 24 h at 37 °C, 5% CO2. Then, wash the cells with pre-warmed phosphate-buffered saline (PBS) and incubate with gold nanoparticles (1:100 dilution in cell culture medium, stock concentration: 2.3 x 1010 particles/mL, diameter: 60 nm) for 4 h.
- Prior to imaging, wash the cells with ice-cold PBS and fix them with 4% paraformaldehyde in PBS for 15 min at room temperature (RT). Wash the cells with PBS 3x, and then mount between two coverslips for imaging.
- Culture 4T1 mouse mammary carcinoma cells in RPMI-1640 supplemented with 10% Fetal Bovine Serum (FBS). Change the medium every 2 days and passage the cells at 70%–80% confluence.
- Place a droplet of distilled water on top of the water immersion objective that focuses the laser beams into to the sample between the objective and the sample. Then, place the sample on the microscope stage.
- A condenser with NA 1.4 collects the forward propagated light. Apply a droplet of water between the condenser and the sample for imaging. Adjust the height of the condenser to approximately 1 mm above the sample to collect the maximum amount of light.
- The SRS detector is on a moveable mount, which allows it to be slide in and out from the collection optical pathway. In order to focus on the sample using white light, move the SRS detector out of the beam path.
- Place the detector back in the beam path, ready for SRS imaging.
7. Changing the Raman shift and collecting a hyperspectral data stack
NOTE: The Raman shift at which the imaging takes place is dependent on the delay stage position in the SF-TRU box and the wavelength of the pump beam from the laser system.
- For large changes in the Raman shift, change the laser wavelength. For example, for imaging CH vibrations between 2,800–3,100 cm-1, select 802 nm, whereas, for imaging around 1,600 cm-1 for the amide I peak, select the pump beam at 898 nm. Adjust this via the software.
- To achieve small adjustments in the Raman shift, scan the delay stage in the SF-TRU unit.
- The SF-TRU gives delay stage positions in millimeters (mm). To convert the delay stage position from mm to cm-1, perform the calibration hyperspectral scan using a sample of polystyrene beads and compare the peak positions found in the calibration scan with those taken on a spontaneous Raman setup. This will provide a linear equation, which converts the delay stage position in mm to Raman shift in cm-1.
- To generate a hyperspectral scan, take a time series of images at different delay stage positions.
- To synchronize Image acquisition and movement of the delay stage, use the analog unit to send and receive TTL signals to and from the SF-TRU system.
NOTE: To do this, the analog box has the following connections: (i) TRIG OUT VD1—this is connected to the SF-TRU via a BNC and sends a TTL signal to tell the delay stage to move +1 increment. (ii) TRIG In 1—here, a signal is received via BNC when the delay stage has finished its movement, and this is used to trigger the next image in the time series. - For the hyperspectral data set, select the following settings in the confocal microscope software:
- In the Trigger tab, select Start by TTL trigger (ON) and Trigger (every frame).
- In the Series tab, set the time series ON and the z series OFF.
- Set the number of frames in the time series to match the number of steps in the ATM software (101 steps is used here).
- In the ATM software, go to the Run tab.
- Set the delay stage positions in millimeters for the start and stop of the hyperspectral scan. For CH high wavenumber region, use the start position = 90.25 mm and the stop position = 92.25 mm.
- Ensure that the number of steps matches the number of frames in the confocal microscope software.
- After checking the correct settings, start the hyperspectral stack in the confocal microscope software first. Go to Acquire and click on Start Scan. Then, in the ATM software, click on Start. This initiates the collection of a series of images, with incremental increases in the delay stage position between the selected start and stop positions
- Export the hyperspectral image series as a multi-frame .tif file and use a suitable software package to carry out the calibration.
- Once the calibration is completed, use the linear calibration equation to convert the delay stage position to the Raman shifts.
- For switching between imaging in the CH vibrational region (2,800–3,100 cm-1) to the amide I vibrational region 1,500–1,700 cm-1), do the following.
- Change the pump wavelength in the laser software from 802 nm to 898 nm.
- Change the Stokes dispersion setting in the ATM software from 30 mm to 5 mm.
- Make a small adjustment to the mirror in the pump beam dispersion pathway inside the SF-TRU box. This is done by adjusting the lower knob on the mirror mount, which changes the mirror angle by an incremental amount.
- When performing a hyperspectral scan over the amide I region, set the start and stop positions in the ATM software to 89 and 91mm, respectively
CAUTION: Laser safety goggles must be worn for this step as the laser beam will be exposed.
Representative Results
The Spectral Focusing Timing and Recombination Unit (SF-TRU) module is introduced between the dual-output femtosecond laser and the modified laser scanning microscope. The tunable ultrafast laser system used in this study has two output ports delivering one beam at a fixed 1,045 nm wavelength and the other beam tunable in the range of 680–1,300 nm. A detailed schematic of the SF-TRU module and multimodal imaging platform is depicted in Figure 1. The SF-TRU is employed to chirp two femtosecond laser beams to picosecond pulses and overlaps two beams spatially and temporally. Hyperspectral stimulated Raman scattering (SRS) imaging is performed by scanning the delay between the picosecond pump and Stokes pulses.
All the samples are illuminated by pulsed lasers through a high numerical aperture (NA) objective on a modified inverted microscope. The SRS and TPEF signals are collected in the forward direction with a high NA condenser. For SRS microscopy, the tunable (680–1,300 nm) and fixed (1,045 nm) laser outputs are used as the pump and the Stokes beams, respectively. The combination of a large-area photodiode and lock-in amplifier is used to perform SRS imaging, while the Stokes beam is blocked with two filters (890/210 band-pass and 950 nm short-pass filters). An important advantage of the spectral focusing SRS approach over the picosecond laser-based systems is the ability to tune the Raman shift of interest by simply controlling the time delay between the chirped pump and Stokes pulses. This approach allows fast probing of Raman shifts within a range of several hundred wavenumbers without changing the pump and Stokes wavelengths, allowing much faster hyperspectral imaging.
To validate the performance of spectral resolution and chemical selectivity, the sample of polystyrene (PS) microspheres is imaged first (Figure 2). The laser powers at the sample (after 60x NA 1.2 water immersion objective) are 10 mW for the 1,045 nm Stokes beam and 20 mW for the 802 nm pump beam. The SRS spectrum can be extracted at every pixel in the frames. Figure 2B shows the hyperspectral SRS spectrum of PS beads. For comparison, the spontaneous Raman spectrum of PS beads is shown in Figure 2A. The SRS and spontaneous Raman spectra of PS microspheres are almost identical except for relative intensity differences at the sides. These spectroscopic measurements allow converting optical delay line stage (mm) to Raman wavenumbers (cm-1) for SRS imaging.
Since the Raman spectra of major biomolecules in the carbon-hydrogen stretching vibrational band is in the range of 2,800–3,050 cm−1, the SRS imaging of 4T1 cancer cells is performed at 2,852 cm−1 (lipid), 2,930 cm−1 (protein), 2,968 cm−1 (DNA), and the overlay images as shown in Figure 3. SRS images of different Raman bands are acquired with a pixel dwell time of 4 μs at a frame size of 512 x 512 pixels.
Finally, this method has further extended to multimodal imaging of 4T1 cancer cells dosed with gold nanoparticles (AuNPs), allowing us to image the AuNPs’ distributions in cancer cells more precisely. The acquired SRS (CH2 and CH3 channels), TPEF (LysoTracker fluorescent probes), and TA (off-resonance SRS channel) images are depicted in Figure 4.
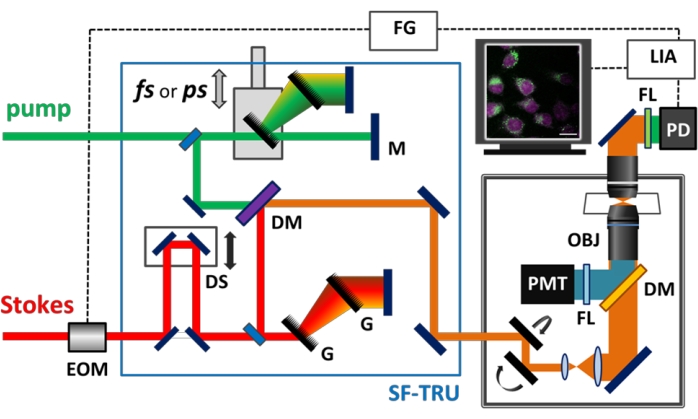
Figure 1: Schematic of the hyperspectral multimodal imaging platform. Illustration of multimodal nonlinear optical microscopy. DM, dichroic mirror; DS, delay stage; EOM, electro-optical modulator; FG, function generator; FL, filter; G, grating; LIA, lock-in amplifier; M, mirror; OBJ, objective; PD, photodiode; PMT, photomultiplier tube; SF-TRU, Spectral Focusing Timing, and Recombination Unit. Please click here to view a larger version of this figure.
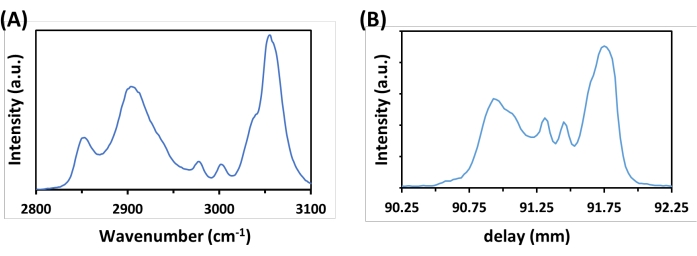
Figure 2: Spectra of polystyrene (PS) microspheres. The spontaneous (A) Raman spectrum of PS microspheres shows good agreement with the (B) hyperspectral SRS spectrum. Please click here to view a larger version of this figure.
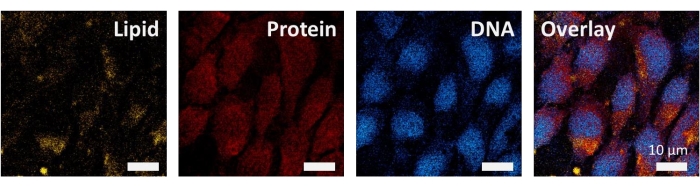
Figure 3: Spectroscopic SRS imaging of 4T1 cancer cells. SRS images at 2,852 cm−1, 2,930 cm−1, 2,968 cm−1, and overlay image. Scale bar: 10 µm. Please click here to view a larger version of this figure.
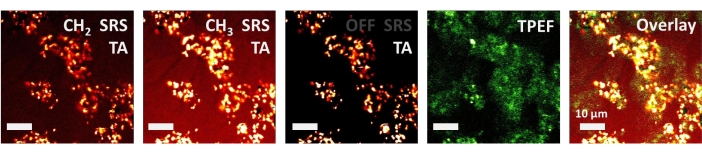
Figure 4: Multimodal imaging of gold nanoparticles uptake by 4T1 cancer cells. Hyperspectral SRS images at 2,852 cm−1 (CH2), 2,928 cm−1 (CH3), 3,080 cm−1 (off-resonance, only TA signal left), TPEF, and overlay image. Scale bar: 10 µm. Please click here to view a larger version of this figure.
Discussion
This study has presented the combination of SF-TRU module and ultrafast dual-output laser system demonstrated its applications for multimodal microspectroscopy. With its ability to investigate gold nanoparticles’ (AuNPs’) uptake by cancer cells, the multimodal imaging platform can visualize the cellular responses to hyperthermic cancer treatments when laser beams are absorbed by AuNPs.
Moreover, rapid chemically specific imaging and high spectral resolution are achieved by employing the spectral focusing technique, using two sets of grating pairs to control the chirps in each laser beam. Compared to the usage of fixed-length glass rods for chirping, grating pairs allow matching the spectral resolution and the line widths of the Raman lines of interest19.
Furthermore, this system can be easily switched from femtosecond to chirped picosecond regimes by utilizing specially designed manual sliders to remove the gratings from the beam paths without affecting alignment inside the microscope. In order to achieve high sensitivity and spectral resolution, SRS is typically implemented using the narrowband picosecond pump and Stokes pulses. However, picosecond lasers are not sufficient for multiphoton excitation that require higher peak power levels, which are achievable with femtosecond lasers. The multimodal capabilities allow using the method for a variety of applications such as multiphoton imaging20, coherent Raman scattering21, and pump- probe spectroscopy.
There are several strategies that can be used to further improve the performance of the demonstrated device. For example, a faster-motorized delay line stage and data-acquisition software offer a higher imaging rate, which can increase the throughput of the multimodal imaging platform. In addition, the current setup covered ~300 cm−1 of spectral range with a spectral resolution up to 5 cm−1. To further enlarge the spectral range, a modified laser system with a broader spectral bandwidth has been employed for hyperspectral SRS microscopy22.
Collectively, this multimodal and non-invasive imaging platform provides new insights into nanomedicine and opens a new avenue to high-content multimodal molecular imaging of cells, biological tissues, and nanoparticles23,24,25.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This research was supported by EPSRC Grants: Raman Nanotheranostics (EP/R020965/1) and CONTRAST facility (EP/S009957/1).
Materials
| APE SRS Detection Unit | APE (Angewandte Physik & Elektronik GmbH) | APE Lock-in Module | Combined system containing a large area Si photo-diode for detecting the pump beam along with a Lock-In amplifier for detecting the beam modulations |
| Confocal Scanning Unit | Olympus | FV 3000 | Confocal scanning unit used for imaging |
| CML Latex Beads, 4% w/v, 1.0 µm | Invitrogen | C37483 | Polystyrene microspheres |
| Coverslips | Thorlabs | CG15CH2 | 22 mm x 22 mm coverslips for seeding cells |
| FBS | Gibco | 10500-064 | Foetal Bovine Serum (Heat Inactivated) |
| Flouview | Olympus | FV31S-SW | Laser scanning microscope control software |
| Function Generator | BX precision | 40543 | Used to generate square wave function which is fed to EOM in SF-TRU to produce modulations in the stokes beam |
| FV3000 | Olympus | IX83P2ZF | Other microscope frames can be used. |
| Gold Nanoparticles | Nanopartz | A11-60 | Spherical gold nanoparticles, 60 nm diameter |
| Input Output Interface | Olympus | FV30 ANALOG | This unit allows voltage readouts from PMT and LockIn to be fed into the confocal scanning software and allows timing pulses to be sent between the olympus microscope and the SF-TRU unit. |
| InSight X3 | Newport | Spectra-Physics | Dual-output femtosecond pulsed laser. Tunable (680–1300 nm) and fixed (1045 nm) laser outputs with the repetition rate of 80 MHz. |
| Microscope Frame | Olympus | IX83 | Inverted microscope |
| Mouse 4T1 cells | ATCC | CRL-2539 | Mouse breast cancer cells |
| NA 1.2 Water Immersion Objective | Olympus | UPLSAPO60XW/IR | The multiphoton 60x Objective has a 0.28 mm working distance. Other similar objectives can be used. |
| NA 1.4 Condenser | Nikon | CSC1003 | Other condensers with NA higher than the excitation objective can also be used. |
| PMT | Hamamatsu | R3896 | PMT used for detecting anti-stokes photos for CARS micrsocopy |
| PMT Connector | Hamamatsu | C13654-01-Y002 | Connector for PMT |
| Power Supply | RS | RSPD-3303 C | Programmable power supply which is used for providing the correct voltage to the PMT |
| RPMI-1640 | Gibco | A10491-01 | Roswell Park Memorial Institute (RPMI) 1640 Medium has since been found suitable for a variety of mammalian cells. |
| SF-TRU | Newport Spectra Physics | SF-TRU | System designed for controlling the time delay and dispersion of the 2 laser outputs and for performing the beam modulations required for SRS |
References
- Tabish, T. A., et al. Smart gold nanostructures for light mediated cancer theranostics: Combining optical diagnostics with photothermal therapy. Advanced Science. 7 (15), 1903441 (2020).
- Tian, F., et al. Gold nanostars for efficient in vitro and in vivo real-time SERS detection and drug delivery via plasmonic-tunable Raman/FTIR imaging. Biomaterials. 106, 87-97 (2016).
- Jenkins, S. V., et al. Enhanced photothermal treatment efficacy and normal tissue protection via vascular targeted gold nanocages. Nanotheranostics. 3 (2), 145-155 (2019).
- Huang, J., et al. Rational design and synthesis of gammaFe2 O3 @Au magnetic gold nanoflowers for efficient cancer theranostics. Advanced Materials. 27 (34), 5049-5056 (2015).
- Dilipkumar, A., et al. Label-free multiphoton endomicroscopy for minimally invasive in vivo imaging. Advanced science. 6 (8), 1801735 (2019).
- Wang, C. -. C., et al. Differentiation of normal and cancerous lung tissues by multiphoton imaging. Journal of Biomedical Optics. 14 (4), 044034 (2009).
- Chrabaszcz, K., et al. Comparison of standard and HD FT-IR with multimodal CARS/TPEF/SHG/FLIMS imaging in the detection of the early stage of pulmonary metastasis of murine breast cancer. The Analyst. 145 (14), 4982-4990 (2020).
- Tsai, T. H., et al. Visualizing radiofrequency-skin interaction using multiphoton microscopy in vivo. Journal of Dermatological Science. 65 (2), 95-101 (2012).
- Wang, C. -. C., et al. Early development of cutaneous cancer revealed by intravital nonlinear optical microscopy. Applied Physics Letters. 97 (11), 113702 (2010).
- Li, F. -. C., et al. Dorsal skin fold chamber for high resolution multiphoton imaging. Optical and Quantum Electronics. 37 (13), 1439-1445 (2005).
- Tong, L., et al. Label-free imaging of semiconducting and metallic carbon nanotubes in cells and mice using transient absorption microscopy. Nature Nanotechnology. 7 (1), 56-61 (2011).
- Chong, S., Min, W., Xie, X. S. Ground-state depletion microscopy: Detection sensitivity of single-molecule optical absorption at room temperature. The Journal of Physical Chemistry Letters. 1 (23), 3316-3322 (2010).
- Chen, T., et al. Transient absorption microscopy of gold nanorods as spectrally orthogonal labels in live cells. Nanoscale. 6 (18), 10536-10539 (2014).
- Liu, J., Irudayaraj, J. M. Non-fluorescent quantification of single mRNA with transient absorption microscopy. Nanoscale. 8 (46), 19242-19248 (2016).
- Freudiger, C. W., et al. Label-free biomedical imaging with high sensitivity by stimulated Raman scattering microscopy. Science. 322 (5909), 1857-1861 (2008).
- Wang, C. -. C., et al. In situ chemically specific mapping of agrochemical seed coatings using stimulated Raman scattering microscopy. Journal of Biophotonics. 11 (11), 201800108 (2018).
- Wang, C. -. C., Yoong, F. -. Y., Penfield, S., Moger, J. Visualization of active ingredients uptake in seed coats with stimulated Raman scattering microscopy. Proceedings SPIE 10069, Multiphoton Microscopy the Biomedical Sciences XVII. , 1006928 (2017).
- Hu, F., Shi, L., Min, W. Biological imaging of chemical bonds by stimulated Raman scattering microscopy. Nature Methods. 16 (9), 830-842 (2019).
- Zeytunyan, A., Baldacchini, T., Zadoyan, R. Module for multiphoton high-resolution hyperspectral imaging and spectroscopy. Proceedings SPIE 10498, Multiphoton Microscopy in the Biomedical Sciences XVIII. , 104980 (2018).
- Wang, C. -. C., Wu, R. -. J., Lin, S. -. J., Chen, Y. -. F., Dong, C. -. Y. Label-free discrimination of normal and pulmonary cancer tissues using multiphoton fluorescence ratiometric microscopy. Applied Physics Letters. 97 (4), 043706 (2010).
- Wang, C. -. C., Chandrappa, D., Smirnoff, N., Moger, J. Monitoring lipid accumulation in the green microalga botryococcus braunii with frequency-modulated stimulated Raman scattering. Proceedings SPIE 9329, Multiphoton Microscopy in the Biomedical Sciences XV. , 9329 (2015).
- Figueroa, B., et al. Broadband hyperspectral stimulated Raman scattering microscopy with a parabolic fiber amplifier source. Biomedical Optics Express. 9 (12), 6116-6131 (2018).
- Cui, L., et al. In situ plasmon-enhanced CARS and TPEF for Gram staining identification of non-fluorescent bacteria. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy. 264, 120283 (2022).
- Ma, J., Sun, M. Nonlinear optical microscopies (NOMs) and plasmon-enhanced NOMs for biology and 2D materials. Nanophotonics. 9 (6), 1341-1358 (2020).
- Sun, L., Chen, Y., Sun, M. Exploring nonemissive excited-state intramolecular proton transfer by plasmon-enhanced hyper-Raman scattering and two-photon excitation fluorescence. The Journal of Physical Chemistry C. 126 (1), 487-492 (2022).

