Robotic Myotomy and Partial Fundoplication for Achalasia
Summary
Surgical myotomy with a partial fundoplication may be used in selected patients as a definitive treatment for achalasia. This article provides a step-by-step description of a robotic myotomy and partial fundoplication in a 32-year-old patient with megaesophagus.
Abstract
Laparoscopic Heller myotomy is currently considered the standard definitive treatment of achalasia. With the advancements in technology, robotic Heller myotomy has emerged as an alternative approach to traditional laparoscopy due to three-dimensional (3D) visualization, fine motor control, and improved ergonomics provided by the robot.
Although there is a lack of randomized controlled trials, robotic-assisted Heller myotomy seems to be associated with lower rates of intraoperative perforations compared to the laparoscopic approach. A robotic approach may also improve surgical outcomes by providing a more complete myotomy.
Here, we describe the detailed steps of robotic myotomy and partial fundoplication for achalasia.
Introduction
Achalasia is a primary neurodegenerative esophageal motility disorder characterized by abnormal peristalsisand failure of the lower esophageal sphincter to relax1. Treatment of achalasia aims to reduce the resting pressure of the lower esophageal sphincter, thereby allowing esophageal emptying2. There are multiple options for treating achalasia, such as oral pharmacologic therapy, endoscopic pharmacologic therapy3, pneumatic dilatation4, peroral endoscopic myotomy (POEM)5, and surgical myotomy6.
Surgical myotomy, in which the muscle fibers of the lower esophageal sphincter are divided, has been described as one of the three definitive therapies for non-advanced achalasia, along with pneumatic dilatation and peroral endoscopic myotomy7,8. The addition of a fundoplication is performed as an anti-reflux procedure since the myotomy reduces the pressure of the lower esophageal sphincter, which can result in potential gastroesophageal reflux disease9,10.
Laparoscopic Heller myotomy has become the most common surgical procedure for treating achalasia due to decreased postoperative pain and reduced morbidity compared to other surgical approaches, such as thoracotomy, laparotomic, and thoracoscopic11,12. Robotic Heller myotomy has emerged as a minimally invasive alternative to laparoscopy for treating achalasia because of mechanical advantages provided by the robotic approach, such as magnified high-resolution three-dimensional visualization and minimized physiological tremor13,14,15.
This article presents a case of a 32-year-old patient with chronic dysphagia, regurgitation, and weight loss. The dysphagia was initially associated with solids, slowly progressing to liquids as well. The patient denied other clinical symptoms, such as pyrosis, epigastric pain, and postprandial fullness. An endoscopic evaluation was initially performed in order to exclude malignancy (Figure 1). The exam revealed dilatation and tortuosity of the esophagus, as well as retention of food, which was completely aspirated with the endoscope. Thickening of the mucosa was also identified, and no neoplastic lesions were detected. Narrow band imaging showed normal vascular and mucosal patterns. The gastroesophageal junction was located at the level of the diaphragmatic crus.
The investigation then proceeded with an esophageal manometry (Figure 2) and a barium esophagram (Figure 3). The manometry showed impaired gastroesophageal junction relaxation and esophagus with the absence of peristalsis. Barium esophagram findings were esophageal dilatation and delayed emptying of the barium. The diagnosis of achalasia was then established by the findings on the manometry and barium esophagram. The patient was considered eligible for robotic-assisted myotomy and partial fundoplication.
The aim of this article is to provide a step-by-step description of a robot-assisted Heller myotomy, performed at the University of São Paulo.
Protocol
The recording of the surgical procedure and the use of its content for scientific and educational reasons were explained to the patient; he then signed a consent form, according to the Institution's human ethics committee. Written informed consent for the surgical and anesthetic procedures was also obtained.
NOTE: Patients with a confirmed diagnosis of achalasia by manometry and barium esophagram findings can be included in the protocol for robotic myotomy and partial fundoplication. A preoperative pre-anesthesia evaluation was performed, and patients with increased surgical risk were excluded. Patients who failed to meet the diagnostic criteria of achalasia and/or presented other esophageal motility disorders were excluded. Failure to sign both anesthetic and surgical forms also implied exclusion.
1. Operative setting and trocar placement
- Once under general anesthesia, place the patient in a supine position.
- Create a pneumoperitoneum using a Veress needle. Insert the needle above the umbilical scar.
- Use four 8 mm robotic trocars, one 12 mm trocar and one 5 mm trocar in this procedure. Place the 12 mm trocar in the supraumbilical area, left to the midline, for the robotic camera system. This positioning allows better visualization of the gastroesophageal junction (GEJ).
- Place the remaining trocars along a straight line above the umbilicus: two 8 mm trocars, one at the left and one at the right midclavicular line subcostal margin, and the remaining two trocars at the left and right lateral abdominal walls (Figure 4). A 5 mm trocar is placed in a subxiphoid position and then replaced by a Nathanson liver retractor.
2. Dissection of the lower esophagus and division of the short gastric vessels
- Place a Nathanson liver retractor in a subxiphoid position in order to elevate the left lobe of the liver.
- Start the operation by dividing the short gastric vessels, starting from the middle of the great curvature of the stomach all the way to the angle of His.
- Perform full mobilization of the gastric fundus, using an ultrasonic harmonic scalpel to free it from the retroperitoneum while an atraumatic grasper retracts the stomach.
- Divide the gastrohepatic ligament – below the hepatic branch of the vagus – and progressively dissect it in order to identify and expose both diaphragmatic crura.
- Place tape around the esophagus, allowing gentle traction.
- Proceed with the isolation of the esophagus by separating it from the left and right crura by blunt dissection. During the dissection, identify and preserve the posterior and anterior vagal trunks and both pleurae. The anterior vagal trunk is adhered to or embedded in the esophageal wall, and the posterior trunk lies on a layer of adipose tissue posterior to the esophagus.
- Mobilize the esophagus circumferentially, creating a longer intra-abdominal esophagus.
3. Heller myotomy
- Remove the fat pad by holding the adipose tissue that lies above the pharyngoesophageal ligament and separate it circumferentially from the esophagus wall with a harmonic scalpel in order to expose and better visualize the gastroesophageal junction.
- Before performing the myotomy, mark the esophageal wall with the bipolar forceps in the anterior midline of the esophagus, with an extension approximately 6 cm above the gastroesophageal junction.
- Grasp the borders of the myotomy and disrupt the muscular layer while moving the borders away from each other until exposure of the submucosal layer.
- Proceed with the myotomy with the harmonic scalpel by interposing the inactive jaw of the scalpel between the submucosal (pinkish) and muscular layers (whitish) and activating the trigger in order to cut and cauterize.
- During this dissection, make sure to separate the muscle edges of the myotomy laterally, thus avoiding it to fuse when healing. Use a sterile measuring stripe to ensure the length of the myotomy is 6 cm above the gastroesophageal junction and 3 cm below it.
- Continue the demarcation and myotomy below the gastroesophageal junction laterally into the stomach as the muscle fibers change direction from circular to oblique. Adequate extension of myotomy is crucial to achieve good postoperative results and avoid the recurrence of dysphagia.
4. Creation of the partial fundoplication
- Perform the repair of the hiatus with the figure of eight stitches, with 2.0 cotton sutures, although silk or polyester sutures may also be suitable options. Be aware not to tighten the hiatus, which may lead to postoperative dysphagia.
- Proceed with the Heller Pinotti fundoplication. That technique involves creating an anterior-lateral-posterior fundoplication using three suture lines to connect the gastric fundus and esophagus.
- Mobilize the gastric fundus and bring it around the back of the esophagus in order to perform the suturing of the gastric fundus to the posterior wall of the distal esophagus.
- Perform interrupted stitches with 2-0 cotton sutures, and attach the seromuscular layer of the stomach to the muscular layer of the esophagus. The extension of the suture should correspond to that of the myotomy; usually 2-3 stitches are enough. The first stitch of that row should also incorporate the hiatus. Although this maneuver does not prevent the migration of the valve, it restrains its rotation.
- Resect a muscular strip over the gastroesophageal junction, when needed.
- Next, perform the second row of sutures from the gastric fundus to the left edge of the myotomy. As with the first row, the initial stitch should also be attached to the hiatus.
- Perform the third and last row of sutures from the gastric fundus to the right edge of the myotomy. By the end of these steps, the exposed submucosa of the esophagus should be covered by gastric serosa.
Representative Results
Representative results are shown in Table 1. The operation time was 112 min with a measured blood loss of 20 mL. The postoperative course was uncomplicated. The post-operative care was carried out in a regular hospital room. There was no need for an intensive care unit since there were no complications. A liquid diet was started after day one of the surgery – the patient did not report dysphagia. The patient was discharged in good condition on postoperative day 2, on a liquid diet. Soft food was gradually introduced, after day 5. The patient did not develop any complications during the follow-up. A barium esophagram was performed 30 days after surgery, (Figure 5). The exam showed an adequate emptying of the barium, with no contrast retention and a normal aspect of the fundoplication.
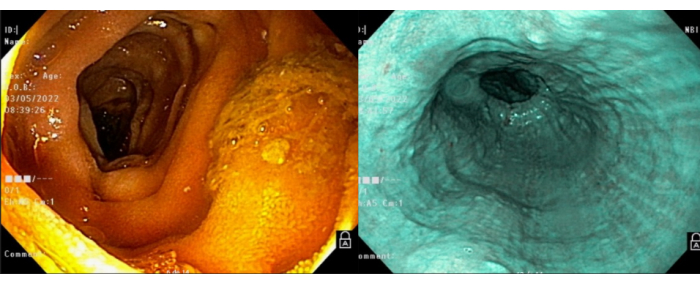
Figure 1: Endoscopy. The preoperative endoscopy showed no signs of malignancy or other diseases of the esophagus. Please click here to view a larger version of this figure.
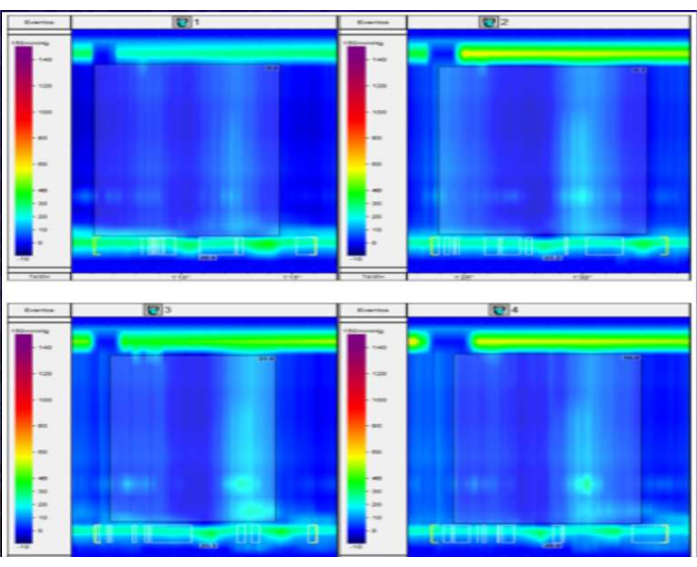
Figure 2: Manometry. This figure displays the results of a high-resolution esophageal manometry study in a patient with suspected esophageal motility disorder. The plot shows the pressure waves detected along the length of the esophagus during a series of swallows. In this case, there is a complete absence of peristaltic contractions and no detectable pressurization of the esophageal lumen. These findings are consistent with a diagnosis of achalasia type I. Please click here to view a larger version of this figure.
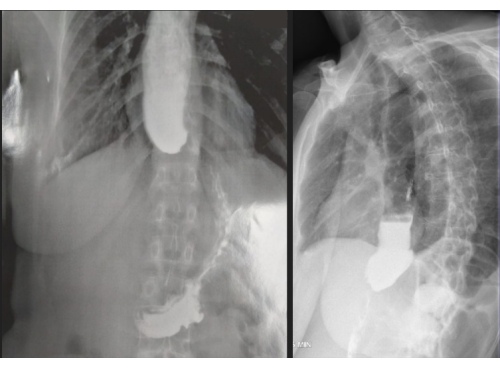
Figure 3: Barium esophagram. On the preoperative esophagogram, typical signs of achalasia were observed, including dilation of the distal esophagus, stasis of contrast material, and absence of air in the stomach. These findings are consistent with impaired esophageal emptying due to a failure of the lower esophageal sphincter to relax properly, which is characteristic of achalasia. The esophagus in this patient dilates up to 6 cm, indicating a grade III megaesophagus according to the Rezende-Mascarenhas classification. Please click here to view a larger version of this figure.
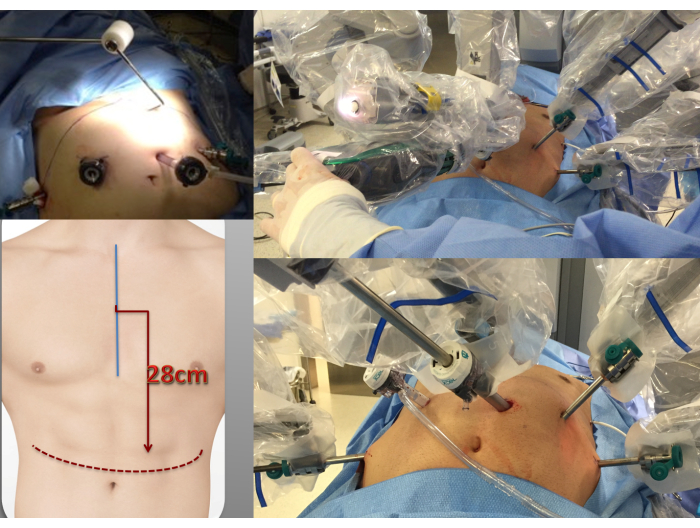
Figure 4: Trocar placement. The trocars are positioned in a linear arrangement directly above the umbilicus, with a distance of 28cm from the sternal angle. Please click here to view a larger version of this figure.
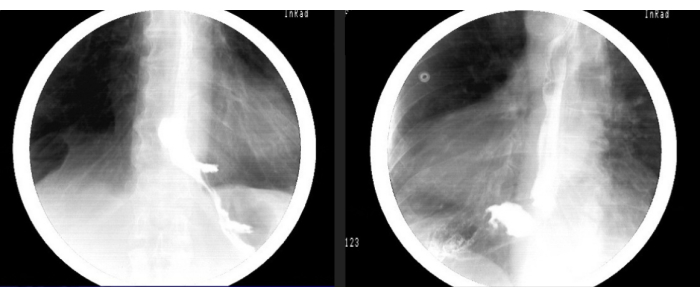
Figure 5: Postoperative barium esophagram. The postoperative esophagogram reveals improved contrast clearance from the esophagus to the stomach, along with a reduction in esophageal stasis. These findings suggest that the surgical intervention has been successful in improving esophageal motility and facilitating the emptying of ingested material into the stomach. Please click here to view a larger version of this figure.
| Operation time | 112 minutes |
| Blood loss | 20 ml |
| Hospital discharge | 2 days |
Table 1: Postoperative outcomes.
Discussion
This protocol describes robotic myotomy and partial fundoplication as a treatment for achalasia. The article highlights a Heller Pinotti fundoplication, which consists of a variation of the classic Dor fundoplication. This technique, presented in the article, demonstrates the performance of three rows of sutures, instead of the classic two sutures performed in the Dor fundoplication. The optimal type of fundoplication, including total, anterior, or posterior has been extensively studied in the literature, but there is still controversy surrounding the best approach for improving outcomes17. Modifications have been implemented to the technique over time, for example, the introduction of a posterior suture line to the classic Dor fundoplication, resulting in a postero-lateral-anterior fundoplication that provides enhanced reflux control compared to the anterior fundoplication alone. This technique, widely known as the Heller Pinotti procedure, has gained popularity in Brazil17.
Critical steps during the procedure include the proper placement of the robotic arms and trocars and careful dissection of the muscular layers of the esophagus. While laparoscopic Heller myotomy is currently considered the gold standard treatment for achalasia, robotic-assisted myotomy can also be a successful alternative in the hands of experienced surgeons.
Possible reasons for the low rate of robotic-assisted operations for Heller myotomy and other gastro-esophageal procedures have already been presented by several authors16 those include, mostly, a longer learning curve and cost. Although the robotic myotomy technique requires specialized equipment and may be more expensive than other minimally invasive procedures, it is expected that these limitations will become less significant over time as robotic technology becomes more widely available and costs decrease. It is the authors’ opinion that robotic assisted Heller myotomy should be considered as a feasible treatment option, in the field of minimally invasive treatments, for achalasia.
Divulgations
The authors have nothing to disclose.
Acknowledgements
None.
Materials
| Da Vinci Surgical System | Intuitive Surgical | ||
| Needle driver | Intuitive Surgical | ||
| Bipolar forceps | Intuitive Surgical | ||
| Bipolar Fenestrated Grasper | Intuitive Surgical | ||
| Ultracision | Johnson & Johnson |
References
- Moonen, A., Boeckxstaens, G. Current diagnosis and management of achalasia. Journal of Clinical Gastroenterology. 48 (6), 484-490 (2014).
- Tuason, J., Inoue, H. Current status of achalasia management: a review on diagnosis and treatment. Journal of Gastroenterology. 52 (4), 401-406 (2017).
- Pasricha, P. J., Rai, R., Ravich, W. J., Hendrix, T. R., Kalloo, A. N. Botulinum toxin for achalasia: long-term outcome and predictors of response. Gastroenterology. 110 (5), 1410-1415 (1996).
- Eckardt, V., Gockel, I., Bernhard, G. Pneumatic dilation for achalasia: late results of a prospective follow up investigation. Gut. 53 (5), 629-633 (2004).
- Inoue, H., et al. Peroral endoscopic myotomy (POEM) for esophageal achalasia. Endoscopy. 42 (4), 265-271 (2010).
- Doubova, M., et al. Long-term symptom control after laparoscopic heller myotomy and dor fundoplication for achalasia. Annals in Thoracic Surgery. 111 (5), 1717-1723 (2021).
- Vaezi, M. F., Pandolfino, J. E., Yadlapati, R. H., Greer, K. B., Kavitt, R. T. ACG clinical guidelines: Diagnosis and management of achalasia. American Journal of Gastroenterology. 115 (9), 1393-1411 (2020).
- Triadafilopoulos, G., et al. The Kagoshima consensus on esophageal achalasia. Diseases of Esophagus. 25 (4), 337-348 (2012).
- Richards, W. O., et al. Heller myotomy versus Heller myotomy with Dor fundoplication for achalasia: A prospective randomized double-blind clinical trial. Annals of Surgery. 240 (3), 405-415 (2004).
- Wang, X. H., Tan, Y. Y., Zhu, H. Y., Li, C. J., Liu, D. L. Full-thickness myotomy is associated with a higher rate of postoperative gastroesophageal reflux disease. World Journal of Gastroenterology. 22 (42), 9419-9426 (2016).
- Ali, A., Pellegrini, C. A. Laparoscopic myotomy: technique and efficacy in treating achalasia. Gastrointestinal Endoscopy Clinics of North America. 11 (2), 347-358 (2001).
- Spiess, A. E., Kahrilas, P. J. Treating achalasia: from whalebone to laparoscope. JAMA. 280 (7), 638-642 (1998).
- Xie, J., Vatsan, M. S., Gangemi, A. Laparoscopic versus robotic-assisted Heller myotomy for the treatment of achalasia: A systematic review with meta-analysis. International Journal of Medical Robotics and Computer Assisted Surgery. 17 (4), e2253 (2021).
- Kim, S. S., Guillen-Rodriguez, J., Little, A. G. Optimal surgical intervention for achalasia: laparoscopic or robotic approach. Journal of Robotic Surgery. 13 (3), 397-400 (2019).
- Pallabazzer, G., et al. Clinical and pathophysiological outcomes of the robotic-assisted Heller-Dor myotomy for achalasia: a single-center experience. Journal of Robotic Surgery. 14 (2), 331-335 (2020).
- Baek, S. J., Kim, S. H. Robotics in general surgery: an evidence-based review. Asian Journal of Endoscopic Surgery. 7 (2), 117-123 (2014).
- Bianchi, E. T., et al. Heller-Pinotti, a modified partial fundoplication associated with myotomy to treat achalasia: technical and final results from 445 patients. Mini-invasive Surgery. 1, 153-159 (2017).

