The Chick Chorioallantoic Membrane (CAM) Model as a Tool to Study Ovarian Tissue Transplantation
Summary
Here, we describe a protocol for developing a chick chorioallantoic membrane (CAM) xenografting model for human ovarian tissue and demonstrate the effectiveness of the technique, the graft revascularization time frame, and the tissue viability across a 6 day grafting period.
Abstract
Ovarian tissue cryopreservation and transplantation is an effective strategy for preserving fertility but has one major drawback, namely massive follicle loss occurring shortly after reimplantation due to abnormal follicle activation and death. Rodents are benchmark models for investigating follicle activation, but the cost, time, and ethical considerations are becoming increasingly prohibitive, thus driving the development of alternatives. The chick chorioallantoic membrane (CAM) model is particularly attractive, being inexpensive and maintaining natural immunodeficiency up to day 17 postfertilization, making it ideal to study short-term xenografting of human ovarian tissue. The CAM is also highly vascularized and has been widely used as a model to explore angiogenesis. This gives it a remarkable advantage over in vitro models and allows the investigation of mechanisms affecting the early post-grafting follicle loss process. The protocol outlined herein aims to describe the development of a CAM xenografting model for human ovarian tissue, with specific insights into the effectiveness of the technique, the graft revascularization time frame, and the tissue viability across a 6 day grafting period.
Introduction
The demand for fertility preservation for oncological and benign indications, as well as social reasons, has dramatically increased over recent decades. However, various treatments used to cure malignant and non-malignant diseases are highly toxic to the gonads and can result in iatrogenic premature ovarian insufficiency, ultimately leading to infertility1. Established techniques for fertility preservation include embryo cryopreservation, immature or mature oocyte vitrification, and ovarian tissue cryopreservation2,3,4. Ovarian tissue freezing is the only available option for preserving fertility in prepubertal girls or women who require immediate cancer therapy. The restoration of endocrine function following ovarian tissue transplantation occurs in over 95% of subjects, with live birth rates ranging from 18% to 42%5,6,7,8,9.
Although the transplantation of frozen-thawed ovarian tissue has proven successful, there is still room for improvement. Indeed, as ovarian cortical fragments are transplanted without vascular anastomosis, they experience a period of hypoxia during which graft revascularization takes place10,11,12. The vast majority of studies investigating human ovarian tissue transplantation have used a xenografting model, in which ovarian tissue is transplanted to immunodeficient mice. The complete revascularization of the xenografts takes around 10 days, with both the host and graft vessels contributing to the formation of functional vessels12,13,14. Around 50%-90% of the follicle reserve is lost during this hypoxic window before the completion of graft revascularization10,15,16. It has been strongly suggested that this massive follicle loss occurs through both direct follicle death, as demonstrated by a decrease in the absolute follicle numbers left after grafting, and the activation of primordial follicle growth, as indicated by changes in follicle proportions towards increased rates of growing follicles17,18.
Interestingly, previous research works using various animal ovarian tissues grafted to chick chorioallantoic membrane (CAM), which has a constitution mimicking the typical grafting site of the peritoneum, have reported the inhibition of spontaneous follicle activation, with the primordial follicle reserve staying intact for up to 10 days19,20,21,22. Our team previously demonstrated that the grafting of frozen-thawed human ovarian tissue to CAM constituted a reliable approach for investigating human ovarian tissue transplantation in its first ischemic stages23 and recently showed that this grafting method was able to counteract follicle activation24.
The CAM model is especially appealing not only because eggs are much cheaper than mice but also because of the highly vascularized nature of CAM, allowing scrutiny of the association between follicle activation and ovarian graft revascularization. The avian system is, indeed, one of the most common and versatile ways of studying angiogenesis25. Chick embryo development (ED) takes 21 days until hatching, and the CAM is formed within the first 4-5 days through the fusion of the allantois and chorion26. Notably, the chick embryo is a naturally immunodeficient host until day 17 of ED, so xenografting experiments can be performed without any risk of graft rejection27,28. Moreover, the CAM model approach does not raise any ethical or legal concerns in terms of European law29, making it an attractive alternative to other animal models. With regard to breeding conditions, chick embryos only need an incubator set at 37 °C with a relative air humidity of 40%-60%. These limited experimentation requirements significantly reduce the research costs compared to use of immunodeficient mice.
The protocol presented herein aims to describe the development of a CAM xenografting model for human ovarian tissue and provide specific insights into the effectiveness of the technique, the time frame of graft revascularization, and the tissue viability over a 6 day grafting period. This protocol could be of great interest for investigating the mechanisms behind early post-grafting follicle loss and studying the impact of several agents (growth factors, hormones, etc.) on this phenomenon.
Protocol
The use of human tissue was approved by the Institutional Review Board of the Catholic University of Louvain. The patients gave their written informed consent for the use of their ovarian tissue for research purposes.
1. Ordering day 0 eggs that are highly likely to be embryonated
- Find a certified laboratory-grade Lohman-selected white Leghorn egg supplier that reports high rates of embryonated eggs, which is primarily dependent on the age of the chicks.
2. Preparing the eggs for incubation
- Prior to the arrival of the eggs, assemble and equilibrate the egg incubator to 37 °C in 40%-60% relative air humidity. Monitor the temperature and air humidity by inserting a thermometer and hygrometer into the incubator. Ensure the lid of the incubator is equipped with a glass window to allow for checking the internal parameters of the incubator without having to open it (Figure 1).
- Having received the day 0 eggs from Lohman-selected white Leghorn chicks from a certified laboratory-grade egg supplier, clean the surface of the shells with humidified paper, and dry them immediately.
NOTE: Since the eggshell membrane is porous, autoclaved water may be used to clean egg surfaces and control humidity within the incubator in order to minimize contamination risks. - Label the eggs using a marker (e.g., date and egg number).
- Incubate the eggs with the pointed end facing down, and rotate them to allow the CAM to develop. Manually rotate the eggs by 180° two to three times per day or use an automatic rotator.
NOTE: In order to ensure the eggs are indeed being rotated, the eggshell may be marked with an "X" and "O" on the two opposing lateral sides using a pencil or marker. The glass window in the lid of the incubator allows one to check the rotation of the eggs without opening the device.
3. Opening the eggshell on day 3 of ED
NOTE: A rectangular window is made in the eggshell on day 3 of ED.
- Prepare the laminar flow hood in order to work in sterile conditions. Place the following instruments under the hood, and disinfect them in 70% ethanol (if not already sterilized):
-Egg rack
-Egg candler or focal cold light source
-Marker
-Sterile straight pin
-Sterile 19 G needle
-5 mL sterile syringe
-Scroll saw blade
-Sterile forceps
-Adhesive tape - Transfer one egg from the incubator to the egg rack placed under the hood, and turn off the egg rotator.
- In the dark, identify the air pocket of the egg by placing the egg candler (or focal cold light source) against the eggshell. The air pocket is localized at the blunt end of the egg. Use a marker to pinpoint the center of the air pocket on the eggshell.
- Turn on the lights. Make a small hole in the eggshell where it is marked by gently rotating a sterile straight pin. A tiny opening of around 1 mm in diameter is usually sufficient.
NOTE: Be careful not to push the pin all the way through the egg. If this happens, discard the egg. - Connect a sterile 19 G needle to a sterile 5 mL syringe.
- In the dark, locate the yolk sac using the egg candler (or focal cold light source), and puncture the egg through the hole made in step 3.4 with the sterile 19 G needle angled at 45° toward the bottom of the egg; take great care not to disrupt the yolk sac. Aspirate between 1.5-2 mL of albumen to detach the CAM from the shell, and then close the hole with a piece of adhesive tape.
NOTE: If the yolk sac is disrupted during aspiration, the aspirated liquid in the syringe will be yellow-colored rather than transparent (albumen). If this happens, replace the needle and syringe. If a relatively large amount of egg yolk is sucked up, it may jeopardize the viability of the embryo. - Turn on the lights. Place the egg horizontally, and draw a rectangular window measuring 1 cm x 1.5 cm with a marker. Do not make the window larger than the usual width of standard adhesive tape.
- Hold the egg in one hand, and gently saw the previously drawn window into the eggshell using a scroll saw blade. Ensure that the shell does not crack and do not cut all the way down to the depressed CAM. Blow regularly to remove shell dust and debris.
- Slide sterile forceps under the rectangular piece of sawed shell, and grasp deftly to remove it cleanly without damaging the CAM. Additionally, discard the white outer shell membrane to be able to see the embryo and its CAM.
NOTE: If some eggshell dust or debris falls onto the CAM, it may be removed using sterile forceps, being very careful not to tear the CAM. - Identify viable embryonated eggs. They are discernible by their clear albumen and the vascular ring around the embryo, where a beating heart may sometimes be detected even at this stage (day 3 of ED). Discard non-fertilized or dead embryos.
- Place adhesive tape over the newly created window to avoid dehydration. Be sure to fold one end over itself for ease of removal.
- Put the egg back in the incubator with the opened window facing up and the tape not touching the CAM. Use a folded piece of paper or part of the egg rack to stop the egg from rolling. Make sure the rotating tray is off.
- Repeat steps 3.2-3.12 to open the remaining eggs.
4. Grafting frozen-thawed human ovarian tissue to the CAM
NOTE: Transplantation to the CAM should ideally be initiated between days 7-10 of ED.
- Thaw cryopreserved ovarian cortical strips under the laminar flow hood following the protocol described elsewhere30.
- Cut the cortical strips into three fragments of 4 mm x 2 mm x 1 mm. Use one piece as a non-grafted control and the other two for xenografting for 1 day or 6 days.
NOTE: If enough tissue is available, work in duplicate. - Transfer one egg from the incubator to the egg rack positioned under the hood, with the window facing up.
- Peel the tape off the window, and ensure that the embryo is viable. At this stage, viable embryos feature extensive vasculature, a clear albumen, a visible heartbeat, and some embryo movement.
- Prepare the grafting site by gently traumatizing a small area of the CAM by laying a 1 cm2 strip of sterile ether-extracted lens paper onto the epithelial surface and removing it immediately.
NOTE: The CAM is an impenetrable barrier unless the membrane has been traumatized by the removal of the upper peridermal part of the double epithelial layer, leaving the basal layer intact. This technique also enhances the revascularization process by activating wound healing. If the sterile ether-extracted lens paper stays on the CAM too long, the membrane may adhere to the lens paper and tear. If this happens, discard the egg. - Grasp one frozen-thawed ovarian cortical strip (4 mm x 2 mm x 1 mm) with microsurgical forceps, and place it onto the traumatized CAM with the medullary side against the CAM. Graft one tissue piece per egg.
- Cover the window with adhesive tape, and carefully return the egg to the incubator. Ensure that the eggshell opening sits upright and the egg is secure.
- Repeat steps 4.1-4.7 for the grafting of all the remaining tissues.
NOTE: The implants should be checked on each grafting day to assess the embryo viability and monitor changes in the vasculature.
5. Harvesting the grafts
NOTE: Xenografts should be harvested at the latest by day 17 of ED, since the immune system of the embryo becomes mature and competent from day 18.
- Place the egg on a rack, and enlarge the window in the eggshell to allow for better graft visualization and easier manipulation.
- Evaluate the graft macroscopically, and pay particular attention to the vascular reaction of the CAM towards the graft. Take digital photographs or videos for the record.
- Grasp the tissue or the surrounding CAM with forceps, and use scissors or a scalpel to excise the graft from the CAM carefully.
NOTE: The grafts become covered with a second layer of CAM at around day 3 of grafting and eventually become encapsulated. They may also have moved inside the egg by day 6, making it difficult to retrieve them in some cases. - Analyze the excised tissues by any method appropriate for the given experiment. In the present study, tissue pieces were fixed in paraformaldehyde, paraffin-embedded and stained with hematoxylin and eosin following the steps outlined below:
- Fix the fragments in 4% paraformaldehyde for 24 h, and embed them in paraffin using an automatic embedding device with the steps mentioned in Table 1.
- Leave the paraffin-embedded blocks overnight at 4°C before cutting them into 5 µm thick sections with a microtome.
- Spread the tissue sections on a glass slide placed on a hot plate (30 °C), and leave them to dry for 2 h, followed by 24 h in an oven at 37 °C.
- Afterwards, stain the slides with hematoxylin and eosin following a protocol described elsewhere31. Subsequently, digitize the samples using a slide scanner, and analyze them using an image analysis software.
Representative Results
Chick embryo survival rates
The embryo survival rate from windowing (day 3 of ED) to ovarian tissue grafting (day 7 of ED) was 79% (33/42). Since the percentage of embryonated day 0 eggs is unknown, supernumerary day 0 eggs from Lohman-selected white Leghorn chickens were ordered to ensure sufficient embryonated eggs would be available for grafting. A total of 23 viable day 7 eggs were used for grafting, one of which perished during the first 24 h, resulting in an overall embryo survival rate of 96% after transplantation (22/23).
Macroscopic aspect of the grafts
After 1 day of grafting, the grafts looked viable in 100% (10/10) of cases and were already at least partially adherent to the CAM, showing a wheel-spoke pattern of blood vessels needed for their vascularization (Figure 2A,B). After 6 days of grafting, all the implants were still adherent (Figure 2C), apart from two that did not attach to the CAM. They took on a necrotic appearance and were excluded from further analysis (Figure 3), resulting in a grafted tissue survival rate of 83% (10/12) after 6 days. All in all, the viability assessment of the human ovarian grafts revealed an overall tissue survival rate of 91% (20/22), irrespective of the day of grafting (Table 2).
Around day 3 post-transplantation, the grafts were found to be covered with a second layer of CAM, and they eventually became encapsulated, leading to even better graft vascularization (Figure 2C). Overall, 80% of the transplants had also penetrated the egg (Figure 2D), in some cases making it hard to retrieve them on day 6.
Microscopic assessment of the grafts
A total of 30 frozen-thawed human ovarian fragments obtained from five different patients were analyzed and fixed in 4% paraformaldehyde on grafting day 0, day 1, or day 6, embedded in paraffin, and serially cut into 5 µm sections for histological evaluation (Figure 4).
In order to assess the ovarian follicle survival rates, follicles were counted in 12 random hematoxylin and eosin-stained sections per time point and per patient. Only morphologically normal follicles with a visible oocyte were taken into consideration for analysis32. Healthy follicles were observed at all time points (Figure 4B–D). The follicle survival rates were calculated by determining the remaining follicle density (number of follicles/mm3) after grafting and normalizing it to the follicle density in non-grafted controls (considered 100%). The follicle densities tended to decrease after transplantation, but not enough to reach statistical significance (p > 0.8) (Table 2). Similarly, the follicle survival rates were maintained during the grafting period, standing at 95% ± 19% on day 1 and 83% ± 27% on day 6 (p > 0.5).
The revascularization process was further investigated by the detection of avian red blood cells in vessels from the xenografted ovarian tissue. Avian erythrocytes are easily discernible since they are nucleated (Figure 4A,C,D). Surprisingly, we were able to observe avian erythrocytes in ovarian vessels in 30% of implants after only 1 day of transplantation and in all the implants by day 6, exhibiting very rapid revascularization (Table 2).

Figure 1: Egg incubator. Please click here to view a larger version of this figure.
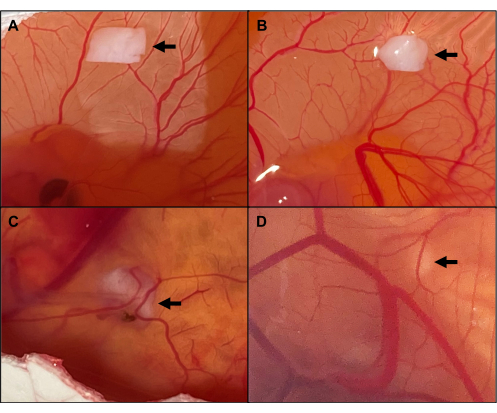
Figure 2: Macroscopic aspect of viable implants. (A) Grafting day 0. (B) Implants on day 1, with the CAM showing a wheel-spoke pattern of blood vessels towards the ovarian tissue. (C) Implants encapsulated by CAM on day 6, leading to even better graft vascularization. (D) Graft penetrating the egg. The arrows point to the grafted ovarian tissue. Please click here to view a larger version of this figure.
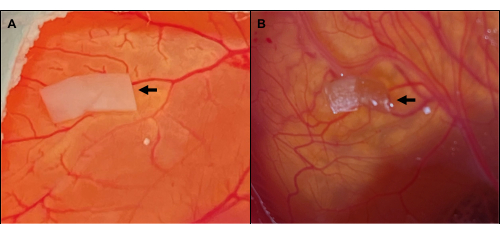
Figure 3: Macroscopic aspect of necrotic implants. (A) Healthy-looking ovarian tissue on day 0 of grafting. (B) Necrotic aspect of the implant 6 days later. The arrows point to the ovarian tissue. Please click here to view a larger version of this figure.
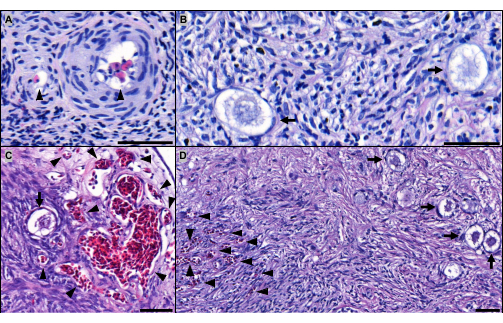
Figure 4: Microscopic aspect of implants. Hematoxylin and eosin-stained sections of implants grafted for (A,B) 1 day and for (C,D) 6 days. The arrowheads point to avian red blood cells perfusing the ovarian vessels after (A) 1 day and (C,D) 6 days of grafting. The arrows indicate healthy-looking primordial follicles in implants grafted for (B) 1 day and (C,D) 6 days. Scale bar: 50 µm. Please click here to view a larger version of this figure.
| Steps | Baths | Time |
| Second fixation | 1 Formalin 10% bath | 2 h |
| Dehydration | 7 methanol baths | 7x 1 h |
| Clarification | 3 toluene baths | 3x 1 h |
| Impregnation | 3 liquid paraffin baths (60°C) | 15 min – 30 min – 30 min |
Table 1: Paraffin embedding steps.
| Non-grafted | Grafting day 1 | Grafting day 6 | P-value | |
| Macroscopic aspect of grafts | ||||
| Tissue survival rate | – | 100% (10/10) | 83% (10/12) | / |
| Microscopic aspect of grafts | ||||
| Follicle density (n/mm3) | 848 ± 272 | 817 ± 370 | 684 ± 236 | p > 0.8 |
| Follicle survival rate | 100% | 95% ± 19% | 83% ± 27% | p > 0.5 |
| Avian red blood cells present in implants | 0% (0/10) | 30% (3/10) | 100% (10/10) | / |
Table 2: Results. Statistical analysis conducted using one-way ANOVA, followed by Sidak correction. Results are expressed as mean ± standard deviation.
Discussion
The most challenging part of the protocol described here is making the small hole required to aspirate the albumen in order to detach the CAM from the eggshell prior to creating a window. Applying too much pressure can result in overpenetration or may even crack and destroy the egg, causing irrevocable damage to the CAM and its vasculature. To keep mistakes to a minimum during initial attempts to separate the CAM, it is strongly advised to practice making small holes in the eggshell of non-fertilized, grocery-bought eggs using a straight pin. Furthermore, given the natural variability of fertility across batches, along with the fact that not all embryos survive the procedure, obtaining supernumerary eggs from the egg supplier is recommended. In order to achieve optimal embryo survival rates and a well-developed CAM, the eggs need to be incubated in ideal conditions. In the case of low viability, different aspects may be to blame and need investigating. The egg supplier should be contacted, as the viability of embryos from the supplier may simply be lower at this point in time. Low egg viability could also be due to inaccurate or inappropriate incubator settings, which may compromise the CAM development. The use of an independent hygrometer and thermometer can ensure that the settings are accurate and stable, but detailed troubleshooting instructions should be provided by the manufacturer. Finally, it has also been suggested that checking transplanted eggs too often may result in temperature and humidity fluctuations, which could have deleterious effects on the grafted embryos33.
In the present protocol, the windowing of the eggshell was performed on day 3 of ED by aspirating around 2 mL of albumen from the embryo to detach the CAM from the shell, which, in turn, allowed the creation of a window without damaging the CAM. The embryo survival rate 4 days later was 79%, consistent with previous studies34,35. Other methods have been reported, with varying degrees of success. One alternative is to prise the CAM away from the shell by applying suction to the air sac, typically around day 7-10 of ED. This method does not require the egg to be punctured33,36. Studies reporting embryo survival rates using the latter technique are few and far between in the literature, with one team reporting up to 90% of embryo survival36. Another option to consider is shell-less chick embryo culture, also known as ex ovo culture. Removing the eggshell provides unrestricted access to the embryo, thus facilitating embryo manipulation and surgery and also allowing the use of high-resolution imaging techniques for live experiments, such as fluorescence microscopy and microcomputed topography37,38. In order to conduct ex ovo culture, the embryo, including its extraembryonic membranes, is transferred to a specific culture containment between day 2 and day 4 of ED. However, this technique is highly invasive, with embryos usually dying around day 12 and fewer than 18% of them surviving until day 1538,39.
During the xenografting experiments reported herein, human ovarian implants were grafted to CAMs that had been gently traumatized by applying a small strip of sterile ether-extracted lens paper to the surface of the epithelium and removing it immediately. Overall, the embryo survival rates were excellent and reached 96% during the grafting period, which is consistent with previous studies involving the transplantation of ovarian tissue fragments to CAMs23,40. Moreover, this approach has been shown to increase graft adhesion rates and enhance the subsequent establishment of neovascularization, with angiogenesis likely induced by the initial wound-healing process23. Indeed, xenografting rat ovarian tissue to murine wound-healing granulation tissue was found to improve graft vascularization41. Likewise, in the first live birth reported after orthotopic transplantation of cryopreserved human ovarian tissue, Donnez et al. created a peritoneal window and coagulated its margins 7 days before implantation to stimulate angiogenesis and neovascularization in the transplantation site42.
With regard to the histological assessment of grafted human ovarian tissue, the follicle survival rates were encouraging, with more than 80% of follicles remaining intact after 6 days of grafting. Interestingly, the follicle survival rates reported in the literature after the transplantation of human frozen-thawed ovarian tissue to the peritoneum of immunodeficient mice are only around 30%10,14,18,43,44, which is much lower than in the present study. Our findings of enhanced follicle survival after the grafting of human ovarian tissue to the CAM corroborate our recently published data, in which we demonstrate that CAM inhibits follicle activation and apoptosis24. Moreover, avian blood vessels were able to penetrate the implants, as demonstrated by the presence of avian erythrocytes after just 1 day of transplantation. All in all, these results demonstrate that the CAM approach is a suitable model for further scrutinizing the transplantation of human ovarian tissue, since it supports the survival of the tissue.
The major drawback of the CAM model is the limited period during which grafting is possible. Indeed, transplantation can be performed at the earliest on day 7 of ED, as soon as the CAM has developed sufficiently, until day 17, before the chick embryos acquire immunocompetence and hatch. Despite the short grafting duration, the CAM model is still a useful tool for studying human ovarian tissue transplantation in its first ischemic stages. One can also use this model to test the impact of proangiogenic factors, protective antioxidant agents, cytokines, growth- and reproduction-related hormones, and factors controlling follicle activation in grafted human ovarian tissue, all under experimental conditions that can be easily controlled25,45.
Divulgations
The authors have nothing to disclose.
Acknowledgements
The authors thank Mira Hryniuk, BA, for reviewing the English language of the article.
Materials
| Agani hypodermic needle, 19 G | Terumo Europe | AN*1950R1 | 19 G needle to aspirate albumen |
| Terumo syringe, 5 mL concentric Luer lock | Terumo Europe | SS*05LE1 | 5-mL sterile syringe |
| Caseviewer v2.2 | 3DHISTECH | Image analysis software | |
| Diethyl ether | Merck Chemicals | 603-022-00-4 | Sterile ether to traumatize the CAM |
| Eosin Y aqueous solution 0.5% | Merck | 1098441000 | Staining solution |
| Formaldehyde 4% aqueous solution buffered (Formalin 10%) | VWR | 97139010 | Formaldehyde used for tissue fixation |
| Fridge | Liebherr | 7081260 | Fridge at 4 °C used for paraffin-embedding |
| Heating plate | Schott | SLK2 | Hot plate used to dry the slides |
| Incubator | Thermo Forma Scientific 3111 | 10365156 | Oven used for slide incubation |
| Leica CLS 150 XE microscope cold light source | Leica | CLS 150 XE | Focal cold light source to candle the eggs |
| Lens cleaning tissue, grade 541 | VWR | 111-5003 | Tissue to soak in sterile ether to traumatize the CAM |
| Mayer's hematoxylin | MERCK | 1092491000 | Staining solution |
| Methanol | VWR | 20847307 | Methanol |
| Microtome | ThermoScientific-MICROM | HM325-2 | Microtome |
| Pannoramic P250 Flash III | 3DHISTECH | / | Slide scanner at 20x magnification |
| Paraformaldehyde | Merck | 1,04,00,51,000 | Paraffin-embedding solution |
| Paraplast Plus R | Sigma | P3683-1KG | Paraffin |
| Petri dish, 60×15 mm, sterile | Greiner | 628161 | Sterile petri dish |
| Pin holder | Fine Science Tools | 26016-12 | Pin holder |
| Polyhatch | Brinsea | CP01F | Egg incubator with automatic rotator |
| Scroll saw blade, 132 mm | Sencys | / | Saw blade to create a window in the eggshell |
| Stainless steel insert pins | Fine Science Tools | 26007-02 | Straight pin to make a hole in the eggshell |
| Steril-Helios | Angelantoni Industrie | ST-00275400000 | Laminar flow hood |
| Superfrost Plus bords rodés 90° | VWR | 631-9483 | Glass slides |
| Tissue-Tek VIP 6Al | Sakura | 60320417-0711 VID6E3-1 | Automatic embedding device |
| Titanium forceps | Fine Science Tools | 11602-16 | Forceps for eggshell removal and ovarian tissue manipulation |
| Toluene, pa | VWR | 28701364 | Paraffin-embedding solution |
References
- Cacciottola, L., Donnez, J., Dolmans, M. M. Ovarian tissue and oocyte cryopreservation prior to iatrogenic premature ovarian insufficiency. Best Practice & Research Clinical Obstetrics & Gynaecology. 81, 119-133 (2022).
- Dolmans, M. -. M., Hossay, C., Nguyen, T. Y. T., Poirot, C. Fertility preservation: How to preserve ovarian function in children, adolescents and adults. Journal of Clinical Medicine. 10 (22), 5247 (2021).
- Donnez, J., Dolmans, M. -. M. Fertility preservation in women. New England Journal of Medicine. 377 (17), 1657-1665 (2017).
- Donnez, J., Dolmans, M. -. M. Fertility preservation in women. Nature Reviews Endocrinology. 9 (12), 735-749 (2013).
- Dolmans, M. -. M., et al. Transplantation of cryopreserved ovarian tissue in a series of 285 women: a review of five leading European centers. Fertility and Sterility. 115 (5), 1102-1115 (2021).
- Shapira, M., Dolmans, M. -. M., Silber, S., Meirow, D. Evaluation of ovarian tissue transplantation: results from three clinical centers. Fertility and Sterility. 114 (2), 388-397 (2020).
- Jensen, A. K., et al. Outcomes of transplantations of cryopreserved ovarian tissue to 41 women in Denmark. Human Reproduction. 30 (12), 2838-2845 (2015).
- Vander Ven, H., et al. Ninety-five orthotopic transplantations in 74 women of ovarian tissue after cytotoxic treatment in a fertility preservation network: Tissue activity, pregnancy and delivery rates. Human Reproduction. 31 (9), 2031-2041 (2016).
- Diaz-Garcia, C., et al. Oocyte vitrification versus ovarian cortex transplantation in fertility preservation for adult women undergoing gonadotoxic treatments: A prospective cohort study. Fertility and Sterility. 109 (3), 478-485 (2018).
- Nisolle, M., Casanas-Roux, F., Qu, J., Motta, P., Donnez, J. Histologic and ultrastructural evaluation of fresh and frozen-thawed human ovarian xenografts in nude mice. Fertility and Sterility. 74 (1), 122-129 (2000).
- Van Eyck, A. -. S., et al. Electron paramagnetic resonance as a tool to evaluate human ovarian tissue reoxygenation after xenografting. Fertility and Sterility. 92 (1), 374-381 (2009).
- Van Eyck, A. -. S., et al. Both host and graft vessels contribute to revascularization of xenografted human ovarian tissue in a murine model. Fertility and Sterility. 93 (5), 1676-1685 (2010).
- Hossay, C., et al. Can frozen-thawed human ovary withstand refreezing-rethawing in the form of cortical strips. Journal of Assisted Reproduction and Genetics. 37 (12), 3077-3087 (2020).
- Manavella, D. D., et al. Two-step transplantation with adipose tissue-derived stem cells increases follicle survival by enhancing vascularization in xenografted frozen-thawed human ovarian tissue. Human Reproduction. 33 (6), 1107-1116 (2018).
- Rahimi, G., et al. Re-vascularisation in human ovarian tissue after conventional freezing or vitrification and xenotransplantation. European Journal of Obstetrics & Gynecology and Reproductive Biology. 149 (1), 63-67 (2010).
- Cacciottola, L., Manavella, D. D., Amorim, C. A., Donnez, J., Dolmans, M. -. M. In vivo characterization of metabolic activity and oxidative stress in grafted human ovarian tissue using microdialysis. Fertility and Sterility. 110 (3), 534-544 (2018).
- Gavish, Z., et al. Follicle activation is a significant and immediate cause of follicle loss after ovarian tissue transplantation. Journal of Assisted Reproduction and Genetics. 35 (1), 61-69 (2018).
- Masciangelo, R., Hossay, C., Donnez, J., Dolmans, M. -. M. Does the Akt pathway play a role in follicle activation after grafting of human ovarian tissue. Reproductive BioMedicine Online. 39 (2), 196-198 (2019).
- Fortune, J. E., Cushman, R. A., Wahl, C. M., Kito, S. The primordial to primary follicle transition. Molecular and Cellular Endocrinology. (1-2), 53-60 (2000).
- Cushman, R. A. Bovine ovarian cortical pieces grafted to chick embryonic membranes: A model for studies on the activation of primordial follicles. Human Reproduction. 17 (1), 48-54 (2002).
- Gigli, I., Cushman, R. A., Wahl, C. M., Fortune, J. E. Evidence for a role for anti-Müllerian hormone in the suppression of follicle activation in mouse ovaries and bovine ovarian cortex grafted beneath the chick chorioallantoic membrane. Molecular Reproduction and Development. 71 (4), 480-488 (2005).
- Qureshi, A. I., et al. Testosterone selectively increases primary follicles in ovarian cortex grafted onto embryonic chick membranes: relevance to polycystic ovaries. Reproduction. 136 (2), 187-194 (2008).
- Martinez-Madrid, B., et al. Chick embryo chorioallantoic membrane (CAM) model: A useful tool to study short-term transplantation of cryopreserved human ovarian tissue. Fertility and Sterility. 91 (1), 285-292 (2009).
- Hossay, C., et al. Follicle outcomes in human ovarian tissue: Effect of freezing, culture, and grafting. Fertility and Sterility. 119 (1), 135-145 (2023).
- Ribatti, D. . The Chick Embryo Chorioallantoic Membrane in the Study of Angiogenesis and Metastasis. , (2010).
- Pawlikowska, P., et al. Exploitation of the chick embryo chorioallantoic membrane (CAM) as a platform for anti-metastatic drug testing. Scientific Reports. 10 (1), 16876 (2020).
- Davison, T. F. The immunologists’ debt to the chicken. British Poultry Science. 44 (1), 6-21 (2003).
- Schilling, M. A., et al. Transcriptional innate immune response of the developing chicken embryo to Newcastle disease virus infection. Frontiers in Genetics. 9, 61 (2018).
- Directives originating from the EU. Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes (Text with EEA relevance). The National Archives Available from: https://www.legislation.gov.uk/eudr/2010/63/contents (2010)
- Dolmans, M. -. M., et al. A review of 15 years of ovarian tissue bank activities. Journal of Assisted Reproduction and Genetics. 30 (3), 305-314 (2013).
- Fischer, A. H., Jacobson, K. A., Rose, J., Zeller, R. Hematoxylin and eosin staining of tissue and cell sections. Cold Spring Harbor Protocols. 2008 (5), 4986 (2008).
- Anderson, R. A., McLaughlin, M., Wallace, W. H. B., Albertini, D. F., Telfer, E. E. The immature human ovary shows loss of abnormal follicles and increasing follicle developmental competence through childhood and adolescence. Human Reproduction. 29 (1), 97-106 (2014).
- Sharrow, A. C., Ishihara, M., Hu, J., Kim, I. H., Wu, L. Using the chicken chorioallantoic membrane in vivo model to study gynecological and urological cancers. Journal of Visualized Experiments. (155), e60651 (2020).
- Lokman, N. A., Elder, A. S. F., Ricciardelli, C., Oehler, M. K. Chick chorioallantoic membrane (CAM) assay as an in vivo model to study the effect of newly identified molecules on ovarian cancer invasion and metastasis. International Journal of Molecular Sciences. 13 (8), 9959-9970 (2012).
- Sys, G. M. L., et al. The In ovo CAM-assay as a xenograft model for sarcoma. Journal of Visualized Experiments. (77), e50522 (2013).
- Li, M., et al. The in ovo chick chorioallantoic membrane (CAM) assay as an efficient xenograft model of hepatocellular carcinoma. Journal of Visualized Experiments. (104), e52411 (2015).
- Cloney, K., Franz-Odendaal, T. A. Optimized ex-ovo culturing of chick embryos to advanced stages of development. Journal of Visualized Experiments. (95), e52129 (2015).
- Yalcin, H. C., Shekhar, A., Rane, A. A., Butcher, J. T. An ex-ovo chicken embryo culture system suitable for imaging and microsurgery applications. Journal of Visualized Experiments. (44), e2154 (2010).
- Schomann, T., Qunneis, F., Widera, D., Kaltschmidt, C., Kaltschmidt, B. Improved method for ex ovo-cultivation of developing chicken embryos for human stem cell xenografts. Stem Cells International. 2013, 960958 (2013).
- Beck, K., Singh, J., Dar, M. A., Anzar, M. Short-term culture of adult bovine ovarian tissues: chorioallantoic membrane (CAM) vs. traditional in vitro culture systems. Reproductive Biology and Endocrinology. 16 (1), 21 (2018).
- Israely, T., Nevo, N., Harmelin, A., Neeman, M., Tsafriri, A. Reducing ischaemic damage in rodent ovarian xenografts transplanted into granulation tissue. Human Reproduction. 21 (6), 1368-1379 (2006).
- Donnez, J., et al. Livebirth after orthotopic transplantation of cryopreserved ovarian tissue. The Lancet. 364 (9443), 1405-1410 (2004).
- Dolmans, M. -. M., et al. Short-term transplantation of isolated human ovarian follicles and cortical tissue into nude mice. Reproduction. 134 (2), 253-262 (2007).
- Baird, D. T., Webb, R., Campbell, B. K., Harkness, L. M., Gosden, R. G. Long-term ovarian function in sheep after ovariectomy and transplantation of autografts stored at −196 C. Endocrinology. 140 (1), 462-471 (1999).
- Ribatti, D., Vacca, A., Roncali, L., Dammacco, F. The chick embryo chorioallantoic membrane as a model for in vivo research on anti-angiogenesis. Current Pharmaceutical Biotechnology. 1 (1), 73-82 (2000).

