Management of the Uncinate Process in No-Touch Laparoscopic Pancreaticoduodenectomy
Summary
Complete resection of the uncinate process and mesopancreas is one of the most important and difficult processes in laparoscopic pancreatoduodenectomy (LPD). This article presents a method for managing the uncinate process in no-touch LPD using the median-anterior and left-posterior approaches to the superior mesenteric artery (SMA).
Abstract
Laparoscopic pancreatoduodenectomy (LPD) is a demanding abdominal operation that necessitates meticulous surgical skills and teamwork. The management of the pancreatic uncinate process is one of the most important and difficult processes in LPD because of its deep anatomical location and difficult exposure. Complete resection of the uncinate process and mesopancreas has become the cornerstone of LPD. In particular, it is even more difficult to avoid positive surgical margins and incomplete lymph node dissection when the tumor is located in the uncinate process. No-touch LPD, which is an ideal oncological operation process fitting the “tumor-free” principle, has been reported by our group previously. This article introduces the management of the uncinate process in no-touch LPD. Based on the multi-angle arterial approach, in this protocol, the median-anterior and left-posterior approaches to the SMA are used to correctly deal with the important vascular structure, the inferior pancreaticoduodenal artery (IPDA), in order to ensure the safe and complete excision of the uncinate process and mesopancreas. For the achievement of the no-touch isolation technique in LPD, the pancreatic head and the blood supply to the duodenal region must be severed at the very early stage of the operation; after that, the tumor can be isolated intact, resection can be performed in situ, and finally, the tissue can be removed en bloc. This paper aims to show the distinctive ways to manage the uncinate process in no-touch LPD and investigate the viability and safety of this approach. Moreover, the technique may increase the R0 resection rate.
Introduction
The pancreatic uncinate process is the protrusion from the bottom left of the pancreatic head that is situated behind the SMA and superior mesenteric vein (SMV)1. The management of the uncinate process is a challenging area in pancreatic surgery due to its deep anatomy and challenging exposure; therefore, especially for pancreatic cancer located in the uncinate process, surgery is prone to result in a positive surgical margin, incomplete lymph node dissection, and poor prognosis2. Therefore, improved surgical techniques and strategies are urgently required.
Most resection procedures of the pancreatic head and uncinate process are performed along the right side of the SMV and SMA3. Although this approach largely works for ampullary tumors, it has drawbacks for pancreatic head cancer, particularly for large tumors in the uncinate process2. During the procedure, it is frequently required to spin and pull the SMV and SMA in order to expose them. The IPDA cannot be dissected clearly with such approaches, which usually results in bleeding or insufficient cleaning of the area. In this work, the authors describe an approach that can support the early control of the IPDA, which results in better bleeding control, less blood loss, and better dissection of the uncinate process.
Meanwhile, conventional laparotomy or laparoscopy pancreatoduodenectomy requires a Kocher maneuver for the wide mobilization of the duodenum and pancreatic head4. However, with this maneuver, the tumor cells have the potential capacity to metastasize via the portal vein (PV), as the surgeon might squeeze the tumor while holding it during surgery4,5,6. The no-touch isolation technique is one of the popular concepts in pancreatoduodenectomy. Although it has not been proven by large clinical trials whether this surgical intervention could enhance the cancer-related prognosis of pancreatic cancer patients, the study of Hirota et al.7 reported the significance of no-touch technology in preventing cancer cell metastasis by using molecular markers (CEA mRNA) to detect cancer cells in the portal vein blood. In their study, the no-touch technique group had a lower spread rate of portal vein cancer cells and a higher 3 year survival rate than the conventional technique group. No-touch LPD has been reported by the team of the current authors previously as an ideal oncological operation process fitting the "tumor-free" principle8,9.
This article presents the management of the uncinate process in no-touch LPD. The median-anterior and left-posterior approaches to the SMA were performed to deal precisely with the IPDA. To achieve the no-touch isolation technique in LPD, the blood supply to the duodenum and pancreatic heads must be cut off at the very early stage of the operation, after which the tumor can be isolated intact, resected in situ, and finally, removed en bloc.
The purpose and advantages of this strategy are to ensure the safe and complete excision of the uncinate process and mesopancreas based on a multi-angle arterial approach10. This article aims to explore the efficacy and safety of this technique for the management of the uncinate process in no-touch LPD, which might improve the R0 resection rate11.
Protocol
This study was approved by the Ethics Committee of the Second Affiliated Hospital of Guangzhou University of Chinese Medicine, and informed written consent was obtained from the patients involved in this study.
1. Preoperative work-up
- Perform the high-resolution contrast-enhanced CT12,13 (see Table of Materials) to assess the tumor's extent and search for any abnormal vasculature.
- Determine the inclusion and exclusion criteria for patients.
- Select patients meeting the following criteria: (1) a tumor located in the uncinate process that needs radical resection; (2) resectable cases.
- Exclude the following cases: (1) metastatic tumor; (2) patients with poor cardio-pulmonary function that may not tolerate laparoscopic surgery; (3) a tumor that has invaded the major vessels such as SMA, common hepatic artery (CHA), or celiac artery (CA).
2. Anesthetization steps
- Use 0.3 µg/kg sufentanil, 3 µg/mL propofol, and 0.15 mg/kg cis-atracurium (see Table of Materials) for anesthesia induction.
- Perform endotracheal intubation14 after the muscle relaxant takes effect.
- Puncture and catheterize the radial artery and central vein under the guidance of ultrasound.
- Use target-controlled infusion (TCI) of 1-2 µg/mL propofol, 1-3 ng/mL remifentanil, 1.53 µg/kg/min cis-atracurium, and sevoflurane (see Table of Materials) with an inhaled concentration of 1%-2% to maintain anesthesia, and keep the depth of anesthesia between 37 and 64 on the Nacrotrend Index15.
NOTE: The Nacrotrend anesthesia monitoring system (see Table of Materials) is an anesthesia depth monitoring method developed in recent years. It obtains the original EEG, analyzes the changes in frequency and power in the EEG, integrates the factors of phase and harmonics, and expresses the anesthesia depth in the form of a value from 0-99. - Set the ventilator parameters to volume control ventilation mode, with a tidal volume of 8 mL/kg, an inhalation oxygen saturation of 60%, a gas flow of 2 L/min, and an end-expiratory carbon dioxide range of 35-45 mmHg.
3. Installation
- Refer to our previously published8 article (Figure 1) for details on the patient position and trocar (see Table of Materials) location.
- Surgeon position: Ensure that the operator surgeon and the first assistant stand on the patient's right and left sides, respectively. Ensure that the holder of the laparoscope stands between the patient's legs.
4. Surgery techniques
- Inspect the peritoneal surfaces and intraperitoneal organs for any potential extrapancreatic metastases. Remove the greater omentum for better exposure, and open the lesser sac by severing the gastrocolic ligament.
- Dissect the gallbladder triangle with an ultrasonic knife, then resect and sever the gallbladder artery and cystic duct, and finally, remove the gallbladder, and suspend the liver to expose the hepatoduodenal ligament and hilum (Figure 2).
- Explore the gap between the pancreatic neck and the SMV in order to assess the surgical resectability.
NOTE: The preferred gap is the space between the posterior neck of the pancreas and the SMV, which is difficult to open and expose if the tumor has invaded the SMV. - Suspend the transverse colon and its mesentery on the cephalic side to establish effective exposure.
- Using the ileocolonic artery as the marker, open the transverse mesocolon. Protect the ileocolon artery and middle colonic artery.
- Expose the second and third segments of the duodenum, dissect along the right side of the SMV, and separate the second and third segments of the duodenum from the transverse mesocolon.
- Expose the main trunk of SMV and the proximal-dorsal jejunal vein (PDJV) between the SMV and SMA (Figure 3), and then ligate and dissect the inferior pancreaticoduodenal vein (IPDV).
NOTE: Be careful not to apply too much pressure, as this might cause the IPDV to hemorrhage. - Perform the median-anterior approach to the SMA following steps 4.8.1-4.8.4.
- Start the median-anterior approach to the SMA from the inferior colon region.
- Pull the SMV just to the right, widen the operation field, and expose the SMA pulsation to determine its trajectory.
- Expose the right half of the SMA, and dissect outside the arterial sheath of the SMA on the anterior-right margin in order to separate the SMA from the pancreatic mesopancreas.
NOTE: The middle colonic artery (MCA), which runs along the side of the vascular arch of the margin of the transverse mesocolon, can be directly linked to the trunk of the SMA in cases where it is difficult to identify the SMA, such as when it is difficult to assess arterial visual pulsation in obese patients. The best place to locate and identify the trunk of the SMA is where the MCA begins in the inferior colonic region. If required, color ultrasound assistance could be used. - Through the median-anterior approach to the SMA approach, expose the main branches of the IPDA or jejunal artery (JA) along the right side of the SMA, and then conduct a further follow-up to confirm them (Figure 4).
NOTE: The authors' experience is that IPDA and JA can be effectively identified by the subsequent left-posterior approach to the SMA.
- Perform the left-posterior approach to the SMA following steps 4.9.1-4.9.9.
- Place the entire small bowel to the right side to facilitate space exposure between the fourth segment of the duodenum and the inferior vena cava.
- Dissect the fusion fascia, and expose the left renal vein (LRV) to establish the posterior space of the SMA.
- Use an ultrasonic scalpel to dissect the ligament of Treitz between the beginning part of the jejunum and the mesentery of the transverse colon.
- Use a stapler to divide the proximal jejunum.
- Place an Fr8 catheter for suspending the dorsal sides of the SMA and SMV.
- To facilitate SMA dissection on its periadventitial plane on the anterior-left border and its detachment from the mesopancreas, pull the catheter to the upper right side. Establish an effective exposure space at the left-posterior aspect of the SMA (Figure 5).
- Trace up the SMA to the root along the first jejunal artery (FJA).
- Dissect the SMA on its periadventitial plane on the anterior-left margin in order to separate it from the pancreatic mesopancreas. Circumferentially dissect the SMA.
- Ligate and dissect the IPDA (Figure 6). Sacrifice the FJA if it is involved in the tumor.
- Using the median-anterior and left-posterior SMA approaches, separate the SMA and SMV from the uncinate process and mesopancreas completely (Figure 7).
- Return to the superior region of the colon following steps 4.11.1-4.11.16.
- Draw the proximal jejunum from the dorsal of the SMA to the right.
- Reveal the right gastrointestinal vein, the collateral right colonic vein, and the SMV trunk using an ultrasonic knife (see Table of Materials).
- Expose Henle's gastrocolic trunk vein to disconnect its distal and proximal ends on the right side of the SMV trunk.
NOTE:Find Henle's gastrocolic trunk vein along the right reticular vein on the right side of the SMV. - Divide the stomach 3-5 cm away from the pylorus using a stapler device (see Table of Materials).
- Expose the CHA at the superior margin of the pancreatic neck.
- Dissect along the CHA toward the first hepatic hilar to reveal the proper hepatic artery (PHA) and the right gastric artery (RGA). Ligate and sever the RGA.
- Use laparoscopic bulldog clamps (see Table of Materials) to temporarily occlude the common bile duct (CBD) after separating the CBD.
- Dissect the hepatoduodenal ligament. Execute a lymphadenectomy along the CHA, PV, and PHA.
- Ligate and dissect the right gastric artery.
- Locate the gastroduodenal artery (GDA) at the junction of the PHA with the CHA, and meticulously ligate or suture (5-0, see Table of Materials) the GDA to reduce the possibility of an ensuing erosion or hemorrhage.
- Explore the tunnel between the pancreatic neck and the SMV. Ligate and dissect the dorsal pancreatic artery (DPA).
- Place two Fr14 catheters to block the pancreatic neck blood supply in order to reduce bleeding (Figure 8).
- Dissect the parenchyma of the pancreatic neck using scissors. Suture (5-0, see Table of Materials) the bleeding point of the resection surface.
NOTE: Examine a fast-frozen pathological specimen of the pancreatic stump to prove the negative margin. - Suspend the splenic vein (SV) to establish exposure after the left first branch of the SMV has been ligated and dissected (Figure 9).
- Ligate and dissect the uncinate process artery behind the SV. Identify and retain the alternative right hepatic artery originating from the SMA from the uncinate process artery.
NOTE: At this point, all the blood supplies to the tumor are completely occluded. - Ligate and dissect the branches of the PV.
- Using the Kocher maneuver, cut off the lymphatics, and dissect the duodenum from the retroperitoneum. Resect the tumor in situ, and remove it en bloc following the oncologic principles of no-touch16,17.
- Use Child's method for the reconstruction of the digestive tract8,18.
- Perform a single-layer running suture hepaticojejunostomy from end to side using 4-0 absorbable sutures. Utilize an internal stent to perform a duct-to-mucosal, end-to-side pancreaticojejunostomy.
5. Postoperative management
- If the gastric tube drainage is <200 mL, remove the tube on the next day.
- Start appropriate physical exercise if the vital signs are stable and the patient is generally in good condition.
- Assess the amylase level of the drainage liquid on postoperative day 3, and review the abdominal CT on postoperative day 7. Remove the drainage tubes if there is no evidence of biochemical leakage, lymphatic fistula, or hemorrhage.
Representative Results
A male patient aged 59 years old with symptoms including upper abdominal pain and jaundice was hospitalized in our department. He had no previous medical history and a relatively moderate normal body mass index (23.94 kg/m2). A contrast-enhanced CT scan was performed, and a tumor with a diameter of approximately 5.5 cm x 4.6 cm x 6.3 cm was found at the head and uncinate process of the pancreas (Figure 10). No evidence of distant metastasis was found, and percutaneous transhepatic cholangical drainage19 was performed before the operation. The total bilirubin declined from 201.4 µmol/mL to 36.0 µmol/mL.
Representative results are shown in Table 1. The operation lasted 314 min, and the total blood loss was 80 mL. The pancreatic fistula was not found, as the amylase level of the abdominal drainage fluid was normal throughout the postoperative period, and the drainage tubes were removed before postoperative day 8. No other significant morbidity occurred, and this patient was discharged on the 11th postoperative day. Histopathology confirmed a pancreatic intraductal papillary mucinous neoplasm with focal moderately differentiated invasive adenocarcinoma (Figure 11). The surgical margins were microscopically negative (R0), and none of the 18 lymph nodes were involved.
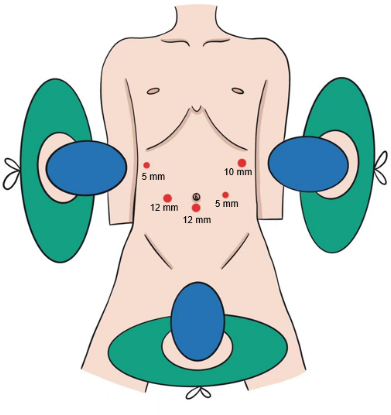
Figure 1: Distribution of the trocars. The five-port method was used. The patient was laid down in a supine position with his legs spread out. Please click here to view a larger version of this figure.
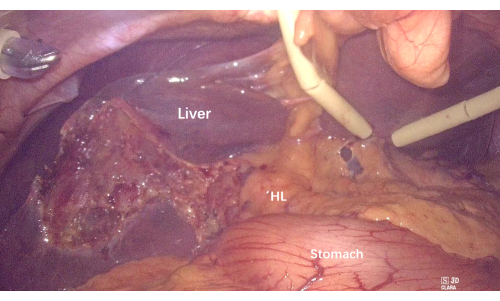
Figure 2: Removal of the gallbladder and suspending the liver to expose the hepatoduodenal ligament and hilum. Abbreviation: HL = hepatoduodenal ligament. Please click here to view a larger version of this figure.
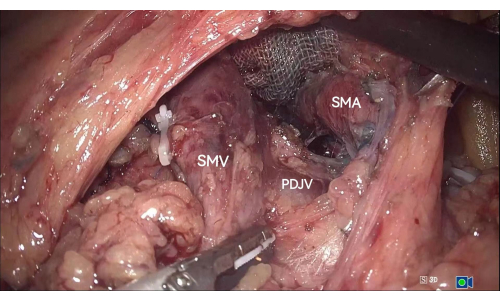
Figure 3: The PDJV between the SMV and the SMA is exposed in the inferior colonic region. Abbreviations: PDJV = proximal-dorsal jejunal vein; SMV = superior mesenteric vein; SMA = superior mesenteric artery. Please click here to view a larger version of this figure.
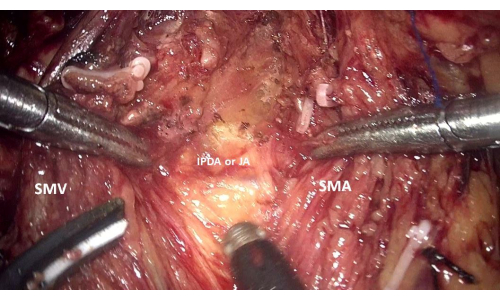
Figure 4: Exposure of the main branches of the IPDA and JA along the right side of the SMA through the median-anterior approach to the SMA. Abbreviations: IPDA = inferior pancreaticoduodenal artery; JA = jejunal artery; SMV = superior mesenteric vein; SMA = superior mesenteric artery. Please click here to view a larger version of this figure.
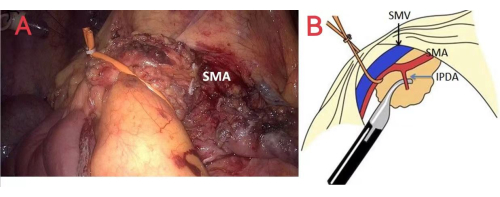
Figure 5: Method of hanging the SMA and SMV. (A) and (B) represent the method of hanging the SMA and SMV to establish a sufficient view of the left-posterior surface of the SMA. Abbreviations: IPDA = inferior pancreaticoduodenal artery; SMA = superior mesenteric artery; SMV = superior mesenteric vein. Please click here to view a larger version of this figure.
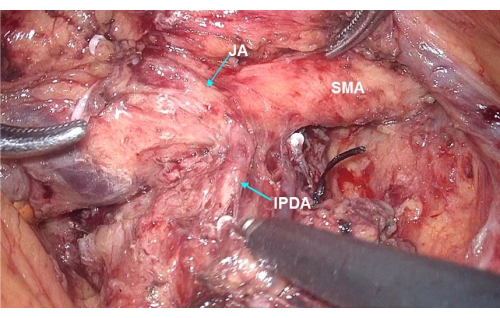
Figure 6: Dissection along the SMA circumferentially exposing the IPDA, which originates directly from the SMA. Abbreviations: IPDA = inferior pancreaticoduodenal artery; JA = jejunal artery; SMA = superior mesenteric artery. Please click here to view a larger version of this figure.
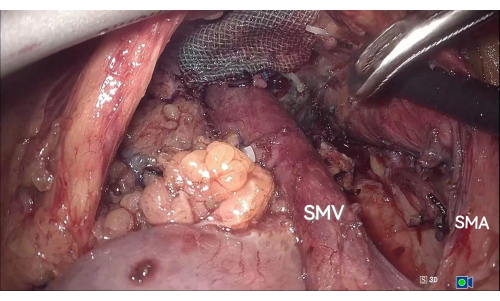
Figure 7: Separating the SMA and SMV from the uncinate process and mesopancreas. Using the median-anterior and left-posterior approaches to the SMA, the SMA and SMV are completely separated from the uncinate process and mesopancreas. Abbreviations: SMA = superior mesenteric artery; SMV = superior mesenteric vein. Please click here to view a larger version of this figure.
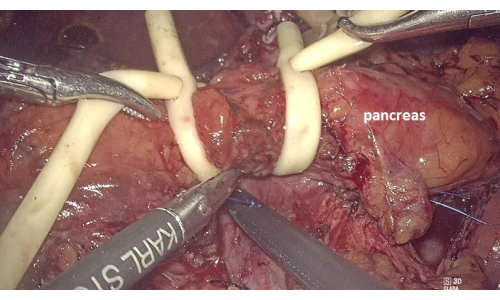
Figure 8: Placing two Fr14 catheters to block the pancreatic neck blood supply and reduce bleeding. Please click here to view a larger version of this figure.
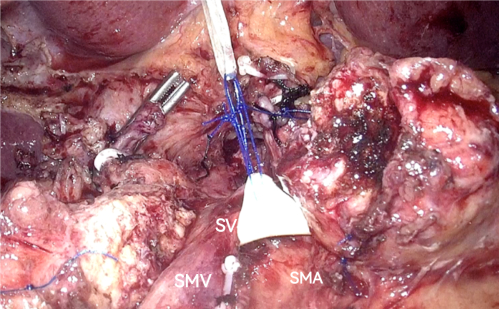
Figure 9: Suspending the SV to establish the exposure. Abbreviations: SV = splenic vein; SMA = superior mesenteric artery; SMV = superior mesenteric vein. Please click here to view a larger version of this figure.
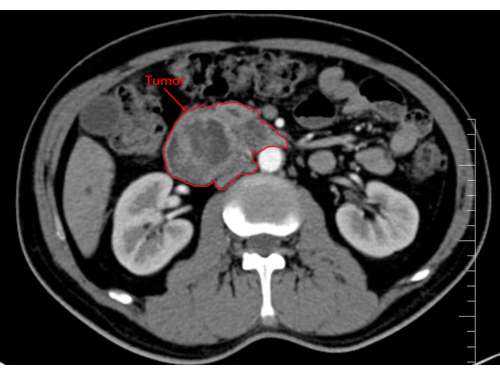
Figure 10: Image showing the tumor in the head and uncinate process of the pancreas. Please click here to view a larger version of this figure.
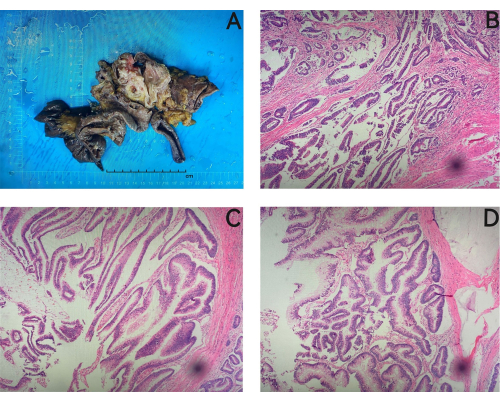
Figure 11: Histopathological study. Histopathology confirmed a pancreatic intraductal papillary mucinous neoplasm with focal moderately differentiated invasive adenocarcinoma. (A) Postoperative paraffin specimens in which the size of the stomach is 7 cm x 5 cm x 2.5 cm, the length of the small intestine is about 22 cm, the size of the pancreas is 6 cm x 4 cm x 3 cm, and there is a gray mass in the head of the pancreas, which is about 6 cm x 3 cm x 1.5 cm in size. (B) Atypical glands, suggesting a focally infiltrating adenocarcinoma. (C,D) An intraductal papillary mucinous tumor. Magnifications: B, 200x; C, 400x; D, 300x. Please click here to view a larger version of this figure.
| Variable | Outcome |
| Intraoperative | |
| operative time, min | 314 |
| Intraoperative blood loss, mL | 80 |
| Postoperative | |
| Postoperative pancreatic fistula | Not found |
| Drain removal, postoperative day | 8 |
| Postoperative hospital stay, days | 11 |
| Pathological diagnosis | Pancreatic intraductal papillary mucinous neoplasm with focal moderately differentiated invasive adenocarcinoma |
Table 1: Representative results of the surgery.
Discussion
With intestinal rotation, the ventral pancreas flips to the dorsal side and unites with the dorsal pancreas throughout the development of the human embryo, and the ventral pancreas also grows into the lower portion of the pancreatic head and the uncinate process1. The pancreatic uncinate process is described as the protrusion from the bottom left of the pancreatic head that is situated behind the SMA and the SMV1.
In the meantime, the mesopancreas rotates around the SMA into a spiral along with intestinal rotation20. As the IPDA and IPDV arise from the posterior area of the SMA and SMV, their lengths are also relatively shorter due to the rotation of the mesenteric vascular axis. These vessels may be inadvertently injured and lead to massive bleeding when dissecting the pancreatic uncinate process and mesopancreas from the SMA. Due to this issue, the management of the pancreatic uncinate process is one of the most important and difficult processes in LPD. Importantly, the complete resection of the uncinate process and mesopancreas has become a cornerstone of LPD.
This study provides a unique strategy for the management of the uncinate process in no-touch LPD, which might potentially develop into a reproducible, standardized, and effective oncological procedure for the safe and complete excision of the uncinate process and mesopancreas. The median-anterior and left-posterior approaches to the SMA are used to ligate and dissect the IPDA, IPDV, and uncinate process artery in situ. The blood supply to the pancreatic head and duodenal region must be severed at the very early stage of the operation so that the tumor can be isolated intact, and then SMA and SMV can be separated from the uncinate process and mesopancreas completely, and the specimen can be resected en bloc using the Kocher maneuver.
The pancreatic uncinate process is situated behind the SMA1. Preoperative high-resolution contrast-enhanced CT should be performed to classify the anatomical features of the interaction between them into one of two types: type I, when the uncinate process is located at the right side of the SMA (this type is common in ampullary tumors and some cases of pancreatic head tumor); and type II, when the uncinate process extends to the left side of the SMA (this type is more commonly seen when the tumor is located in the uncinate process of pancreas)21. It is relatively easy to dissociate the uncinate process and mesopancreas from the SMA in type I. However, the SMA, the SMV, and their branches are presented on the ventral side of the uncinate process and tumor in type II. This kind of uncinate process excision is challenging and calls for a multi-angle combined arterial approach and advanced surgical skills. In these situations, dissecting the 14th group of lymph nodes is challenging. Using the median-anterior and left-posterior approaches to the SMA is beneficial and necessary.
The goal and benefit of this method are the demonstration of the feasibility and safety of the management of the uncinate process in no-touch LPD. This complex procedure is recommended to be performed in relatively high-volume centers by experienced surgical teams. The learning curve is inevitably steep as this operation strategy requires effective cooperation among the surgical teams, comprehensive anatomical knowledge of the epigastrium, and a flexible response capability against unexpected variations. Furthermore, a limited number of cases are suitable for this procedure, and randomized clinical trials are difficult to design and implement; as a result, high-level evidence regarding the short-term and long-term outcomes of this strategy is difficult to establish.
Divulgations
The authors have nothing to disclose.
Acknowledgements
Our article #64904 was supported by the Research project of Traditional Chinese Medicine Bureau of Guangdong Province (ID:20222077).
Materials
| 3D Laparoscope | STORZ | TC200,TC302 | |
| Cisatracurium Besylate Injection | Hengrui Pharma | H20183042 | |
| Dual-source dual-energy CT | SOMATOM | Definition FLASH | |
| Echelon Flex Powered Plus Articulating Endoscopic Linear Cutter and Endopath Echelon Endoscopic Linear Cutter Reloads with Gripping Surface Technology | Ethicon Endo-Surgery | ECR60G/GST60G | |
| ENDOPATH XCEL Trocars | Johnson & Johnson | CB5LT/CB12LT | |
| HARMONIC ACE Ultrasonic Surgical Devices | Ethicon Endo-Surgery | HAR36 | |
| Laparoscopic bulldog clamps | Aesculap | FB367R,FB369R | |
| Ligating Clips | Teleflex Medical | 5,44,22,05,44,23,05,44,000 | |
| Nacrotrend anaesthesia monitoring system | Monitor Technik,Bad Bramsted | ||
| PROLENE Polypropylene Nonabsorbable Suture/5/0 | Johnson & Johnson | W8556 | |
| Propofol Injectable Emulsion | Aspen Pharma Trading Limited | H20171275 | |
| Remifentanil Hydrochloride for injection | Humanwell Healthcare | H20030197 | |
| Sevoflurane for Inhalation | Hengrui Pharma | H20070172 | |
| Sufentanil Citrate Injecton | Humanwell Healthcare | H20054171 | |
| Trocars | AOFO | FQ-D1/5.5mm 10.5mm | |
| Ultrasonic Surgical & Electrosurgical Generator | Ethicon Endo-Surgery | GEN11CN |
References
- Léger, L. Pancreatic Surgery. Chugai-Igakusha. , (1984).
- O’Sullivan, A. W., Heaton, N., Rela, M. Cancer of the uncinate process of the pancreas: Surgical anatomy and clinicopathological features. Hepatobiliary and Pancreatic Diseases International. 8 (6), 569-574 (2009).
- Corcione, F., et al. Laparoscopic pancreaticoduodenectomy: Experience of 22 cases. Surgery Endoscopy. 27 (6), 2131-2136 (2013).
- Kuroki, T., Eguchi, S. No-touch isolation techniques for pancreatic cancer. Surgery Today. 47 (1), 8-13 (2017).
- Gall, T. M., et al. Reduced dissemination of circulating tumor cells with no-touch isolation surgical technique in patients with pancreatic cancer. Journal of the American Medical Association Surgery. 149 (5), 482-485 (2014).
- Tien, Y. W., et al. A high circulating tumor cell count in portal vein predicts liver metastasis from periampullary or pancreatic cancer: A high portal venous CTC count predicts liver metastases. Médecine. 95 (16), 3407 (2016).
- Hirota, M., et al. Pancreatectomy using the no-touch isolation technique followed by extensive intraoperative peritoneal lavage to prevent cancer cell dissemination: A pilot study. Journal of the Pancreas. 6 (2), 143-151 (2005).
- Shen, Z., et al. Laparoscopic pancreatoduodenectomy for pancreatic cancer using in-situ no-touch isolation technique. Journal of Visualized Experiments. (180), e63450 (2022).
- Tan, Z. J., et al. Clinical experience of laparoscopic pancreatoduodenectomy via orthotopic resection. Zhonghua Wai Ke Za Zhi. 58 (10), 782-786 (2020).
- Nagakawa, Y., et al. Surgical approaches to the superior mesenteric artery during minimally invasive pancreaticoduodenectomy: A systematic review. Journal of Hepatobiliary Pancreatic Sciences. 29 (1), 114-123 (2022).
- Hermanek, P., Wittekind, C. Residual tumor (R) classification and prognosis. Seminars in Surgical Oncology. 10 (1), 12-20 (1994).
- Chu, A. J., et al. Dual-source dual-energy multidetector CT for the evaluation of pancreatic tumours. The British Journal of Radiology. 85 (1018), 891-898 (2012).
- Mao, G. -. Q., et al. The application value of spectral imaging in diagnosis of pancreatic adenocarcinoma by dual source CT. Journal of Medical Imaging. 26 (07), 1243-1246 (2016).
- Higgs, A., et al. Guidelines for the management of tracheal intubation in critically ill adults. British Journal of Anaesthesia. 120 (2), 323-352 (2018).
- Schultz, A., et al. Comparison of Narcotrend Index, Bispectral Index, spectral and entropy parameters during induction of propofol-remifentanil anaesthesia. Journal of Clinical Monitoring and Computing. 22 (2), 103-111 (2008).
- Cole, W. H. Problems in operability. The Ulster Medical Journal. 23 (2), 102-116 (1954).
- Liu, K. J., Zhao, X. X. Application of tumor-free technique in laparoscopic surgery for malignant tumors. Chinese Journal of Laparoscopic Surgery. 11 (01), 17-19 (2018).
- Osada, S., et al. Reconstruction method after pancreaticoduodenectomy. Idea to prevent serious complications. Journal of the Pancreas. 13 (1), 1-6 (2012).
- Li, M. Q. Clinical practice guidelines for the interventional treatment of advanced pancreatic carcinoma (on trial) (6th edition). Journal of Clinical Hepatology. 38 (06), 1242-1251 (2022).
- Sugiyama, M., et al. Vascular anatomy of mesopancreas in pancreatoduodenectomy using an intestinal derotation procedure. World Journal of Surgery. 44 (10), 3441-3448 (2020).
- Zhu, C. -. F., et al. Novel morphological classification of the normal pancreatic uncinate process based on computed tomography. Journal of International Medical Research. 48 (9), 300060520957453 (2020).

