Noninvasive Intratracheal Lipopolysaccharide Instillation in Mice
Summary
Here, we propose a protocol for intratracheal lipopolysaccharide (LPS) delivery via noninvasive oropharyngeal endotracheal intubation. This method minimizes the trauma of the surgical procedure for the animal and accurately delivers LPS to the trachea and then to the lungs.
Abstract
The acute lung injury (ALI) mouse model induced by lipopolysaccharide (LPS) or endotoxin is still among the most commonly used models in animal studies of acute lung injury or acute inflammation. The current most commonly used methods in acute lung injury mouse models are an intraperitoneal injection of LPS and tracheostomy for the tracheal infusion of LPS. However, the former method lacks lung targeting and damages other organs, and the latter method induces operative trauma, infection risk, and a low survival rate. Here, we recommend a noninvasive oropharyngeal endotracheal intubation method for LPS instillation in mice. In this method, LPS is noninvasively introduced into the trachea through the oropharyngeal cavity to be instilled into the lung with the help of an apparatus for endotracheal intubation. This method not only assures lung targeting but also avoids damage and the risk of death in the animals. We expect that this approach will become widely used in the field of acute lung injury.
Introduction
Acute lung injury (ALI) is a common clinical syndrome. Under a variety of pathogenic factors, the disruption of the physiological barrier of the lung epithelial cells and vascular endothelial cells leads to increased alveolar permeability, thereby causing decreased lung compliance, pulmonary edema, and severe hypoxemia1. Acute respiratory distress syndrome (ARDS) is the most severe form of ALI. Uncontrolled inflammation and oxidative stress damage are considered to be the main causes of ALI and the more severe ARDS2. When alveolar epithelial cells are directly injured due to trauma, the inflammatory response chain of alveolar macrophages is activated, leading to inflammation in the lung3. Globally, there are more than 3 million patients with acute ARDS per year, and they account for approximately 10% of intensive care unit admissions; additionally, the mortality rate in severe cases is as high as 46%4,5,6. Therefore, there is a need to establish a suitable animal model of ALI to study its pathogenesis. The mouse is the most commonly used experimental animal in the study of ALI since its respiratory tract can simulate the human respiratory tract well for ALI studies. Furthermore, ALI manifests as massive inflammatory cell infiltration, increased pulmonary vascular permeability, and pulmonary edema. The changes in inflammatory cytokines in serum and the lung dry-wet weight ratio reflect the degree of ALI7.
At present, the main methods for modeling LPS-induced ALI in mice include intranasal and surgical tracheal intubation8,9. Here, we propose a new method to deliver LPS into the trachea via noninvasive oropharyngeal intubation. This method uses an illuminated intubator to find the trachea of the mouse and then delivers LPS into the trachea and lung. This method delivers LPS to the lungs more accurately than the intranasal method of delivery. Compared with surgical tracheal intubation, this method does not require surgery, avoids causing wounds, and reduces pain in mice10. Therefore, this method can be used to establish a more convincing mouse model of ALI.
Protocol
The animal experiment protocol was reviewed and approved by the Management Committee of Chengdu University of Traditional Chinese Medicine (Record No. 2021-11). Male C57/BL mice (20-25 g, 6-8 weeks old) were used for the present study. The mice were kept in an animal chamber and were free to drink and eat during the experiment.
1. Preparation
- Ensure that the intubation platform consists of a base, a riser, a paper clip, two rubber bands, and some strings. Take a string, pass the string through the two holes at the top of the riser, and tie the two ends of the string, respectively, to the small protrusions at the top of the riser.
NOTE: Leave room for the mouse's head to pass through between the string and the two holes. - Tie two rubber bands to each end of the paper clip, and tape the paper clip with the rubber bands to the back of the riser. Finally, fix the riser to the base at 90° (Figure 1).
- Select a cannula of the appropriate size and length. For a 20−30 g mouse, a 22 G catheter (2.5−3.8 cm long) can be used11. Assemble the cannula onto a cannula pen, and turn on the pen's light (Figure 2).
- Prepare small surgical forceps and a Pasteur pipette by disinfecting them with 70% alcohol.
2. Preparation of the test compound
- Weight and dissolve 3 mg of LPS in 1 mL of phosphate-buffered saline (PBS, pH 7.2) to form an LPS solution with a concentration of 3 mg/mL.
- Weigh and dissolve 10 mg of pentobarbital sodium in 1 mL of normal saline to form a 1% pentobarbital sodium solution. Filter-sterilize the solution using a 0.45 µm syringe filter.
3. Noninvasive oropharyngeal instillation
- Anesthetize the mice with an intraperitoneal injection of 1% pentobarbital sodium at a dose of 50 mg/kg12,13. Determine the depth of anesthesia by a lack of response to the righting reflex.
- Place the anesthetized mouse on the intubation platform. Fix the top front teeth with the thread and the two forefeet with the rubber bands (Figure 3).
- Pull the tongue out with tweezers, and hold it with the left hand. Push the cannula slowly, along the mouth, with the right hand up, to the mandibular epiglottis. Use the light of the cannula pen to find the trachea, and slowly insert it into the trachea (Figure 4).
- After the cannula has been inserted into the trachea, withdraw the intubation pen slowly, and leave the cannula inside. Insert the Pasteur pipette into the cannula joint, and press the head (Figure 5).
NOTE: If the chest of the mouse bulges, then the intubation is successful (Figure 6). - After successful endotracheal intubation, instill the mice with 3 mg/mL LPS at 3 mg/kg through the cannula using a flat-head microsyringe14,15 (Figure 7).
- Once done, remove the cannula and the microsyringe. Remove the mouse from the scaffold, and place it in a cage separately to recover. Observe the respiratory state of the mouse until it has recovered and regained conciousness.
NOTE: At 12 h after LPS tracheal instillation, euthanize the mice following the procedure approved by the animal ethics committee. Serum TNF-α assays and dry-wet lung weight measurements were performed using standard procedures.
Representative Results
The proposed method for LPS instillation in mice was verified by evaluating the expression of the inflammatory cytokine TNF-α and the lung dry-wet weight ratio 12 h after LPS instillation. There were four groups in the experiment: blank control (without any treatment), surgical intubation16, intranasal17,18, and noninvasive oropharyngeal intubation (n = 6). Compared with the blank control group, the serum TNF-α levels in the noninvasive oropharyngeal intubation group were significantly increased (Figure 8A). The lung dry-wet weight ratio was also increased (Figure 8B), reaching the same level as that in the surgical tracheal intubation group. The datasets were statistically analyzed with an unpaired ANOVA and post-hoc multiple-comparisons Tukey Kramer tests. All data are presented as mean ± SEM, and a level of p < 0.05 was considered statistically significant.

Figure 1: Intubation platform fittings and assembly. The platform consists of a base, a riser, a paper clip, two rubber bands, and some strings. Please click here to view a larger version of this figure.
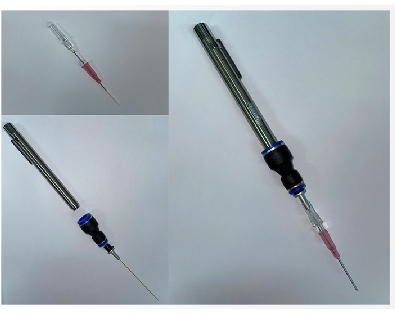
Figure 2: Intubation kit. This figure shows the intubation kit and its assembly. This includes a pen lamp, an optical fiber, and a cannula. Please click here to view a larger version of this figure.
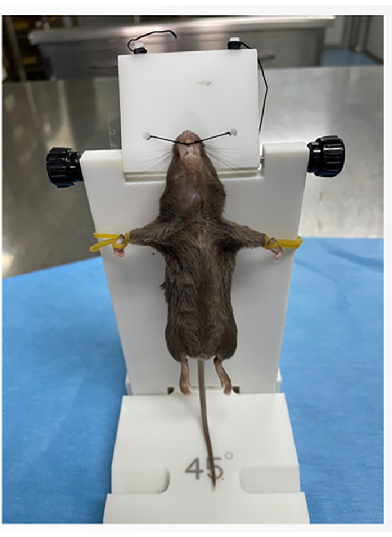
Figure 3: Mouse fixation. Please click here to view a larger version of this figure.
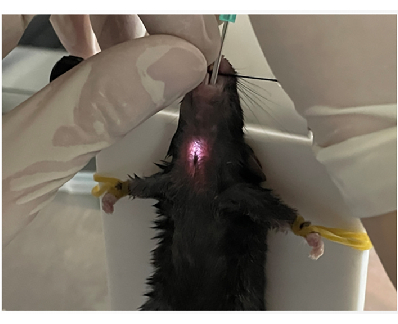
Figure 4: Locating the trachea. Please click here to view a larger version of this figure.
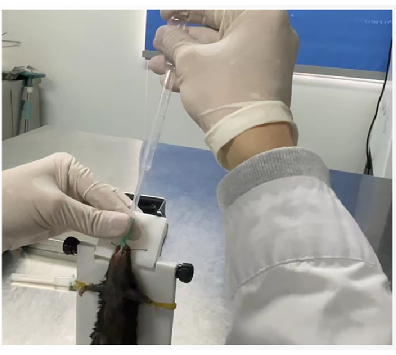
Figure 5: Pasteur pipette pump verification. Please click here to view a larger version of this figure.

Figure 6: Before and after image of the chest showing successful intubation. (A) Chest before intubation. (B) Chest after intubation; the area showing the bulging of the chest is marked with a red circle. Please click here to view a larger version of this figure.

Figure 7: Flat head microsampler for delivering LPS. Please click here to view a larger version of this figure.
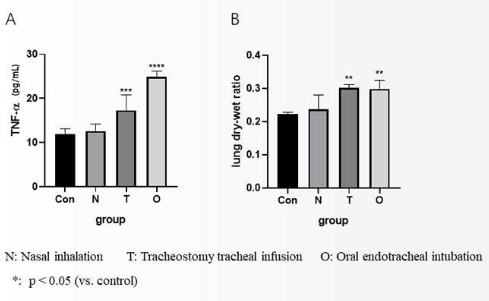
Figure 8: Assessment of the validity of the noninvasive LPS instillation. (A) Expression of TNF-α in the serum of C57BL/6 mice 12 h after an endotracheal injection of LPS. (B) Data analysis of the lung tissue dry-wet weight ratio. Please click here to view a larger version of this figure.
Discussion
Initially, we looked inside the oral cavity to find the location of the trachea19. However, during this process, we discovered that the trachea of C57/BL mice is narrow, which makes it difficult to find the correct location by this method without the help of equipment such as an endoscope20. Upon further exploration, we found that the light from the intubator lamp could penetrate the surface of the body, allowing the operator to determine the position of the cannula21.
To check whether the tube had entered the trachea, initially, we tried using a small mirror, which was cooled by placing it on ice. After intubation, we used a mirror to approach the cannula opening. If mist appeared on the mirror, the intubation was considered successful. However, we found that this examination method could not accurately determine whether the cannula had entered the trachea. First, the cannula head was close to the mouth of the mouse, and it could not be determined whether the mist that appeared on the mirror was caused by exhaled gas from the mouth. Second, the mirror needed to be cooled. Upon constant use, the time required to cool the mirror also led to an increase in the experiment time. We then used a Pasteur pipette to pump air into the trachea; the chest of the mouse would swell if the cannula was inserted into the trachea, and if it was inserted into the esophagus, the right lower abdomen would swell22. Therefore, we used this method as the basis for judging whether the intubation was successful.
Compared with surgical tracheal intubation, noninvasive oropharyngeal intubation avoids surgical wounds and improves the survival rate of the experimental animals23. Compared with intranasal intubation, noninvasive oropharyngeal intubation leads to more accurate entry of the cannula into the bronchus and lungs24. However, mastering these technicalskills requires much practice. In the case of mice with small body sizes, inserting the cannula into the trachea is difficult, and one may easily scratch the trachea during the operation. Therefore, we suggest that mice with larger body sizes should be selected for the experiment.
The method can also be used to deliver other liquid drugs to the bronchus and the lung, thus meaning it has wide application potential25,26.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No.: 81903902), the China Postdoctoral Science Foundation (No.: 2019M663457), the Sichuan Science and Technology Program (No.: 2020YJ0172), and the Xinglin Scholar Research Premotion Project of Chengdu University of TCM (No.: QJRC2022053).
Materials
| Lipopolysaccharide | MERK | L4130 | LPS |
| Microliter Syringes | SHANGHAI GAOGE INDUSTRY AND TRADE CO., LTD | 10028505008124 | To deliver LPS |
| Mouse cannula | RWD Life Science | 803-03008-00 | Mouse cannula |
| Mouse intubation kit | RWD Life Science | 903-03027-00 | Including a base, a riser, a intubator, a surgical forceps and some strings |
| Pasteur pipette | Biosharp life science | BS-XG-03 | To verify the success of intubation |
| Pentobarbital sodium | Beijing Chemical Co., China | 20220918 | To anesthetize mice |
References
- Xia, Y., et al. Protective effect of human amnion epithelial cells through endotracheal instillation against lipopolysaccharide-induced acute lung injury in mice. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 33 (1), 7-11 (2017).
- Butt, Y., Kurdowska, A., Allen, T. C. Acute lung injury: A clinical and molecular review. Archives of Pathology and Lab Medicine. 140 (4), 345-350 (2016).
- Ware, L. B., Matthay, M. A. The acute respiratory distress syndrome. The New England Journal of Medicine. 342 (18), 1334-1349 (2000).
- Fan, E., Brodie, D., Slutsky, A. S. Acute respiratory distress syndrome: Advances in diagnosis and treatment. JAMA. 319 (7), 698-710 (2018).
- Meyer, N. J., Gattinoni, L., Calfee, C. S. Acute respiratory distress syndrome. Lancet. 398 (10300), 622-637 (2021).
- An, N., Yang, T., Zhang, X. X., Xu, M. X. Bergamottin alleviates LPS-induced acute lung injury by inducing SIRT1 and suppressing NF-κB. Innate Immunity. 27 (7-8), 543-552 (2021).
- Liu, L., et al. Comparative study of trans-oral and trans-tracheal intratracheal instillations in a murine model of acute lung injury. The Anatomical Record. 295 (9), 1513-1519 (2012).
- Virag, J. A., Lust, R. M. Coronary artery ligation and intramyocardial injection in a murine model of infarction. Journal of Visualized Experiments. (52), e2581 (2011).
- Li, J., et al. Panaxydol attenuates ferroptosis against LPS-induced acute lung injury in mice by Keap1-Nrf2/HO-1 pathway. Journal of Translational Medicine. 19 (1), 96 (2021).
- Zhou, Y., et al. Soluble epoxide hydrolase inhibitor attenuates lipopolysaccharide-induced acute lung injury and improves survival in mice. Shock. 47 (5), 638-645 (2017).
- Nosaka, N., et al. Optimal tube length of orotracheal intubation for mice. Laboratory Animals. 53 (1), 79-83 (2019).
- Cicero, L., Fazzotta, S., Palumbo, V. D., Cassata, G., Lo Monte, A. I. Anaesthesia protocols in laboratory animals used for scientific purposes. Acta Bio-Medica: Atenei Parmensis. 89 (3), 337-342 (2018).
- Ehrentraut, H., Weisheit, C. K., Frede, S., Hilbert, T. Inducing acute lung injury in mice by direct intratracheal lipopolysaccharide instillation. Journal of Visualized Experiments. (149), e59999 (2019).
- Yang, H., et al. STAT6 inhibits ferroptosis and alleviates acute lung injury by regulating P53/SLC7A11 pathway. Cell Death & Disease. 13 (6), 530 (2022).
- Aramaki, O., et al. Induction of operational tolerance and generation of regulatory cells after intratracheal delivery of alloantigen combined with nondepleting anti-CD4 monoclonal antibody. Transplantation. 76 (9), 1305-1314 (2003).
- Lan, W. Activation of mammalian target of rapamycin (mTOR) in a murine model of lipopolysaccharide (LPS) -induced acute lung injury (ALI). Peking Union Medical College. , (2010).
- Lv, H., et al. Tenuigenin ameliorates acute lung injury by inhibiting NF-κB and MAPK signalling pathways. Respiratory Physiology & Neurobiology. 216, 43-51 (2015).
- Yang, H., Lv, H., Li, H., Ci, X., Peng, L. Oridonin protects LPS-induced acute lung injury by modulating Nrf2-mediated oxidative stress and Nrf2-independent NLRP3 and NF-κB pathways. Cell Communication and Signaling. 17 (1), 62 (2019).
- Im, G. H., et al. Improvement of orthotopic lung cancer mouse model via thoracotomy and orotracheal intubation enabling in vivo imaging studies. Laboratory Animals. 48 (2), 124-131 (2014).
- Hamacher, J., et al. Microscopic wire guide-based orotracheal mouse intubation: description, evaluation and comparison with transillumination. Laboratory Animals. 42 (2), 222-230 (2008).
- Nelson, A. M., Nolan, K. E., Davis, I. C. Repeated orotracheal intubation in mice. Journal of Visualized Experiments. (157), e60844 (2020).
- Zhang, S., et al. Microvesicles packaging IL-1β and TNF-α enhance lung inflammatory response to mechanical ventilation in part by induction of cofilin signaling. International Immunopharmacology. 63, 74-83 (2018).
- Feng, Z. Comparative study of different intratracheal instillation in acute lung injury model of mice. Jilin University. , (2010).
- Chen, J. H., et al. Comparison of acute lung injury mice model established by intranasal and intratracheal instillation of lipopolysaccharide. Pharmacology and Clinics of Chinese Materia Medica. 38 (02), 222-227 (2022).
- Luckow, B., Lehmann, M. H. A simplified method for bronchoalveolar lavage in mice by orotracheal intubation avoiding tracheotomy. BioTechniques. 71 (4), 534-537 (2021).
- Cai, Y., Kimura, S. Noninvasive intratracheal intubation to study the pathology and physiology of mouse lung. Journal of Visualized Experiments. (81), e50601 (2013).

