In Vivo Osteo-organoid Approach for Harvesting Therapeutic Hematopoietic Stem/Progenitor Cells
Summary
Here, we established in vivo osteo-organoids triggered by bone morphogenetic protein-2-loaded gelatin scaffolds to harvest therapeutic hematopoietic stem/progenitor cells for the reconstruction of a damaged hematopoietic and immune system. Overall, this approach can provide a promising cell source for cell therapies.
Abstract
Hematopoietic stem cell transplantation (HSCT) requires a sufficient number of therapeutic hematopoietic stem/progenitor cells (HSPCs). To identify an adequate source of HSPCs, we developed an in vivo osteo-organoid by implanting scaffolds loaded with recombinant human bone morphogenetic protein-2 (rhBMP-2) into an internal muscle pouch near the femur in mice. After 12 weeks of implantation, we retrieved the in vivo osteo-organoids and conducted flow cytometry analysis on HPSCs, revealing a significant presence of HSPC subsets within the in vivo osteo-organoids.
We then established a sublethal model of hematopoietic/immune system injury in mice through radiation and performed hematopoietic stem cell transplantation (HSCT) by injecting the extracted osteo-organoid-derived cells into the peripheral blood of radiated mice. The effect of hematopoietic recovery was evaluated through hematological, peripheral blood chimerism, and solid organ chimerism analyses. The results confirmed that in vivo osteo-organoid-derived cells can rapidly and efficiently reconstruct damaged peripheral and solid immune organs in irradiated mice. This approach holds potential as an alternative source of HSPCs for HSCT, offering benefits to a larger number of patients.
Introduction
Hematopoietic stem cell transplantation stands as the conventional therapy for a variety of hematological malignancies, as well as numerous inherited and autoimmune disorders1,2,3,4. Nevertheless, the restricted quantity and origin of hematopoietic stem/progenitor cells (HSPCs) have emerged as a substantial impediment to the clinical implementation of hematopoietic stem cell transplantation (HSCT)5,6.
Large-scale in vitro cell expansion is a commonly employed method for harvesting therapeutic cells5,7. Various studies have developed conditions that stimulate HSPCs to self-renew in vitro, typically through the use of a combination of self-renewal agonists (such as cytokines and growth factors) and serum albumin, resulting in the ex vivo expansion of HSPCs8. However, it is important to note that current methods are still time-consuming and challenging to maintain the self-renewal capacity of expanded HSPCs9.
In contrast to the aforementioned method, harvesting cells in vivo presents a new and innovative strategy. This approach involves the establishment of an in vivo osteo-organoid that mimics the native bone marrow structure10,11. To achieve this, we form in vivo osteo-organoids using bone morphogenetic protein-2 (BMP-2)-loaded gelatin scaffolds to obtain abundant and high-quality autologous cell cocktails, including HSPCs. By therapeutically applying these osteo-organoids, we have successfully treated irradiation damage and demonstrated that HSPCs derived from the osteo-organoids can rapidly and stably reconstitute the experimentally impaired immune system.
Protocol
Male and female C57BL/6 mice, aged 8-10 weeks, were included in the study. All mice were housed in the animal facility of East China University of Science and Technology. All the experimental procedures were approved by the Institutional Animal Care and Use Committees of East China University of Science and Technology (ECUST-21010).
1. Fabrication of bioactive scaffold
- Preparation
- Put the cleaned and dried surgical scissors and tweezers into a 1,000 mL beaker. Wrap the mouth of the beaker with absorbent paper and tie it tightly with cotton thread.
- Place the beaker in the high-pressure sterilizer, close the lid tightly, set the sterilization program to 121 °C for 30 min, and start the sterilization program.
- After the completion of the sterilization process, remove and transfer the sterilized beaker to a drying oven set at 60 °C for drying. Once completely dried, spray the beaker with 75% (v/v) alcohol and place it on the clean bench.
- Thaw the rhBMP-2 stock solution (1.0 mg/mL) 2 h before the procedure.
- Once thawed to room temperature, transfer the rhBMP-2 stock solution and unopened gelatin sponge to the sterile clean bench for the fabrication of bioactive scaffolds.
- Cut the gelatin sponge (60 mm x 20 mm x 5 mm) into 48 uniformly sized pieces (5 mm x 5 mm x 5 mm) (Figure 1B) with the sterilized scissors and transfer them to a 48-well plate.
- Mix the rhBMP-2 stock solution by pipetting up and down gently to ensure even mixing, taking care to avoid bubbles. Aspirate 30 µL of the solution and add it dropwise to the gelatin sponge.
- Seal the 48-well plate using one layer of parafilm to facilitate the subsequent freeze-drying process. Take the plate containing the gelatin sponge out from the clean bench. Place it at -20 °C to be frozen. Simultaneously, turn on the freeze-dryer and precool it to a temperature range of -50 °C to -60 °C.
- After freezing for 2 h, place the well plate with gelatin sponges in the freeze-dryer for 12 h. To maintain the dryness and sterility of the samples, apply an additional layer of parafilm to seal the well plate after freeze-drying. Store it in a -20 °C refrigerator.
2. Surgical implantation of bioactive scaffolds
NOTE: All surgical instruments are sterilized for use.
- Fast the animals for 8-12 h before anesthesia to reduce the risk of vomiting during induction and emergence, as well as the risk of aspiration of foreign matter. Apply a lubricating vet ointment around the eyes of mice to reduce postoperative discomfort caused by dryness in the eye area. Anesthetize the mice by intraperitoneal injection of 1% (w/v) solution of pentobarbital sodium (at a dose of 48 mg/kg).
NOTE: This protocol uses the ethically approved method of anesthesia with sodium pentobarbital intraperitoneal injection. Other methods, such as isoflurane inhalation or intraperitoneal injection of ketamine-xylazine, can also be used after approval. - Confirm the depth of anesthesia in mice by looking for the following signs in a supine mouse: steady heartbeat and respiration, relaxed muscles, immobile limbs, whiskers unresponsive to touch, and absence of a pedal reflex. In addition, pinch the skin of the mice with toothed forceps or stimulate the toes or paws of mice. If the mice display sensitive reactions, the anesthesia is too light; administer additional anesthesia. After complete anesthesia, transfer the mice to the surgical area in the right lateral recumbent position to implant the bioactive scaffold into the left leg easily.
- Shave the hair of the left leg using a shaving machine, disinfect the shaved area with alcohol wipes, and notch the skin at the location of the muscular thigh pouch approximately 3.0 mm using ophthalmic scissors.
NOTE: To maintain sterile conditions during the surgery, the used surgical items should be stored in designated locations. Sterile supplies should be placed in a clean area to prevent contact with individuals other than the operator. Reusable surgical instruments should be promptly disinfected with 75% (v/v) ethanol. Minimize the duration of surgery to reduce the exposure of wounds to air and decrease the risk of infection. - Insert ophthalmic scissors into the muscle of the pouch in the direction of the tibia to provide space for subsequent material implantation.
- Pinch the bioactive scaffold into a cylindrical shape using ophthalmic forceps. Implant the scaffold into the muscle pocket along the muscle slit created by ophthalmic scissors and close the wound using a 4-0 suture.
NOTE: The occurrence of convulsions or urination in animals during the surgical procedure indicates excessively deep anesthesia, necessitating prompt implementation of emergency measures. - Repeat steps 2.3-2.5 and implant another bioactive scaffold in the right femoral muscle pouch of the mice. Use iodine swabs to clean the surgical and injection sites of the mice after the surgery to prevent infection.
- Place the mice on a constant temperature platform at 37 °C until they awake after the experiment. Turn the mice over every 10-15 min to prevent blood pooling or collapse of lung lobes. After surgery, in addition to maintaining warmth, monitor the postoperative mice without leaving the animal unattended until it has regained sufficient consciousness to maintain sternal recumbency. Pay close attention to important indicators of mice, such as heart rate and respiratory rate.
NOTE: Trembling of the forelimbs in mice signifies the initiation of the recovery phase.2.8. Provide sterile cages and a comfortable, quiet individual housing environment. Do not place the animal that has undergone surgery with other animals until it has fully recovered. In addition, check the animal and the wound regularly during the 72 h post surgery period to identify any potential adverse effects. If infection is detected later, euthanize the animals by first anesthetizing the mice via intraperitoneal injection of 1% (w/v) solution of pentobarbital sodium (at a dose of 48 mg/kg), followed by cervical dislocation after anesthesia.
3. Characterization of in vivo osteo-organoids 12 weeks after implantation
NOTE: The bioactive scaffolds will develop in vivo to form osteo-organoids after implantation.
- Collection of in vivo osteo-organoids
- When the in vivo osteo-organoids have been implanted for 12 weeks, anesthetize the mice by intraperitoneal injection of 1% (w/v) solution of pentobarbital sodium (at a dose of 48 mg/kg), followed by cervical dislocation after anesthesia.
NOTE: To minimize harm to animals, mice should be anesthetized prior to euthanasia by cervical dislocation. - Spray 75% (v/v) alcohol on the skin at the site of dissection.
- Disconnect the hind limb from the trunk and remove the muscle from the osteo-organoids using miniature scissors carefully.
- Remove the attached soft tissues with gauze.
- Fix some osteo-organoids in a 4% (v/v) paraformaldehyde (PFA) solution for 24 h at room temperature for histological analysis. Utilize the remaining samples for macroscopic photography and flow cytometry analysis of HPSCs.
- When the in vivo osteo-organoids have been implanted for 12 weeks, anesthetize the mice by intraperitoneal injection of 1% (w/v) solution of pentobarbital sodium (at a dose of 48 mg/kg), followed by cervical dislocation after anesthesia.
- Macroscopic images
- Place the osteo-organoids on a blue background plate and photograph them with a digital camera.
- Histological analysis
- Preparation for histological staining
- Transfer the osteo-organoids from the PFA solution to ultrapure water and soak for 1 h.
- Replace ultrapure water with 0.5 M ethylenediamine tetraacetic acid (EDTA) solution for decalcification for 1 week.
- Place decalcified tissues in an embedding cassette and submerge the cassette in ultrapure water for 1 h.
- For gradient dehydration, immerse the embedding cassette in ethanol at varying concentrations (v/v): 75%, 80%, 90%, and 95% for a duration of 1 h each, followed by immersion in 100% (v/v) ethanol I and II for 1 h each.
NOTE: "100% (v/v) ethanol I and II" indicates that the embedding cassette should be immersed once in the "container I" filled with 100% (v/v) ethanol and then immersed a second time in the "container II" filled with 100% (v/v) ethanol. - After dehydration, place the embedded tissues sequentially in a mixture of 100% (v/v) xylene and absolute ethanol (xylene: ethanol volume ratio of 1:1) for 1 h and 100% (v/v) xylene I and II for 30 min each.
- Immerse the tissues in paraffin I and II at a constant temperature of 60 °C for 2 h each.
- To embed the tissues soaked in wax using an embedding machine, place the melted wax into the embedding mold. Before the wax solidifies, remove the tissues from the dehydration container and place them in the embedding mold; label them accordingly. Cool the mold at -20 °C freezing condition and after the wax solidifies, remove the wax block from the embedding mold and trim it.
- Slice the embedded wax blocks into 4.5 µm thick sections using a paraffin slicer and then affix the sections to glass slides.
- Briefly bake the sections at 40 °C and then collect and store them in a sample box.
- H&E staining
- Place the sections in a sample rack and bake in a drying oven at 60 °C for 30 min.
- Deparaffinize the sections in xylene and ethanol. Sequentially place the paraffin sections into xylene I for 10 min, xylene II for 10 min, xylene III for 10 min, absolute ethanol I for 5 min, absolute ethanol II for 5 min, 90% (v/v) ethanol for 5 min, 80% (v/v) ethanol for 5 min, 70% (v/v) ethanol for 5 min, 50% (v/v) ethanol for 5 min and transfer into pure water for a gradient dewaxing process.
- Immerse the sections in the hematoxylin staining solution for 2 min, then rinse them in running water for 10 min.
- Immerse the sections in eosin staining solution for 2 min.
- Sequentially place the sections into 75% (v/v) ethanol for 2 min, 85% (v/v) ethanol for 2 min, absolute ethanol for 5 min, absolute ethanol for 5 min, and xylene for 5 min for dehydration.
- Take the sections out of xylene and seal them with neutral balsam.
- Observe the sections under the microscope and capture representative images at a magnification of 10x after the neutral balsam has dried.
- Preparation for histological staining
- Flow cytometry analysis for HPSCs
- Place the in vivo osteo-organoids obtained from step 3.1 in a mortar and add an appropriate amount of staining buffer. Cut the osteo-organoids into fragments with a diameter of 1-3 mm using ophthalmic scissors and crush them in the staining buffer using the mortar and pestle. Vertically press rather than grind the fragments of the in vivo osteo-organoids using a pestle to prevent a decline in cell survival rate.
- Collect the cell suspension into a centrifuge tube and centrifuge them at 300 × g for 5 min at 4 °C.
- Remove the supernatant and add 1 mL of red blood cell lysis buffer to lyse the cells for 3-5 min.
- Wash the cells with 1 mL of staining buffer and filter them through a 300 mesh nylon filter into a 5 mL flow cytometry tube.
- Centrifuge at 300 × g for 5 min at 4 °C and discard the supernatant. Stain the remaining approximately 100 µL (cells) at the bottom of the flow cytometry tube at 4 °C for 30 min using antibodies, including a live/dead staining kit, Peridinin chlorophyll protein-Cyanine5.5 (PerCp-Cy5.5)-anti-lineage cocktail (1:10), Phycoerythrin-Cyanine5 (PE-Cy5)-anti-c-kit (1:200), Alexa Fluor 700(AF700)-anti-Stem cells antigen-1(Sca-1) (1:200), Brilliant Violet 711(BV711)-anti-CD16/32 (1:200), bio-anti-CD34 (1:200), BV421-anti-CD127 (1:200), PE-CF594-anti-CD135 (1:200), BV510-anti-CD48 (1:200), and PE-Cy7-anti-CD150 (1:200).
- Resuspend the cells with 1 mL of Hanks' Balanced Salt Solution (HBSS) without Ca2+ and Mg2+ and centrifuge at 300 × g for 5 min at 4 °C.
- Aspirate the supernatant and stain with PE-streptavidin secondary antibody (1:200) for an additional 60 min at 4 °C.
- Resuspend the cells with 1 mL of HBSS (no Ca2+, Mg2+) and centrifuge at 300 × g for 5 min at 4 °C.
- Aspirate the supernatant and resuspend the cells with 300 µL of HBSS (no Ca2+, Mg2+).
- Vortex the solution to perform further flow cytometry analysis. Further analyze the obtained data using flow cytometry software to obtain quantitative data on HSPCs.
NOTE: The data collection and analysis of flow cytometry refer to our previous articles and supporting materials (Supplemental Figure S1) for the typical gating strategies used to identify HSPCs10. - Establish a side scatter-area (SSC-A) vs forward scatter-area (FSC-A) dot plot and gate the surrounding region of the cell population of interest (P1) in the SSC-A vs FSC-A dot plot during data acquisition on a flow cytometer. Double-click on the P1 gate; select the forward scatter-height (FSC-H) for the X-axis and the forward scatter-width (FSC-W) for the Y-axis to perform the first cell adhesion treatment. Double-click on the single cells gate; select the side scatter-height (SSC-H) for the X-axis and the side scatter-width (SSC-W) for the Y-axis to perform the second cell adhesion treatment. Use the rectangular gating tool to select the single cells.
- Exclude dead cells. Distinguish live cells from dead cells by detecting viability markers. Establish a dot plot of SSC-A vs Live/Dead and gate the region corresponding to live cells, naming it "live cells".
- Screen the cells labeled with specific antibodies in "live cells". Exclude the cell population that may be affected by the positive expression of lineage-specific antibodies in the myeloid hematopoietic stem/progenitor cell lineage by using lineage-negative (lin–) gating. Gate the lin– cells.
- Screen for different subpopulations of cells within the lin– cells. Double-click on the lin– cells gate; select Sca-1-AF700 for the X-axis and c-kit-PE-Cy5 for the Y-axis. Gate the c-kit+Sca-1– cells as the LKS– cells, the c-kit+Sca-1+ cells as the LKS+ cells, and the cells with low expression of c-kit and Sca-1 as the LKloSlo cells. Double-click on the LKS– gate and select CD34-PE for the X-axis and FCγR-BV711 for the Y-axis. Double-click on the LKS+ gate; select FIt3-PE-CF594 for the X-axis and IL7Rα-BV421 for the Y-axis and select CD150-PE-Cy7 for the X-axis and CD48-BV510 for the Y-axis.
- Based on the specificity of antibodies, identify positive or negative gates to distinguish cell subpopulations: hematopoietic stem cells (Lin−c-kit+Sca-1+CD48−CD150+) and differentiated hematopoietic progenitors: multipotent progenitors (Lin−c-kit+Sca-1+CD48−CD150−), common lymphoid progenitors (Lin−c-kit–Sca-1–Flt3+IL7Rα+), common myeloid progenitors (Lin−c-kit+Sca-1−CD34+FCγR−), granulocyte-monocyte progenitors (Lin−c-kit+Sca-1−CD34+FCγR+), and megakaryocyte erythroid progenitors (Lin−c-kit+Sca-1−CD34−FCγR−).
4. Mouse irradiation model
NOTE: C57BL/6 mice were irradiated with X-rays using an X-ray irradiator.
- To achieve sublethal impairment of the hematopoietic/immune system, expose wild-type (WT) mice in the 5 Gray (Gy) phosphate buffered saline (PBS)-treated group, the 5 Gy native bone marrow (BM)-treated group, and the 5 Gy osteo-organoids-treated group to 5 Gy irradiation (1.25 Gy/min) 24 h prior to transplantation.
NOTE: Make sure that WT mice in the 0 Gy PBS-treated group are treated with PBS and do not undergo irradiation.
5. Cell therapy process
NOTE: All procedures should be done in sterile conditions.
- Acquisition of in vivo osteo-organoid-derived- and native bone marrow- derived cells
- Implant two bioactive scaffolds into the muscle pouch of C57BL/6.SJL-Ptprca Pepcb/BoyJ (CD45.1) mice and incubate them for 12 weeks to form the in vivo osteo-organoids. Harvest two in vivo osteo-organoids and two femurs from one CD45.1 mice mentioned above. Subsequently, clear the soft tissue on the surface of the osteo-organoids and femurs using forceps and gauze.
- Separately place the osteo-organoids and femurs into a mortar and add 1 mL of HBSS (no Ca2+, Mg2+).
NOTE: HBSS without Ca2+ and Mg2+ can reduce cell aggregation. The solution used for transplantation must not contain serum components to avoid any potential rejection reactions. - Cut the samples up with surgical scissors and gently crush them in a mortar and pestle.
- Filter the cell suspension through a 40 µm cell strainer and centrifuge at 300 × g for 5 min at 4 °C.
- Remove the supernatant and resuspend the cell pellets with 300 µL of HBSS (no Ca2+, Mg2+). Mix thoroughly to obtain a suspension of osteo-organoid-derived cells or whole bone marrow cells.
NOTE: To fully utilize the cell cocktails derived from the in vivo osteo-organoid, we chose to use whole bone marrow cells for therapy. The typical number of total cells in a 30 µg BMP-2 induced in vivo osteo-organoid is 30,000,000-40,000,000.
- Orbital venous sinus injection
NOTE: CD45.1+ whole bone marrow cells collected from in vivo osteo-organoids or femurs were injected through the orbital venous sinus into the sublethally irradiated C57BL/6 (CD45.2) recipient, respectively. Orbital venous sinus injection and tail vein injection are commonly used methods in animal experiments. Orbital venous sinus injection was chosen due to its simplicity and low failure rate for beginners. During the process of orbital injection, certain protective measures need to be taken, such as pre-anesthetizing the mice, using a heated plate to reduce the risk of hypothermia, and restraining mechanically the mice to prevent movement.- Place the CD45.2 mice in a preanesthesia chamber and induce anesthesia using isoflurane. Apply a lubricating vet ointment around the eyes of mice to prevent dryness and counteract the ocular irritations caused by isoflurane.
NOTE: To minimize harm to the animals, isoflurane anesthesia is administered to mice before orbital venous sinus injection. - Transfer the mice to the anesthesia table for further anesthesia.
- To reduce the likelihood of hypothermia in mice, place the irradiated, anesthetized mouse on a thermostatic heating pad set at 37 °C.
- Place the mouse in left lateral recumbency with its head facing to the right after being fully anesthetized.
- To protrude the eye, place a finger on the top of the head and along the jawline. Gently pull the skin back and down.
- Use a 1 mL syringe fitted with a 25 G needle to aspirate 200 µL of the in vivo osteo-organoid-derived cell suspension or native bone marrow-derived cell suspension of CD45.1+ mice obtained in step 5.1.5.
- Carefully introduce the needle, bevel down, at an angle of approximately 30°, into the medial canthus.
NOTE: Injections are usually given with the needle bevel up, but for retro-orbital injections, it is better to position the needle bevel down to reduce the risk of eye damage. - Use the needle to follow the edge of the eyeball down until the needle tip is at the base of the eye.
- Slowly and smoothly inject the injectate.
- After the injection is complete, place the mouse back into its cage for recovery.
- Place the CD45.2 mice in a preanesthesia chamber and induce anesthesia using isoflurane. Apply a lubricating vet ointment around the eyes of mice to prevent dryness and counteract the ocular irritations caused by isoflurane.
6. Evaluation of treatment effects
- Hematological analysis
NOTE: To analyze the hematology of recipients in immune system reconstitution assays, we collected peripheral blood samples from recipients subjected to different treatments: 0 Gy PBS treatment, 5 Gy PBS treatment, 5 Gy native BM treatment, and 5 Gy osteo-organoid treatment. The blood samples were obtained through retro-orbital puncture 2 days before and 2, 4, 8, 12, and 16 weeks after transplantation. A portion of the blood sample of the same group was used for hematological analysis, while the other part was used for chimerism analysis. There are different blood sampling methods, such as through submandibular vein or the tail vein. Retro-orbital puncture is one of the common methods for blood sampling in mice and is permitted under ethical conditions.- Repeat steps 5.2.1-5.2.5 to anesthetize the mice and protrude the eyes of the mice.
NOTE: To mitigate harm to the animals, isoflurane anesthesia is administered to the mice before retro-orbital puncture blood sampling. - Position the capillary tube with a diameter of 0.5 mm in the medial canthus of the eye and direct it caudally at a 30°-45° angle relative to the plane of the nose.
- Rotate the capillary tube gently while applying pressure, allowing it to penetrate through the conjunctival membranes. Allow the blood to flow into the capillary tube via capillary action.
- Apply continuous pressure after the blood starts flowing to sustain the flow.
- After drawing 100 µL of blood, release the pressure on the neck immediately. Simultaneously, remove the capillary tube to allow the eye to return to the normal position to prevent postoperative bleeding from the puncture site.
- Promptly expel the blood into a centrifuge tube that had been washed with 1.5% (w/v) ethylenediamine tetraacetic acid dipotassium salt dihydrate (EDTA-K2) solution and then had been added 10 µL of 1.5% (w/v) EDTA-K2 solution. Gently shake the centrifuge tube to ensure thorough mixing of EDTA-K2 solution and blood, preventing clotting.
- After wiping the wound with sterile cotton and applying slight pressure to stop bleeding, observe the mice for 1-2 min after hemostasis to ensure there are no abnormalities and then return them to their cages.
- Perform hematological analysis on a portion of each group's blood samples using a hematology analyzer, including the counts of white blood cells (WBC), red blood cells (RBC), and platelets (PLT).
NOTE: Analyze all blood samples within 2 h of collection.
- Repeat steps 5.2.1-5.2.5 to anesthetize the mice and protrude the eyes of the mice.
- Analysis of peripheral blood chimerism
NOTE: Similar to hematological analysis, peripheral blood samples from 5 Gy native BM-treated and 5 Gy osteo-organoid-treated groups were collected by retro-orbital puncture at 2, 4, 8, 12, and 16 weeks after transplantation.- Use the other portion of the peripheral blood sample obtained from step 6.1 for analysis of peripheral blood chimerism.
- Add 1 mL of erythrocyte lysis buffer to the sample and lyse for 3-5 min. Invert the sample upside down gently to facilitate better lysis of red blood cells.
- Centrifuge at 300 × g for 5 min at 4 °C.
- Discard the supernatant and add 1 mL of HBSS (no Ca2+, Mg2+) to resuspend the pellet.
- Centrifuge at 300 × g for 5 min at 4 °C and discard the supernatant. Stain the remaining approximately 100 µL of cells at the bottom of the centrifuge tube at 4 °C for 30 min using antibodies, including a live/dead staining kit, PE-anti-CD45.1 (1:200), Fluorescein Isothiocyanate (FITC)-anti-CD45.2 (1:200), PE-Cy7-anti-B220 (1:200), AF700-anti-CD11b (1:200), PerCp-Cy5.5-anti-CD8a (1:200), PE-Dazzle594-anti-CD4 (1:200), and Allophycocyanin (APC)-anti-CD3e (1:200).
- Resuspend the cells with 1 mL of HBSS (no Ca2+, Mg2+) and centrifuge at 300 × g for 5 min at 4 °C.
- Aspirate the supernatant and resuspend the cells with 300 µL of HBSS (no Ca2+, Mg2+).
- Vortex the suspension to perform further flow cytometry analysis.
- Further analyze the obtained data using flow cytometry software to obtain information about the chimerism rate of HSPCs in the peripheral blood of recipient mice in both the 5 Gy native BM-treated group and the 5 Gy osteo-organoid-treated group.
NOTE: For the analysis of flow cytometric data, refer to Supplemental Figure S2 for the typical gating strategies used to identify donor-derived CD45.1+ cells in peripheral blood of CD45.2 recipients. - Repeat steps 3.4.11-3.4.12 to gate the region corresponding to live cells.
- Analyze peripheral blood chimerism by initially identifying cell populations and subsequently discerning donor and recipient cells.
- Screen specific antibody-labeled cells within live cells. Select CD11b-AF700 for the X-axis and B220-PE-Cy7 for the Y-axis. Gate the B220+CD11b– cells as B cells, the B220–CD11b+ cells as myeloid cells, and the B220–CD11b– cells as double-negative (DN) cells.
- Double-click on the DN cells gate, using CD3-APC for the X-axis and SSC-A for the Y-axis. Gate the CD3+ cells as T cells.
- Double-click on the CD3+ cells gate, selecting CD4-PE-Dazzle594 for the X-axis and CD8-PerCp-Cy5.5 for the Y-axis. Gate the CD8+CD4– cells as cytotoxic T cells (CTL) and the CD8–CD4+ cells as regulatory T cells (Treg).
- Finally, peripheral blood chimerism analysis is performed on the donor and recipient cells. Within the designated cell populations, double-click to select CD45.1-PE for the X-axis and CD45.2-FITC for the Y-axis. Gate the CD45.1+ cells and perform cell counting to determine the proportion of CD45.1+ cells within the cell population, thus reflecting the chimerism rate.
- Analysis of solid organ chimerism
NOTE: At 16 weeks post transplantation, BM, spleen, and thymus chimeras were analyzed in the 5 Gy BM-treated group and the 5 Gy osteo-organoid-treated group.- Repeat steps 5.1.1-5.1.4 to obtain the cell pellets derived from the BM.
- Place a 300 mesh nylon filter above a cell culture dish (6 cm) containing 1 mL of HBSS (no Ca2+, Mg2+).
- Place the dissected spleen or thymus on the nylon filter.
- Grind the mentioned organs using the front rubber stopper of a 5 mL syringe until no obvious tissue structure remains, only residual fibrous tissue. Handle the cells gently to ensure cell viability.
- Collect the filtrate and centrifuge at 300 × g for 5 min at 4 °C.
- Remove the supernatant to obtain the cell precipitates derived from the spleen or thymus.
- Add 1 mL of red blood cell lysis buffer to the cell pellets obtained in steps 6.3.1 and 6.3.6, and lyse for 3-5 min.
- Repeat steps 6.2.3-6.2.8 for staining and flow cytometry analysis.
- Further analyze the obtained data using flow cytometry software to obtain information about the chimerism rate of HSPCs in the solid organs (BM, spleen, and thymus) of recipient mice in both the 5 Gy native BM-treated group and the 5 Gy osteo-organoid-treated group.
NOTE: For the analysis of flow cytometric data, refers to Supplemental Figure S2 for the typical gating strategies used to identify donor derived CD45.1+ cells in BM, spleen, and thymus of CD45.2 recipients. - Replace peripheral blood in steps 6.2.10-6.2.12 with BM, spleen, and thymus to obtain the chimerism rate of solid organs.
Representative Results
As per the protocol, we have created a bioactive scaffold by dripping BMP-2 into a degradable gelatin sponge under sterile conditions. The scaffold was then implanted into the lower limb muscles of mice to establish in vivo osteo-organoids. After an incubation period of 12 weeks, we conducted macroscopic photography, histological analysis, and flow cytometry analysis on the osteo-organoids (Figure 1A). The gelatin sponge was cut into cubes with dimensions of 5 mm x 5 mm x 5 mm, and after absorbing 30 µL of BMP-2 stock solution (1.0 mg/mL), a bioactive scaffold was obtained. The bioactive scaffold retained its original shape even after freeze-drying (Figure 1B). Scanning electron microscopy (SEM) images depict a porous structure within the bioactive scaffold that promotes cell adhesion and proliferation (Figure 1C). The interconnections among various pores could enhance blood vessel sprouting and invasion, facilitating nutrient transport to support cell growth and aiding in osteo-organoid formation. After 12 weeks of induction in vivo, the implanted osteo-organoids exhibited a deep red color (Figure 1D). H&E-stained image showed that the bioactive scaffold induced the formation of a natural bone marrow-like structure composed of osteocytes and bone marrow cells, further confirming the formation of in vivo osteo-organoids (Figure 1E).
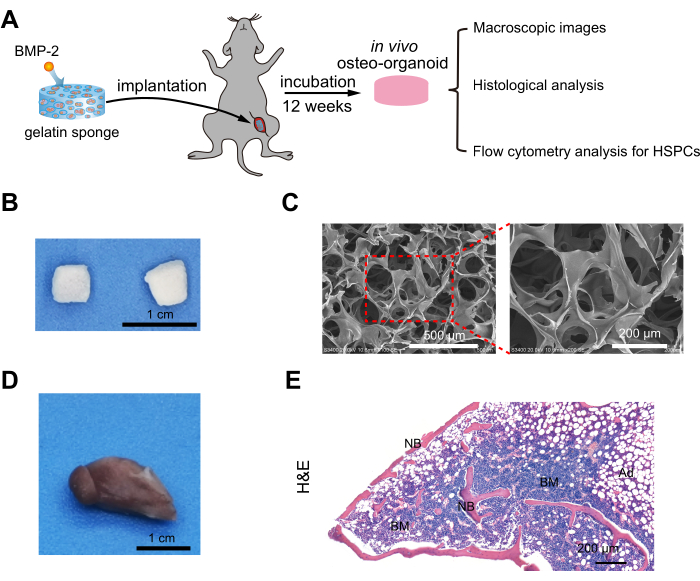
Figure 1: Formation and characterization of in vivo osteo-organoids. (A) Schematic diagram for harvesting and evaluating in vivo osteo-organoids induced by bioactive scaffolds. Freeze-dried gelatin scaffolds loaded with BMP-2 were surgically implanted into the lower limb muscles of mice. The in vivo osteo-organoids were explanted after 12 weeks of incubation for subsequent analysis. (B) Macroscopic images and (C) scanning electron microscope images of the bioactive scaffold. Figure 1A–C are reproduced from Dai et al.11. (D) Macroscopic image and (E) H&E-stained sections of the osteo-organoid 12 weeks after implantation. Figure 1D,E are reproduced from Dai et al.10. n = 4 biological replicates. Bone marrow and new bone are shown. Scale bars = 200 µm (C, right; E), 500 µm (C, left), 1 cm (B,D). Abbreviations: BMP-2 = bone morphogenetic protein-2; H&E = hematoxylin and eosin; BM = bone marrow; NB = new bone. Please click here to view a larger version of this figure.
To assess the long-term therapeutic cell-generating potential of osteo-organoids, we analyzed single-cell suspensions from the osteo-organoids using flow cytometry for quantitative analysis of HSPCs (Supplemental Figure S1). We analyzed the types of HSPC subsets 12 weeks after implantation (Figure 2A). Hematopoietic stem cells (HSCs; Lin−c-kit+Sca-1+CD48−CD150+) and their differentiated hematopoietic progenitor cells (HPCs), including multipotent progenitors (MPPs; Lin−c-kit+Sca-1+CD48−CD150−), common lymphoid progenitors (CLPs; Lin−c-kit–Sca-1–Flt3+IL7Rα+), common myeloid progenitors (CMPs; Lin−c-kit+Sca-1−CD34+FCγR−), granulocyte-monocyte progenitors (GMPs; Lin−c-kit+Sca-1−CD34+FCγR+), and megakaryocyte erythroid progenitors (MEPs; Lin−c-kit+Sca-1−CD34−FCγR−) were all present in the osteo-organoids. We then conducted further quantitative analysis of the cell numbers in different cell populations within the osteo-organoids (Figure 2B). After 12 weeks of implantation, the numbers of HSCs, MMPs, CLPs, CMPs, GMPs, and MEPs in the osteo-organoids were approximately 530, 860, 2750, 80,000, 43,000, and 55,000, respectively. In conclusion, these results supported that osteo-organoids contained an abundant supply of HSPCs for cell therapy.
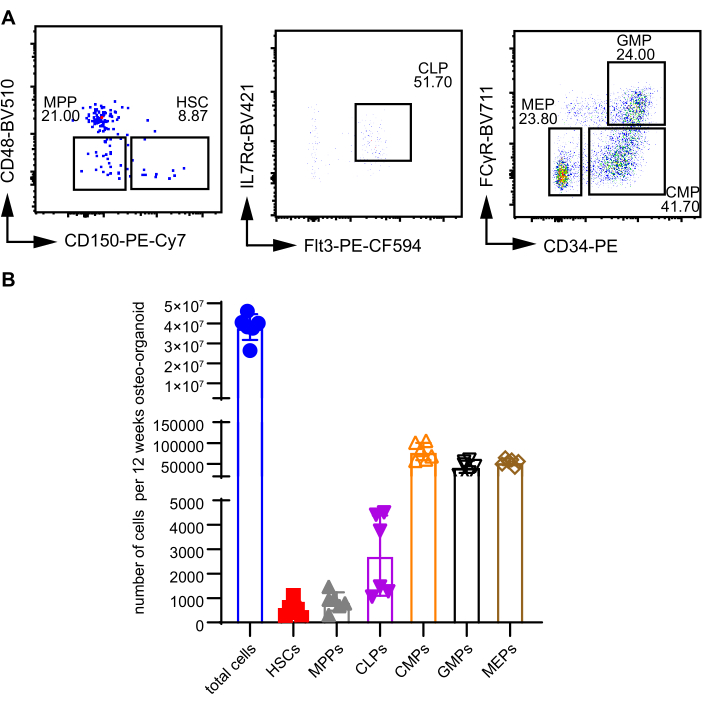
Figure 2: In vivo osteo-organoids contain abundant HSPCs. (A) Representative flow cytometric plots of HSPCs in the osteo-organoids 12 weeks after implantation. (B) Absolute numbers of total cells, Hematopoietic stem cells (Lin−c-kit+Sca-1+CD48−CD150+) and differentiated hematopoietic progenitors: multipotent progenitors (Lin−c-kit+Sca-1+CD48−CD150−), common lymphoid progenitors (Lin−c-kit–Sca-1–Flt3+IL7Rα+), common myeloid progenitors (Lin−c-kit+Sca-1−CD34+FCγR−), granulocyte-monocyte progenitors (Lin−c-kit+Sca-1−CD34+FCγR+), and megakaryocyte erythroid progenitors (Lin−c-kit+Sca-1−CD34−FCγR−). Figure 2A,B was modified from Dai et al.10. n = 6 biological replicates. Data are presented as means ± SD. Statistical differences among groups are identified by one-way ANOVA, followed by Tukey's multiple comparison tests. *P < 0.05, **P < 0.01, and ***P < 0.001. Abbreviations: HSPCs = hematopoietic stem/progenitor cells; HSCs = Hematopoietic stem cells; MPPs = multipotent progenitors; CLPs = common lymphoid progenitors; CMPs = common myeloid progenitors; GMPs = granulocyte-monocyte progenitors; MEPs = megakaryocyte erythroid progenitors; PE = phycoerythrin. Please click here to view a larger version of this figure.
HSCT has proven to be an effective cell therapy for treating radiation-induced immune system injury12,13,14. We transplanted cells derived from in vivo osteo-organoids (incubated for 12 weeks) into sublethally irradiated mice and collected samples from different parts of the mice at indicated time points for subsequent analysis (Figure 3A). Hematological analysis showed that, compared to the native BM-treated group, the WBC count in the 5 Gy osteo-organoid-treated group significantly increased at the 4th week post transplantation, suggesting an enhanced WBC recovery capability of the osteo-organoid-derived cells (Figure 3B). In the second week after transplantation, the RBC count of both the 5 Gy osteo-organoid-treated group and the 5 Gy native BM-treated group returned to normal levels (Figure 3C). At 2 weeks post transplantation, the PLT count was significantly increased in the 5 Gy osteo-organoid-treated group and the 5 Gy native BM-treated group compared to the 5 Gy PBS-treated group (Figure 3D). In conclusion, these results confirmed that cells derived from osteo-organoids exerted a more robust capacity to enhance hematopoietic recovery compared to native BM cells.
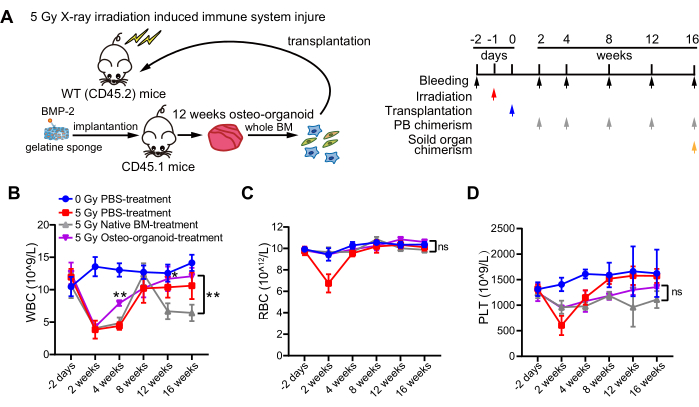
Figure 3: Cell therapy from osteo-organoids for the treatment of sublethally irradiated mice. (A) Experimental scheme of HSCT. The osteo-organoids were generated by implanting bioactive scaffolds into the femur muscle pocket of mice (donors, CD45.1) for 12 weeks. The osteo-organoids were collected and digested to obtain a whole bone marrow cell suspension, which was then transplanted to WT mice (recipients, CD45.2) with immune system injury induced by 5 GY X-ray. peripheral blood was collected at 2, 4, 8, 12, and 16 weeks after transplantation for subsequent analysis of peripheral blood chimerism. Solid organ was collected at 16 weeks after transplantation for subsequent analysis of solid organ chimerism. (B–D) peripheral blood cells from the four groups were analyzed at indicated time points. WT mice in the 0 Gy PBS-treated group did not receive irradiation and received PBS. WT mice in the 5 Gy PBS-treated, 5 Gy native BM-treated, and 5 Gy osteo-organoid-treated groups received 5 Gy irradiation 1 day prior to transplantation and were transplanted with PBS, native BM cells, or osteo-organoid-derived cells, respectively. n = 4 to 5 biological replicates. Figure 3A–D was modified from Dai et al.10. Data are presented as means ± SD. (B–D) Significant differences among groups are identified by two-way ANOVA followed by Bonferroni's post hoc test. **P < 0.01. Abbreviations: HSCT = Hematopoietic stem cell transplantation; BMP-2 = bone morphogenetic protein-2; WT = wild type; PB = peripheral blood; PBS = phosphate-buffered saline; BM = bone marrow; WBC = white blood cell; RBC = red blood cell; PLT = platelet. Please click here to view a larger version of this figure.
To assess the contribution of transplanted cells to the peripheral immune system reconstitution in irradiated mice, we performed quantitative analysis of CD3+ T cells, B220+ B cells, and CD11b+ myeloid cells using flow cytometry. This allowed us to evaluate the chimerism of osteo-organoid-derived and native BM-derived cells in the peripheral blood of the recipients. (Figure 4A and Supplemental Figure S2). We collected peripheral blood samples at 2, 4, 8, 12, and 16 weeks after cell therapy in different groups (Figure 3A). We observed sustained high levels of chimerism in the peripheral blood, with donor-derived CD3+ T cells, B220+ B cells, and CD11b+ myeloid cells consistently exceeding 80% in both the 5 Gy osteo-organoid treated groups and the 5 Gy native BM treated groups. (Figure 4B and Figure 4E,F). Consistent with hematological analysis, the osteo-organoid-derived cells exhibited enhanced regenerative capacity in the T cell subset chimerism in comparison to the native BM-derived cells (Figure 4C,D). The CD3+ T cell subset from the donor source achieved stable chimerism at 8 weeks post transplantation, while the B220+ B cell and CD11b+ myeloid cell subsets from the donor source achieved stable chimerism as early as 2 weeks after transplantation (Figure 4B–F). These data substantiated the role of osteo-organoid-derived cells from the donor in accelerating the recovery of T cell subsets in peripheral blood compared to the native BM-derived cells from the donor.
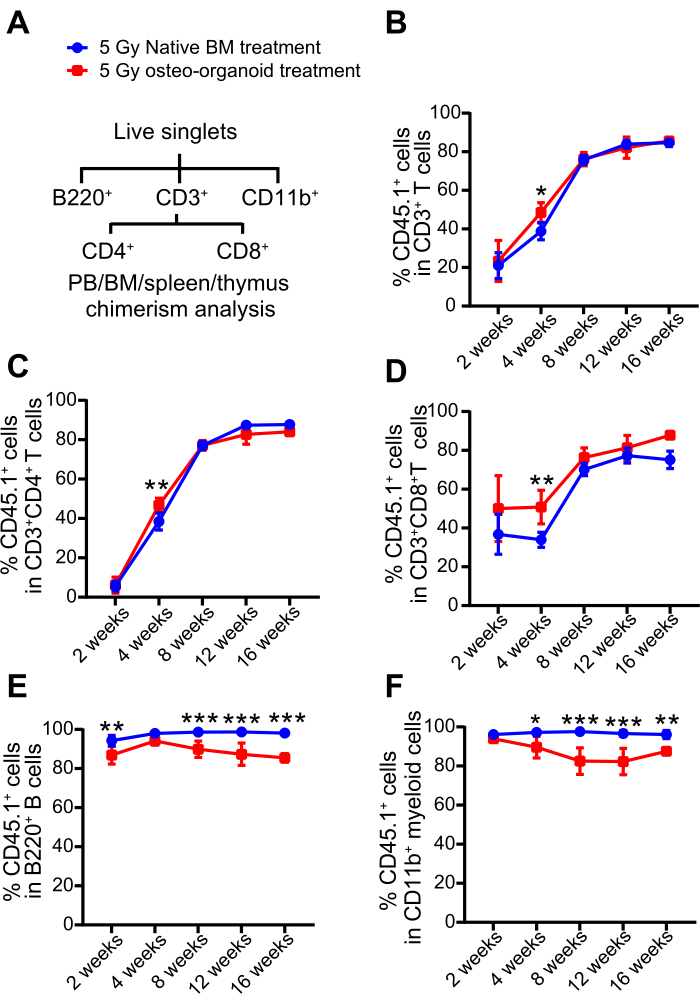
Figure 4: Reconstitution of the peripheral immune system in sublethally irradiated mice by osteo-organoid-derived cells. (A) The flow cytometric gating strategy for the chimerism analysis of recipient mice peripheral blood, BM, spleen, and thymus. (B–F) peripheral blood chimerism from the 5 Gy native BM-treated and 5 Gy osteo-organoid-treated groups was analyzed by the proportion of T cells, B cells, and myeloid cells at the indicated time points. Figure 4A–F was reproduced from Dai et al.10. n = 4 to 5 biological replicates. Data are presented as means ± SD. (B–F) Significant differences among groups were identified by two-way ANOVA followed by Bonferroni's post hoc test. *P < 0.05, **P < 0.01, and ***P < 0.001. Abbreviations: PB = peripheral blood; BM = bone marrow. Please click here to view a larger version of this figure.
To assess the regenerative capacity of transplanted cells in the irradiated mice's solid immune organs, we utilized the same flow cytometry gating strategy to analyze the chimerism of both native BM-derived and osteo-organoid-derived cells in the recipients' BM, spleen, and thymus (Figure 4A). At 16 weeks after cell therapy, the chimerism of T cells derived from donor osteo-organoid cells in recipients' BM was found to be increased, while the chimerism of B cells and myeloid cells was observed to be reduced, as compared to the chimerism of native BM cells from the donor (Figure 5A). Donor cells from both the 5 Gy osteo-organoid-treated group and the 5 Gy native BM-treated group exhibited pronounced chimerism in the spleen and thymus of the recipients, characterized by high levels of T cells, B cells, and myeloid cells (typically exceeding 80%) (Figure 5B,C). These findings provided evidence that osteo-organoid-derived and native BM-derived cells efficiently reconstituted the damaged immune system in mice exposed to irradiation.
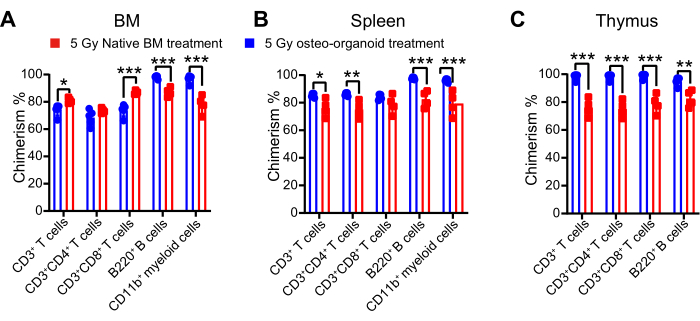
Figure 5: Reconstitution of the solid immune organ in sublethally irradiated mice by osteo-organoid-derived cells. (A–C) BM, spleen, and thymus chimerism from the 5 Gy native BM-treated and 5 Gy osteo-organoid-treated groups were analyzed by the chimerism rates of T cells, B cells, and myeloid cells at 16 weeks after transplantation. Figure 5A–C were reproduced from Dai et al.10. n = 4 to 5 biological replicates. Data are presented as means ± SD. Statistical differences among groups are identified by two-way ANOVA followed by Bonferroni's post hoc test. *P < 0.05, **P < 0.01, and ***P < 0.001. Abbreviation: BM = bone marrow. Please click here to view a larger version of this figure.
Supplemental Figure S1: Gating strategy used to identify HSPCs in the osteo-organoid. Single-cell suspensions from the osteo-organoid were subjected to flow cytometry for quantitative analysis of HSPCs. This figure was reproduced from Dai et al.10. Please click here to download this File.
Supplemental Figure S2: Gating strategy used to identify donor derived CD45.1+ cells in PB, BM, spleen, and thymus of CD45.2 recipients. Single-cell suspensions from PB, BM, spleen, and thymus of the CD45.2 recipients were analyzed via flow cytometry for the quantitative analysis of donor chimera. This figure was reproduced from Dai et al.10. Abbreviations: PB = peripheral blood; BM = bone marrow. Please click here to download this File.
Discussion
In this protocol, we present an approach to establish in vivo osteo-organoids with bone marrow-like structures by implanting gelatin sponge scaffolds loaded with BMP-2. We demonstrate that these in vivo osteo-organoids can stably produce therapeutic HSPCs over a long period of time (more than 12 weeks). Compared to existing in vitro expansion or in vivo incubation methods that load cells, this protocol can obtain cell cocktails with diverse cell types, including HSPCs and various immune cells, within 3 weeks10. Animal experiments have also shown that transplantation of in vivo osteo-organoid-derived cells can accelerate hematopoietic recovery in irradiated mice. Given that gelatin sponges loaded with BMP-2 can retain their osteogenic activity for over 12 months after lyophilization when stored at -20 °C and that the material does not necessitate the incorporation of any cells, this variety of gelatin sponge infused with BMP-2 has the potential to be fashioned into an off-the-shelf product for immediate utilization.
There are two pivotal stages in the protocol that require special attention. First, during the material preparation process, it is crucial to maintain sterility to avoid bacterial contamination, which could result in bone formation failure and the inability to attain in vivo osteo-organoids. Second, after lyophilization, it is imperative to store the material at a low temperature to preserve the activity of BMP-2 protein. Additionally, the process of obtaining single-cell suspensions for transplantation must be carried out in a gentle and prompt manner to ensure cell viability. If the cell suspension is placed on ice, we recommend completing the transplantation within 4 h.
Although various biocompatible materials, such as hydroxyapatite, decalcified bone matrix, and matrix gel, have been reported in the literature as scaffold materials for loading BMP-2 to establish in vivo organoids15, we chose gelatin sponges as the scaffold material due to their availability and clinical translational needs. In addition, we chose to lyophilize the gelatin sponge after loading it with BMP-2 solution. This approach can increase the convenience of use and control the release ability of BMP-2 to a certain extent, reducing the dosage of BMP-2 used. From this perspective, this will significantly reduce the side effects of current clinical products that mainly consist of BMP-2 solution. In the cell transplantation process, in vivo organoid-derived cells can be transplanted after simple filtration without lysis of red blood cells, further simplifying the experimental process. In addition to routine peripheral blood chimerism analysis, we also systematically evaluated the engraftment levels of in vivo osteo-organoid derived cells in multiple solid organs of recipient mice, including bone marrow, spleen, and thymus. This approach will provide a more comprehensive evaluation of the therapeutic potential of in vivo osteo-organoid-derived cells in terms of hematopoietic function.
Although in vivo organoid derived cells can accelerate hematopoietic recovery in mice with bone marrow injuries, the number of HSPCs obtained is still lower than that obtained from single femurs. How to modify the material system to obtain a higher quantity and stronger functional HSPCs is an area that needs further exploration. We recently found that sulfated polysaccharides (SCS), especially sulfated chitooligosaccharides (SCOS), can regulate the function of HSPCs in in vivo osteo-organoids11. This result also suggests that the complexation of BMP-2 with more functional molecules should be a key research direction in the future.
Animal experiments have shown that functional HSPCs can be obtained through the in vivo osteo-organoid approach. However, in large animals such as miniature pigs or macaques, it is still unclear how high doses of BMP-2 can induce functional HSPCs, which is an important step for clinical translation and requires further research.
In summary, the in vivo osteo-organoid approach proposed in this protocol can efficiently and stably produce therapeutic HSPCs in vivo and provide a stable cell source for hematopoietic stem cell transplantation. It is worth exploring more HSPC enhancement factors to benefit more patients. In the following clinical application, we are investigating the use of immediate family members of patients as biogenerators to establish the in vivo osteo-organoid for therapeutic cell provision. This approach has the potential to minimize the risk of immunological rejection and eliminate potential ethical issues.
Divulgations
The authors have nothing to disclose.
Acknowledgements
This research was supported by the Basic Science Center Program (No. T2288102), the Key Program of the National Natural Science Foundation of China (No. 32230059), the National Natural Science Foundation of China (No. 32301123), the Foundation of Frontiers Science Center for Materiobiology and Dynamic Chemistry (No. JKVD1211002), the Wego Project of Chinese Academy of Sciences (No. (2020) 005), the Project of National Facility for Translational Medicine (Shanghai) (No. TMSK-2021-134), and the China Postdoctoral Science Foundation (No. 2022M721147).
Materials
| AF700-anti-CD11b (M1/70) | eBioscience | 56-0112-82 | Store at 4 °C. Dilute 1:200 for staining. |
| AF700-anti-Sca-1 (D7) | BioLegend | 108141 | Store at 4 °C. Dilute 1:200 for staining. |
| APC-anti-CD3e (145-2C11) | Tonbo | 20-0031-U100 | Store at 4 °C. Dilute 1:200 for staining. |
| bio-anti-CD34 (RAM34) | eBioscience | 13-0341-82 | Store at 4 °C. Dilute 1:200 for staining. |
| BV421-anti-CD127 (IL-7Rα) | BioLegend | 135023 | Store at 4 °C. Dilute 1:200 for staining. |
| BV510-anti-CD48 (HM48-1) | BioLegend | 103443 | Store at 4 °C. Dilute 1:200 for staining. |
| BV711-anti-CD16/32 (93) | BioLegend | 101337 | Store at 4 °C. Dilute 1:200 for staining. |
| Capillary tube | Shanghai Huake Labware Co. | DC616297403604-100mm/0.5mm | |
| Cell strainer | CORNING | 352340 | |
| Ethanol | GENERAL-REAGENT | 01158566 | |
| Ethylenediamine tetraacetic acid (EDTA) solution | Servicebio | G1105 | |
| Ethylenediaminetetraacetic acid dipotassium salt dihydrate (EDTA-K2) | Solarbio | E8651 | |
| FITC-anti-CD45.2 (104) | BioLegend | 109806 | Store at 4 °C. Dilute 1:200 for staining. |
| Flowjo | Becton, Dickinson & Company | A flow cytometry. | |
| Gelatin sponge | Jiangxi Xiangen Co. | Use under sterile conditions. | |
| HBSS without Ca2+ and Mg2+ | Gibco | 14170112 | HBSS without Ca2+ and Mg2+ can prevent cell aggregation. |
| Hematology analyzer | Sysmex | pocH-100i Diff | |
| iodine swabs | Xiangtan Mulan Biological Technology Co., Ltd. | 01011 | To prevent the infection after operation. |
| Isoflurane | RWD | R510-22-10 | To avoid adverse effects of anesthesia waste gases on the environment and laboratory personnel, a gas recovery system should be used in conjunction. |
| Kraft paper | absorbent paper | ||
| LIVE/DEAD Fixable Near IR Dead Cell Staining Kit(used in 3.4.5) | Thermo Fisher Scientific | L34962 | A live/dead staining kit. Store at -20 °C. Dissolve in 50 μL of DMSO for working solution. |
| lubricating vet ointment | Pfizer | To prevent dryness and counteract the ocular irritations caused by isoflurane. | |
| Neutral balsam | Solarbio | G8590 | |
| nylon filter | Shanghai Shangshai Wire Mesh Manufacturing Co., Ltd. | Used for cell filtration. | |
| Paraffin liquid | Macklin | P815706 | |
| Paraformaldehyde (PFA) solution | Servicebio | G1101 | Immersion fixation is used for routine animal tissues. The volume of fixative used is generally 10-20 times the tissue volume, and fixation at room temperature for 24 hours is sufficient. |
| PE-anti-CD45.1 (A20) | BioLegend | 110708 | Store at 4 °C. Dilute 1:200 for staining. |
| PE-CF594-anti-CD135 (A2F10.1) | BD Biosciences | 562537 | Store at 4 °C. Dilute 1:200 for staining. |
| PE-Cy5-anti-c-kit (2B8) | BD Biosciences | 105809 | Store at 4 °C. Dilute 1:200 for staining. |
| PE-Cy7-anti-B220 (RA3-6B2) | BioLegend | 103222 | Store at 4 °C. Dilute 1:200 for staining. |
| PE-Cy7-anti-CD150 (TC15-12F12.2) | BioLegend | 115914 | Store at 4 °C. Dilute 1:200 for staining. |
| PE-Dazzle594-anti-CD4 (GK1.5) | BioLegend | 100456 | Store at 4 °C. Dilute 1:200 for staining. |
| Pentobarbital sodium salt | Sigma-Aldrich | 57-33-0 | Prepare for use at a concentration of 1% (w/v). |
| PerCp-Cy5.5-anti-CD8a (53-6.7) | BioLegend | 100734 | Store at 4 °C. Dilute 1:200 for staining. |
| PerCp-Cy5.5-anti-lineage cocktail | BD Biosciences | 561317 | Store at 4 °C. Dilute 1:10 for staining. |
| Red blood cell lysis buffer | Beyotime | C3702 | Store at 4 °C. Use in clean bench. |
| rhBMP-2 | Shanghai Rebone Biomaterials Co. | The concentration of rhBMP-2 in the stock solution is 1.0 mg/mL. | |
| Staining buffer | BioLegend | 420201 | Store at 4 °C. |
| Xylene | GENERAL-REAGENT | 01018114 | |
| Zombie UV Fixable Viability Kit (used in 6.2.5) | BioLegend | 423108 | A live/dead staining kit. For reconstitution, bring the kit to room temperature; add 100 µL of DMSO to one vial of Zombie UV dye until fully dissolved. |
References
- Copelan, E. A. Hematopoietic stem-cell transplantation. New England Journal of Medicine. 354 (17), 1813-1826 (2006).
- Vittayawacharin, P., et al. Autologous hematopoietic stem cell transplantation for a patient with multiple autoimmune diseases. American Journal of Hematology. 98 (10), 1659-1662 (2023).
- Xu, L. -. P., et al. Hematopoietic stem cell transplantation activity in China 2019: a report from the Chinese Blood and Marrow Transplantation Registry Group. Bone Marrow Transplantation. 56 (12), 2940-2947 (2021).
- Gratwohl, A., et al. Hematopoietic stem cell transplantation: a global perspective. JAMA. 303 (16), 1617-1624 (2010).
- Sakurai, M., et al. Chemically defined cytokine-free expansion of human haematopoietic stem cells. Nature. 615 (7950), 127-133 (2023).
- Pineault, N., Abu-Khader, A. Advances in umbilical cord blood stem cell expansion and clinical translation. Experimental Hematology. 43 (7), 498-513 (2015).
- Liang, G., Liu, F. Long-term expansion of human hematopoietic stem cells. Cell Regeneration. 12 (1), 18 (2023).
- Wilkinson, A. C., Igarashi, K. J., Nakauchi, H. Haematopoietic stem cell self-renewal in vivo and ex vivo. Nature Reviews Genetics. 21 (9), 541-554 (2020).
- Kumar, S., Geiger, H. HSC niche biology and HSC expansion ex vivo. Trends in Molecular Medicine. 23 (9), 799-819 (2017).
- Dai, K., et al. A BMP-2-triggered in vivo osteo-organoid for cell therapy. Science Advances. 9 (1), 1541 (2023).
- Dai, K., Zhang, W., Deng, S., Wang, J., Liu, C. Sulfated polysaccharide regulates the homing of HSPCs in a BMP-2-triggered in vivo osteo-organoid. Advanced Science. 10 (24), e2301592 (2023).
- Alexander, T., Greco, R. Hematopoietic stem cell transplantation and cellular therapies for autoimmune diseases: overview and future considerations from the Autoimmune Diseases Working Party (ADWP) of the European Society for Blood and Marrow Transplantation (EBMT). Bone Marrow Transplantation. 57 (7), 1055-1062 (2022).
- Ding, L., et al. Infusion of haploidentical HSCs combined with allogenic MSCs for the treatment of ALL patients. Bone Marrow Transplantation. 57 (7), 1086-1094 (2022).
- Sharrack, B., et al. Autologous haematopoietic stem cell transplantation and other cellular therapy in multiple sclerosis and immune-mediated neurological diseases: updated guidelines and recommendations from the EBMT Autoimmune Diseases Working Party (ADWP) and the Joint Accreditation Committee of EBMT and ISCT (JACIE). Bone Marrow Transplantation. 55 (2), 283-306 (2020).
- Nelson, M. R., Roy, K. Bone-marrow mimicking biomaterial niches for studying hematopoietic stem and progenitor cells. J Mater Chem B. 4 (20), 3490-3503 (2016).

.
