Production of Cardiac Extracellular Matrix from Adult Human Fibroblasts for Culture Dish Coating
Summary
Fibroblasts isolated from the adult human heart were cultured to confluence on gelatin-coated dishes to produce the myocardium-specific extracellular matrix. After decellularization, this substrate can be used for the culture and study of other cardiac cells and cell-matrix interactions.
Abstract
The myocardium is composed of cardiomyocytes and an even greater number of fibroblasts, the latter being responsible for extracellular matrix production. From the early stages of heart development throughout the lifetime, in both normal and pathological conditions, the composition of the extracellular matrix changes and influences myocardium structure and function. The purpose of the method described here is to obtain the substrate for the culture of cardiac cells in vitro (termed cardiac ECM), mimicking the myocardial extracellular matrix in vivo. To this end, fibroblasts isolated from the adult human heart were cultured to confluence on gelatin-coated dishes to produce the myocardium-specific extracellular matrix. The subsequent removal of cardiac fibroblasts, while preserving the deposited cardiac ECM, produced the substrate for studying the influence of the myocardium-specific extracellular matrix on other cells. Importantly, the composition of the fibroblast-derived coating of the culture dish changes according to the in vivo activity of the fibroblasts isolated from the heart, allowing subsequent studies of cell-matrix interactions in different normal and pathological conditions.
Introduction
All cells are located in vivo in a specialized microenvironment in which they can survive and carry out their specific functions. Within any given tissue, the cells are surrounded by an extracellular matrix composed of fibrillar and non-fibrillar proteins, and fundamental substances rich in glycosaminoglycans1. The qualitative and quantitative changes in the matrix content influence cell biology, controlling processes such as cell proliferation, apoptosis, migration, or differentiation. Hence, efforts are invested in recreating this microenvironment for in vitro studies of cells from different tissues2,3.
The myocardium consists of cardiomyocytes and an even larger quantity of fibroblasts that play a critical role in producing and maintaining the extracellular matrix within the myocardium4. Throughout life, the composition of the extracellular matrix can change in response to various normal and pathological factors. These modifications in extracellular matrix composition have a significant impact on the structure and biomechanical characteristics of the myocardium5. Accordingly, it should be advantageous for understanding cell-matrix interactions within the human myocardium if the microenvironment specific to different ages or pathological conditions was reproduced in vitro6,7.
The method described here aims to obtain the substrate for the culture of cardiac cells in vitro (termed cardiac ECM), mimicking the myocardial extracellular matrix in vivo.
Cardiovascular research presents specific challenges, including the difficulty in obtaining samples from living donors or patients and culturing human cardiac cells8. The method presented here addresses these challenges by enabling the acquisition of cardiac fibroblasts even from small bioptic fragments of human myocardium and culturing isolated cardiac cells in vitro on their native extracellular matrix typical of the human myocardium.
While current efforts focus on developing 3D scaffolds of biofartificial synthetic or natural polymers that mimic the biomechanical properties of normal myocardium9, they overlook the cell-matrix interactions and signaling that occur in both normal and pathological conditions. Since the cardiac ECM is synthesized by cardiac fibroblasts derived from the human heart, its composition is determined by the activity of these cells, which changes in response to various physiological and pathological conditions, thereby allowing the study of its specific influence on cardiac cell biology10.
The current protocol was specifically designed for human cardiac tissue, but its scientific basis should also apply to other organs, especially those with low regeneration potential, intense fibrosis, and scarring influencing overall structure and function, as well as limited sample numbers and sizes.
Protocol
Cardiac tissues were obtained from patients with end-stage heart failure due to ischemic cardiopathy who were undergoing heart transplantation. All specimens used for the experiments were collected with patient consent and without patient identifiers, following the protocols approved by the ethical committee of the University of Naples Federico II and in accordance with the principles outlined in the Declaration of Helsinki. The details of all the reagents and equipment used for the study are listed in the Table of Materials.
1. Experiment preparation
- Prepare sterile tools/apparatus: One large pair of surgical scissors, two sets of fine forceps, two pairs of microdissecting scissors, 1 L sterile bottle, 500 mL sterile bottle, and a 250 mL sterile bottle.
- Disinfect the 22 mm x 22 mm cover glasses with 70% ethanol for a few seconds. Aspirate the ethanol using a 10 mL serological pipette, let the cover glasses dry in the oven at 37 °C, and sterilize them in an autoclave.
- Dissolve commercially available powdered salts in sterile double-distilled water. In a 1 L sterile bottle, add 0.35 g of sodium bicarbonate powder to prepare 1 L of Hank's balanced salt solution (HBSS) at pH 7.4. Sterilize the solution using 0.22 µm filter membranes under a sterile hood and store at 4 °C until use.
- Weigh 0.1 g of potassium phosphate monobasic, 4.0 g of sodium chloride, 0.1 g of potassium chloride, and 0.575 g of sodium phosphate dibasic to prepare 500 mL of 1x phosphate-buffered saline (PBS). Dissolve the components in sterile double-distilled water and then check the pH value (7.4). Sterilize under a sterile hood by filtration and store at 4 °C until use.
- Add 5 mL of fetal bovine serum (FBS) to 45 mL of HBSS to prepare a final volume of 50 mL of trypsin stop solution (TSS). Store at 4 °C until use.
- Measure 223.75 mL of Dulbecco's modified Eagle medium (DMEM) supplemented with 25 mL of FBS (final concentration, 10%) and add 1.25 mL penicillin and streptomycin solution (pen/strep, final concentration, 0.5%) to prepare DMEM for fibroblast isolation and culture. Store at 4 °C until use.
- Prepare 0.2% (w/v) gelatin solution by adding 0.2 g of gelatin from porcine skin powder to 100 mL of PBS.
NOTE: The components and the respective concentrations for each solution utilized in the study are provided in Table 1.
2. Primitive culture of human cardiac fibroblasts by outgrowth from atrial myocardium fragments
NOTES: Steps 2.2-2.3 should be performed in sterile conditions.The protocol is validated for atrial samples approximately 0.3 cm in diameter, which is a typical dimension for biopsy specimens and allows the isolation of approximately 2 x 106 fibroblasts.
- Wash the freshly obtained sample of the atrium in a 100 mm glass plate with sterile HBSS, gently shaking it to remove the blood. Repeat this step three times, changing HBSS and the plate at each wash.
- Place the sample in a 100 mm plate previously wetted with a small volume of HBSS and chop finely with crossed scalpels to about 2 mm cubes.
- Place four small fragments of myocardium in a 35 mm plate, close enough to lie approximately at the angles of a 22 mm x 22 mm cover glass, place the sterile coverglass over the fragments, apply gentle pressure, and add 1.5 mL of DMEM.
- Incubate the culture plates at 37 °C in 5% CO2 for about 15 days or until cells reach 85% confluence, checking daily the outgrowth of cells at an inverted phase-contrast microscope and changing the culture medium every 3 days.
3. Subculture of human cardiac fibroblasts by warm trypsinization
NOTES: The steps reported below must be performed in sterile conditions. As the fibroblasts can migrate from the explanted tissue along the surface of the cover glass and culture plate, it is recommended to process both for the first subculture.
- Lift the cover glass with fine, sterile forceps, place it upside-down in a new 35 mm plate, and rinse with sterile PBS.
- Remove myocardium fragments, discard them, and rinse the 35 mm plate with outgrown cells with sterile PBS.
- Discard the rinse and add 1 mL of 0.25% trypsin-EDTA (Ethylenediaminetetraacetic acid) to each plate. Incubate for 5 min at 37 °C with 5% CO2. Confirm cell detachment at a microscope. The cells should be fully rounded up and detached, forming small clumps.
- Block the trypsinization by adding 2 mL of TSS to each 35 mm plate and collect the suspension into a sterile 15 mL tube, centrifuge at 400 x g for 5 min at 4 °C.
- Aspirate the supernatant using a 5 mL serological pipette, resuspend the pellet in 3 mL of DMEM and place cell suspension into a 60 mm plate.
- Incubate such obtained cells in subculture 1 (or passage 1) in 3 mL of DMEM at 37 °C with 5% CO2, changing the medium every three days until the cells reach 75% confluence.
- Subculture the fibroblasts that reached 75% confluence, repeating warm trypsinization and splitting the cells 1:3 into new 60 mm plates up to two times (passages 2 and 3).
4. Extracellular matrix deposition
- Prepare gelatin-coated dishes by adding 3 mL of sterile 0.2% gelatin solution to each 60 mm plate and incubating at 37 °C for 2 h; then aspirate the solution and add 3 mL of sterile PBS until use.
- Detach the cardiac fibroblasts by warm trypsinization and subculture them at high density (15 x 103 cells per cm2) on 60 mm gelatin-coated plates.
- Culture the fibroblasts in a confluent state for up to 21 days, allowing for extracellular matrix synthesis and deposition. Replace 50% of the medium with fresh medium every 3 days.
5. Extracellular matrix decellularization
NOTE: Steps 5.3-5.4 should be performed with gentle shaking to dislodge only cells without detaching the extracellular matrix.
- Prepare a decellularization solution containing 0.25% Triton X-100 and 10 mM NH4OH in PBS (calculate the final volume, considering 1 mL for every 60 mm plate).
- Remove the culture medium and rinse the plates with PBS. Discard the rinse.
- Add 1 mL of decellularization solution and observe the plates at an inverted phase-contrast microscope; after 1-2 min, when the cells are no longer discernible, gently add 4 mL of PBS to dilute the decellularization solution.
- Remove the diluted solution and gently rinse the plates with PBS; discard the rinse.
- Add 1 mL of PBS and store the ECM-coated plates at 4 °C until further use.
Representative Results
The outgrowth of fibroblasts from the small fragments of native myocardium placed in culture was observed within 3-5 days (Figure 1).
In the subsequent days, the number of fibroblasts continued to increase, possibly due to sustained outgrowth from the cardiac tissue specimen and the proliferation of migrated fibroblasts on the dish surface. It should not be expected that all myocardium fragments obtained by mechanical disaggregation with a scalpel yield the same number of fibroblasts within the same time, as the resulting tissue fragments can have slightly different volumes and cell survival rates. Notably, myocardium fragments with confirmed outgrowth activity can be retained and replated, thus allowing them to be used at least twice for fibroblast isolation.
The fibroblasts can be subcultured for several passages to increase the number of cells available for further experiments. However, once they reach confluence, fibroblasts cease to proliferate in vitro and assume synthesis and secretion activity, depositing extracellular matrix. Subsequent decellularization leaves the surface of the plate coated with cardiac ECM mesh (Figure 2). Visual observation of the culture dish, with the naked eye and using an inverted phase-contrast microscope, during fibroblast removal is essential to minimize the time of incubation in the decellularization solution to the necessary and sufficient minimum. The presence of a floating veil during incubation indicates permeabilization and detachment of the monolayer of fibroblasts, and the decellularization solution should be diluted and removed immediately after.
The composition of the fibroblast-derived cardiac ECM depends on the source of the fibroblasts. In our experiments, the cardiac ECM produced by fibroblasts isolated from normal human hearts and from human hearts with ischemic cardiopathy had different protein contents and exerted different influences on primitive cardiac cell proliferation, apoptosis, and migration11,12. Although ECM protein loss is inevitable during the decellularization process, the method employed in the present protocol preserved the composition and architecture of the in vitro produced cardiac ECM to a large extent (Figure 3).
In this protocol, atrial tissue was used because it can be obtained from both normal (donor) and pathological (recipient) hearts. While the whole pathological (recipient) heart can be sectioned and used for research (following the principles of the Declaration of Helsinki and with local ethical committee approval), it is not possible to obtain samples of ventricles from the donor heart at the time of transplantation. For this reason, if the scope of the study is to investigate differences between cardiac tissue in normal and pathological conditions, the best source of sample is the atrial wall. Depending on the availability of other viable cardiac tissue sources, samples from atrial or ventricular walls of the normal or pathological hearts can be used following the same protocol for cardiac ECM dish coating.
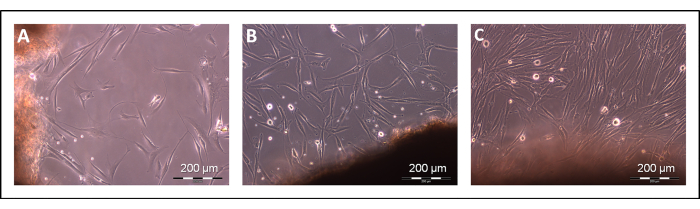
Figure 1: Isolation of cardiac fibroblasts. Fibroblast outgrowth from myocardium fragments after 4 days (A), 8 days (B), and 12 days (C) days of culture. Scale bars: 200 µm. Please click here to view a larger version of this figure.

Figure 2: Decellularization of extracellular matrix. Fibroblasts after approximately 10 s (A), 20 s (B), 30 s (C), 40 s (D), 50 s (E), and 60 s (F) of incubation in the decellularization solution. Scale bars: 200 µm. Please click here to view a larger version of this figure.
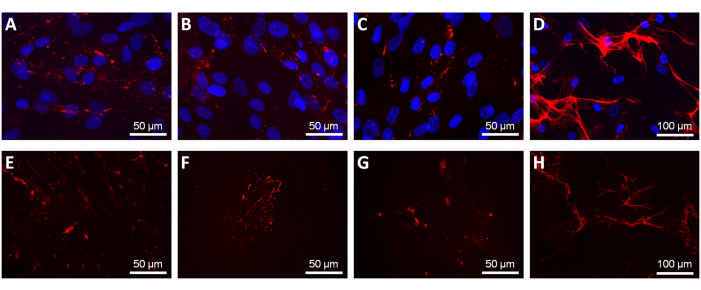
Figure 3: Extracellular matrix composition before and after decellularization. Indirect immunofluorescence staining of representative cardiac extracellular matrix proteins, including type III (A,E) and type IV (B,F) collagen, laminin (C,G), and fibronectin (D,H), before (A–D) and after (E–H) fibroblast removal. Cell nuclei were stained with DAPI (blue). Scale bars: (A–C,E–G) 50 µm; (D,H) 100 µm. Please click here to view a larger version of this figure.
| Solutions | Composition of solutions | Final volume | Comments | |
| Hank's Balanced Salt Solution (HBSS) | Commercially powered salts + double-distilled sterile water + 0.35 g sodium bicarbonate | 1 L | Sterilized by filtration/store at 4 °C | |
| Decellularization solution | 0.25% Triton X-100 + 10 mM NH4OH+ PBS | 50 mL | Sterilized by filtration/store at 4 °C | |
| Dulbecco's Modified Eagle Medium (DMEM) | 223.75 mL DMEM + 25 mL fetal bovine serum + 1.25 mL penicillin/streptomycin solution | 250 mL | Store at 4 °C | |
| Gelatin solution | 0.2 g gelatin powder + 100 mL Phosphate-buffered-saline | 100 mL | Sterilized by filtration/store at 4 °C | |
| Phosphate-buffered-saline (1x PBS) | 0.1 g potassium phosphate monobasic + 0.1 g potassium chloride + 4.0 g sodium chloride + 0.575 g sodium phosphate dibasic + double-distilled sterile water | 500 mL | Sterilized by filtration/store at 4 °C | |
| Trypsin stop solution (TSS) | 5 mL fetal bovine serum + 45 mL Hank's Balanced Salt Solution | 50 mL | Store at 4 °C | |
Table 1: The components and the respective concentrations for each solution utilized in the study.
Discussion
The fibroblasts isolated from human heart samples were cultured to confluence for 21 days to synthesize and deposit the extracellular matrix, forming a cohesive layer firmly adherent to the surface of the culture plate. Subsequent removal of cardiac fibroblasts, while preserving the deposited cardiac ECM, produced the substrate for studying the influence of myocardium-specific extracellular matrix on other cells within the cardiac tissue.
The concept of using a natural and tissue-specific substrate for in vitro cell culture has attracted the interest of biologists, leading to several attempts at developing such a substrate using fibroblasts isolated from neonatal animal hearts. As early as 1996, Vanwinkle et al.13 used neonatal rat cardiac fibroblasts to produce extracellular matrix coating on glass coverslips using EDTA, a calcium chelating agent, for cell separation. More recently, EDTA has been used in combination with a basic solution of Triton X-100 at lower concentrations14. Calcium ions mediate interactions between cells and extracellular matrix, as well as between different extracellular matrix molecules15. However, using such chelating agents can lead to matrix protein unfolding and loss of interactions between its different fibrillar and amorphous components, inevitably modifying the cardiac ECM obtained in vitro. In our experience, incubation with 0.5 mM EDTA required a lengthy time, with the first signs of cell detachment observable at the inverted phase-contrast microscope not appearing until after 2 h.
A similar method for in vitro decellularization was published by Hellewell et al.16 However, in the present protocol, a lower concentration of ammonium hydroxide (10 mM instead of 20 mM) was used, and Triton X-100 was added as a decellularization agent. While the authors of the mentioned protocol tested its application in three commercially available cell types, primitive cardiac fibroblasts were not included. Given the longstanding difficulty in obtaining and culturing adult human cardiomyocytes or cardiac fibroblasts in vitro, the focus here was on cardiac tissue and cells, which are of primary interest to cardiovascular research groups and cardiovascular regenerative medicine in general. It is possible that this solution may be suitable for other cell types as well.
While denaturation is necessary for protein solubilization, the use of a non-denaturing decellularization agent is essential for the quantitative and qualitative preservation of extracellular matrix components for further in vitro use. Therefore, Triton X-100 was used instead of SDS to remove the fibroblasts, leaving the intact cardiac ECM coating on the surface of the Petri dish. Incubation with a basic solution of Triton X-100 induces membrane permeabilization and cell lysis, resulting in fibroblast removal. If nuclear debris is an issue (for example, when subsequent experiments require cell transfection or nucleic acid molecular analysis), it is possible to minimize DNA content with DNase I before seeding other cell types on cardiac ECM.
The most significant advantage of the method detailed here is the ability to obtain cardiac ECM typical of different conditions related to human heart structure and function, as the same fibroblasts influenced by those changing conditions in vivo deposit the extracellular matrix in vitro. Thus, the composition of the fibroblast-derived coating of the culture dish changes according to the in vivo activity of the fibroblasts isolated from the heart, allowing subsequent studies of cell-matrix functional interactions in various normal and pathological conditions17,18.
Although it may be tempting to stimulate fibroblast activity in vitro by adding ascorbic acid, known to stimulate the production of collagen and glycosaminoglycans, such selective stimulation is discouraged, as it could potentially modify the composition of the obtained cardiac ECM towards a fibrotic phenotype19. Instead, depending on the study's aim, the composition of the cardiac ECM can be influenced in vitro by the addition of factors such as angiotensin peptides or adrenaline20,21, known to play a crucial role in the pathogenesis and pathophysiology of specific cardiovascular disorders.
The critical step in the process of extracellular matrix deposition, followed by fibroblast layer removal from culture, is the adhesion and preservation of fibroblast-derived matrix deposited on the dish surface. Unlike previously discussed studies where neonatal fibroblasts were cultured for only a few days, adult human cardiac fibroblasts survive in culture in the confluent state for as long as 21 days, continuously synthesizing and secreting extracellular matrix. It is essential to use gelatin-coated culture dishes and visually control fibroblast detachment, reducing the time of incubation in the decellularization solution to the necessary minimum.
In the present protocol, a feasible method for obtaining cardiac extracellular matrix in vitro is described. The 2D monolayer system reflects the in vivo microenvironment thanks to the presence of the native extracellular matrix produced by the same cells that are responsible for extracellular matrix synthesis and maintenance, namely fibroblasts, with the composition influenced by the biological cues present in vivo. Other biomimetic and 3D cell culture substrates or systems, such as hydrogels, spheroids, organoids, and 3D bioprinting methods, have attracted considerable attention as a strategy to mimic in vivo conditions, allowing the study of the complex spatial cell-cell and cell-ECM interactions22,23. These approaches address the spatial and biomechanical cues for in vitro cell growth and differentiation24,25, but they present strong limitations with regard to the composition and characteristics of the produced substrates, which hardly resemble those of the native tissue. Based on the scope of the study and its applications, the 2D cardiac ECM or 3D biomimetic scaffolds can be used, and together, they represent a complementary method for enhancing the current knowledge of cell biology and the capabilities of tissue regeneration.
Limitations
The method described is subject to certain limitations, particularly concerning the decellularization process designed to eliminate cellular elements. This process could inadvertently disturb the natural architecture or composition of the ECM. However, the ECM obtained from normal and pathological heart-derived fibroblasts differently influences the proliferation, apoptosis, and migration of cultured cells11,18, indicating that the described method preserves at least the main determinants of these biological processes.
Similarly, cardiac ECM obtained from different donors may have different compositions, even if the main cardiac pathology is the same. As with all human samples, there is great variability resulting from environmental factors, genetic determinants, pharmacological or other therapies, among others26. The cardiac ECM obtained by the described method should not always be expected to have the same composition and characteristics. If qualitative reproducibility is essential, it may be worth considering expanding the isolated fibroblasts up to two times (as described in the present protocol) to produce ECM on numerous Petri dishes or using fibroblasts obtained from different donors in the same Petri dish for ECM production.
Divulgations
The authors have nothing to disclose.
Acknowledgements
None.
Materials
| 1 L laboratory bottle | VWR | 215-1595 | Clean and autoclave before use |
| 10 mL serological pipet | Falcon | 357551 | Sterile, polystyrene |
| 100 mm glass plate | VWR | 391-0578 | Clean and autoclave before use |
| 100 mm plates | Falcon | 351029 | Treated, sterile cell culture dish |
| 15 mL sterile tubes | Falcon | 352097 | Centrifuge sterile tubes, polypropylene |
| 22 mm x 22 mm cover glasses | VWR | 631-1570 | Autoclave before use |
| 25 mL serological pipet | Falcon | 357525 | Sterile, polystyrene |
| 250 mL laboratory bottle | VWR | 215-1593 | Clean and autoclave before use |
| 35 mm plates | Falcon | 353001 | Treated, sterile cell culture dish |
| 5 mL serological pipet | Falcon | 357543 | Sterile, polystyrene |
| 50 mL sterile tubes | Falcon | 352098 | Centrifuge sterile tubes, polypropylene |
| 500 mL laboratory bottle | VWR | 215-1594 | Clean and autoclave before use |
| 60 mm plates | Falcon | 353004 | Treated, sterile cell culture dish |
| Ammonium hydroxide (NH4OH) | Sigma- Aldrich | 338818 | Liquid |
| Disposable scalpels | VWR | 233-5526 | Sterile and disposable |
| Dulbecco's Modified Eagle Medium (DMEM) | Sigma- Aldrich | D6429-500ml | Store at 2-8 °C; avoid exposure to light |
| Fetal Bovine Serum (FBS) | Sigma- Aldrich | F9665-500ml | Store at -20 °C. The serum should be aliquoted into smaller working volumes |
| Fine forceps | VWR | 232-1317 | Clean and autoclave before use |
| Gelatin from porcine skin | Sigma- Aldrich | G1890-100G | Commercial Powder |
| Hank's Balanced Salt Solution (HBSS) | Sigma- Aldrich | H1387-1L | Powder |
| Large surgical scissors | VWR | 233-1211 | Clean and autoclave before use |
| Microdissecting scissors | Sigma- Aldrich | S3146 | Clean and autoclave before use |
| Penicillin and Streptomycin | Sigma- Aldrich | P4333-100ml | Store at -20°C. The solution should be aliquoted into smaller working volumes |
| Potassium Chloride | Sigma- Aldrich | P9333 | Powder |
| Potassium Phosphate Monobasic | Sigma- Aldrich | P5665 | Powder |
| Sodium Chloride | Sigma- Aldrich | S7653 | Powder |
| Sodium Phosphate Dibasic | Sigma- Aldrich | 94046 | Powder |
| Stericup Filters | Millipore | S2GPU05RE | Sterile and disposable 0.22 mm filter membranes |
| Triton X-100 | Sigma- Aldrich | 9002-93-1 | Liquid |
| Trypsin-EDTA | Sigma- Aldrich | T4049-100ml | Store at -20 °C. It should be aliquoted into smaller working volumes |
References
- Gattazzo, F., Urciuolo, A., Bonaldo, P. Extracellular matrix: A dynamic microenvironment for stem cell niche. Biochim Biophys Acta. 1840 (8), 2506-2519 (2014).
- Zhang, Y., et al. Tissue-specific extracellular matrix coatings for the promotion of cell proliferation and maintenance of cell phenotype. Biomaterials. 30 (23-24), 4021-4028 (2009).
- Xing, H., Lee, H., Luo, L., Kyriakides, T. R. Extracellular matrix-derived biomaterials in engineering cell function. Biotechnol Adv. 42, 107421 (2020).
- Andrés-Delgado, L., Mercader, N. Interplay between cardiac function and heart development. Biochim Biophys Acta. 1863 (7), 1707-1716 (2016).
- Hall, C., Gehmlich, K., Denning, C., Pavlovic, D. Complex relationship between cardiac fibroblasts and cardiomyocytes in health and disease. J Am Heart Assoc. 10 (5), e019338 (2021).
- Castaldo, C., Chimenti, I. Cardiac progenitor cells: The matrix has you. Stem Cells Transl Med. 7, 506-510 (2018).
- Nurzynska, D., Iruegas, M. E., Castaldo, C., Müller-Best, P., Di Meglio, F. Application of biotechnology in myocardial regeneration-tissue engineering triad: cells, scaffolds, and signaling molecules. Biomed Res Int. 2013, 236893 (2013).
- Giacca, M. Cardiac regeneration after myocardial infarction: An approachable goal. Curr Cardiol Rep. 22 (10), 122 (2020).
- Spedicati, M., et al. Biomimetic design of bioartificial scaffolds for the in vitro modeling of human cardiac fibrosis. Front Bioeng Biotechnol. 10, 983782 (2022).
- Souders, C. A., Bowers, S. L., Baudino, T. A. Cardiac fibroblast: The renaissance cell. Circ Res. 105 (12), 1164-1176 (2009).
- Castaldo, C., et al. Cardiac fibroblast-derived extracellular matrix (biomatrix) as a model for the studies of cardiac primitive cell biological properties in normal and pathological adult human heart. Biomed Res Int. 2013, 352370 (2013).
- Nurzynska, D., et al. In vitro produced cardiac extracellular matrix for studies of myocardium regeneration potential. Tissue Eng Part A. 21, 77 (2015).
- VanWinkle, W. B., Snuggs, M. B., Buja, L. M. Cardiac ECM: A biosynthetic extracellular matrix for cardiomyocyte culture. In Vitro Cell Dev Biol Anim. 32, 478-485 (1996).
- Santhakumar, R., Vidyasekar, P., Verma, R. S. Cardiac ECM: A nano-matrix scaffold with potential application in cardiac regeneration using mesenchymal stem cells. PLoS One. 9 (12), e114697 (2014).
- Gopal, S., Multhaupt, H. A. B., Couchman, J. R. Calcium in cell-extracellular matrix interactions. Adv Exp Med Biol. 1131, 1079-1102 (2020).
- Hellewell, A. L., Rosini, S., Adams, J. C. A Rapid, scalable method for the isolation, functional study, and analysis of cell-derived extracellular matrix. J Vis Exp. (119), e55051 (2017).
- Nurzynska, D., Di Meglio, F., Montagnani, S., Castaldo, C., Hayat, M. A. Cardiac stem cells derived from epithelial-mesenchymal transition of epicardial cells: role in heart regeneration (method). Stem Cells and Cancer Stem Cells, vol. 5: Therapeutic Applications in Disease and Injury. , 109-115 (2012).
- Pagano, F., et al. Normal versus pathological cardiac fibroblast-derived extracellular matrix differentially modulates cardiosphere-derived cell paracrine properties and commitment. Stem Cells Int. 2017, 7396462 (2017).
- Xu, Y., et al. Vitamin C regulates the profibrotic activity of fibroblasts in in vitro replica settings of myocardial infarction. Int J Mol Sci. 24 (9), 8379 (2023).
- Jaffré, F., et al. Serotonin and angiotensin receptors in cardiac fibroblasts coregulate adrenergic-dependent cardiac hypertrophy. Circ Res. 104 (1), 113-123 (2009).
- Błyszczuk, P., et al. Activated cardiac fibroblasts control contraction of human fibrotic cardiac microtissues by a β-adrenoreceptor-dependent mechanism. Cells. 9 (5), 1270 (2020).
- Duval, K., et al. Modeling physiological events in 2D vs. 3D cell culture. Physiol. 32 (4), 266-277 (2017).
- Ungerleider, J. L., et al. Fabrication and characterization of injectable hydrogels derived from decellularized skeletal and cardiac muscle. Methods. 84, 53-59 (2015).
- Seo, Y., et al. Decellularized heart ECM hydrogel using supercritical carbon dioxide for improved angiogenesis. Acta Biomater. 67, 270-281 (2018).
- Liguori, G. R., et al. Molecular and biomechanical clues from cardiac tissue decellularized extracellular matrix drive stromal cell plasticity. Front Bioeng Biotechnol. 8, 520 (2020).
- Silva, A. C., Pereira, C., Fonseca, A. C. R. G., Pinto-do-Ó, P., Nascimento, D. S. Bearing my heart: The role of extracellular matrix on cardiac development, homeostasis, and injury response. Front Cell Dev Biol. 8, 621644 (2021).

.
