Coronary Angiography During Ex-Situ Heart Perfusion in a Porcine Model
Summary
Screening for coronary artery disease could increase the procurement of donor hearts with extended criteria. A protocol for performing reproducible coronary angiography during ex-situ heart perfusion is described herein in a porcine model.
Abstract
Heart transplantation is the gold standard treatment for advanced heart failure. The procurement of extended criteria donors (ECD) increases due to the current organ shortage. Coronary angiography is recommended in ECD at risk for coronary artery disease but is not systematically performed. These hearts are, therefore, either declined for transplant or procured without screening for coronary artery disease. Coronary angiography during normothermic ex-situ heart perfusion (NESP) could be an interesting approach to enhance the rate of ECD procurement and to reduce the risk of primary graft failure in the absence of coronary angiography in ECD. The present protocol aims to provide material details along with optimal imaging views for coronary angiography during NESP. Reproducible angiographic views were observed, including one dedicated to the right coronary artery, two for the left anterior descending artery, two for the circumflex artery, and a spider view. Continuous lactate extraction was observed in all procedures with a final median concentration of 1.10 mmol/L (0.61-1.75 mmol/L) two hours after coronary angiography, consistent with myocardial viability. The median contrast agent volume used for ex-situ imaging of the isolated perfused heart was 48 mL (38-108 mL). This protocol was reproducible for coronary artery imaging and did not impair myocardial viability during NESP.
Introduction
Heart transplantation is the gold standard therapy for advanced heart failure1,2. However, this treatment is limited by the current organ shortage, leading to increased allocation of extended criteria donors (ECD), who are more frequently exposed to coronary artery disease (CAD). Coronary angiography is recommended in France to prevent the risk of primary graft failure related to CAD if the donor's age is greater than 55 years or 45 years with two cardiovascular risk factors3. Even though coronary angiography increases the chances of graft acceptance by 9%, this procedure is performed in only 33% of cases in France4. Hearts from ECD at risk for CAD are therefore either declined for transplant or procured without screening for coronary artery disease.
The Organ Care System (OCS) is the only clinically available machine perfusion (MP) for Normothermic Ex-situ heart perfusion (NESP). This technology allows for longer preservation before transplantation by perfusing the donor heart with warm, oxygenated blood. Continuous metabolic assessment is performed during NESP by monitoring the lactate profile5. The efficacy and safety of NESP are now demonstrated by several studies, including a recent meta-analysis6,7,8. Performing coronary angiography during NESP would be of major interest to increase the probability of transplanting hearts from ECDs at risk for CAD.
Previously published experiences of ex-situ coronary angiography9,10,11 suggest that conventional views are not suitable due to the inverted position of the heart in the perfusion module. In addition, OCS components visible with X-rays may impair imaging findings. Lastly, the toxicity of contrast agents on the myocardium during isolated heart perfusion remains questionable. The present report aims to provide a safe and reproducible approach to performing coronary angiography during NESP.
Protocol
All pigs received humane care in compliance with the 'Principles of Laboratory Animal Care' formulated by the National Society for Medical Research and the 'Guide for the Care and Use of Laboratory Animals' prepared by the Institute of Laboratory Animal Resources and published by the National Institutes of Health (NIH Publication No. 86-23, revised 1996). This study design was approved by the French Ministry of Research for the use of animals for scientific purposes (protocol APAFiS#23716-2020012112432923v1). Eight large-white male pigs (60 kg, 6 months) were used for the present study. The animals were housed for one week before surgery with congeners at room temperature with a day-night light shift. Food and water were freely available. The reagents and equipment used for this study are listed in the Table of Materials.
1. Animal preparation
- Premedicate animals with an intramuscular injection of Tiletamine and Zolazepam (2 mg/kg).
- Complete anesthesia with an intravenous injection of propofol (3 mg/kg).
- After mask ventilation using 100% FiO2, perform oro-tracheal intubation with a 7.5 mm probe using a laryngoscope dedicated to large animals.
- Maintain general anesthesia by inhalation of 2% isoflurane, continuous infusion of sufentanil (10 µg/kg/h), and intermittent injections of cisatracurium (0.3 mg/kg every 30 min).
- Perform a transthoracic echocardiogram to look for any cardiac disease that may contraindicate ex-situ perfusion of the heart. Significant aortic regurgitation does not allow for efficient NESP in Langendorff mode since coronary flow will be impaired. Retrograde aortic flow will mainly go to the left ventricle in this condition.
NOTE: Animals presenting with cardiac abnormalities, including significant aortic regurgitation, should be excluded from the protocol (anesthesia is stopped to allow transfer to the animal facility).
2. Heart retrieval
- Perform a midline sternotomy. Use a scalpel (No. 23) to open the skin. Open the subcutaneous tissues using a monopolar cautery. Open the sternum using a semilunar sternal saw and complete the sternotomy using Mayo scissors.
- Dissect the thymus, open it, and expose the pericardium with Metzenbaum scissors to avoid ventricular fibrillation induced by the electric field of the monopolar cautery.
- Inject Heparin (300 IU/kg) directly into the right atrium.
- Perform purse-string stitches using 4/0 polypropylene sutures on the right atrium and aortic root to prepare for cannulation.
- Carefully detach the aorta from the main pulmonary artery to prepare for clamping of the aorta.
- Control the superior and inferior vena cava with a surgical loop.
- Introduce a 9-French antegrade long cannula into the aortic root 3 min after heparin administration.
- Purge the cardioplegia line and connect it to the aortic cannula, then clamp the cardioplegia line before blood collection.
NOTE: Blood collection results in a significant volume drop that may impair appropriate cannulation of the ascending aorta. Therefore, it is easier to do it before blood collection. - Canulate the right atrium with a 32-French two-stage venous cannula.
- Connect a sterilized bag previously heparinized (10,000 IU) to the right atrium cannula and collect 1500 mL of blood.
NOTE: The collected blood is leukodepleted by another operator using a leukocyte filter and then placed directly in the machine perfusion circuit. - Clamp the aorta and infuse 1000 mL of cold (4 °C) cardioplegia solution (Table 1).
- Secure the superior and inferior vena cava with a surgical loop.
- Stop mechanical ventilation. Open the inferior vena cava and the left superior pulmonary vein to unload the heart. Place a cold saline solution in the pericardium.
- Remove the aortic and venous cannulas. Explant the heart by sectioning the left azygos vein, the two vena cavae, the distal ascending aorta, the pulmonary artery trunk, and the four pulmonary veins.
NOTE: The left azygos vein is specific to porcine anatomy. This vein goes directly to the coronary sinus. The left azygos vein must be transected between two ligatures before the heart is harvested.
3. Heart preparation for NESP
- Place the heart in 4 °C saline solution.
- Investigate the aortic valve to confirm normal coaptation and close a patent foramen ovale if present using a 4-0 polypropylene suture.
- Complete the separation between the aorta and the pulmonary artery.
- Place four pledgeted sutures (4/0 polypropylene) around the distal section of the ascending aorta.
- Insert the appropriate aortic cannula and fix it with a hose clamp. Four sizes of aortic cannula are available: 3/4 inch, 7/8 inch, 1 inch, and 5/4 inch.
- Insert the 30 Fr pulmonary artery cannula and secure it with a 4/0 purse-string suture.
- Place a 20-French intracardiac sump through the mitral orifice to vent the left ventricle.
4. Normothermic ex-situ heart perfusion
NOTE: A detailed composition of the cardioplegia solution, priming solution, epinephrine, and adenosine infusions is provided in Table 1.
- Prime the machine perfusion (MP) with the collected blood (1500 mL) and add the priming solution (500 mL).
- Oxygenate with a membrane oxygenator and warm the blood to 34 °C using a heater-cooler.
- Place a pressure probe on the aortic line above the aorta.
- Place a flowmeter around the pulmonary artery cannula.
NOTE: Continuous infusion of epinephrine (0.5 mL/h) is initiated after connection to the aortic line. Adenosine infusion will be adjusted to maintain mean aortic pressure between 65 mmHg and 80 mmHg. - Increase the pump speed to reach a flow rate of 2500 mL/min.
- Collect an initial blood sample for blood gas analysis, ionogram, and lactate level. Adjust potassium, calcium, glycemia, and pH if necessary.
NOTE: All these steps are performed simultaneously with heart retrieval. - Before positioning the heart in the MP, reduce the pump flow to 1000 mL/min and remove the shunt between the aortic and venous lines.
- De-air the aorta and connect the aortic cannula to the aortic line.
- Place the heart for optimal NESP (following the manufacturer's instructions). The postero-inferior ventricular wall will be in front of the operator, and the pulmonary artery cannula will be placed behind the aortic cannula.
- Perform gentle cardiac massage until the heart is warmed.
- Deliver a 20 J shock in case of ventricular fibrillation.
- Pace the heart at 80 beats per minute with an external pacemaker.
- Slowly increase pump flow to reach an aortic pressure of 65 mmHg.
- Close the inferior vena cava with a 4/0 polypropylene running suture.
- Purge and connect the pulmonary artery cannula to the venous line to continuously assess coronary flow.
NOTE: The coronary flow should be between 650 mL/min and 850 mL/min. The mean aortic pressure should be maintained between 70 mL/min and 85 mmHg. Assess blood gas and ionogram every hour and lactates every 30 min. If venous lactate is higher than arterial lactate, the first step is to ensure the normality of the ionogram and correct any biochemical abnormality. If pressure and coronary flow are low, increase the pump speed. If the pressure is high and the coronary flow is low, increase adenosine to vasodilate the coronary arteries. Stop adenosine infusion if aortic pressure is below 70 mmHg.
5. Coronary angiography during NESP
- Experimental setup
NOTE: The setup is depicted in Figure 1.- Ensure sufficient space is available for the machine perfusion (MP) system.
- Elevate the MP by approximately 25 cm using a platform. Rotate the MP 90° to the left from the initial position.
- Perform coronary angiography (Figure 2).
- Flush the 5 French introducer and the 5 French Judkins right 3.5 coronary angiography catheter with serum.
- Insert a 5 French introducer through the valved orifice of the MP.
NOTE: The distal part of the introducer has to be placed just above the aorta. - Advance the guide wire into the coronary angiography catheter up to the distal orifice. Insert the catheter and guide wire into the introducer up to the aortic root.
- Perform catheterization of the right coronary artery (RCA) using the following views: RAO 35/ CAU 44 or RAO 135/ CAU 62. Capture images of the RCA network using the views described in Table 2.
- Perform catheterization of the left coronary artery with the following view: RAO 135/ CAU 62. Capture images of the left coronary artery network using the views described in Table 2.
- Remove the coronary angiography catheter and the introducer.
NOTE: Use approximately 50 mL of Iomeprol without exceeding 100 mL.
Representative Results
The outlined protocol was designed for easy reproducibility in clinical practice. Minimal modifications were made to the available module for Normothermic Ex-situ Heart Perfusion (NESP). The only adjustments were made to connect the module to an extracorporeal life support pump, an oxygenator, and a heater-cooler unit to replace the machine perfusion (MP).
Two test procedures were initially conducted to assess the coronary angiography views used in this protocol. Six coronary angiographies were then performed on ex-situ perfused porcine hearts. Hearts were perfused for 240 min, and coronary angiography was performed after 120 min. The initial 120-min period was defined to replicate the average time observed in clinical practice between the beginning of NESP and the return to the transplant center where coronary angiography will be performed; the last two hours represented the duration needed for coronary angiography and preparation of the receiver in the operating room. Coronary angiography procedures were performed by two experienced interventional cardiologists. Mean aortic pressure and coronary flow were continuously monitored. Lactate levels were measured every 30 min to assess myocardial viability. Iomeprol volume and exposure to radiation were also recorded.
Baseline parameters are presented in Table 3. All hearts showed a favorable metabolic profile with preserved lactate extraction and were successfully preserved for 4 h on NESP. There were no premature endings due to perfusion failure. There was a failure in catheterizing the left coronary artery during the second procedure. In the third case, the views between the left and right coronary arteries were switched. The median volume of the contrast agent was 48 mL (min 38 mL-max 108 mL). The median dose of ionizing radiation was 3.30 mGy (min 1.0 mGy-max 17 mGy), with a median Dose-Area Product (DAP) of 0.55 Gy/cm2 (min 0.18 Gy/cm2-max 2.05 Gy/cm2). The median exposure time to ionizing radiation was 9.50 min (IQR: 6.75-12.75). The average procedure duration was 34.5 min (± 21). Lactate profiles, coronary flow, aortic pressure, and metabolic results are shown in Table 4 and Figure 3.

Figure 1: MP and fluoroscopic arm setup. (A) The MP is elevated by a platform so that the heart is within the X-ray field of the fluoroscopic arm to access the coronary angiography images. (B) After a 10 cm elevation, the MP is within the X-ray field, and coronary angiography images can be captured. (C) Operators performing coronary angiography through the valved orifice. Please click here to view a larger version of this figure.
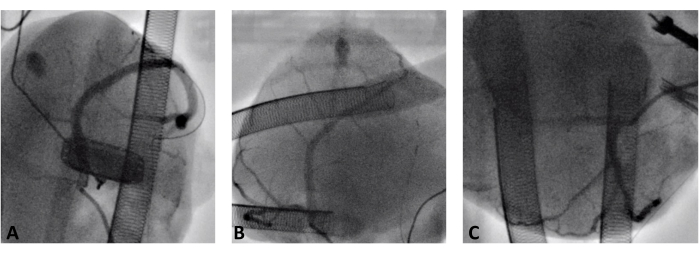
Figure 2: Coronary angiography during normothermic ex-situ heart perfusion. (A) Right coronary artery. (B) Left anterior descending artery. (C) Circumflex artery. Please click here to view a larger version of this figure.
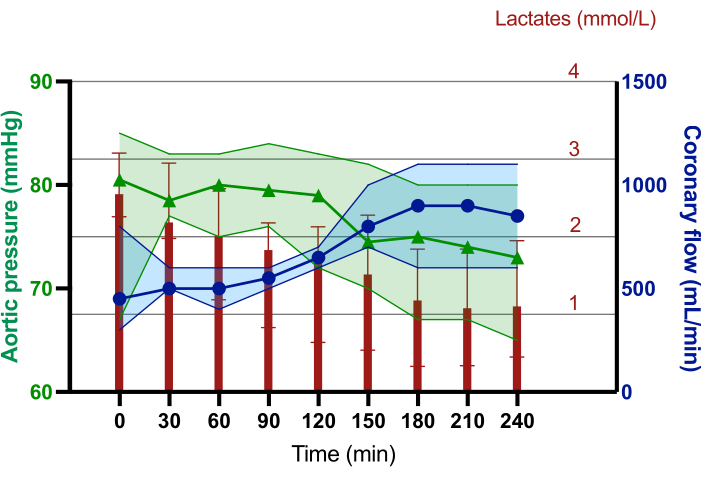
Figure 3: Median lactate levels, coronary flow, and aortic pressure trends during the 240 min of normothermic ex-situ heart perfusion. The red diagram bars represent the median lactate concentration in mmol/L. The blue line with dots represents the median coronary flow in mL/min. The green line with triangles represents the median aortic pressure in mmHg. The error bars represent 95% CI. Please click here to view a larger version of this figure.
| Solution | Components |
| Cardioplegia | 500 mL of Ringer solution |
| 10 mL of 10% KCl | |
| 3 mL of 2% Xylocaïne | |
| 6 mL of 20% Mannitol | |
| 6 mL of 8.4% Sodium Bicarbonate | |
| 7 mL of 15% Magnesium Sulfate | |
| Priming | 500 mL NaCl solution |
| 150 mg of Magnesium | |
| 250 mg of Methylprednisolone | |
| 1 g of Cefotaxime | |
| 50 UI of Insuline | |
| 5 mL of 30% Glucose | |
| Epinephrine infusion | 500 mL of 5% Glucose |
| 0.25 mg of epinephrine | |
| Adenosine infusion | 60 mg of Adenosine |
| 40 mL of NaCl solution |
Table 1: A detailed composition of the cardioplegia solution, priming solution, epinephrine, and adenosine infusions.
| Coronary arteries | Lateral rotation | Supero-inferior rotation |
| RCA | RAO 35 | CAU 44 |
| Proximal RCA | RAO 64 | CAU 38 |
| Distal RCA | RAO 65 | CAU 66 |
| Spider | RAO 70 | CAU 52 |
| LAD | RAO 135 | CAU 62 |
| LAD | RAO 167 | CAU 79 |
| Marginal artery | RAO 44 | CAU 51 |
| Cx | LAO 125 | CAU 83 |
Table 2: Coronary angiography views. RCA: Right Coronary Artery; LAD: Left Anterior Descending coronary artery; Cx: Circumflex artery; RAO: Right Anterior Oblique view; LAO: Left Anterior Oblique view; CAU: Caudal view.
| T0 | |
| Weight | |
| Pigs (kg) † | 51.9 (50.1—53.9) |
| Hearts (g) ‡ | 228.5 (± 30.1) |
| Timing (min) ‡ | |
| Heart retrieval | 13.5 (± 5.4) |
| Cold ischemic time | 41.8 (± 9.8) |
| Back-table time | 29.3 (± 8.2) |
| Perfusion parameters † | |
| Aortic Pressure (mmHg) | 80.50 (75.25—83.50) |
| Coronary flow (L/min) | 0.450 (0.375– 0.650) |
| Biological results † | |
| Lactates (mmol/L) | 2.59 (2.87—2.32) |
| pH ‡ | 7.44 (± 0.05) |
| pO2 (mmHg) | 523 (466–631) |
| pCO2 (mmHg) | 30.1 (26.2–35.8) |
| HCO3– (mmol/L) | 22.10 (17.55–24.37) |
| Natremia (mmol/L) | 143 (138.7–145.0) |
| Kaliemia (mmol/L) ‡ | 2.97 (± 0.60) |
| Calcemia (mmol/L) | 1.11 (1.09–1.16) |
| Glycemia (mmol/L) | 1.17 (1.12–1.34) |
Table 3: Baseline characteristics. †Data are expressed as medians (with IQR interquartile range).
‡Data are expressed as means (with standard derivations). T0 = at the beginning of the normothermic ex-situ heart perfusion; pO2 = oxygen partial pressure; pCO2 = carbon dioxide partial pressure; HCO3– = Bicarbonates.
| T120 | T240 | |
| Perfusion parameters | ||
| Aortic Pressure (mmHg) | 79.00 (74.2 – 82.2) | 73.00 (69.50 – 76.25) |
| Coronary flow (L/min) | 0.65 (0.60 – 0.70) | 0.85 (0.67 – 1.10) |
| Biological results | ||
| Lactates (mmol/L) | 1.68 (0.92 – 2.08) | 1.10 (0.61 – 1.75) |
| pH | 7.39 (7.35 – 7.42) | 7.36 (7.32 – 7.43) |
| HCO3- (mmol/L) | 22.10 (20.10 – 23.12) | 21.20 (19.22 – 22.92) |
| Natremia (mmol/L) | 144.5 (144.00 – 146.25) | 142.5 (137.50 – 145.25) |
| Kaliemia (mmol/L) | 3.55 (3.12 – 3.82) | 3.10 (2.97 – 3.62) |
| Calcemia (mmol/L) | 1.13 (1.11 – 1.16) | 1.10 (1.08 – 1.16) |
Table 4: Perfusion parameters and biological results before and 2 h after coronary angiography. Data are expressed as medians (with IQR interquartile range). T120 = 120 min after the beginning of the normothermic ex-situ heart perfusion, just before coronary angiography; T240 = 240 min after the beginning of the normothermic ex-situ heart perfusion, 120 min after coronary angiography; HCO3– = Bicarbonates.
Discussion
This study describes, for the first time, a protocol for coronary angiography on an isolated beating heart without compromising myocardial metabolic viability during Normothermic Ex-situ Heart Perfusion (NESP). The views were reproducible, allowing for a comprehensive anatomical analysis of the coronary network. The required contrast agent volume was approximately 50 mL and rarely exceeded 100 mL. In comparison, the average volume used for in vivo coronary angiography is twice as much, ranging between 100 mL and 150 mL. The volume of the contrast agent was associated with the duration of the procedure. The experience gained by interventional cardiologists led to a reduction in exposure to X-rays as well as the volumes of the contrast agent.
Coronary angiographies showed that artifacts related to machine perfusion (MP) did not impair imaging interpretation. The unusual cardiac position during NESP led to anatomical views different from those observed in clinical practice during in vivo coronary angiography. The RAO 135/CAU 62 view was appropriate for catheterizing both the right and left coronary arteries. However, the RAO 35/CAU 44 incidence also facilitated easy catheterization of the RCA. This view is usually the first to be observed in our experience. Thus, catheterizing the RCA using RAO 35/CAU 44 as the first option is recommended. In case of technical difficulty, the RAO 135/CAU 62 view can be used as a secondary option. The 25 cm elevation of the MP requires meticulous care to prevent inclinations exceeding 45°, as this may introduce air into the perfusion circuit. Sterility during the procedure is mandatory. An appropriate sterile field and disinfection of the valve orifice of the aortic line are highly recommended. Additionally, the operator is very close to the fluoroscopic arm in this configuration. Protection against X-ray exposure has to be adapted.
Limited literature exists on coronary angiography during Normothermic Ex-situ Heart Perfusion (NESP). The first description was reported in 2014 by Anthony et al. at St. Vincent Hospital in Australia9. Ex-situ coronary angiography identified coronary artery disease in the three main arteries, and the graft was not transplanted. A second description by the same team in 2020 reported the success of two donations after circulatory death (DCD) heart transplantations, including coronary angiography during NESP10. In both publications, the authors did not describe the volume and nature of the contrast agent they used or the imaging views. Another case of NESP coronary angiography on a DCD heart was reported by Mendirichaga et al. in San Diego in April 202212,13.
To the best of our knowledge, this study is the first to investigate the reproducibility of coronary angiography during NESP. A detailed protocol for coronary angiography during NESP is provided that could be applied for clinical use. These results obtained in a porcine model are reproducible in clinical practice, as porcine hearts share anatomical and physiological similarities with the human heart. Additionally, instrumentation and resuscitation of porcine hearts on machine perfusion are similar to clinical practice on the human heart.
This protocol could enhance the assessment of extended criteria donors (ECD), particularly within the European population, since donors are older and thus more exposed to coronary artery disease (CAD) compared to North America. This demographic difference underscores the importance of considering all available donor hearts, including those without coronary angiography, before organ procurement1. In France, Ivanes et al. showed that coronary angiography was not performed in 55% of ECDs. Among these hearts without coronary angiography, 88% were offered, and 57% were accepted4. In the US, 30% of ECD hearts are accepted without coronary angiography since the donor pool is younger with fewer donors at risk for CAD14.
This procedure has some limitations. It only allows for the investigation of the main coronary vessels. However, distal stenosis or single-vessel stenosis does not necessarily rule out heart transplantation. Hearts with mild stenosis can be accepted for unstable patients (such as those requiring VA-ECMO, Bi-VAD, etc.) in urgent need of heart transplantation15. Currently, no coronary artery revascularization has been performed on the Organ Care System (OCS) to date, and coronary artery bypass grafting is not a conventional procedure during heart transplantation16,17. The current gold standard approach remains to perform coronary angiography before organ procurement due to its cost-effectiveness and more sensitive analysis of the coronary network. Ex-situ coronary angiography represents an interesting alternative option if coronary angiography cannot be performed in extended criteria donors (ECD) at high risk for CAD while the recipient needs urgent transplantation.
Other less invasive methods for coronary assessment are currently under development, such as dynamic speckle laser imaging18. This technology employs low-energy laser illumination to achieve high-resolution real-time imaging of coronary microvascularization. However, this method still requires validation in isolated ex-situ perfused hearts. Previous work has described cardiac echography during Normothermic Ex-situ Heart Perfusion (NESP), requiring heart perfusion in a working mode state19. Screening for CAD in ECD could expand the pool of donor hearts. This protocol would also allow for the assessment of the coronary network in donation after circulatory death (DCD) donors. In this category of donors, coronary angiography cannot be performed before organ retrieval due to ethical considerations. Performing coronary angiography during NESP in this setting could enhance the safety of these transplantations. NESP is a major technological advancement in the field of heart transplantation. This technology paves the way for a better assessment of donor hearts prior to transplantation.
Divulgations
The authors have nothing to disclose.
Acknowledgements
We gratefully acknowledge the Fondation Cœur Recherche and ADETEC for their financial support. We would also like to express our gratitude to Marie Lannelongue's lab team for their contributions to this project: Antoine Agathon, Laure Aubrège, Aimée Alméras, Catarina Bastos Dias, Antoine Grussé, Anselme Limousin, and Slimane Idir.
Materials
| 3T Heater Cooler System | Liva Nova, Châtillon, France | IM-00727 A | Extracorporeal Heater Cooler device |
| 4-0 polypropylene suture | Peters, bobigny, France | 20S15B | sutures |
| 5-0 polypropylene suture | Peters, bobigny, France | 20S10B | sutures |
| Adenosine | Efisciens BV, Rotterdam, Netherlands | 9088309 | Drugs for the ex-vivo perfusion |
| Adrenaline | Aguettant, Lyon, France | 600040 | Drugs for the ex-vivo perfusion |
| ADW software | General Electric Healthcare, Chicago, Ill, USA | ADW_IGS730/ M5173653 | Software used for the coronary angiography visualization |
| Atracurium | Pfizer Holding France, Paris, France | 582547 | Drugs for the induction of the anesthesia |
| CG4 + | Abbott, Chicago, Ill, USA | 03P85-25 | Lactates and blood gas cartridge for the blood analyzer machine |
| CG8 + | Abbott, Chicago, Ill, USA | 03P88-25 | Ionogram cartridge for the blood analyzer machine |
| DeltaStream | Fresenius Medical Care, L’Arbresle, France | MEH2C4024 | Extracorporeal blood pump |
| Discovery IGS 7 | General Electric Healthcare, Chicago, Ill, USA | Discovery IGS7.3 / M5173652 | Angiography system for hybrid operating room |
| DLP Aortic Root Cannula | Medtronic Inc. Minneapolis, Minneapolis, USA | ref. 11012, 12.5 inch | Aortic Root Cannula |
| External pacemaker | Medtronic Inc. Minneapolis, Minneapolis, USA | 5392 | Pacemaker device |
| Fresinus Agilia injectomat | Fresenius vial , Brezins, France | 018010/20640320 | Syringe driver |
| Glucose 5% | B.Braun Melsungen AG, Melsungen, Germany | 3400891780017 | Drugs for the priming solution |
| Haemonetics leukocyte filter | Haemonetics, Boston, Massachusetts, USA | RS1VAE | Leukocyte Filter |
| Heart Perfusion Set, Organ Care System | Transmedics, Andover, MA, USA | Ref#1200 | Normothermic ex-vivo heart perfusion device, including, the perfusion module, pulmonary artery and aortic cannula |
| Intellivue MX550 | Philips Healthcare, Suresnes, France | NA | Permanent monitoring system |
| Iomeron 350 | Bracco imaging, Massy, France | 3400933794606.00 | Iomeprol contrast agent product |
| istat 1 | Abbott, Chicago, Ill, USA | 714336-03O | Blood Analyzer machine |
| Judkins Right 3.5 | Boston Scientific, Marlborough, Massachusetts, USA | H74934358202 | Coronary angiography catheterization probe |
| Magnesium | Aguettant, Lyon, France | 564 780-6 | Drugs for the cardioplegia |
| Magnesium Sulfate | Aguettant, Lyon, France | 600111 | Drugs for the cardioplegia |
| Mannitol 20% | Macopharma, Mouvoux, France | 3400891694567.00 | Drugs for the cardioplegia |
| Methylprednisolone | Mylan S.A.S, Saint Priest, France | 400005623 | Drugs for the priming solution |
| MWI software | General Electric Healthcare, Chicago, Ill, USA | NA | software used for the Ultrasound echocardiographic machine |
| Orotracheal probe | Smiths medical ASD, Inc., Minneapolis, Minneapolis, USA | 100/199/070 | probe for the intubation during anesthesia |
| Potassium chloride 10% | B.Braun Melsungen AG, Melsungen, Germany | 3400892691527.00 | Drugs for the cardioplegia |
| Propofol | Zoetis France, Malakoff, France | 8083511 | Drugs for the induction of the anesthesia |
| Quadrox-I small Adult Oxygenator | Getinge, Göteborg, Sweden | BE-HMO 50000 | Extracorporeal blood oxygenator |
| Ringer solution | B.Braun Melsungen AG, Melsungen, Germany | DKE2323 | Drugs for the cardioplegia |
| Sodium Bicarbonate | Laboratoire Renaudin, itxassou, France | 3701447 | Drugs for the cardioplegia |
| Sodium chloride | Aguettant, Lyon, France | 606726 | Drugs for the priming solution |
| Swan Ganz Catheter | Merit Medical, south jordan, utah, USA | 5041856 | Right pressure and cardiac output probe |
| Tiletamine | Virbac France, Carros, France | 3597132126021.00 | Drugs for the induction of the anesthesia |
| Two staged Venous Cannula | Medtronic Inc. Minneapolis, Minneapolis, USA | 91240, 32 french | Venous Cannula |
| Vivid E95 ultraSound Machine | General Electric Healthcare, Chicago, Ill, USA | NA | Ultrasound echocardiographic machine |
| Xylocaïne 2% | Aspen, Reuil-malmaison, France | 600550 | Drugs for the cardioplegia |
| Zolazepam | Cardinal Health LLC, Waukegan, Illinois, USA | 31050522 | EKG detection electrodes |
| Zolazepam | Virbac France, Carros, France | 3597132126021.00 | Drugs for the induction of the anesthesia |
References
- Khush, K. K., et al. The International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation: 37th adult heart transplantation report-2020; focus on deceased donor characteristics. J Heart Lung Transplant. 39 (10), 1003-1015 (2020).
- Khush, K. K., et al. The International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation: Thirty-fifth Adult Heart Transplantation Report-2018; Focus Theme: Multiorgan Transplantation. J Heart Lung Transplant. 37 (10), 1155-1168 (2018).
- Dorent, R., et al. Assessment of potential heart donors: A statement from the French heart transplant community. Arch Cardiovasc Dis. 111 (2), 126-139 (2018).
- Ivanes, F., et al. Performing diagnostic coronary angiography to evaluate high-risk cardiac donors: A French nationwide cohort study. Int J Cardiol. 277, 71-78 (2019).
- Hassanein, W. H., et al. Continuous perfusion of donor hearts in the beating state extends preservation time and improves recovery of function. J Thorac Cardiovasc Surg. 116 (5), 821-830 (1998).
- Ardehali, A., et al. Ex-vivo perfusion of donor hearts for human heart transplantation (PROCEED II): a prospective, open-label, multicentre, randomised non-inferiority trial. Lancet. 385 (9987), 2577-2584 (2015).
- McCurry, K., et al. 294: Prospective Multi-Center Safety and Effectiveness Evaluation of the Organ Care System Device for Cardiac Use (PROCEED). J Heart Lung Transplant. 27 (2), S166 (2008).
- Langmuur, S. J. J., et al. Normothermic ex-situ heart perfusion with the organ care system for cardiac transplantation: A Meta-analysis. Transplantation. 106 (9), 1745-1753 (2022).
- Anthony, C., et al. Ex vivo coronary angiographic evaluation of a beating donor heart. Circulation. 130 (25), e341-e343 (2014).
- Nadel, J., et al. Successful transplantation of high-risk cardiac allografts from DCD donors following ex vivo coronary angiography. J Heart Lung Transplant. 39 (12), 1496-1499 (2020).
- Dang Van, S., et al. Ex vivo perfusion of the donor heart: Preliminary experience in high-risk transplantations. Arch Cardiovasc Dis. 114 (11), 715-726 (2021).
- Mendirichaga, R., et al. Ex vivo coronary angiography and intravascular ultrasound of a donor heart in the organ care system. JSCAI. 1 (4), 100342 (2022).
- Schibilsky, D., et al. Coronary angiography and intravascular ultrasound in an ex-vivo perfused heart using the Organ Care System (OCS). J Heart Lung Transplant. 38 (4), 271 (2019).
- Davis, M., et al. Coronary angiography of older donor hearts during evaluation for transplantation. J Heart Lung Transplant. 33 (4), S161-S162 (2014).
- Grauhan, O., et al. Coronary atherosclerosis of the donor heart – impact on early graft failure. Eur J Cardiothorac Surg. 32 (4), 634-638 (2007).
- Abid, Q., Parry, G., Forty, J., Dark, J. H. Concurrent coronary grafting of the donor heart with left internal mammary artery: 10-year experience. J Heart Lung Transplant. 21 (7), 812-814 (2002).
- Pinto, C. S., Prieto, D., Antunes, M. J. Coronary artery bypass graft surgery during heart transplantation. Interact Cardiovasc Thorac Surg. 16 (2), 224-225 (2013).
- Plyer, A., Colin, E., Orlik, X., Akamkam, A., Guihaire, J. Imaging the vasculature of a beating heart by dynamic speckle: the challenge of a quasiperiodic motion. J Biomed Opt. 28 (04), 046007 (2023).
- Dang Van, S., Brunet, D., Akamkam, A., Decante, B., Guihaire, J. Functional assessment of the donor heart during ex situ perfusion: Insights from pressure-volume loops and surface echocardiography. J Vis Exp. 188, 63945 (2022).

.
