Síntesis de un metaloceno Ti(III) utilizando la técnica de línea de Schlenk
English
Diviser
Vue d'ensemble
Fuente: Tamara M. Powers, Departamento de química, Texas A & M University
Químicos inorgánicos a menudo trabajan con altamente sensibles al aire y agua compuestos. Los dos métodos más comunes y prácticos para aire libre síntesis utilizan líneas de Schlenk o guanteras. Este experimento demostrará cómo realizar manipulaciones simples en una línea de Schlenk con énfasis en preparación solvente y transferencia. A través de la síntesis de un complejo reactivo del metaloceno Ti(III), le mostraremos un método nuevo, simple para desgasificar solventes, así como la forma para transferir el solvente cánula y la jeringa en una línea de Schlenk.
La síntesis de un metaloceno Ti(III) compuesto 3 se muestra en la figura 1. 1 3 compuesto es altamente reactivo con O2, (ver oxidación de 3 compuesto metaloceno Ti(IV) 4 que se muestra en la figura 1). Por lo tanto, es importante para que funcione la síntesis bajo condiciones anaeróbicas. La síntesis del compuesto puede 3 objetivo controlarse visualmente y progresa a través de un color adicional cambiar antes de llegar al producto deseado, que es azul en color. Si durante el experimento hay un cambio de color observado de azul a amarillo (o verde = azul + amarillo), esto es una indicación que O2 entraron en el frasco y eso oxidación indeseada de 3 compuesto para el Ti(IV) analógico (compuesto 4) ha ocurrido.
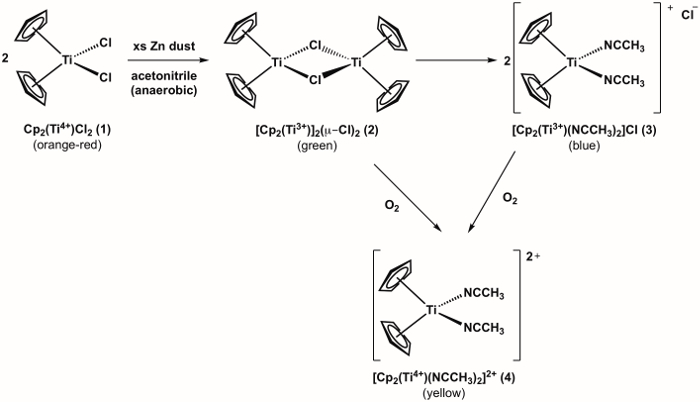
Figura 1. Síntesis de metaloceno Ti(III) compuesto de 3 y de reacción con el O2.
Principles
Procédure
Résultats
Upon addition of the acetonitrile in step 4, the solution should change color from orange, to green, to blue (Figure 4). Failure to obtain the blue color indicates a leak in the system. Addition of acetonitrile by syringe in step 6 should result in no color change if anaerobic conditions are maintained. If oxygen is present, the solution will turn from blue, to green, to orange.



Figure 4. Three color stages during the synthesis of Ti(III) metallocene compound 3.
Applications and Summary
Here, we demonstrated standard Schlenk line technique to synthesize an air-sensitive Ti(III) metallocene complex. The solvent was degassed by bubbling N2 through the liquid in a Schlenk flask. We also demonstrated how to set up a reaction under anaerobic conditions on the Schlenk line and transfer solvent anaerobically by cannula transfer as well as by syringe.
Inorganic chemists use Schlenk line technique in the synthesis of air- and water-sensitive compounds. The solvent used in the synthesis of highly-reactive materials can be prepared using the Schlenk line. Air-sensitive reactions can also be set up and worked up using a Schlenk line. The Schlenk line technique is a powerful method for air-free manipulations used in synthesis, purification (i.e.,distillation, sublimation, and crystallization), catalysis, and gas reactions. In the next module, we will demonstrate how to use a glovebox for air-free synthesis. While some air-free manipulations are easier to perform in a glovebox, there are certain situations when one cannot use a glovebox and must rely on Schlenk line technique (such as heating a reaction). Some metallocene complexes (metal compounds featuring typically two cyclopentadienyl anions (Cp, C5H5–)) exhibit catalytic properties. For example, titanocene is a catalyst used in olefin metathesis.
The Ti(III) metallocene synthesized herein can be used on the Schlenk line or in the glove box as an atmospheric test. Oxidation of the Ti(III) metallocene by O2 on the Schlenk line or in the glove box would result in a color change and would provide a visual indication that the atmosphere contains O2.
References
- Burgmayer, S. N. Use of a Titanium Metallocene as a Colorimetric Indicator for Learning Inert Atmosphere Techniques. J Chem Educ. 75, 460 (1998).
Transcription
Chemists frequently encounter air-sensitive chemical reagents and reactions, and thus have to apply special techniques when working with them.
The slightest trace of air in a chemical reaction would likely result in unwanted side products. To avoid this, first traces of oxygen are removed by purging equipment and reagents.
Then, in order to maintain an oxygen-free atmosphere, reagents are handled in a glovebox, or transferred from one closed system to another by cannulation using a Schlenk line.
This video will illustrate a procedure for purging oxygen from a reaction mixture and maintaining an air-free atmosphere in the synthesis of a Ti(III) metallocene. This will be followed by a few examples demonstrating the application of this technique.
Inorganic chemical reactions, such as the conversion of titanocene dichloride to its dimeric form and the final Ti(III) metallocene, are highly sensitive to oxygen, and therefore must be carried out in air-free conditions.
To start, in a fume hood equipped with a Schlenk line, also known as a double manifold, weigh Cp2(Ti4+)Cl2 and zinc dust into a 200 mL Schlenk flask equipped with a stir bar, labeled as “A”. Seal the flask with a greased glass stopper and secure with a rubber band. Attach Tygon tubing from the Schlenk line to flask sidearm.
Open the stopcock to vacuum and evacuate for 5 min, then close the stopcock to the flask, switch to N2, and make at least five rapid 180 ° turns before slowly opening to fill the flask with N2.
In a separate Schlenk flask labeled “B”, measure 15 mL of acetonitrile and seal with a rubber septum. Attach Tygon tubing from the Schlenk line to the flask sidearm, then evacuate the tubing for 5 min. Refill the tubing with N2.
Attach a long needle to a second Tygon tube on the Schlenk line, and purge with N2 for several minutes. Insert the purged needle into the Schlenk flask containing acetonitrile, followed by the venting needle. Bubble N2 into the solvent for 15 min, then open the flask stopcock to N2 and remove the needles.
With Schlenk flask A under N2, remove the glass stopper and replace it with a rubber septum. With the two Schlenk flasks open to N2, insert one end of the cannula into the donor flask, above the level of the solvent, and determine whether N2 is flowing through the other end. Then insert the other end of the cannula into the receiving flask containing the reagents, close the receiving flask’s stopcock, and attach a venting needle.
Lower the cannula into the solvent, and allow all of the acetonitrile to drip or slowly flow along the sides of the receiving flask. Once the addition is complete, reopen the receiving flask stopcock to N2, and remove the cannula and venting needle.
After the solvent is added, vigorously stir the reaction mixture of acetonitrile, zinc dust, and Cp2(Ti4+)Cl2 until it turns blue, indicating formation of Ti(III) metallocene complex.
If the reaction mixture remains green after 15 min, keep the stopcock open to positive N2 pressure, remove the septum and add 1-2 equivalents of zinc dust. If the mixture is still green or has turned yellow, it is likely that oxygen has entered the system, which results in further oxidation to the Ti(IV) metallocene complex.
Now you know how to use a cannula transfer, but in case this is not possible, the solvent can be added via a syringe. First, make sure both the receiving and donor flasks are open to N2.
Insert the needle fitted to a 12 mL syringe into either flask and pull only N2 into it. Remove the needle and eject the N2 into the hood.
Once the needle and syringe are purged, insert the needle into the donor flask and pull up the desired volume of solvent. Then, raise the needle slightly, bend it to an arch and pull up 1 mL of N2. Keep the needle arched and syringe pointing up and remove it from the donor flask.
Insert the arched needle into the receiving flask. Slowly add the solvent, and remove the syringe needle from receiving flask when finished.
Now that we have discussed a procedure for an air-free synthesis, let’s look at a few applications.
Cadmium selenide quantum dots are semiconductor nanocrystals composed of a cadmium selenide core and a ligand shell. These multicomponent structures are capable of manipulating electrons at the nanoscale.
The synthesis of these nanocrystals requires precise reaction conditions, especially an oxygen-free atmosphere.
Titanocene dichloride, the reagent used in this video, is an organotitanium compound commonly used in organic and organometallic synthesis. The compound itself is synthesized by reacting 2 equivalents of sodium cyclopentadiene (NaCp) with TiCl4 in anhydrous, oxygen-free THF. Titanocene dichloride is also used for the production of the Petasis reagent, which is a useful reagent applied in the conversion of esters to vinyl ethers.
Another titanocene dichloride reagent, called the Tebbe reagent, is applied to convert various carbonyl functional groups to alkenes, or also known as methylenation.
You’ve just watched JoVE’s introduction to Synthesis of a Ti(III) metallocene using the Schlenk Line Technique. You should now understand how to perform degassing as well as cannula transfer, and some of its applications. Thanks for watching!
