One-anastomosis Gastric Bypass (OAGB) in Rats
PRÉPARATION DE L'INSTRUCTEUR
concepts
PROTOCOLE ETUDIANT
All animal use conformed to the European Community guidelines and was approved by the local ethics committee (Comité d'éthique en expérimentation animale N° 121) and the Ministry of Higher Education and Research (Ref #02285.03).
1. Animals and Diets
- Rats
- Use male Wistar rats weighing 260 – 320 g, started on a high-fat diet (HFD) at 7 weeks of age.
- Diet
- Induce a diet-induced obesity, with at least 12 preoperative weeks of the HFD. Give the rats free access to water and the HFD.
NOTE: The HFD consists of a purified laboratory hypercaloric rodent diet containing 45% fat (see Table of Materials). - Ensure that the rats are obese at the time of surgery, with a weight between 600 – 800 g.
- Induce a diet-induced obesity, with at least 12 preoperative weeks of the HFD. Give the rats free access to water and the HFD.
2. Material
- Surgical equipment
- Autoclave all surgical instruments before surgery.
- Lay out all the surgical instruments needed for the procedure (see Table of Materials).
- Perform the anastomosis under magnifying glasses, using conventional and microsurgical instruments.
- Ensure that the rats are positioned on the warming plate at a temperature of 37 °C and under a heat lamp. Position the isothermal electric board as needed.
- Anesthesia and animal preparation
- Anesthetize the rat with an inhalation of 3% vol isoflurane in air for the induction of the anesthesia in the rodent box.
- Transfer the rat to the warming plate and maintain the anesthesia with the facemask at the same flow rate of 3%. Ensure that the anesthetization is complete by pinching the tail of the animal.
- Shave the anterior fur, corresponding to the laparotomy's incision zone, a ~3 cm-wide zone from the xiphoid to the animal pelvis, using an electric razor.
- Shave a 3 cm-wide zone on the back of the animal, centered on the neck of the animal. This step facilitates monopolar power conduction.
- Spray an alcohol-based disinfectant solution over the operation site.
3. Surgery
- Laparotomy
- Apply alcohol-based local antiseptic on the shaved skin.
- Open the abdomen with a midline cutaneous incision from the xiphoid process of about 3 cm in length with a scalpel. Open the fascia and the peritoneum along the linea alba with a monopolar scalpel.
- Ensure that the peritoneal cavity is opened on the entire length of the cutaneous incision.
Expose the peritoneal cavity with a self-retractor.
- Greater curve and hiatal dissection
- Dissect the greater gastric curve from the junction between the glandular and nonglandular stomach to the left side of the abdominal esophagus with surgical scissors.
- Progressively coagulate the gastrosplenic vessels with the monopolar scalpel, in order to fully liberate the great gastric curve to the His angle. Then expose the hiatal area by sectioning of thin ligaments.
- Dissect the right side of the abdominal esophagus, preserving the left gastric artery and vagal nerve, using a manual dissector.
- Gastric pouch confection
- Nonglandular stomach ablation
- Apply a 35 mm staple gun (see Table of Materials) at the junction between the glandular and nonglandular stomach to the left side of the gastroesophageal junction. Ensure that the nonglandular stomach is on the right side of the stapler.
- Fire the gun and then remove the resected nonglandular stomach.
- Gastric pouch
- Apply a second stapler using a thoraco-abdominal (TA) device (3 – 3.5 mm) (see Table of Materials), parallel to the first stapling, on the right side of the gastroesophageal junction to exclude the antrum and a part of the body of the stomach.
- Ensure that the result of the two staple lines is a gastric pouch about 0.5 cm wide.
- Nonglandular stomach ablation
- OAGB
- Small bowel measurement
- Locate the duodenojejunal angle. The proximal jejunum is the small bowel portion located under the medial portion of the colon; it is the only nonmobile part of the small bowel in the rat.
- With a 5 cm-long tape, manually measure 35 cm from the duodenojejunal angle. Then perform a punctiform enterotomy on the antimesenteric side of the jejunum.
- Gastrojejunal anastomosis
- Perform a punctiform gastrotomy with the monopolar scalpel on the greater curve side of the gastric pouch, precisely between the two previous staple lines. If the gastric pouch is not totally empty, perform gentle manual pressure on the gastric pouch to empty it.
- Perform an end-to-side gastrojejunal anastomosis with nontraumatic forceps and a small needle-holder.
- Start the suture with two extremity stitches of 7-0 isotactic polypropylene (see Table of Materials), suturing the two extremities of the gastrotomy with the extremities of the enterotomy.
- Then, with the same threads used for the two extremity stitches, perform two running sutures on each side of the anastomosis. If needed, return the anastomosis to facilitate the maneuver. Ensure the quality of the anastomosis by placing close stitches, at least seven or eight on each side of the anastomosis.
- Take 1 mm-wide serosa in every stitch. Avoid taking any mucosa in the stitch in order to gently bury the enteric and gastric mucosa inside the suture.
NOTE: The result of this is a terminolateral gastrojejunal anastomosis with a biliopancreatic limb on the left and an alimentary (AL)/CL on the right. Ensure that the anastomosis is permeable with gentle pressure on the gastric pouch.
- Small bowel measurement
- Closure and immediate care
- Rinse the peritoneal cavity with 0.9% NaCl solution and manually replace the abdominal organs into their physiological positions.
- Close the abdominal wall with one running suture of braided polyester suture 2-0 (see Table of Materials) with a classical needle-holder.
- Close the skin with one running suture of polyglactin 910 4-0 (see Table of Materials).
- Perform two parietal injections of lidocaine 1% on each side of the laparotomy. Administer a total dose of 1 mL/kg of lidocaine 1% into the rectus abdominal muscle aponeurosis.
4. Postoperative Care
- Perform an intramuscular injection of 20,000 units/kg penicillin G.
- Perform a subcutaneous injection of 10 mL of isotonic polyionic solution potassium (2 g of KCl/L, 4 g of NaCl/L, and 50 g of glucose/L). In this injection, it is possible to mix with suitable analgesics such as buprenorphine (0.1mg/kg) and meloxicam (2mg/kg).
- Place the rats in individual cages warmed up by a lamp until they are fully awake.
- Frequently inspect the animals in order to administer postoperative treatment with suitable analgesia and hydration (isotonic polyionic solution).
- Inject suitable analgesic, buprenorphine (0.1 mg/kg) and meloxicam (2 mg/kg), subcutaneously from the immediate postoperative period till day 4, every 12 h.
- Keep the rats with free access to water during day 1. On day 2/3, give them free access to a liquid diet. On day 4, give them ad libitum access to a normal diet.
One-anastomosis Gastric Bypass (OAGB) in Rats
Learning Objectives
This protocol is reproducible and mimics OAGB as practiced on humans. The safety of this protocol is supported with published data23,24 with a low mortality rate between 12% and 25%. Different surgeons in the research team with a similar mortality rate have practiced this protocol.
Nonetheless, learning rodent surgery is complex, and the use of magnifying glasses is mandatory. Indeed, the critical step of the construction of a microscopic anastomosis requires a learning period from around 15 to 20 operations to reach an optimal dexterity. A surgical defect can lead to an anastomotic leakage of a gastric obstruction, which is the main death cause in our experience. Another key to success is the postoperative nutrition; indeed, an early postoperative solid nutrition can be dangerous. This protocol also provides a precise postoperative nutrition protocol, which has been demonstrated to be safe. The concept of this surgery on humans and on rats is illustrated in Figure 1. A result of the operation after sacrifice is provided in Figure 2.
Results obtained on weight loss after operation on obese rats were quite similar to those observed in humans, with a body weight loss of about 20%, stable after the operation. The results are detailed in Figure 3. Better insulin sensitivity and glucose tolerance were observed as soon as 2 weeks after the surgery, as assayed by an oral glucose test (OGTT) and an insulin tolerance test (ITT). These results are detailed in Figure 4. HFD-induced obesity has been described to promote insulin resistance and glucose intolerance in Wistar rats25; comparable data have been gathered in our unit (unpublished). Wistar rats are not a model of type 2 diabetes.
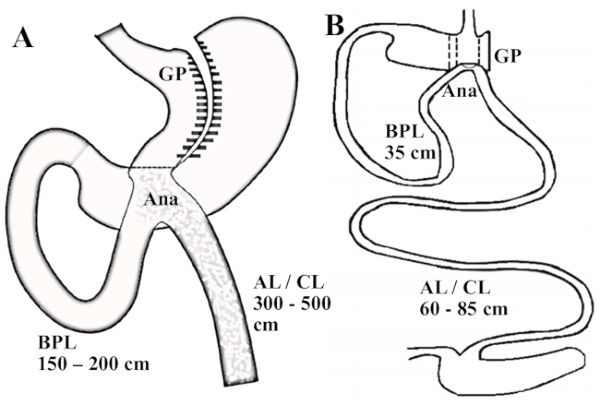
Figure 1: Illustration of OAGB on humans and on this rat model. (A) Scheme of an OAGB in humans; the gastric pouch is long and tubular and the BPL measures between 150 and 200 cm. (B) Rat model of an OAGB. The ratio of the BPL and the AL/CL reproduce the one practiced for human surgery; the BPL measures 35 cm. Abbreviations: GP = gastric pouch; Ana = anastomosis; AL = alimentary limb; CL = common limb. Please click here to view a larger version of this figure.
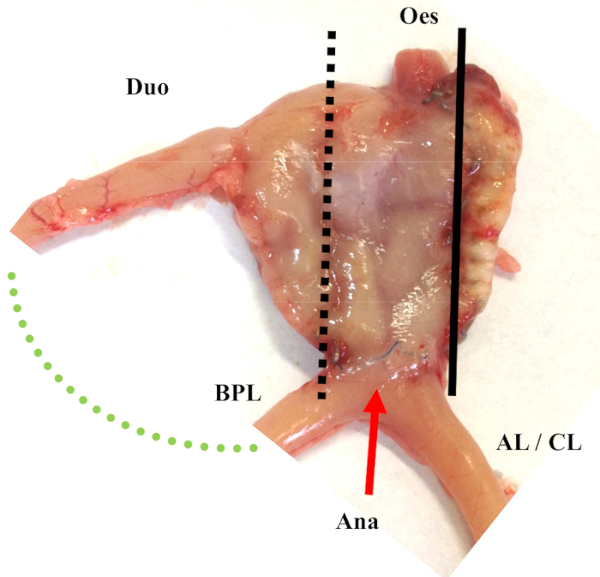
Figure 2: Illustration of OAGB on rats after sacrifice. Abbreviations: GP = gastric pouch; Ana = anastomosis; AL = alimentary limb; CL = common limb; Oes = Esophagus; Duo = duodenum Please click here to view a larger version of this figure.
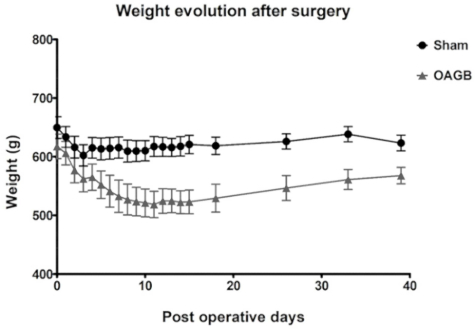
Figure 3: Postoperative weight evolution (median ± SD) after OAGB and sham surgery. The sham surgery consisted of a laparotomy and a pinch on the great gastric curve with non-traumatic forceps. Please click here to view a larger version of this figure.
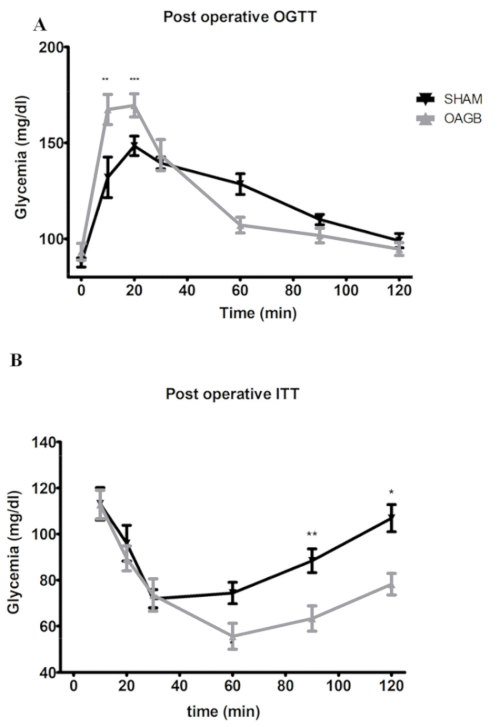
Figure 4: Glucose metabolism after OAGB and sham surgery; post-operative OGTT and ITT. (A) Post-operative OGTT. OAGB and sham rats were made to fast before being subjected to an oral glucose tolerance test (OGTT: 1 g/kg of body weight). (B) Post-operative ITT: an injection of 1 U insulin/kg of body weight. Rats were made to fast 4 h before receiving the insulin injection. Blood was harvested in the tail vein at t = 0 and at 10, 20, 30, 60, and 120 min after the insulin or glucose administration. Glucose was measured using a blood glucose meter. Glycemia was expressed in mg/dL ± standard deviation. Abbreviations:OGTT = oral glucose tolerance test; ITT = insulin tolerance test; BW = body weight. * p < 0.05, ** p < 0.01, *** p < 0.001; after two-way ANOVA statistical test. The sham surgery consisted of a laparotomy with a nontraumatic pinch of the great gastric curve using non-traumatic forceps. All data are presented as mean ± SD. Please click here to view a larger version of this figure.
List of Materials
| High-Fat Diet, 45 % fat | Genestil, Royaucourt, France | C 1090-45 | http://www.genestil.com/ |
| Metzenbaum Scissors | World Precision instrument, Sarasota, US | 501739 | https://www.wpi-europe.com |
| Dumont Tweezers | World Precision instrument, Sarasota, US | 14098 | https://www.wpi-europe.com |
| Iris Forceps | World Precision instrument, Sarasota, US | 15915 | https://www.wpi-europe.com |
| Iris Scissors 10cm | World Precision instrument, Sarasota, US | 14218 | https://www.wpi-europe.com |
| Needle Holder | World Precision instrument, Sarasota, US | 14110 | https://www.wpi-europe.com |
| Alm Self Retaining Retractor | World Precision instrument, Sarasota, US | 14240 | https://www.wpi-europe.com |
| Baby Mixter Hemostatic Forceps, Right Angle | World Precision instrument, Sarasota, US | 501240 | https://www.wpi-europe.com |
| Non woven sterile swabs | LCH Medical Products, Paris, France | SN40-0755 | www.lch-medical.com |
| Galilean Binocular Loupe 2.5x | World Precision instrument, Sarasota, US | 504056 | https://www.wpi-europe.com |
| Digestive thread: Isotactic polypropylene, monofil Prolene 7-0 | Ethicon, Issy les Moulineaux, France | EH7813E | www.ethicon.com |
| Parietal thread : Coated Braided Polyester Ti-Cron 2-0 | Covidien, Mannsfield, MA, USA | 3003-52 | www.covidien.com |
| Cutaneous thread : Polyglactin, VICRYL RAPIDE 4-0 | Ethicon, Issy les Moulineaux, France | VR3100 | www.ethicon.com |
| ETS-Flex 35- mm staple gun | Ethicon, Issy les Moulineaux, France | ATS45 | www.ethicon.com |
| Proximate Reloadable Staplers | Ethicon, Issy les Moulineaux, France | XR30V | www.ethicon.com |
| TA-DST 30mm-3.5mm | Ethicon, Issy les Moulineaux, France | TX30B | www.ethicon.com |
| Alcoholic 5% Betadine | MEDA Pharma, Merignac, France | 41085 | www.medapharma.fr |
| LIDOCAINE AGUETTANT 20 mg/ml | LABORATOIRE AGUETTANT, Lyon, France | 3400940000000.0 | https://www.aguettant.fr |
| Penicillin G 5MUI | Panpharma, Luitre, France | 3.40E+12 | https://www.panpharma.eu/fr |
| Bionolyte G5 ; Sodium chloride 0.4%, glucose 5%, potassium chloride 0.2%) | Baxter, Maurepas, France | ||
| Liquid diet | Genestil, Royaucourt, France | C-0200T | http://www.genestil.com/ |
| Isoflurane 100% | Centravet, Plancoet, France | ISO007 | http://cooperative.centravet.net |
Lab Prep
The goal of this protocol is to set up a preclinical model of bariatric surgery and, more specifically, OAGB in obese rats. Based on this preclinical model, longitudinal studies can be carried out to provide an improved understanding of the mechanisms underlying the outcomes seen after bariatric surgery in humans. For this purpose, rats are operated on through a laparotomy under general anesthesia with isoflurane. First, the surgeon creates a long and tubular gastric pouch: after greater curve and hiatal dissection, the nonglandular stomach is stapled and removed. Then, the remaining stomach is also stapled in order to create a gastric tube and exclude the antrum of the stomach. After that, the surgeon performs a single end-to-side gastrojejunostomy 35 cm from the duodenojejunal angle. This limb length has been chosen in order to reproduce the same ratio between the biliopancreatic limb (BPL) and common limb (CL) length as in human bariatric surgery. The operation ends by aponeurotic and cutaneous closure. The early postoperative management consists of subcutaneous hydration, an intramuscular prophylactic antibiotic injection, a parietal injection of xylocaine, the administration of painkillers, and a progressive reintroduction of diet.
The goal of this protocol is to set up a preclinical model of bariatric surgery and, more specifically, OAGB in obese rats. Based on this preclinical model, longitudinal studies can be carried out to provide an improved understanding of the mechanisms underlying the outcomes seen after bariatric surgery in humans. For this purpose, rats are operated on through a laparotomy under general anesthesia with isoflurane. First, the surgeon creates a long and tubular gastric pouch: after greater curve and hiatal dissection, the nonglandular stomach is stapled and removed. Then, the remaining stomach is also stapled in order to create a gastric tube and exclude the antrum of the stomach. After that, the surgeon performs a single end-to-side gastrojejunostomy 35 cm from the duodenojejunal angle. This limb length has been chosen in order to reproduce the same ratio between the biliopancreatic limb (BPL) and common limb (CL) length as in human bariatric surgery. The operation ends by aponeurotic and cutaneous closure. The early postoperative management consists of subcutaneous hydration, an intramuscular prophylactic antibiotic injection, a parietal injection of xylocaine, the administration of painkillers, and a progressive reintroduction of diet.
Procédure
The goal of this protocol is to set up a preclinical model of bariatric surgery and, more specifically, OAGB in obese rats. Based on this preclinical model, longitudinal studies can be carried out to provide an improved understanding of the mechanisms underlying the outcomes seen after bariatric surgery in humans. For this purpose, rats are operated on through a laparotomy under general anesthesia with isoflurane. First, the surgeon creates a long and tubular gastric pouch: after greater curve and hiatal dissection, the nonglandular stomach is stapled and removed. Then, the remaining stomach is also stapled in order to create a gastric tube and exclude the antrum of the stomach. After that, the surgeon performs a single end-to-side gastrojejunostomy 35 cm from the duodenojejunal angle. This limb length has been chosen in order to reproduce the same ratio between the biliopancreatic limb (BPL) and common limb (CL) length as in human bariatric surgery. The operation ends by aponeurotic and cutaneous closure. The early postoperative management consists of subcutaneous hydration, an intramuscular prophylactic antibiotic injection, a parietal injection of xylocaine, the administration of painkillers, and a progressive reintroduction of diet.
