A Hepatocellular Cancer Patient-Derived Organoid Xenograft Model to Investigate Impact of Liver Regeneration on Tumor Growth
Summary
Organoids generated from patient tumors are orthotopically injected into the mouse liver. The resection of non-tumorous liver tissue leads to a regenerative environment in the liver tissue where the tumor is located.
Abstract
Recurrence poses a notable challenge after hepatocellular carcinoma (HCC) treatment, impacting more than 70% of patients who undergo surgical resection. Recurrence stems from undetected micro-metastasis or de novo cancer, potentially triggered by postsurgical liver regeneration. Prior research employed HCC cell lines in orthotopic models to study the impact of liver regeneration, but their limited validity prompted the need for a more representative model. Here, we introduce a novel approach utilizing patient-derived HCC organoids to investigate the influence of liver regeneration on HCC.
Patient tumor tissues are processed to create tumor organoids, embedded in a three-dimensional basement membrane matrix, and cultured in a liver-specific medium. One million organoids are injected into the right superior lobe (RSL) of immunodeficient mice, confirming macroscopic tumor growth through sonography. The intervention group undergoes resection of the left lateral lobe (LLL) (30% of total liver volume) or additionally, the middle lobe (ML) (65% of total liver volume) to induce liver regeneration within the tumor site. The control group experiences re-laparotomy without liver tissue resection. After 2 weeks, both groups undergo tumor and normal tissue explantation.
In conclusion, this patient-derived HCC organoid model offers a robust platform to investigate the impact of liver regeneration post-cancer resection. Its multi-cellular composition, genetic diversity, and prolonged culture capabilities make it an invaluable tool for studying HCC recurrence mechanisms and potential interventions.
Introduction
Recurrence following hepatocellular carcinoma (HCC) treatment is a significant challenge, impacting over 70% of patients undergoing surgical resection1,2,3. This recurrence may result from undetected micro-metastasis (multicentric tumor) or the development of de novo cancer4. Both clinical and experimental investigations propose that the liver regeneration process triggered by surgical resection could activate latent micro-metastases, contributing to tumor recurrence.
Further research is necessary to compare the gene and protein expression in normal and malignant liver cells within a regenerative liver environment. In addition to elucidating the mechanisms underlying recurrence, identifying targetable signaling pathways specific to cancer cells holds the potential for substantial therapeutic advancements. This approach aims to reconcile the seemingly contradictory notions of an anti-tumor microenvironment and favorable conditions for liver regeneration.
Previous models have used orthotopic injection of HCC cell lines to investigate the impact of liver regeneration5. This pioneering model employs patient-derived HCC organoids, offering several advantages over traditional cell lines. Organoids maintain diverse cell populations, mirroring the model template's structure and function, unlike cell lines and spheroids. Their genetic diversity enhances representativeness. Additionally, organoids provide stability for extended culture, enabling prolonged intervention studies. We recommend RSL injection due to its superior regeneration potential compared to the right inferior lobe (RIL). The method described here provides a platform using cancer organoids with pronounced advantages compared to cancer cell lines to investigate the aforementioned research question.
Protocol
Liver samples used for organoid generation were obtained from patients operated at the University Hospital of Basel (USB) following written informed consent under approval by the Ethical Committee of Northwestern and Central Switzerland (BASEC number 2019-02118). All mouse experiments were approved by and performed in accordance with the guidelines and regulations of the Animal Care Committee of Kanton Basel-Stadt, Switzerland (3123-33896). All mice were of the non-obese diabetic (NOD) SCID gamma mouse strain and therefore genetically modified to be immunosuppressed; only male animals were used. See Table 1 and Table of Materials for details related to solutions, reagents, materials, and instruments used in the protocol. See Figure 1 for an overview of the experiment.
1. Mouse husbandry
- Breed and maintain mice in the animal facility under specific pathogen-free conditions on a 12 h day and 12 h night schedule with ad libitum access to food and drinking water.
NOTE: Implantation of the organoids is performed at an age of 6-8 weeks.
2. Patient and clinical samples
- Place 12-, 24-, and 6-well plates in the incubator at 37 °C before starting. If possible, do this 1 day in advance.
- Collect the tissue within 1 h after resection in RPMI 1640 medium and transport it to the laboratory to start the dissociation process immediately. Place the tissue in a 10 cm Petri dish (prewarmed dish) and add a sufficient amount of sterile saline to avoid the tissue drying out.
- Liver cancer tissue dissociation for the generation of organoids
- Mince the tissue into small fragments of 1-2 mm2 using scalpels. Use additional tissue forceps to stabilize the tissue in case cutting is difficult.
- Place the tissue pieces in a 15 mL tube with 5 mL of dissociation buffer (physiological pH). Place at 37 °C (warming cabinet/incubator) in a rotation device.
- Mix by pipetting every 10 min and leave for approximately 1-1.5 h, repeating the mixing process every 10 min.
NOTE: Mixing may require cutting the end of the pipette to increase the orifice diameter, thereby preventing oversize tissue from getting stuck in the pipette tip. - Collect 10 µL of cell-dissociation buffer mixture from step 2.2.3 in a 0.5 mL plastic tube, add 10 µL of Trypan Blue, and use the automated cell counter to assess cell viability and cell clusters. If clusters are visible, repeat the mixing process (step 3.3.); otherwise, proceed with the next step.
- Take a 50 mL tube, place a 100 µm cell strainer on the top, and pass the mix through it. With the plunger of a 5 mL syringe, smash all remaining tissue fragments through the filter. Wash everything with 5 mL of Ad-DF+++ and be sure to collect the drop from the bottom of the filter.
- Transfer the filtrate to a 15 mL conical tube. Centrifuge for 15 min at 300 × g.
- Aspirate the supernatant carefully, taking care to not touch the pellet. Add 5 mL of Red Blood cell lysis buffer. Incubate for 5-10 min at room temperature.
- Centrifuge for 5 min at 300 × g. Remove the supernatant and resuspend in 1 mL of advanced medium.
- Counting cells and assessing cell viability
- Add 10 µL of Trypan blue to a 1.5 mL tube and mix with 10 µL of the cell suspension. Place 10 µL of the mix on the counting slides and count the cells.
- Separate the necessary number of cells for the organoid generation and centrifuge for 5 min at 300 × g. Generally, seed 80,000 cells per dome and 20 µL of basement membrane matrix per dome (10 drops per well per 6-well plate).
- If any excess cells are remaining in suspension, suspend the pellet in Cell Suspension Freezing medium (10% DMSO in FBS), transfer it into a cryotube, and place it in a freezing container. Immediately transfer the tube to a -80 °C freezer overnight. The next day, transfer into liquid nitrogen until needed.
- Plating for organoid generation
- Remove the supernatant. Place the tube on ice and resuspend the pellet with a basement membrane matrix.
- Take the prewarmed plate out of the incubator (prewarmed overnight). Make small drops in the plate (20 µL of basement membrane matrix per dome [10 drops per well]). Invert the plate and leave the plate for 5 min in the flow cabinet.
- After 5 min, transfer the plate to the 37 °C incubator and leave the plate for ~20 min, allowing the matrix to solidify. Add 2 mL of organoid culture medium6,7.
- Maintenance of organoid culture
- Replace the medium every 2 days. Once the organoids are confluent in the basement membrane matrix dome and ready for expansion, calculate the splitting ratio e.g., 2x or 3x accordingly.
NOTE: Record this information; it will be needed in the next steps. - Remove the medium from the corner of the well using a P1000 tip, taking care not to touch the matrix domes.
- Add 100 µL of 0.25% trypsin-EDTA per dome, scrape it, collect it into a 15 mL tube, and add again a small amount of trypsin into the well to collect everything. Pipette it ~10x.
- Incubate for 3 min in the 37 °C water bath and pipette 10x to dissociate the cells with a P1000 tip. Repeat this step every 3 min for a maximum of 10 min.
- Add at least the same volume of Dulbecco's modified Eagle's medium (DMEM)/10% Fetal bovine serum (FBS)/1% Penicillin-Streptomycin (Pen-Strep) medium and mix up and down slowly 2x. Fill up with DPBS.
- Centrifuge at 300 × g for 5 min and carefully remove the supernatant. Resuspend according to the splitting ratio decided based on the organoid confluency e.g., 2x or 3x the initial amount of basement membrane matrix.
- Take the prewarmed plate out of the incubator (prewarmed overnight). Make small drops in the plate (20 µL of basement membrane matrix per dome [10 drops per well]). Invert the plate and leave the plate for 5 min in the flow cabinet.
- After 5 min, transfer the plate to the 37 °C incubator. Leave the plate for ~20 min, allowing the matrix to solidify. Add 2 mL of organoid culture medium.
- Replace the medium every 2 days. Once the organoids are confluent in the basement membrane matrix dome and ready for expansion, calculate the splitting ratio e.g., 2x or 3x accordingly.
- Preparation of organoids for making a formalin-fixed paraffin-embedded (FFPE) block
- Remove the medium from the corner of the well using a P1000 tip, taking care not to touch the matrix domes. Add 200 µL of Dispase (2 mg/mL) and disrupt the drops with a P1000 pipette tip. Leave in the incubator for 80 min.
- Add 2 mL of Dulbecco's phosphate-buffered saline (DPBS) to the well, transfer it to a 15 mL conical tube, and fill it up with DPBS. Centrifuge at 300 × g for 5 min, aspirate the supernatant, and resuspend the pellet in 1 mL of 4% paraformaldehyde. Refrigerate for at least 1 h.
- Centrifuge at 300 × g for 5 min, aspirate the paraformaldehyde, and resuspend the pellet with the specimen-processing gel to create a drop. Wait for 40 min for the drops to solidify. Place the gel drop containing the cells in a histology cassette. Move the cassette to dehydrate and afterward embed it in paraffin for cutting and H/E staining8,9.
- Preparation of organoids for injection
- Take the plate containing the growing organoid out of the incubator and place it in the flow cabinet.
- Follow steps 2.5.2 to 2.5.4.
- Add 2 mL of DMEM and mix.
- Add 10 µL of Trypan blue to a 1.5 mL tube and mix with 10 µL of cell suspension.
- Place 10 µL of the mix on the counting slides and count the cells. Calculate the average cell count of both chambers. If the difference is too big between the two chambers, consider redoing the count.
- Calculate the volume of cell suspension required for 106 cells and dispense this volume to 1.5 mL tubes. Centrifuge at 300 × g for 5 min and place the tubes on ice.
- Carefully remove the supernatant and add 5 µL of DMEM and 5 µL of basement membrane matrix. Carefully mix the suspension by pipetting while keeping the suspension cold during the process.
- Place the 1.5 mL tubes containing the organoids on ice during the transport to the mice and proceed with injection.
3. Injection of organoid into mouse
- Perioperative steps
- Remove the experimental animal from the main cage and place it into an isolated, fresh cage with water.
- Anesthetize the mouse with 1.5% to 2.5% Isoflurane.
- Perform a subcutaneous injection into the neck with 10 µL of Buprenorphine (0.3 mg/mL) 30 min prior to the start of the surgery.
- Immediately prior to the start of the surgery, carefully paint the eyes of animals with Carbomerum (e.g., Lacrinorm) to avoid any dryness of the eyes during surgery. Do not touch the cornea during the application to avoid any erosion of the cornea.
- Place the animals onto a clean and sterile surface and handle them with fresh plastic gloves, hairnet, and a special laboratory working coat used only in the mouse facility.
- Use two heat-emitting headlights close to the animal to provide a sufficient view. If there is any doubt or concern about hypothermia during the surgery, use another heating lamp or heating mat.
- Laparotomy
- Place the anesthetized mouse in the supine position, extend the four extremities, and fix them with tape. Remove the fur by applying a depilatory cream and use a wet tissue to remove the excess cream and fur.
NOTE: Depilation works best if a cotton swab is used to massage the cream into the fur against the grain. - Localize the linea alba, which is the target for the laparotomy. Identify the superior and inferior epigastric arteries as well. Take care not to damage the vessels to avoid injuries.
- Make an incision along the linea alba with forceps and microsurgical scissors from the lower abdomen to the xiphoid cartilage.
- Insert the retractors and adjust the direction of traction for optimal exposure of the upper abdomen (Figure 2A,B).
NOTE: We use bent paper clips as retractors with attached rubber bands for fixation.
- Place the anesthetized mouse in the supine position, extend the four extremities, and fix them with tape. Remove the fur by applying a depilatory cream and use a wet tissue to remove the excess cream and fur.
- Injection of organoids
- Place a clean napkin right next to the incision and put some drops of saline onto it. Exteriorize the small intestine and the cecum and place it on the wet napkin.
- Expose the right superior lobe (RSL) until an injection is possible.
- Prepare the Hamilton syringe, rinse it with water, and place a mark 2-3 mm from the tip as a reference for the injection.
- Draw up the syringe with the prepared organoid mixture and inject the needle until the mark. Slowly inject the organoids in a steady position; do not move the needle during this time.
- Wait for 15-20 s to avoid backflow of the mixture. Remove the needle and put some pressure with a cotton swab on the injection site. Check the abdomen for a rupture of the liver lobe and backflow. Rinse the abdomen with 0.5 mL of sterile saline and close the abdomen with a running suture using a resorbable suture.
4. Minor resection (30% of liver tissue)
- Perform the laparotomy as stated above in step 3.2.
- Use moist cotton swabs and push the ML cranially to create more space in the surgical field of the LLL.
- Flip the LLL cranially and expose the caudate lobe (CL) and ligament, which is habitually present between the CL and LLL (Figure 2C).
- Cut the ligament completely and insert the ligature (non-resorbable thin thread, e.g. Prolene 5-0) below the LLL. To ensure that it stays in this position, moisten the ligature and move it towards the right upper quadrant.
- Flip the LLL caudally. Guide the left free end of the ligature around the left tip of the LLL. Guide the right free end of the ligature around the right tip of the LLL. Apply caudal traction on the LLL with a cotton swab and ensure the correct and central positioning of the ligature.
- Tie a double half knot. Secure the knot with a second half knot tied in the other direction without fully tightening it. Tighten the knots a little bit, check the correct placement of the ligature, and reposition it to guarantee good positioning.
- Tighten the knot completely by holding the cranial free end in position with blunt-tipped forceps and pull the caudal free end with another pair of forceps (Figure 2D). Secure the knot with a second half knot tied in the opposite direction.
- Resect the liver lobe. Cut close to the ligature without cutting it.
5. Major resection (65% of liver tissue)
- Perform the minor resection of the LLL as stated above (steps 3.4.1-3.4.8).
- Transect the falciform ligament to mobilize the ML. Flip the lobe cranially. Insert the ligature below the lobe and flip the lobe caudally.
- Loop the free ends around the lobule. Repeat placement of the ligature in case of misplacement during manipulation. Once the ligature is positioned correctly, place a double half knot, tie it slowly, and reposition the ligature if needed.
- Tie a second half knot in the opposite direction and tighten it completely. Add a third half knot in the opposite direction.
- Resect the lobe.
- Flush the abdomen with sterile saline solution and place the intestines in their anatomical place.
6. Closure of the abdomen
- Grab the skin and peritoneum with forceps and make a stitch with a resorbable suture (e.g., Monocryl 5-0) on the top end of the incision line. Be sure to exit on the peritoneal side.
- Perform the second stitch on the counterpart of the incision, exiting on the skin side. Tie the knot tightly while leaving the free end without the needle short and the end with the needle long. Cut the short end to a length of 2 mm.
- Continue the running suture and finish with a knot at the bottom end of the incision.
- Clean the abdomen of the mouse of any remaining blood.
7. Postoperative care
- After the closure of the abdomen, inject another 10 µL of Buprenorphine (0.3 mg/mL) subcutaneously into the neck. Place the animals in the previously used, isolated cage, partially placed on a heating mat. Provide some easily accessible food on the floor of the cage.
NOTE: Do not leave the animal unattended until it has regained consciousness. - Carry out postoperative controls at 2 h and 6 h after the surgery. Closely observe the behavior with special attention to any sign of pain; administer another 10 µL of Bupivacaine if needed.
- Once the mouse is fully recovered, but not prior to 6 h after surgery and not later than 24 h after surgery, re-introduce it to the main cage. Closely monitor the behavior of all the involved animals regarding any aggression or other suspicious behavior. When in doubt of any postoperative weakness of the animal, leave it isolated overnight to recover until final re-introduction.
- After the final postoperative control 1 day after the surgery, proceed with the regular weighing of the animals.
8. Monitoring tumor growth
- Immobilize the mice using isoflurane anesthesia and use sonography to visualize tumor implantation beginning 2 weeks after injection10.
- Euthanize the mice using CO2.
Representative Results
We looked at the contribution of the single liver lobes to the total liver volume and found that the LLL represents 33% of the total liver volume. The ML represents 32% of the total liver volume, the RSL 13%, the RIL 10%, and the CL 10% (Table 2). The boxplots show that the relative contribution of the liver lobes is comparable between the different mice of the same strain (Figure 3B). Hence, a resection of the LLL results in a resection of 35% of the total liver volume, and a resection of the LLL and ML results in a resection of 66% of the total liver volume. As resection of the independent liver lobes to measure weight for contribution to total liver volume occurred postmortem, the value measured is most likely a slight overrepresentation of actual resection weight that can be achieved when performing surgery on a living mouse. This is because the ligature is placed slightly decentral (especially when performing a resection of the ML) to prevent obstruction of the vena cava.
Further experiments show that resection has an impact on the weight of the reference lobe, which is the RIL. The mean weight of the RIL was 0.49% of the total body weight in those mice that did not receive a resection. After performing a resection of the LLL (= Minor Resection), the mean weight increased to 0.82% of the total body weight; this difference was significant (p < 0.0001). After performing a resection of the LLL and ML (= Major Resection), the weight of the RIL increased to a mean of 0.99% of the total body weight. The difference between no resection and a large resection (LLL + ML resection) was also significant (p < 0.0001). However, the difference between the resection of the LLL and the combined LLL + ML resection was not significant (Figure 3C).
In a further experiment, we looked at the increase in the weight of the different liver lobules in addition to the increase of the RIL weight after a minor or major resection. This is of pronounced interest as we chose to inject the tumor organoids into the RSL. After performing a major resection, we saw a pronounced increase in the weight of the RSL in relation to the body weight of the mouse compared to the RIL (Figure 3D,E). The mean weight of the RSL was 1.73% of the mouse body weight, which was significantly larger than the mean weight of 0.99% of the mouse body weight for the RIL (p = 0.0053).
Finally, we report the histology and immunohistochemical staining of two separate HCC tumors and the subsequent patient-derived organoid (PDO) line and resulting mouse tumor. The first case depicted in Figure 4A shows a poorly differentiated HCC with neuroendocrine differentiation. The tumor stage after operation was pT2 pN0 (0/15) L0 V1 Pn1 R0. The patient had recurrent disease 3 months after the operation in the remaining liver tissue and distant metastasis in the lung. H/E and immunohistochemistry staining (Figure 4A) typical for HCC showed that the PDO resembles the morphological features of the original patient tumor. The same is also true for the mouse tumor: it resembles the PDO and the original patient tumor. The second case depicted in Figure 4B shows a poorly differentiated HCC (Grade 3 Edmondson). The original Tumor grade after resection was pT4 pN0 (0/5) L1 V1 Pn0 G3 R0. Again, the staining shows that the PDO resembles the original patient's tumor, and the mouse tumor resembles the patient's tumor and the PDO (Figure 4B). Figure 5A-E show PDO development from passages 1 to 18.

Figure 1: Flowchart of the experimental setup. In the first step, the organoids will be injected into the right superior lobe via laparotomy. Until the second step, monitoring of the tumor growth is done with sonography at an interval of 2 weeks. In the second step, mice will be randomized into three groups: control (no resection), 30% resection, and 65% resection. After resection, the tumor is sonographically monitored until it reaches the termination criteria. Please click here to view a larger version of this figure.

Figure 2: Liver lobules and positioning of retractors and ligature for lobe resection. (A) Positioning of retractors that improves the exposure of the cranial abdomen where the liver lobules are located (ventral-dorsal view). (B) Positioning of retractors (caudaln-cranial view). (C) Schematic of liver lobules located in the cranial abdomen as seen during surgery; the ligament between the LLL and the caudate lobe is exposed when the LLL is flipped cranially. It is important to dissect this ligament to prohibit bleeding through tearing forces when left in place and to ensure good central positioning of ligature for removal of the LLL. (D) Positioning of ligature for resection of the LLL (it is important to close the ligature by pulling the caudal free end of the ligature and not both free ends equally in opposite directions. If both ends are pulled equally, there is a risk of perforating the diaphragm with the cranial forceps. It also helps to use blunt forceps to hold the cranial free end in position. Abbreviations: LML = Left Middle Lobe; RML = right middle lobe; RIL = Right Inferior Lobe; RSL = Right Superior Lobe; LLL = Left Lateral Lobe; CL = caudate lobe; Lig = ligament. Please click here to view a larger version of this figure.

Figure 3: Percentage of total liver volume contributed by liver lobules and lobe regeneration after resection. (A) Schematic of liver lobules showing the percentage of total liver volume contributed by each liver lobule. (B) Bar plots showing the contribution of liver lobules to total liver volume, (C) Results showing the increase in weight of the reference lobe (Right Inferior Lobe) after 30% and 65% resection. (D) Regeneration in the RSL after major resection (the regeneration in the RSL is more pronounced compared to the right inferior lobe. Results demonstrate the difference in weight increase between Right superior and Right inferior after 65% resection). (E) Demonstration of size difference between Right superior and Right inferior lobules. Top row corresponds to the Right superior lobe and the bottom row corresponds to the Right inferior lobe. Abbreviations: ML = Middle Lobe; RSL = Right Superior Lobe; RIL = Right Inferior Lobe; LLL = Left Lateral Lobe; AC = Anterior caudate lobe; PC = and Posterior Caudate Lobes; IVC = Inferior Vena Cava. Please click here to view a larger version of this figure.
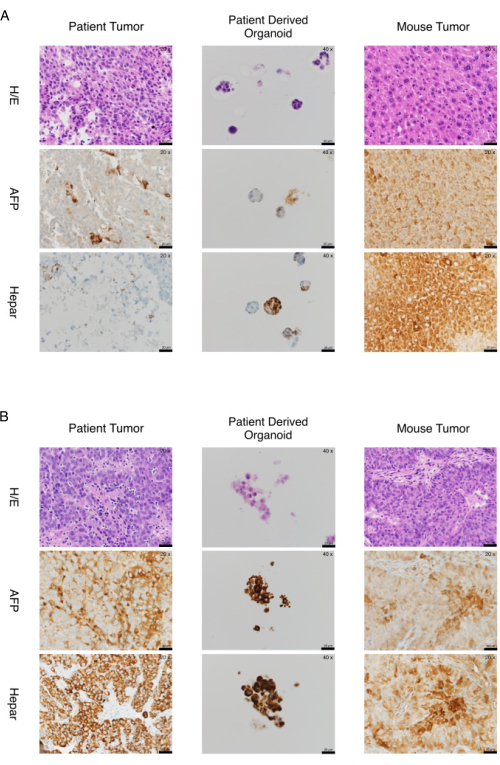
Figure 4: Two cases of marker expression in organoids and tumors from patient-derived xenografts. (A) Expression of markers in organoids and tumors from patient-derived xenograft-a case. The organoids and tumors from the xenograft recapitulate the expression of immunohistochemistry markers of the original tissue. Histology and immunohistochemical staining AFP and Hepar of Case p151-poorly differentiated HCC (Edmondson Grade III) with neuroendocrine differentiation. Microphotographs of patient tumor (20x), organoids (40x), and tumor (20x) obtained from the mouse model are shown in the left, middle, and right columns, respectively. Hematoxylin/Eosin staining and immunohistochemical staining for AFP and Hepar are shown in the top, middle, and bottom rows, respectively. (B) Expression of markers in organoids and tumors from patient-derived xenograft-a second case. The organoids and tumors from the xenograft recapitulate the expression of immunohistochemistry markers of the original tissue. Histology and immunohistochemical staining AFP and Hepar of Case p220-poorly differentiated HCC (Edmondson Grade III). Microphotographs of patient tumor (20x), organoids (40x), and tumor (20x) obtained from the mouse model are shown in the left, middle, and right columns, respectively. Hematoxylin/Eosin staining and immunohistochemical staining for AFP and Hepar are shown in the top, middle, and bottom rows, respectively. Scale bars = 20 µm. Abbreviations: H/E = hematoxylin/eosin; HCC = hepatocellular carcinoma; AFP = alpha fetoprotein. Please click here to view a larger version of this figure.

Figure 5: Brightfield images of HCC cells through several passages. (A) Brightfield image of HCC cells after patient's HCC tissue digestion and after plating at day 0. Magnification 4x, scale bar = 200 µm. (B) Brightfield image of HCC cells 48 h after the first plating. Formation of small organoids. Magnification 4x, scale bar = 200 µm. (C) Brightfield image of HCC organoids at passage 15. Magnification 4x, scale bar = 200 µm. (D) Brightfield image of HCC organoids at passage 15. Magnification 10x, scale bar = 100 µm. (E) Brightfield image of HCC organoids at passage 18 after defrosting. Magnification 4x, scale bar 200 µm. Abbreviation: HCC = hepatocellular carcinoma. Please click here to view a larger version of this figure.
| Dissociation Buffer | Add to 5 mL of Ad-DF+++ : | |||
| o 236 µL of Collagenase IV (50 mg /mL or 11653 U/mL); 202.6 mg of Collagenase IV in 4 mL of 0.15 M NaCl. Filter with 0.22 µm filter and stored at -20 °C. | ||||
| o 10 µL of DNAse (10,000 Kunitz U/mL); 11 mg (2,824 U/mg) in 3 mL of 0.15 M NaCl. Filter with 0.22 µm filter and stored at -20 °C. | ||||
| o 14 µL of Hyaluronidase (339 kU/mL); 191.3 mg in 2 mL of 0.15 M NaCl. Filter with 0.22 µm filter and stored at -20 °C. | ||||
| o 333 µL of BSA; 1 g in 100 mL of 0.15 M NaCl (1% Stock Solution). Filter with 0.22 filter and stored at -20 °C. | ||||
| o 1 µL of 10 mM stock of ROCK Inhibitor | ||||
| Cell suspension freezing medium | To 35 mL of RPMI medium, add 10 mL of FBS and 5 mL of DMSO | |||
| Mix well and aliquot 5 mL in 15 mL tube | ||||
| Store at -20 °C | ||||
| Ad-DF+++ | To 500 mL of adDMEM/F12, add | |||
| o 5 mL of GlutaMAX | ||||
| o 5 mL of HEPES 1 M | ||||
| o 5 mL of Pen-Strep | ||||
| Can be stored at 4 °C for 4 weeks | ||||
Table 1: Solutions for tissue dissociation and organoid generation.
| Mouse Number | Left Lateral | Medial | Superior Right | Inferior Right | Anterior + Posterior Caudate |
| Mouse 1 | 32.317 | 30.894 | 13.618 | 13.211 | 9.959 |
| Mouse 2 | 38.697 | 30.143 | 12.220 | 10.387 | 8.554 |
| Mouse 3 | 32.665 | 32.265 | 12.625 | 11.824 | 10.621 |
| Mouse 4 | 30.000 | 36.721 | 12.623 | 9.508 | 11.148 |
| Mouse 5 | 31.562 | 34.944 | 13.849 | 10.145 | 9.501 |
| Mouse 6 | 32.203 | 35.593 | 14.068 | 9.661 | 8.475 |
| Mouse 7 | 35.591 | 28.504 | 13.071 | 11.496 | 11.339 |
| Mouse 8 | 34.773 | 32.045 | 13.864 | 8.636 | 10.682 |
| Mouse 9 | 33.167 | 41.128 | 7.960 | 7.794 | 9.950 |
| Mouse 10 | 31.751 | 31.751 | 16.367 | 9.493 | 10.638 |
| Median | 32.491 | 32.155 | 13.344 | 9.903 | 10.290 |
| IQR | 2.908 | 4.485 | 1.225 | 2.000 | 1.069 |
Table 2: Results of an experiment showing weight distribution of the different mouse liver lobules. The results demonstrate that the left lateral lobe and middle lobe are consistently the biggest lobules combining to an average of 65% of the total liver volume. Abbreviation: IQR = interquartile range.
Discussion
Critical steps in the protocol
Mouse anesthesia
The goal of the anesthesia is to convert the mouse safely into a state in which it will tolerate surgical manipulation. At the same time, the effect of the anesthesia should not be too strong, to prevent cardiopulmonary arrest and ensuing death. Inhalant anesthesia is easier to dose, allowing the researcher to adjust the dose by manipulating the flow rate of the vaporizer. The effect of the change is quick. This helps prevent a mouse from moving to stage IV anesthesia/asphyxia once the researcher recognizes the symptoms/signs of stage III3 (Depression stage of Anesthesia). Isoflurane is commonly employed as the primary anesthetic in mouse studies for surgical procedures and short-term experiments. This is primarily due to its relatively mild impact on the heart compared to injectable agents like pentobarbital and urethane, or the ketamine/xylazine mixture11,12. According to the literature, 1.5%-2.5% Isoflurane will safely anesthetize most mice13. It is however clearly stated that this is dependent on multiple factors (e.g., age of mice, mouse strain, surgical stimulation) and this fits to our experience.
Generation of organoids
After the tissue harvest and during the handling of the tissue samples, the tissue should not be allowed to dry out. If necessary, the tissue must be remoistened with sterile saline. For HCC tissue or liver tissue in general, no further washing step is required. While dissociating the HCC, the consistency of the tissue may change due to the cutting and the tissue may become viscous or glutinous as a result. This is not considered a problem in the protocol. The digestion of the tissue is a balanced process with either an insufficient dissolution degree (visible pieces of tissue/big cell clusters) or an excessive process leading to cell death/low cell viability. The total time of the incubation must be adjusted accordingly to avoid any loss of cells.
Injection of organoids
There are different possible injection sites for the tumor organoids. The accessibility to these lobes is different and this dictates the difficulty of injection. A schematic of the mouse liver lobes is provided in Figure 3A. We opt to inject into the RSL as this allows us to resect the LLL and the ML. Both resections remove a relevant amount of liver tissue to induce liver regeneration and are fairly easy to perform compared to resections of the smaller lobes. As shown in the representative results section, the hypertrophy of the liver tissue is stronger in the RSL than in the Right inferior lobe (RIL) after a 70% resection. For investigating the impact of liver regeneration on tumor growth, this higher degree of regeneration is favorable.
We suggest taking aside a certain number of cells during the preparation of the injection mixture. We recommend embedding these cells in Histogel and preparing slides for H/E staining. This is a very important moment of quality control as the organoids are passaged several times and there are multiple occasions during which contamination can occur. If this is not done, a different or changed entity from the original PDO can be injected. As it takes the tumor several weeks to grow and there is a marked difference in growth kinetics of the same HCC organoids, confusion can arise.
Surgical method
To achieve a resection of approximately 30% of liver tissue, we perform a resection of the LLL. This is the easiest resection to perform in our opinion. The accessibility is better compared to the middle lobe and the risk of causing damage to vital structures (V. cava) is low. We perform the resection by using a ligature but it is also possible to use clips14.
Regarding the major resection, it must be mentioned that it is more difficult to place the ligature on the middle lobe and this procedure leaves more residual tissue than the resection of the LLL. Tying the knot too close to the V. cava can cause an inflow obstruction to the heart, leading to rapid mortality. It is therefore recommended to accept some degree of residual tissue with the resection of the ML. Furthermore, during the resection of the ML, due to the anatomic relation of the gallbladder and the lobe, the gallbladder is removed with the lobe.
Regarding the closure of the abdomen, we are aware of groups performing two layered closures (peritoneum and skin independently), but we consider this as unnecessary as our method leads to no complications and has the benefit of being quicker. This shortens the operation time and lessens the burden on the mouse by anesthesia15.
Modifications and troubleshooting of the method
There are several modifications of this protocol and the 30% and 65% resections only represent two possible interventions that can be performed once macroscopic tumor growth has been established. Alternatively, Portal Vein Ligation and Associating Liver Partition and Portal Vein Ligation for Staged Hepatectomy (ALPPS) can be performed16,17. In our experience, troubleshooting mainly concerns the maintenance and care of organoids. In particular, the quality control of checking every organoid population, which is injected into the mice every time is of utmost importance.
Limitations of the method
This method aims to investigate the impact of the regenerative environment of the liver on HCC tumor cells. We are using human-derived organoids and injecting them into the liver of a different species (orthotopic xenograft). This aims at recapitulating the surroundings of the original environment but will still harbor significant shortcomings. Does the liver environment, and especially the environment developing during regeneration, resemble that of a human?
Another important shortcoming is that we are investigating the impact of liver regeneration on macroscopic tumors. It cannot (at least in a controlled, evaluable way) show the impact of regeneration with present micro metastases. This represents the more realistic scenario as liver resection aims at clearing the liver of all tumor cells. What cannot be achieved is clearance of occult micro metastases and this is a possible harbor from which recurrence arises.
The significance of the method with respect to existing/alternative methods
To our knowledge, this is the first model using orthotopic injection of organoids. To date, cell lines or spheroids have been injected into the mouse liver to investigate the impact of liver regeneration. Organoids have several advantages compared to cancer cell lines and spheroids. They are more physiologically relevant as they contain multiple cell types (both differentiated and stem cells), recapitulating the structure and function of the organ being modeled. They display genetic diversity, which is not the case for cell lines and spheroids. Finally, they are more stable models, allowing culture for long periods. This is important in the context of this model and allows the performance of different interventions over time.
Importance and potential applications of the method in specific research areas
This method is of utmost importance as the effect of liver regeneration on tumor recurrence remains unclear. Tumor recurrence after liver surgery is a significant problem, reducing long term survival. Discovering targetable mechanisms by using this model is a long-term goal and would improve the quality of treatment for all HCC patients.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We thank everyone assisting in this project, especially Karolina Guja and Dr. Daniela Liberati, for maintaining the organoids and guaranteeing reproducible quality over time. We thank Eva Breuer and Anurag Gupta from the Hepatobiliary and Transplant Surgery Lab of the University Hospital of Zürich for their support concerning anesthesia. This work was supported by the Surgical Department program "Personenförderung" to G.F.H and F.H.; the St. Clara Forschung AG and the "Stiftung zur Krebsbekämpfung" to G.F.H. and University of Basel, Research Fund Junior Researcher (3MS1087) to M.C-LL.
Materials
| Advanced DMEM/F12 (adDMEM/F12) | Life Technologies | 12634-034 | |
| Bovine Serum Albumin (BSA) | Sigma-Aldrich | A3733-50G | |
| Bupaq Buprenorphinum 0.3 mg pro 1 mL | Streuli Tiergesundheit AG | 1121915AB | |
| Centrifuge 5810R | Eppendorf | ||
| Collagenase IV | Worthington | LS004189 | |
| Corning Matrigel Growth Factor Reduced (GFR) Basement Membrane Matrix (10 mL) | Corning (Merk) | CLS356231-1EA | |
| Countess II Automated Cell Counter | Thermo Fisher | ||
| Deoxyribonuclease I Type IV from Bovine (DNAse) | Sigma-Aldrich | D5025-150KU | |
| DMEM (1x) | Gibco | 41965-039 | |
| DPBS, no calcium, no magnesium (500 mL) | Gibco | 14190-094 | |
| Fetal Bovine Serum (FBS) (South America). Heat Inactivated – (500 mL) | Lubio Science | S181H-500 | |
| Glutamax (100x) | Gibco | 9149793 | |
| Grant SUB Aqua Pro Water Bath | Grant Instruments | ||
| HEPES (1 M) | Thermo Fisher | 15630056 | |
| Histogel | Thermo Fisher | R904012 | specimen-processing gel |
| Hyaluronidase Type IV from sheep (Tested) | Sigma-Aldrich | H6254-500MG | |
| Inverted Microscope Olympus CKX53 | Olympus | ||
| MacsMix Tube Rotator | Miltenyi Biotec | ||
| Penicillin-Streptomycin-Glutamine (100x) | Gibco | 10378-016 | |
| Red Blood Cell Lysis | Roche | 11814389001 | |
| RPMI 1640 Medium | Gibco | 72400-021 | |
| Trypsin-EDTA (0.25%), phenol red (100 mL) | Gibco | 25200-056 | |
| Vitaris CO2 Incubator | Vitaris AG | ||
| Y-27632 dihydrochloride (Rock Inhibitor) | Abmole Bioscience | M1817 |
References
- Huang, J., et al. A randomized trial comparing radiofrequency ablation and surgical resection for HCC conforming to the Milan criteria. Annals of surgery. 252 (6), 903-912 (2010).
- Lim, K. -. C., et al. Systematic review of outcomes of liver resection for early hepatocellular carcinoma within the Milan criteria. The British Journal of Surgery. 99 (12), 1622-1629 (2012).
- Roayaie, S., Jibara, G., Taouli, B., Schwartz, M. Resection of hepatocellular carcinoma with macroscopic vascular invasion. Annals of Surgical Oncology. 20 (12), 3754-3760 (2013).
- Sarpel, U., et al. Outcome for patients treated with laparoscopic versus open resection of hepatocellular carcinoma: case-matched analysis. Annals of Surgical Oncology. 16 (6), 1572-1577 (2009).
- Ou, D. -. L., et al. Development of a PD-L1-expressing orthotopic liver cancer model: implications for immunotherapy for hepatocellular carcinoma. Liver Cancer. 8 (3), 155-171 (2019).
- Nuciforo, S., et al. Organoid models of human liver cancers derived from tumor needle biopsies. Cell Reports. 24 (5), 1363-1376 (2018).
- Broutier, L., et al. Human primary liver cancer-derived organoid cultures for disease modeling and drug screening. Nature Medicine. 23 (12), 1424-1435 (2017).
- Qin, C., et al. The cutting and floating method for paraffin-embedded tissue for sectioning. Journal of Visualized Experiments: JoVE. (139), (2018).
- Feldman, A. T., Wolfe, D. Tissue processing and hematoxylin and eosin staining. Methods in Molecular Biology. 1180, 31-43 (2014).
- Bou About, G., et al. Ultrasound-guided approaches to improve orthotopic mouse xenograft models for hepatocellular carcinoma. Current Protocols in Mouse Biology. 9 (2), 62 (2019).
- Janssen, B. J. A., Smits, J. F. M. Autonomic control of blood pressure in mice: basic physiology and effects of genetic modification. American Journal of Physiology. Regulatory, Integrative and Comparative Physiology. 282 (6), R1545-R1564 (2002).
- Janssen, B. J. A., et al. Effects of anesthetics on systemic hemodynamics in mice. American Journal of Physiology. Heart and Circulatory Physiology. 287 (4), H1618-H1624 (2004).
- Navarro, K. L., et al. Mouse anesthesia: the art and Science. ILAR Journal/National Research Council, Institute of Laboratory Animal Resources. 62 (1-2), 238-273 (2021).
- Kamali, C., et al. Extended liver resection in mice: state of the art and pitfalls-a systematic review. European Journal of Medical Research. 26 (1), 6 (2021).
- Kim, M. P., et al. Generation of orthotopic and heterotopic human pancreatic cancer xenografts in immunodeficient mice. Nature Protocols. 4 (11), 1670-1680 (2009).
- Iwakiri, Y., Cadelina, G., Sessa, W. C., Groszmann, R. J. Mice with targeted deletion of eNOS develop hyperdynamic circulation associated with portal hypertension. American Journal of Physiology. Gastrointestinal and Liver Physiology. 283 (5), G1074-G1081 (2002).
- Schlegel, A., et al. ALPPS. Annals of Surgery. 260 (5), 839-847 (2014).

.
